Abstract
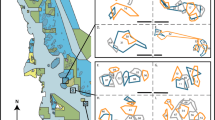
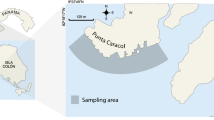
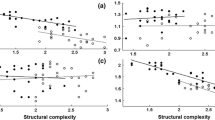
Stoplight parrotfish, Sparisoma viride, are hermaphroditic fish that exhibit a complex social structure in which terminal phase (TP) males control a territory of initial phase (IP) individuals (mostly female, rarely male) called a harem. These fish are prolific herbivores that maintain the health of coral reefs by consuming or removing algae that competes with coral for space. In this study, we estimate the influence of food availability, structural complexity, and conspecific density on territory size in S. viride TP males on reef sites in the middle Florida Keys. Divers estimated the territory sizes of both TP and IP individuals by following fish and dropping markers. Divers also estimated the substrate composition, conspecific density, and physical complexity around these territories. TP territories were roughly circular and only overlapped with IP individuals on the outer edges of the TP territory, resulting in TP territories being significantly larger. While TP territory size was positively influenced by the body size of the TP male and negatively influenced by the number of IP individuals, the average ratio of IP:TP individuals was 2.99:1 (± 0.31 SE, range 1–12) for each TP territory regardless of body size. These results suggest that TP males adjust their territory sizes to maintain an apparent optima for the number of females within harems and that the density of conspecifics can potentially influence the timing of sexual transitions in these fish. These results also suggest that S. viride have a polygynous mating structure that is driven by female defense rather than resource defense.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adam TC, Burkepile DE, Ruttenberg BI, Paddack MJ (2015a) Herbivory and the resilience of Caribbean coral reefs: knowledge gaps and implications for management. Mar Ecol Prog Ser 520:1–20. https://doi.org/10.3354/meps11170
Adam TC, Kelley M, Ruttenberg BI, Burkepile DE (2015b) Resource partitioning along multiple niche axes drives functional diversity in parrotfishes on Caribbean coral reefs. Oecologia 179(4):1173–1185. https://doi.org/10.1007/s00442-015-3406-3
Alevizon WS, Porter JW (2015) Coral loss and fish guild stability on a Caribbean coral reef: 1974–2000. Environ Biol Fish 98(4):1035–1045. https://doi.org/10.1007/s10641-014-0337-5
Allsop DJ, West SA (2003a) Changing sex at the same relative body size. Nature 425(6960):783–784. https://doi.org/10.1038/425783a
Allsop DJ, West SA (2003b) Constant relative age and size at sex change for sequentially hermaphroditic fish. J Evol Biol 16(5):921–929. https://doi.org/10.1046/j.1420-9101.2003.00590.x
Alvarez-Filip L, Dulvy NK, Gill JA, Côté IM, Watkinson AR (2009) Flattening of Caribbean coral reefs: region-wide declines in architectural complexity. Proc Royal Soc B: Biol Sci 276(1669):3019–3025. https://doi.org/10.1098/rspb.2009.0339
Anderson D, Burnham K (2004) Model selection and multi-model inference. Springer-Verlag, New York
Aromaa S, Ilvonen JJ, Suhonen J (2019) Body mass and territorial defence strategy affect the territory size of odonate species. Proc Royal Soc B: Biol Sci 286(1917):20192398. https://doi.org/10.1098/rspb.2019.2398
Bellwood D, Hughes T, Folke C, Nyström M (2004) Confronting the coral reef crisis. Nature 429(6994):827–833. https://doi.org/10.1038/nature02691
Bellwood DR, Hoey AS, Hughes TP (2012) Human activity selectively impacts the ecosystem roles of parrotfishes on coral reefs. Proc Royal Soc B: Biol Sci 279(1733):1621–1629. https://doi.org/10.1098/rspb.2011.1906
Buckman NS, Ogden JC (1973) Territorial behavior of the striped parrotfish, Scarus croicensis, bloch (Scaridae). Ecology 54(6):1377–1382. https://doi.org/10.2307/1934202
Buston PM, Munday PL, Warner RR (2004) Sex change and relative body size in animals. Nature 428(6983):1–1. https://doi.org/10.1038/nature02512a
Catano LB, Barton MB, Boswell KM, Burkepile DE (2017) Predator identity and time of day interact to shape the risk–reward trade-off for herbivorous coral reef fishes. Oecologia 183:763–773. https://doi.org/10.1007/s00442-016-3794-z
Catano LB, Gunn BK, Kelley MC, Burkepile DE (2015) Predation risk, resource quality, and reef structural complexity shape territoriality in a coral reef herbivore. PLoS One 10(2):e0118764. https://doi.org/10.1371/journal.pone.0118764
Davies NB, Krebs JR, West SA (2012) Mating systems. In: Davies NB, Krebs JR, West SA (eds) An introduction to behavioural ecology, 4th edn. John Wiley & Sons, Oxford, UK, pp 254–281
Emlen ST, Oring LW (1977) Ecology, sexual selection, and the evolution of mating systems. Science 197(4300):215–223. https://doi.org/10.1126/science.327542
Eurich JG, Shomaker SM, McCormick MI, Jones GP (2018) Experimental evaluation of the effect of a territorial damselfish on foraging behaviour of roving herbivores on coral reefs. J Exp Mar Biol Ecol 506:155–162. https://doi.org/10.1016/j.jembe.2018.06.009
Genner MJ, Turner GF, Hawkins SJ (1999) Resource control by territorial male cichlid fish in Lake Malawi. J Anim Ecol 68(3):522–529. https://doi.org/10.1046/j.1365-2656.1999.00301.x
Gerking SD (1959) The restricted movement of fish populations. Biol Rev 34(2):221–242. https://doi.org/10.1111/j.1469-185X.1959.tb01289.x
Gintert BE et al (2018) Marked annual coral bleaching resilience of an inshore patch reef in the Florida Keys: a nugget of hope, aberrance, or last man standing? Coral Reefs 37:533–547. https://doi.org/10.1007/s00338-018-1678-x
Grant JWA, Kramer DL (1992) Temporal clumping of food arrival reduces its monopolization and defence by zebrafish Brachydanio rerio. Anim Behav 44(1):101–110. https://doi.org/10.1016/S0003-3472(05)80759-6
Harborne AR, Rogers A, Bozec Y-M, Mumby PJ (2017) Multiple stressors and the functioning of coral reefs. Ann Rev Mar Sci 9:445–468. https://doi.org/10.1146/annurev-marine-010816-060551
Harestad AS, Bunnel FL (1979) Home range and body weight--a reevaluation. Ecology 60(2):389–402. https://doi.org/10.2307/1937667
Hawkins JP, Roberts CM (2004) Effects of fishing on sex-changing Caribbean parrotfishes. Biol Conserv 115(2):213–226. https://doi.org/10.1016/S0006-3207(03)00119-8
Hixon MA (1980) Food production and competitor density as the determinants of feeding territory size. Am Nat 115(4):510–530. https://doi.org/10.1086/283577
Hixon MA, Beets JP (1993) Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol Monogr 63(1):77–101. https://doi.org/10.2307/2937124
Hoey AS, Bonaldo RM (2018) Biology of parrotfishes. CRC Press, Boca Raton
Hughes TP et al (2003) Climate change, human impacts, and the resilience of coral reefs. Science 301(5635):929–933. https://doi.org/10.1126/science.108504
Jackson JB, Donovan MK, Cramer KL, Lam VV (2014) Status and trends of Caribbean coral reefs: 1970-2012. Global Coral Reef Monitoring Network, IUCN, Gland, Switzerland
Jackson JB et al (2001) Historical overfishing and the recent collapse of coastal ecosystems. Science 293(5530):629–637. https://doi.org/10.1126/science.1059199
Keeley MP (1998) Food abundance, intruder pressure, and body size as determinants of territory size in juvenile steelhead trout (Oncorhynchus mykiss). Behaviour 135(1):65–82. https://doi.org/10.1163/156853998793066393
Kohler KE, Gill SM (2006) Coral Point Count with Excel extensions (CPCe): a Visual Basic program for the determination of coral and substrate coverage using random point count methodology. Comput Geosci 32(9):1259–1269. https://doi.org/10.1016/j.cageo.2005.11.009
Larson RJ (1980) Territorial behavior of the black and yellow rockfish and gopher rockfish (Scorpaenidae, Sebastes). Mar Biol 58(2):111–122. https://doi.org/10.1007/bf00396122
Lirman D (2001) Competition between macroalgae and corals: effects of herbivore exclusion and increased algal biomass on coral survivorship and growth. Coral Reefs 19(4):392–399. https://doi.org/10.1007/s003380000125
Mallela J, Fox RJ (2018) The role of parrotfishes in the destruction and construction of coral reefs. In: Howy AS, Bonaldo RM (eds) Biology of parrotfishes. CRC Press, Boca Raton, pp 161–196
Manzello DP, Enochs IC, Kolodziej G, Carlton R (2015) Coral growth patterns of Montastraea cavernosa and Porites astreoides in the Florida Keys: the importance of thermal stress and inimical waters. J Exp Mar Biol Ecol 471:198–207. https://doi.org/10.1016/j.jembe.2015.06.010
McCormick MI (1994) Comparison of field methods for measuring surface-topography and their associations with a tropical reef fish assemblage. Mar Ecol Prog Ser 112(1-2):87–96. https://doi.org/10.3354/meps112087
Molloy PP, Paddack MJ, Reynolds JD, Gage MJG, Côté IM (2011) Relative size-at-sex-change in parrotfishes across the Caribbean: is there variance in a supposed life-history invariant? Evol Ecol 25(2):429–446. https://doi.org/10.1007/s10682-010-9404-3
Muller EM, Sartor C, Alcaraz NI, van Woesik R (2020) Spatial epidemiology of the stony-coral-tissue-loss disease in Florida. Front Mar Sci 7(163):1–11. https://doi.org/10.3389/fmars.2020.00163
Mumby PJ (2006) The impact of exploiting grazers (Scaridae) on the dynamics of Caribbean coral reefs. Ecol Appl 16(2):747–769. https://doi.org/10.1890/1051-0761(2006)016[0747:TIOEGS]2.0.CO;2
Mumby PJ, Steneck RS (2008) Coral reef management and conservation in light of rapidly evolving ecological paradigms. Trends Ecol Evol 23(10):555–563. https://doi.org/10.1016/j.tree.2008.06.011
Mumby PJ, Wabnitz CC (2002) Spatial patterns of aggression, territory size, and harem size in five sympatric Caribbean parrotfish species. Environ Biol Fish 63(3):265–279. https://doi.org/10.1023/A:1014359403167
Munday PL, Buston PM, Warner RR (2006) Diversity and flexibility of sex-change strategies in animals. Trends Ecol Evol 21(2):89–95. https://doi.org/10.1016/j.tree.2005.10.020
Newman SP et al (2015) Reef flattening effects on total richness and species responses in the Caribbean. J Anim Ecol 84(6):1678–1689. https://doi.org/10.1111/1365-2656.12429
Norman M, Jones G (1984) Determinants of territory size in the pomacentrid reef fish, Parma victoriae. Oecologia 61(1):60–69. https://doi.org/10.1007/BF00379090
Paddack MJ, Cowen RK, Sponaugle S (2006) Grazing pressure of herbivorous coral reef fishes on low coral-cover reefs. Coral Reefs 25(3):461–472. https://doi.org/10.1007/s00338-006-0112-y
Paddack MJ, Sponaugle S, Cowen RK (2009) Small-scale demographic variation in the stoplight parrotfish Sparisoma viride. J Fish Biol 75(10):2509–2526. https://doi.org/10.1111/j.1095-8649.2009.02451.x
Pandolfi JM et al (2003) Global trajectories of the long-term decline of coral reef ecosystems. Science 301(5635):955–958. https://doi.org/10.1126/science.1085706
Pandolfi JM et al (2005) Are U.S. coral reefs on the slippery slope to slime? Science 307(5716):1725–1726. https://doi.org/10.1126/science.1104258
Robertson DR, Warner RR (1978) Sexual patterns in the labroid fishes of the western Caribbean, II: the parrotfishes (Scaride). Smithson Cont Zool 255:1–26. https://doi.org/10.5479/si.00810282.255
SAS Institute Inc (1989-2016) JMP® Pro, Version 13.2 edn. SAS Institute, Cary, NC
Shapiro DY (1987) Differentiation and evolution of sex change in fishes. BioScience 37(7):490–497. https://doi.org/10.2307/1310421
Shapiro DY, Lubbock R (1980) Group sex ratio and sex reversal. J Theor Biol 83(3):411–426. https://doi.org/10.1016/0022-5193(80)90048-X
Smith KM, Quirk-Royal BE, Drake-Lavelle K, Childress MJ (2018) Influences of ontogenetic phase and resource availability on parrotfish foraging preferences in the Florida Keys, FL (USA). Mar Ecol Prog Ser 603:175–187. https://doi.org/10.3354/meps12718
Steneck RS, Mumby PJ, MacDonald C, Rasher DB, Stoyle G (2018) Attenuating effects of ecosystem management on coral reefs. Sci Adv 4(5):1–11. https://doi.org/10.1126/sciadv.aao5493
Swihart RK, Slade NA, Bergstrom BJ (1988) Relating body size to the rate of home range use in mammals. Ecology 69(2):393–399. https://doi.org/10.2307/1940437
Tamburello N, Côté IM, Dulvy NK (2015) Energy and the scaling of animal space use. Am Nat 186(2):196–211. https://doi.org/10.1086/682070
Thurber RV et al (2012) Macroalgae decrease growth and alter microbial community structure of the reef-building coral, Porites astreoides. PLoS One 7(9):e44246. https://doi.org/10.1371/journal.pone.0044246
Vallès H, Oxenford HA (2014) Parrotfish size: a simple yet useful alternative indicator of fishing effects on Caribbean reefs? PLoS One 9(1):e86291. https://doi.org/10.1371/journal.pone.0086291
van Rooij JM, de Jong E, Vaandrager F, Videler JJ (1996a) Resource and habitat sharing by the stoplight parrotfish, Sparisoma viride, a Caribbean reef herbivore. Environ Biol Fishes 47(1):81–91. https://doi.org/10.1007/bf00002381
van Rooij JM, Kok JP, Videler JJ (1996b) Local variability in population structure and density of the protogynous reef herbivore Sparisoma viride. Environ Biol Fish 47(1):65–80. https://doi.org/10.1007/BF00002380
van Rooij JM, Kroon FJ, Videler JJ (1996c) The social and mating system of the herbivorous reef fish Sparisoma viride: one-male versus multi-male groups. Environ Biol Fish 47(4):353–378. https://doi.org/10.1007/BF00005050
Warner RR (1984) Mating behavior and hermaphroditism in coral reef fishes. Am Sci 72(2):128–136
Acknowledgements
This research was made possible by the Florida Keys National Marine Sanctuary (permit #FKNMS-2013-077-A1). We would also like to thank Rob Hulsey, Mason Collins, and other members of the Clemson University Conservation of Marine Resources Creative Inquiry team for their assistance in the field and laboratory.
Funding
Funding for this study was provided by Clemson University Creative Inquiry Initiative, the American Society of Ichthyologists and Herpetologists, and Clemson University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This research was based on observations only and was not required to be reviewed by the Institutional Animal Care and Use Committee (IACUC) at Clemson University. All work was permitted by the Florida Keys National Marine Sanctuary (Permit #FKNMS-2013-077-A1).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supporting information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Smith, K.M., Chamberlain, L., Whitaker, S. et al. Factors influencing stoplight parrotfish territoriality and social structure in the middle Florida Keys. Environ Biol Fish 106, 585–595 (2023). https://doi.org/10.1007/s10641-023-01394-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-023-01394-1