Abstract
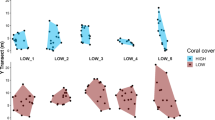
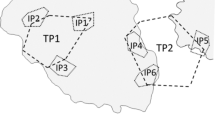
The chevron butterflyfish, Chaetodon trifascialis, is among the most specialised coral-feeding fish, and while it is known to be very susceptible to extensive depletion of its preferred coral prey (tabular Acropora spp.), their specific responses to changing coral cover are poorly understood. The purpose of this study was to test for variation in territorial behaviour and condition of C. trifascialis relative to spatial variation in coral cover across four mid-shelf reefs on the Great Barrier Reef. Explicit consideration was also given to the territorial arrangement and interactions among sympatric individuals, with a view to better understanding the sociality of this species. Variation in overall coral cover (which ranged from 26.5–73.4% among sites) as well as cover of tabular Acropora (13.3–44.8%) had limited effect on the territoriality or body condition of C. trifascialis. Rather, individual variation in territoriality was attributable to differences in gender and size of fish. Male C. trifascialis were generally larger and also had larger territories than female counterparts. They also interacted with conspecifics (and congenerics) much more than females. Taken together, these results support previous assertions that C. trifascialis is haremic. There was, however, limited evidence of male territories encompassing the territories of >1 female. While the sociality of C. trifascialis is clearly atypical of Chaetodon butterflyfishes, more work is needed to understand their reproductive biology as well as their behavioural responses to changing coral cover.





Similar content being viewed by others
References
Alvarez-Filip L, Côté IM, Gill JA, Watkinson AR, Dulvy NK (2011) Region-wide temporal and spatial variation in Caribbean reef architecture: is coral cover the whole story? Glob Chang Biol 17(7):2470–2477. https://doi.org/10.1111/j.1365-2486.2010.02385.x
Bellwood DR, Klanten S, Cowman PF, Pratchett MS, Konow N, Van Herwerden L (2010) Evolutionary history of the butterflyfishes (f: Chaetodontidae) and the rise of coral feeding fishes. J Evol Biol 23(2):335–349. https://doi.org/10.1111/j.1420-9101.2009.01904.x
Berumen ML, Pratchett (2006a) Effects of resource availability on the competitive behaviour of butterflyfishes (Chaetodontidae). Proc 10th Int Coral Reef Symp 1:644–650
Berumen ML, Pratchett (2006b) Recovery without resilience: persistent disturbance and long-term shifts in the structure of fish and coral communities at Tiahura reef, Moorea. Coral Reefs 25(4):647–653. https://doi.org/10.1007/s00338-006-0145-2
Berumen ML, Pratchett MS (2008) Trade-offs associated with dietary specialization in corallivorous butterflyfishes (Chaetodontidae: Chaetodon). Behav Ecol Sociobiol 62(6):989–994. https://doi.org/10.1007/s00265-007-0526-8
Berumen ML, Pratchett MS, McCormick MI (2005) Within-reef differences in diet and body condition of coral-feeding butterflyfishes (Chaetodontidae). Mar Ecol Prog Ser 287:217–227. https://doi.org/10.3354/meps287217
Blowes SA, Pratchett MS, Connolly SR (2013) Heterospecific aggression and dominance in a guild of coral-feeding fishes: the roles of dietary ecology and phylogeny. Am Nat 182(2):157–168. https://doi.org/10.1086/670821
Brandl SJ, Bellwood DR (2015) Coordinated vigilance provides evidence for direct reciprocity in coral reef fishes. Sci Rep 5:1–13. https://doi.org/10.1038/srep14556
Brooker RM, Munday PL, Sj B, Jones GP (2014) Local extinction of a coral reef fish explained by inflexible prey choice. Coral Reefs 33(4):891–896. https://doi.org/10.1007/s00338-014-1197-3
Burnham KP, Anderson DR (2002) Model selection of an appropriate biological model: the key to modern data analysis. In: McCullough DR, Barrett RH (eds) Wildlife 2001: populations. Elsevier, London, pp 16–30. https://doi.org/10.1007/978-94-011-2868-1_3
Cheal AJ, MacNeil MA, Emslie MJ, Sweatman HPA (2017) The threat to coral reefs from more intense cyclones under climate change. Glob Chang Biol 23(4):1511–1524. https://doi.org/10.1111/gcb.13593
Cole AJ, Pratchett MS, Jones GP (2008) Diversity and functional importance of coral-feeding fishes on tropical coral reefs. Fish Fish 9(3):286–307. https://doi.org/10.1111/j.1467-2979.2008.00290.x
Craig SR, MacKenzie DS, Jones G, Gatlin DM III (2000) Seasonal changes in the reproductive condition and body composition of free-ranging red drum, Sciaenops ocellatus. Aquaculture 190(1–2):89–102. https://doi.org/10.1016/S0044-8486(00)00386-0
Darling ES, Graham NA, Januchowski-Hartley FA, Nash KL, Pratchett MS, Wilson SK (2017) Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs 36(2):561–575. https://doi.org/10.1007/s00338-017-1539-z
Duarte CM, Agusti S, Barbier E, Britten GL, Castilla JC, Gattuso JP, Fulweiler RW, Hughes TP, Knowlton N, Lovelock CE, Lotze HK (2020) Rebuilding marine life. Nature 580(7801):39–51. https://doi.org/10.1038/s41586-020-2146-7
Elliott JA, Patterson MR, Staub CG, Koonjul M, Elliott SM (2018) Decline in coral cover and flattening of the reefs around Mauritius (1998–2010). PeerJ 6:e6014. https://doi.org/10.7717/peerj.6014
Emslie MJ, Pratchett MS, Cheal AJ (2011) Effects of different disturbance types on butterflyfish communities of Australia’s great barrier reef. Coral Reefs 30(2):461–471. https://doi.org/10.1007/s00338-011-0730-x
Feary DA, Almany GR, McCormick MI, Jones GP0 (2007) Habitat choice, recruitment and the response of coral reef fishes to coral degradation. Oecologia 153:727–737. https://doi.org/10.1007/s00442-007-0773-4
Feary DA, Bauman AG, Guest J, Hoey AS (2018) Trophic plasticity in an obligate corallivorous butterflyfish. Mar Ecol Prog Ser 605:165–171. https://doi.org/10.3354/meps12771
Godwin J (2009) Social determination of sex in reef fishes. Sem Cell Dev Biol 20(3):264–270. https://doi.org/10.1016/j.semcdb.2008.12.003
Graham NA (2007) Ecological versatility and the decline of coral feeding fishes following climate driven coral mortality. Mar Biol 153(2):119–127. https://doi.org/10.1007/s00227-007-0786-x
Graham NA, Wilson SK, Pratchett MS, Polunin NV, Spalding MD (2009) Coral mortality versus structural collapse as drivers of corallivorous butterflyfish decline. Biodivers Conserv 18:3325–3336. https://doi.org/10.1007/s10531-009-9633-3
Graham NA, McClanahan TR, MacNeil MA, Wilson SK, Cinner JE, Huchery C, Holmes TH (2017) Human disruption of coral reef trophic structure. Curr Biol 27(2):231–236. https://doi.org/10.1016/j.cub.2016.10.062
Harmelin-Vivien ML (1989) Implications of feeding specialisation on the recruitment process and community structure of butterflyfishes. Environ Biol Fish 25(1–3):101–110. https://doi.org/10.1007/978-94-009-2325-6_7
Harmelin-Vivien ML, Bouchon-Navaro Y (1983) Feeding diets and significance of coral feeding among chaetodontid fishes in Moorea (French Polynesia). Coral Reefs 2:119–127. https://doi.org/10.1007/bf02395282
Henderson RJ, Sargent JR, Hopkins CC (1984) Changes in the content and fatty acid composition of lipid in an isolated population of the capelin Mallotus villosus during sexual maturation and spawning. Mar Biol 78(3):255–263. https://doi.org/10.1007/BF00393011
Hing ML, Klanten OS, Dowton M, Brown KR, Wong MY (2018) Repeated cyclone events reveal potential causes of sociality in coral-dwelling Gobiodon fishes. PLoS One 13(9):e0202407. https://doi.org/10.1371/journal.pone.0202407
Hobbs JA, Munday PL, Jones GP (2004) Social induction of maturation and sex determination in a coral reef fish. Proc Roy Soc B 271:2109–2114. https://doi.org/10.1098/rspb.2004.2845
Hoegh-Guldberg O, Bruno FJ (2010) The impact of climate change on the world’s marine ecosystems. Science 328(5985):1523–1528. https://doi.org/10.1126/science.1189930
Hoey J., McCormick M. I., Hoey A. S. (2007) Influence of depth on sex-specific energy allocation patterns in a tropical reef fish. Coral Reefs 26:603–613
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TC (2017) Global warming and recurrent mass bleaching of corals. Nature 543(7645):373–377. https://doi.org/10.1038/nature21707
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM, Baird AH, Baum JK, Berumen ML, Bridge TC, Claar DC (2018a) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359(6371):80–83. https://doi.org/10.1126/science.aan8048
Hughes TP, Kerry JT, Baird AH, Connolly SR, Dietzel A, Eakin CM, Heron SF, Hoey AS, Hoogenboom MO, Liu G, McWilliam MJ (2018b) Global warming transforms coral reef assemblages. Nature 556:492–496. https://doi.org/10.1038/s41586-018-0041-2
Hughes DJ, Alderdice R, Cooney C, Kühl M, Pernice M, Voolstra CR, Suggett DJ (2020) Coral reef survival under accelerating ocean deoxygenation. Nat Clim Chang 10:296–307. https://doi.org/10.1038/s41558-020-0737-9
Irons DK (1989) Temporal and areal feeding behavior of the butterflyfish, Chaetodon trifascialis, at Johnston atoll. Environ Biol Fish 25(1–3):187–193. https://doi.org/10.1007/BF00002211
Keith SA, Baird AH, Hobbs JP, Woolsey ES, Hoey AS, Fadli N, Sanders NJ (2018) Synchronous behavioural shifts in reef fishes linked to mass coral bleaching. Nat Clim Chang 8(11):986–991. https://doi.org/10.1038/s41558-018-0314-7
Khan JA, Goatley CH, Brandl SJ, Tebbett SB, Bellwood DR (2017) Shelter use by large reef fishes: long-term occupancy and the impacts of disturbance. Coral Reefs 36(4):1123–1132. https://doi.org/10.1007/s00338-017-1604-7
Kokita T, Nakazono A (2001) Rapid response of an obligately corallivorous filefish Oxymonacanthus longirostris (Monacanthidae) to a mass coral bleaching event. Coral Reefs 20(2):155–158. https://doi.org/10.1007/s003380100153
Lung MA, Childress MJ (2007) The influence of conspecifics and predation risk on the vigilance of elk (Cervus elaphus) in Yellowstone National Park. Behav Ecol 18(1):12–20. https://doi.org/10.1093/beheco/arl066
MacNeil MA, Chong-Seng KM, Pratchett DJ, Thompson CA, Messmer V, Pratchett MS (2017) Age and growth of an outbreaking Acanthaster cf. solaris population within the great barrier reef. Diversity 9(1):18. https://doi.org/10.3390/d9010018
MacNeil MA, Mellin C, Matthews S, Wolff NH, McClanahan TR, Devlin M, Drovandi C, Mengersen K, Graham NA (2019) Water quality mediates resilience on the great barrier reef. Nat Ecol Evol 3(4):620–627. https://doi.org/10.1038/s41559-019-0832-3
Madin JS, Baird AH, Bridge TC, Connolly SR, Zawada KJ, Dornelas M (2018) Cumulative effects of cyclones and bleaching on coral cover and species richness at Lizard Island. Mar Ecol Prog Ser 604:263–268. https://doi.org/10.3354/meps12735
McWilliam M, Pratchett MS, Hoogenboom MO, Hughes TP (2020) Deficits in functional trait diversity following recovery on coral reefs. Proc Roy Soc B 287(1918):20192628. https://doi.org/10.1098/rspb.2019.2628
Montanari SR, Hobbs JP, Pratchett MS, Bay LK, van Herwerden L (2017) Naturally occurring hybrids of coral reef butterflyfishes have similar fitness compared to parental species. PLoS One 12(3):e0173212. https://doi.org/10.1371/journal.pone.0173212
Nowicki JP, O’Connell LA, Cowman PF, Walker SP, Coker DJ, Pratchett MS (2018a) Variation in social systems within Chaetodon butterflyfishes, with special reference to pair bonding. PLoS One 13(4):e0194465. https://doi.org/10.1371/journal.pone.0194465
Nowicki JP, Walker SP, Coker DJ, Hoey AS, Nicolet KJ, Pratchett MS (2018b) Pair bond endurance promotes cooperative food defense and inhibits conflict in coral reef butterflyfish. Sci Rep 8:6295. https://doi.org/10.1038/s41598-018-24412-0
Ortiz JC, Wolff NH, Anthony KR, Devlin M, Lewis S, Mumby PJ (2018) Impaired recovery of the great barrier reef under cumulative stress. Sci Adv 4:eaar6127. https://doi.org/10.1126/sciadv.aar6127
Pinheiro J, Bates D, DebRoy S, Sarkar D, Core Team R (2020) Nlme: linear and nonlinear mixed effects models. R package version 31-147
Pisapia C, Burn D, Pratchett MS (2019) Changes in the population and community structure of corals during recent disturbances (February 2016-October 2017) on Maldivian coral reefs. Sci Rep 9:8402. https://doi.org/10.1038/s41598-019-44809-9
Pratchett MS (2005) Dietary overlap among coral-feeding butterflyfishes (Chaetodontidae) at Lizard Island, northern great barrier reef. Mar Biol 148(2):373–382. https://doi.org/10.1007/s00227-005-0084-4
Pratchett MS (2014) Feeding preferences and dietary specialisation among obligate coral-feeding butterflyfishes. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of Butterflyfishes. CRC Press, Boca Raton, pp 140–179. https://doi.org/10.1201/b15458-7
Pratchett MS, Wilson SK, Berumen ML, McCormick MI (2004) Sublethal effects of coral bleaching on an obligate coral feeding butterflyfish. Coral Reefs 23(3):352–356. https://doi.org/10.1007/s00338-004-0394-x
Pratchett MS, Pradjakusuma OA, Jones GP (2006a) Is there a reproductive basis to solitary living versus pair-formation in coral reef fishes? Coral Reefs 25(1):85–92. https://doi.org/10.1007/s00338-005-0081-6
Pratchett MS, Wilson SK, Baird AH (2006b) Declines in the abundance of Chaetodon butterflyfishes following extensive coral depletion. J Fish Biol 69(5):1269–1280. https://doi.org/10.1111/j.1095-8649.2006.01161.x
Pratchett MS, Munday PL, Wilson SK, Graham NA, Cinner JE, Bellwood DR, Jones GP, Polunin NV, McClanahan TR (2008) Effects of climate-induced coral bleaching on coral-reef fishes - ecological and economic consequences. Oceanogr Mar Biol Ann Rev 46:257–302
Pratchett MS, Caballes CF, Rivera-Posada JA, Sweatman HPA (2014a) Limits to understanding and managing outbreaks of crown-of-thorns stafish (Acanthaster spp.). Oceanogr Mar Biol Ann Rev 52:133–199
Pratchett MS, Chong-Seng KM, Feary DA, Hoey AS, Fulton CJ, Nowicki JP, Dewan AK, Walker AK, Berumen ML (2014b) Butterflyfishes as a model group for reef fish ecology: important and emerging research topics. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of Butterflyfishes. CRC Press, Boca Raton, pp 310–333
Pratchett MS, Thompson CA, Hoey AS, Cowman PF, Wilson SK (2018) Effects of coral bleaching and coral loss on the structure and function of reef fish assemblages. In: van Oppen MJH, Lough JM (eds) Coral bleaching. Springer.pp, Cham, pp 265–293. https://doi.org/10.1007/978-3-319-75393-5_11
Pratchett MS, McWilliam MJ, Riegl B (2020) Contrasting shifts in coral assemblages with increasing disturbances. Coral Reefs 39:783–793. https://doi.org/10.1007/s00338-020-01936-4
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Reese ES (1989) Orientation behavior of butterflyfishes (family Chaetodontidae) on coral reefs: spatial learning of route specific landmarks and cognitive maps. Env Biol Fish 25(1–3):79–86. https://doi.org/10.1007/BF00002202
Richardson LE, Graham NA, Pratchett MS, Eurich JG, Hoey AS (2018) Mass coral bleaching causes biotic homogenization of reef fish assemblages. Glob Chang Biol 24(7):3117–3129. https://doi.org/10.1111/gcb.14119
Righton D, Mills C (2006) Application of GIS to investigate the use of space in coral reef fish: a comparison of territorial behaviour in two Red Sea butterflyfishes. Int J Geographical Info Sci 20(2):215–232. https://doi.org/10.1080/13658810500399159
Righton D, Miller M, Ormond R (1998) Correlates of territory size in the butterflyfish Chaetodon austriacus (Rüppell). J Exp Mar Biol Ecol 226(2):183–193. https://doi.org/10.1016/S0022-0981(97)00235-9
Roberts CM, Ormond RF (1992) Butterflyfish social behaviour, with special reference to the incidence of territoriality: a review. Environ Biol Fish 34(1):79–93. https://doi.org/10.1007/BF00004786
Roberts CM, McClean CJ, Veron JE, Hawkins JP, Allen GR, McAllister DE, Mittermeier CG, Schueler FW, Spalding M, Wells F, Vynne C (2002) Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295(5558):1280–1284. https://doi.org/10.1126/science.1067728
Robertson DR (1972) Social control of sex reversal in a coral-reef fish. Science 177(4053):1007–1009. https://doi.org/10.1126/science.177.4053.1007
Russ GR, Leahy SM (2017) Rapid decline and decadal-scale recovery of corals and Chaetodon butterflyfish on Philippine coral reefs. Mar Biol 164(1):29. https://doi.org/10.1007/s00227-016-3056-y
Sadovy Y, Shapiro DY (1987) Criteria for the diagnosis of hermaphroditism in fishes. Copeia 1987(1):136–156. https://doi.org/10.2307/1446046
Samways MJ (2005) Breakdown of butterflyfish (Chaetodontidae) territories associated with the onset of a mass coral bleaching event. Aquat Cons Mar FW Ecosys 15(S1):101–107
Sheppard C, Sheppard A, Fenner D (2020) Coral mass mortalities in the Chagos archipelago over 40 years: regional species and assemblage extinctions and indications of positive feedbacks. Mar Pollut Bull 154:111075. https://doi.org/10.1016/j.marpolbul.2020.111075
Shuster SM, Wade MJ (2003) Mating systems and strategies. Princeton University Press, Princeton
Skirving WJ, Heron SF, Marsh BL, Liu G, De La Cour JL, Geiger EF, Eakin CM (2019) The relentless march of mass coral bleaching: a global perspective of changing heat stress. Coral Reefs 38(4):547–557. https://doi.org/10.1007/s00338-019-01799-4
Theilacker GH (1978) Effect of starvation on the histological and morphological characteristics of jack mackerel, Trachurus symmetricus, larvae. Fish Bull 76(2):403–414
Thompson CA, Matthews S, Hoey AS, Pratchett MS (2019) Changes in sociality of butterflyfishes linked to population declines and coral loss. Coral Reefs 38(3):527–537. https://doi.org/10.1007/s00338-019-01792-x
Todd EV, Liu H, Muncaster S, Gemmell NJ (2016) Bending genders: the biology of natural sex change in fish. Sex Dev 10(5–6):223–241. https://doi.org/10.1159/000449297
Tricas TC (1989) Determinants of feeding territory size in the corallivorous butterflyfish, Chaetodon multicinctus. Anim Behav 37(1–2):830–841. https://doi.org/10.1016/0003-3472(89)90067-5
Vanhatalo J, Hosack GR, Sweatman HPA (2017) Spatiotemporal modelling of crown-of-thorns starfish outbreaks on the great barrier reef to inform control strategies. J Appl Ecol 54(1):188–197. https://doi.org/10.1111/1365-2664.12710
Whiteman EA, Côté IM (2004) Monogamy in marine fishes. Biol Rev 79(2):351–375. https://doi.org/10.1017/S1464793103006304
Whiteside MA, Langley EJ, Madden JR (2016) Males and females differentially adjust vigilance levels as group size increases: effect on optimal group size. Anim Behav 118:11–18. https://doi.org/10.1016/j.anbehav.2016.04.025
Wilson SK, Graham NA, Pratchett MS, Jones GP, Polunin NVC (2006) Multiple disturbances and the global degradation of coral reefs: are reef fishes at risk or resilient? Glob Chang Biol 12:2220–2234. https://doi.org/10.1111/j.1365-2486.2006.01252.x
Wilson SK, Burgess SC, Cheal AJ, Emslie M, Fisher R, Miller I, Polunin NV, Sweatman HP (2008) Habitat utilization by coral reef fish: implications for specialists vs. generalists in a changing environment. J Anim Ecol 77:220–228. https://doi.org/10.1111/j.1365-2656.2007.01341.x
Wilson SK, Graham NAJ, Pratchett MS (2014) Susceptibility of butterflyfish to habitat disturbance: do ‘chaets’ ever prosper. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of Butterflyfishes. CRC Press, Boca Raton, pp 226–245
Wilson SK, Robinson JP, Chong-Seng K, Robinson J, Graham NA (2019) Boom and bust of keystone structure on coral reefs. Coral Reefs 38:625–635. https://doi.org/10.1007/s00338-019-01818-4
Wrathall TJ, Roberts CM, Ormond RF (1992) Territoriality in the butterflyfish Chaetodon austriacus. Environ Biol Fish 34(3):305–308. https://doi.org/10.1007/BF00004777
Yabuta S, Berumen M (2014) Social structures and spawning behavior of Chaetodon Butterflyfishes. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of butterflyfishes. CRC Press, Boca Raton, pp 200–225. https://doi.org/10.1201/b15458-9
Yabuta S, Kawashima M (1997) Spawning behavior and haremic mating system in the corallivorous butterflyfish, Chaetodon trifascialis, at Kuroshima Island, Okinawa. Ichthyol Res 44(2):183–188. https://doi.org/10.1007/BF02678696
Acknowledgements
We acknowledge significant contributions by J.-P. Hobbs towards understanding and assessing reproductive staging, S. Reily for histological instruction and advice, and both C. Caballes and S. Wever for field assistance. We also thank two anonymous reviewers for their constructive feedback. This work was conducted aboard the James Cook University Research Vessel, the James Kirby, with significant logistic support provided by staff and crew.
Funding
This study was funded by the ARC Centre of Excellence for Coral Reef Studies.
Author information
Authors and Affiliations
Contributions
CAT and MSP conceived the original idea for this research. CAT designed the research. CAT and VM collected the data. CAT performed the laboratory work. SRM, PCD, and MSP analysed the data. All authors contributed to the preparation of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was conducted in strict accordance with research approvals issued by the Great Barrier Marine Park Authority (permit number G13/35909.1) and the James Cook University Animal Ethics Committee (permit number A2439).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thompson, C.A., Hoey, A.S., Montanari, S.R. et al. Territoriality and condition of chevron butterflyfish (Chaetodon trifascialis) with varying coral cover on the great barrier reef, Australia. Environ Biol Fish 104, 53–69 (2021). https://doi.org/10.1007/s10641-021-01055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-021-01055-1