Abstract
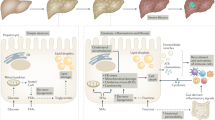
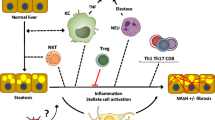
Inflammation and hepatocyte injury and death are the hallmarks of nonalcoholic steatohepatitis (NASH), the progressive form of nonalcoholic fatty liver disease (NAFLD), which is a currently burgeoning public health problem. Innate immune activation is a key factor in triggering and amplifying hepatic inflammation in NAFLD/NASH. Thus, identification of the underlying mechanisms by which immune cells in the liver recognize cell damage signals or the presence of pathogens or pathogen-derived factors that activate them is relevant from a therapeutic perspective. In this review, we present new insights into the factors promoting the inflammatory response in NASH including sterile cell death processes resulting from lipotoxicity in hepatocytes as well as into the altered gut-liver axis function, which involves translocation of bacterial products into portal circulation as a result of gut leakiness. We further delineate the key immune cell types involved and how they recognize both damage-associated molecular patterns or pathogen-associated molecular patterns through binding of surface-expressed pattern recognition receptors, which initiate signaling cascades leading to injury amplification. The relevance of modulating these inflammatory signaling pathways as potential novel therapeutic strategies for the treatment of NASH is summarized.

Similar content being viewed by others
Abbreviations
- NAFLD:
-
Nonalcoholic fatty liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- ROS:
-
Reactive oxygen species
- RIP1 and RIP3:
-
Receptor protein kinases 1 and 3
- TNF-R1:
-
Tumor necrosis factor receptor-1
- DAMPs:
-
Damage-associated molecular patterns
- PRRs:
-
Pattern recognition receptors
- PAMPs:
-
Pathogen-associated molecular patterns
- HMGB1:
-
The high-mobility group box 1
- LPS:
-
Lipopolysaccharide
- TNF-α:
-
Tumor necrosis factor-alpha
- IL:
-
Interleukin
- TLRs:
-
Toll-like receptors
- KCs:
-
Kupffer KCs
- HSC:
-
Hepatic stellate cells
- DCs:
-
Dendritic cells
- NLRs:
-
Nucleotide oligomerization NOD-like receptors domain
- CCL2 and CCL5:
-
C-C motif ligand 2 and 5
- PPAR-δ:
-
Peroxisome proliferator activator receptor delta
- CX3CR1:
-
Fractalkine receptor
- NK:
-
Natural killer
- NKT:
-
Natural killer T
- IB:
-
Intestinal barrier
- GLP-1:
-
Glucagon-like peptide 1
- FGF19:
-
Fibroblast growth factor-19
- IL-4:
-
Interleukin-4
- IFN-γ:
-
Interferon-gamma
- PDGF:
-
Platelet-derived growth factor
- NEU:
-
Neutrophils
- SAMs:
-
Scar-associated macrophages
References
Sayiner M, Koenig A, Henry L, Younossi ZM. Epidemiology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis in the United States and the rest of the world. Clin Liver Dis. (Epub ahead of print). doi:10.1016/j.cld.2015.10.001.
Satapathy SK, Sanyal AJ. Epidemiology and natural history of nonalcoholic fatty liver disease. Semin Liver Dis. 2015;35:221–235.
Yeh MM, Brunt EM. Pathological features of fatty liver disease. Gastroenterology. 2014;147:754–764.
Burt AD, Lackner C, Tiniakos DG. Diagnosis and assessment of NAFLD: definitions and histopathological classification. Semin Liver Dis. 2015;35:207–220.
Marengo A, Jouness EI, Bugianesi E. Progression and natural history of nonalcoholic fatty liver disease in adults. Clin Liver Dis. (Epub ahead of print). doi:10.1016/j.cld.2015.10.010.
Angulo P, Kleiner DE, Dam-Larsen S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149:389–397.
Charlton MR, Burns JM, Pedersen RA, Watt KD, Heimbach JK, Dierkhising RA. Frequency and outcomes of liver transplantation for nonalcoholic steatohepatitis in the United States. Gastroenterology. 2011;141:1249–1253.
Darwish Murad S, Metselaar HJ. The invasion of fatty liver disease in liver transplantation. Transpl Int. 2015. doi:10.1111/tri.12707.
Feldstein AE. Novel insights into the pathophysiology of nonalcoholic fatty liver disease. Semin Liver Dis. 2010;30:391–401.
de la Higuera-Tijera F, Servin-Caamano AI. Pathophysiological mechanisms involved in nonalcoholic steatohepatitis and novel potential therapeutic targets. World J Hepatol. 2015;7:1297–1301.
Peverill W, Powell LW, Skoien R. Evolving concepts in the pathogenesis of NASH: beyond steatosis and inflammation. Int J Mol Sci. 2014;15:8591–8638.
Hirsova P, Gores GJ. Death receptor-mediated cell death and proinflammatory signaling in nonalcoholic steatohepatitis. Cell Mol Gastroenterol Hepatol. 2015;1:17–27.
Trauner M, Arrese M, Wagner M. Fatty liver and lipotoxicity. Biochim Biophys Acta. 2010;1801:299–310.
Ibrahim SH, Kohli R, Gores GJ. Mechanisms of lipotoxicity in NAFLD and clinical implications. J Pediatr Gastroenterol Nutr. 2011;53:131–140.
Zambo V, Simon-Szabo L, Szelenyi P, Kereszturi E, Banhegyi G, Csala M. Lipotoxicity in the liver. World J Hepatol. 2013;5:550–557.
Arguello G, Balboa E, Arrese M, Zanlungo S. Recent insights on the role of cholesterol in nonalcoholic fatty liver disease. Biochim Biophys Acta. 2015;1852:1765–1778.
Seki E, Schwabe RF. Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology. 2015;61:1066–1079.
Feldstein AE, Canbay A, Angulo P, et al. Hepatocyte apoptosis and fas expression are prominent features of human nonalcoholic steatohepatitis. Gastroenterology. 2003;125:437–443.
Luedde T, Kaplowitz N, Schwabe RF. Cell death and cell death responses in liver disease: mechanisms and clinical relevance. Gastroenterology. 2014;147:765–783. (e764).
Wree A, Eguchi A, McGeough MD, et al. NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology. 2014;59:898–910.
Afonso MB, Rodrigues PM, Carvalho T, et al. Necroptosis is a key pathogenic event in human and experimental murine models of nonalcoholic steatohepatitis. Clin Sci (Lond). 2015;129:721–739.
Alkhouri N, Carter-Kent C, Feldstein AE. Apoptosis in nonalcoholic fatty liver disease: diagnostic and therapeutic implications. Expert Rev Gastroenterol Hepatol. 2011;5:201–212.
Moriwaki K, Chan FK. RIP3: a molecular switch for necrosis and inflammation. Genes Dev. 2013;27:1640–1649.
Mehal WZ. The inflammasome in liver injury and nonalcoholic fatty liver disease. Dig Dis. 2014;32:507–515.
Szabo G, Petrasek J. Inflammasome activation and function in liver disease. Nat Rev Gastroenterol Hepatol. 2015;12:387–400.
Sutti S, Bruzzi S, Albano E. The role of immune mechanisms in alcoholic and nonalcoholic steatohepatitis: a 2015 update. Expert Rev Gastroenterol Hepatol. 2015. doi:10.1586/17474124.2016.1111758.
Jindal A, Bruzzi S, Sutti S, et al. Fat-laden macrophages modulate lobular inflammation in nonalcoholic steatohepatitis (NASH). Exp Mol Pathol. 2015;99:155–162.
Ganz M, Szabo G. Immune and inflammatory pathways in NASH. Hepatol Int. 2013;7:771–781.
Kubes P, Mehal WZ. Sterile inflammation in the liver. Gastroenterology. 2012;143:1158–1172.
Miura K, Yang L, van Rooijen N, Brenner DA, Ohnishi H, Seki E. Toll-like receptor 2 and palmitic acid cooperatively contribute to the development of nonalcoholic steatohepatitis through inflammasome activation in mice. Hepatology. 2013;57:577–589.
Garcia-Martinez I, Shaker ME, Mehal WZ. Therapeutic opportunities in damage-associated molecular pattern-driven metabolic diseases. Antioxid Redox Signal. 2015;23:1305–1315.
Uchida K. Natural antibodies as a sensor of electronegative damage-associated molecular patterns (DAMPs). Free Radical Biol Med. 2014;72:156–161.
Huebener P, Pradere JP, Hernandez C, et al. The HMGB1/RAGE axis triggers neutrophil-mediated injury amplification following necrosis. J Clin Invest. 2015;125:539–550.
Seki E, Brenner DA. Toll-like receptors and adaptor molecules in liver disease: update. Hepatology. 2008;48:322–335.
Kesar V, Odin JA. Toll-like receptors and liver disease. Liver Int. 2014;34:184–196.
Petrasek J, Csak T, Szabo G. Toll-like receptors in liver disease. Adv Clin Chem. 2013;59:155–201.
Bieghs V, Trautwein C. Innate immune signaling and gut-liver interactions in nonalcoholic fatty liver disease. Hepatobiliary Surg Nutr. 2014;3:377–385.
Netea MG, van der Meer JW. Immunodeficiency and genetic defects of pattern-recognition receptors. N Eng J Med. 2011;364:60–70.
Takeuchi O, Akira S. Pattern recognition receptors and inflammation. Cell. 2010;140:805–820.
Miura K, Kodama Y, Inokuchi S, et al. Toll-like receptor 9 promotes steatohepatitis by induction of interleukin-1beta in mice. Gastroenterology. 2010;139:323–334. (e327).
Ehses JA, Meier DT, Wueest S, et al. Toll-like receptor 2-deficient mice are protected from insulin resistance and beta cell dysfunction induced by a high-fat diet. Diabetologia. 2010;53:1795–1806.
Szabo G, Petrasek J. Inflammasome activation and function in liver disease. Nat Rev Gastroenterol Hepatol. 2015;1247:387–400.
Wree A, McGeough MD, Pena CA, et al. NLRP3 inflammasome activation is required for fibrosis development in NAFLD. J Mol Med (Berl). 2014;92:1069–1082.
Dixon LJ, Barnes M, Tang H, Pritchard MT, Nagy LE. Kupffer cells in the liver. Compr Physiol. 2013;3:785–797.
Duarte N, Coelho IC, Patarrao RS, Almeida JI, Penha-Goncalves C, Macedo MP. How inflammation impinges on NAFLD: a role for Kupffer cells. Biomed Res Int. 2015;2015:984578.
Lanthier N. Targeting Kupffer cells in nonalcoholic fatty liver disease/nonalcoholic steatohepatitis: why and how? World J Hepatol. 2015;7:2184–2188.
Gadd VL, Skoien R, Powell EE, et al. The portal inflammatory infiltrate and ductular reaction in human nonalcoholic fatty liver disease. Hepatology. 2014;59:1393–1405.
Tacke F, Zimmermann HW. Macrophage heterogeneity in liver injury and fibrosis. J Hepatol. 2014;60:1090–1096.
Marra F, Tacke F. Roles for chemokines in liver disease. Gastroenterology. 2014;147:577–594. (e571).
Leroux A, Ferrere G, Godie V, et al. Toxic lipids stored by Kupffer cells correlates with their pro-inflammatory phenotype at an early stage of steatohepatitis. J Hepatol. 2012;57:141–149.
Sawada K, Ohtake T, Hasebe T, et al. Augmented hepatic Toll-like receptors by fatty acids trigger the pro-inflammatory state of nonalcoholic fatty liver disease in mice. Hepatol Res. 2014;44:920–934.
Fallowfield JA, Mizuno M, Kendall TJ, et al. Scar-associated macrophages are a major source of hepatic matrix metalloproteinase-13 and facilitate the resolution of murine hepatic fibrosis. J Immunol. 2007;178:5288–5295.
Chinetti-Gbaguidi G, Staels B. Macrophage polarization in metabolic disorders: functions and regulation. Curr Opin Lipidol. 2011;22:365–372.
Tacke F, Yoneyama H. From NAFLD to NASH to fibrosis to HCC: role of dendritic cell populations in the liver. Hepatology. 2013;58:494–496.
Lukacs-Kornek V, Schuppan D. Dendritic cells in liver injury and fibrosis: shortcomings and promises. J Hepatol. 2013;59:1124–1126.
Henning JR, Graffeo CS, Rehman A, et al. Dendritic cells limit fibroinflammatory injury in nonalcoholic steatohepatitis in mice. Hepatology. 2013;58:589–602.
Sutti S, Locatelli I, Bruzzi S, et al. CX3CR1-expressing inflammatory dendritic cells contribute to the progression of steatohepatitis. Clin Sci (Lond). 2015;129:797–808.
Almeda-Valdes P, Aguilar Olivos NE, Barranco-Fragoso B, Uribe M, Mendez-Sanchez N. The role of dendritic cells in fibrosis progression in nonalcoholic fatty liver disease. Biomed Res Int. 2015. doi:10.1155/2015/768071.
Xu R, Huang H, Zhang Z, Wang FS. The role of neutrophils in the development of liver diseases. Cell Mol Immunol. 2014;11:224–231.
Rensen SS, Bieghs V, Xanthoulea S, et al. Neutrophil-derived myeloperoxidase aggravates nonalcoholic steatohepatitis in low-density lipoprotein receptor-deficient mice. PLoS One. 2012;7:e52411.
Ibusuki R, Uto H, Arima S, et al. Transgenic expression of human neutrophil peptide-1 enhances hepatic fibrosis in mice fed a choline-deficient, L-amino acid-defined diet. Liver Int. 2013;33:1549–1556.
Talukdar S, da Oh Y, Bandyopadhyay G, et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat Med. 2012;18:1407–1412.
Tian Z, Chen Y, Gao B. Natural killer cells in liver disease. Hepatology. 2013;57:1654–1662.
Geissmann F, Cameron TO, Sidobre S, et al. Intravascular immune surveillance by CXCR6+ NKT cells patrolling liver sinusoids. PLoS Biol. 2005;3:e113.
Kumar V. NKT-cell subsets: promoters and protectors in inflammatory liver disease. J Hepatol. 2013;59:618–620.
Martin-Murphy BV, You Q, Wang H, et al. Mice lacking natural killer T cells are more susceptible to metabolic alterations following high fat diet feeding. PLoS One. 2014;9:e80949.
Elinav E, Pappo O, Sklair-Levy M, et al. Adoptive transfer of regulatory NKT lymphocytes ameliorates nonalcoholic steatohepatitis and glucose intolerance in ob/ob mice and is associated with intrahepatic CD8 trapping. J Pathol. 2006;209:121–128.
Kremer M, Thomas E, Milton RJ, et al. Kupffer cell and interleukin-12-dependent loss of natural killer T cells in hepatosteatosis. Hepatology. 2010;51:130–141.
Tajiri K, Shimizu Y, Tsuneyama K, Sugiyama T. Role of liver-infiltrating CD3+CD56+ natural killer T cells in the pathogenesis of nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2009;21:673–680.
Syn WK, Oo YH, Pereira TA, et al. Accumulation of natural killer T cells in progressive nonalcoholic fatty liver disease. Hepatology. 2010;51:1998–2007.
Tajiri K, Shimizu Y. Role of NKT cells in the pathogenesis of NAFLD. Int J Hepatol. 2012. doi:10.1155/2012/850836.
Kirpich IA, Marsano LS, McClain CJ. Gut-liver axis, nutrition, and nonalcoholic fatty liver disease. Clin Biochem. 2015;48:923–930.
Vajro P, Paolella G, Fasano A. Microbiota and gut-liver axis: their influences on obesity and obesity-related liver disease. J Pediatr Gastroenterol Nutr. 2013;56:461–468.
Federico A, Dallio M, Godos J, Loguercio C, Salomone F. Targeting gut-liver axis for the treatment of nonalcoholic steatohepatitis: translational and clinical evidence. Transl Res (Epub ahead of print). doi:10.1016/j.trsl.2015.08.002.
Kuipers F, Bloks VW, Groen AK. Beyond intestinal soap–bile acids in metabolic control. Nat Rev Endocrinol. 2014;10:488–498.
Liu HX, Keane R, Sheng L, Wan YY. Implications of microbiota and bile acid in liver injury and regeneration. J Hepatol. 2015;63:1502–1510.
Groschwitz KR, Hogan SP. Intestinal barrier function: molecular regulation and disease pathogenesis. J Allergy Clin Immunol. 2009;124:3–20.
Giorgio V, Miele L, Principessa L, et al. Intestinal permeability is increased in children with nonalcoholic fatty liver disease, and correlates with liver disease severity. Dig Liver Dis. 2014;46:556–560.
Luther J, Garber JJ, Khalili H, et al. Hepatic injury in nonalcoholic steatohepatitis contributes to altered intestinal permeability. Cell Mol Gastroenterol Hepatol. 2015;1:222–232.
Teixeira TF, Collado MC, Ferreira CL, Bressan J, Peluzio Mdo C. Potential mechanisms for the emerging link between obesity and increased intestinal permeability. Nutr Res. 2012;32:637–647.
Miele L, Valenza V, La Torre G, et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009;49:1877–1887.
Schneider KM, Bieghs V, Heymann F, et al. CX3CR1 is a gatekeeper for intestinal barrier integrity in mice: limiting steatohepatitis by maintaining intestinal homeostasis. Hepatology. 2015;62:1405–1416.
Vonghia L, Francque S. Cross talk of the immune system in the adipose tissue and the liver in nonalcoholic steatohepatitis: pathology and beyond. World J Hepatol. 2015;7:1905–1912.
Ramadori P, Kroy D, Streetz KL. Immunoregulation by lipids during the development of nonalcoholic steatohepatitis. Hepatobiliary Surg Nutr. 2015;4:11–23.
Moschen AR, Wieser V, Tilg H. Adiponectin: key player in the adipose tissue-liver crosstalk. Curr Med Chem. 2012;19:5467–5473.
Wan J, Benkdane M, Teixeira-Clerc F, et al. M2 Kupffer cells promote M1 Kupffer cell apoptosis: a protective mechanism against alcoholic and nonalcoholic fatty liver disease. Hepatology. 2014;59:130–142.
Arrese M, Cabrera D, Barrera F. Obeticholic acid: expanding the therapeutic landscape of NASH. Ann Hepatol. 2015;14:430–432.
Cariou B, Staels B. GFT505 for the treatment of nonalcoholic steatohepatitis and type 2 diabetes. Expert Opin Invest Drugs. 2014;23:1441–1448.
Acknowledgments
This work was funded by NIH grants R01 DK082451 and U01 AA022489 to A.E.F. and by grants from the Fondo Nacional de Desarrollo Cientıfico y Tecnologico (FONDECYT 1150327 to M.A. and FONDECYT PD3140396 to D.C.) and the Comision Nacional de Investigacion Cientıfica y Tecnologica (grant PFB 12/2007, Basal Centre for Excellence in Science and Technology, M.A.), both from the Government of Chile. A.K. received support from the Millennium Institute on Immunology and Immunotherapy, Santiago Chile. We thank Nicolas Triantafilo, MD, for assistance in providing the figure for this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors state no conflict of interest.
Rights and permissions
About this article
Cite this article
Arrese, M., Cabrera, D., Kalergis, A.M. et al. Innate Immunity and Inflammation in NAFLD/NASH. Dig Dis Sci 61, 1294–1303 (2016). https://doi.org/10.1007/s10620-016-4049-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4049-x