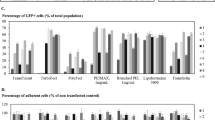
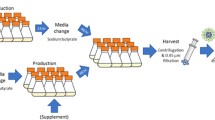
Abstract
Lentivirus and adeno-associated viruses are invaluable tools for biotechnology applications due to their genetic material delivery abilities both in vitro and in vivo. However, their large-scale productions with Good Manufacturing Practices yield low efficiency when adherent and serum dependent HEK293 (Human Embryonic Kidney) cells are used as the host. To increase production efficiency, HEK293 cells are adapted to grow in suspension using commercially available and chemically defined serum-free mediums. Suspended cells can be transiently transfected for viral vector production; however, significant improvements are still needed to increase yield and thereby cost effectiveness. Here, we evaluated four most preferred commercially available mediums that are IVY, FreeStyle293, LV-MAX, and BalanCD HEK293 for the transient transfection feasibility of lentiviral (LV) and adeno-associated virus serotype 2 (AAV2) production in FlorabioHEK293 suspension cells. The highest transfection efficiency was over 90% and obtained by using polyethyleneimine (PEI) 25 K and by media adaptation in IVY without using any transfection enhancer. For the first time the feasibility of HEK293 cells, which were adapted to grow in suspension culture by Florabio and IVY media, were tested for virus production. This study demonstrates the best transfection medium for scalable and optimized production of Lentivirus and Adeno-Associated Virus in suspended HEK293 cell culture.












Similar content being viewed by others
References
Ali RR, Reichel MB, Thrasher AJ et al (1996) Gene transfer into the mouse retina mediated by an adeno-associated viral vector. Hum Mol Genet 5:591–594. https://doi.org/10.1093/HMG/5.5.591
Alton EWFW, Beekman JM, Boyd AC et al (2017) Preparation for a first-in-man lentivirus trial in patients with cystic fibrosis. Thorax 72:137–147. https://doi.org/10.1136/THORAXJNL-2016-208406
Aurnhammer C, Haase M, Muether N et al (2012) Universal real-time PCR for the detection and quantification of adeno-associated virus serotype 2-derived inverted terminal repeat sequences. Hum Gene Ther Methods 23:18–28. https://doi.org/10.1089/HGTB.2011.034
Ausubel LJ, Hall C, Sharma A et al (2012) Production of CGMP-grade lentiviral vectors. Bioprocess Int 10:32–43
Baldi L, Hacker DL, Meerschman C, Wurm FM (2012) Large-scale transfection of mammalian cells. Methods Mol Biol 801:13–26. https://doi.org/10.1007/978-1-61779-352-3_2
Blessing D, Vachey G, Pythoud C et al (2019) Scalable production of AAV vectors in orbitally shaken HEK293 cells. Mol Ther Methods Clin Dev 13:14–26. https://doi.org/10.1016/J.OMTM.2018.11.004
Brunner D, Frank J, Appl H et al (2010) Serum-free cell culture: the serum-free media interactive online database. Altex 27:53–62. https://doi.org/10.14573/altex.2010.1.53
Bucher K, Rodríguez-Bocanegra E, Dauletbekov D, Fischer MD (2021) Immune responses to retinal gene therapy using adeno-associated viral vectors - Implications for treatment success and safety. Prog Retin Eye Res. https://doi.org/10.1016/J.PRETEYERES.2020.100915
Bulcha JT, Wang Y, Ma H et al (2021) Viral vector platforms within the gene therapy landscape. Signal Transduct Target Ther. https://doi.org/10.1038/S41392-021-00487-6
Bundy M, Thompson K, Liu C et al (2020) Optimization of the Gibco™ CTS™ LV-MAX™ Lentiviral production system in stirred tank bioreactors. Cytotherapy 22:S206. https://doi.org/10.1016/J.JCYT.2020.04.087
Cervera L, Fuenmayor J, González-Domínguez I et al (2015) Selection and optimization of transfection enhancer additives for increased virus-like particle production in HEK293 suspension cell cultures. Appl Microbiol Biotechnol 99:9935–9949. https://doi.org/10.1007/S00253-015-6842-4
Chahal PS, Schulze E, Tran R et al (2014) Production of adeno-associated virus (AAV) serotypes by transient transfection of HEK293 cell suspension cultures for gene delivery. J Virol Methods 196:163–173. https://doi.org/10.1016/J.JVIROMET.2013.10.038
Croyle MA, Cheng X, Wilson JM (2001) Development of formulations that enhance physical stability of viral vectors for gene therapy. Gene Ther 8:1281–1290. https://doi.org/10.1038/sj.gt.3301527
De Jesus M, Wurm FM (2011) Manufacturing recombinant proteins in kg-ton quantities using animal cells in bioreactors. Eur J Pharm Biopharm 78:184–188. https://doi.org/10.1016/J.EJPB.2011.01.005
De Molinas LMilagrosB, Beer M, Hesse C et al (2014) Optimizing the transient transfection process of HEK-293 suspension cells for protein production by nucleotide ratio monitoring. Cytotechnology 66:493–514. https://doi.org/10.1007/s10616-013-9601-3
Ferreira MV, Cabral ET, Coroadinha AS (2021) Progress and perspectives in the development of lentiviral vector producer cells. Biotechnol J. https://doi.org/10.1002/BIOT.202000017
Gallaher SD, Berk AJ (2013) A rapid Q-PCR titration protocol for adenovirus and helper-dependent adenovirus vectors that produces biologically relevant results. J Virol Methods 192:28–38. https://doi.org/10.1016/j.jviromet.2013.04.013
Genzel Y, Reichl U (2007) Vaccine production program vaccine production program. In: Pörtner R (ed) Animal cell biotechnology, 2nd edn. Humana Press, pp 457–473
Ghasemi N, Bandehpour M, Ranjbari J (2019) Optimization of key factors in serum free medium for production of human recombinant GM-CSF using response surface methodology. Iran J Pharm Res IJPR 18:146. https://doi.org/10.22037/IJPR.2020.112322.13681
Goradel NH, Alizadeh A, Hosseinzadeh S et al (2022) Oncolytic virotherapy as promising immunotherapy against cancer: mechanisms of resistance to oncolytic viruses. Future Oncol 18:245–259. https://doi.org/10.2217/FON-2021-0802
Graham FL, Smiley J, Russell WC, Nairn R (1977) Characteristics of a human cell line transformed by DNA from human adenovirus type 5. J Gen Virol 36:59–72. https://doi.org/10.1099/0022-1317-36-1-59
Grein TA, Weidner T, Czermak P (2017) Concepts for the production of viruses and viral vectors in cell cultures. In: Gowder SJT (ed) New insights into cell culture technology. IntechOpen, London
Grieger JC, Soltys SM, Samulski RJ (2016) Production of recombinant adeno-associated virus vectors using suspension HEK293 cells and continuous harvest of vector from the culture media for GMP FIX and FLT1 clinical vector. Mol Ther 24:287–297. https://doi.org/10.1038/MT.2015.187
Griffiths JB, Racher AJ (1994) Cultural and physiological factors affecting expression of recombinant proteins. Cytotechnology 15:3–9. https://doi.org/10.1007/BF00762374
Guan J-S, Chen K, Si Y et al (2022) Process improvement of adeno-associated virus production. Front Chem Eng 0:1. https://doi.org/10.3389/FCENG.2022.830421
Haldankar R, Li D, Saremi Z et al (2006) Serum-free suspension large-scale transient transfection of CHO cells in WAVE bioreactors. Mol Biotechnol 34:191–199. https://doi.org/10.1385/MB:34:2:191
Hesse F, Ebel M, Konisch N, et al (2003) Comparison of a Production Process in a Membrane-Aerated Stirred Tank and up to 1000-L Airlift Bioreactors Using BHK-21 Cells and Chemically Defined Protein-Free Medium. Biotechnol Prog 19:833–843. https://doi.org/10.1021/BP0257630
Ho YK, Too HP (2019) Development of a laboratory scalable process for enhancing lentivirus production by transient transfection of HEK293 adherent cultures. Gene Ther 27:482–494. https://doi.org/10.1038/s41434-020-0152-x
Karolewski BA, Watson DJ, Parente MK, Wolfe JH (2004) Comparison of transfection conditions for a lentivirus vector produced in large volumes. Hum Gene Ther 14:1287–1296. https://doi.org/10.1089/104303403322319372
Kissmann J, Ausar SF, Rudolph A et al (2008) Stabilization of measles virus for vaccine formulation. Hum Vaccin 4:350–359. https://doi.org/10.4161/HV.4.5.5863
Ku MW, Bourgine M, Authié P et al (2021) Intranasal vaccination with a lentiviral vector protects against SARS-CoV-2 in preclinical animal models. Cell Host Microbe 29:236–249e6. https://doi.org/10.1016/J.CHOM.2020.12.010
Kumar P, Nagarajan A, Uchil PD (2019) Calcium phosphate-mediated transfection of adherent cells or cells growing in suspension: variations on the basic method. Cold Spring Harb Protoc 2019:705–708. https://doi.org/10.1101/PDB.PROT095455
Legmann R (2020) Transient transfection at large scale for clinical AAV9 vector manufacturing. Cytotherapy 22:S151. https://doi.org/10.1016/J.JCYT.2020.03.312
Liu D, Zhu M, Zhang Y, Diao Y (2021) Crossing the blood-brain barrier with AAV vectors. Metab Brain Dis 36:45–52. https://doi.org/10.1007/S11011-020-00630-2
Longo PA, Kavran JM, Kim MS, Leahy DJ (2013) Transient mammalian cell transfection with polyethylenimine (PEI). Methods Enzymol 529:227. https://doi.org/10.1016/B978-0-12-418687-3.00018-5
Marjanovič I, Kandušer M, Miklavčič D et al (2014) Comparison of flow cytometry, fluorescence microscopy and spectrofluorometry for analysis of gene electrotransfer efficiency. J Membr Biol 247:1259–1267. https://doi.org/10.1007/s00232-014-9714-4
Merten OW, Charrier S, Laroudie N et al (2010) Large-scale manufacture and characterization of a lentiviral vector produced for clinical ex vivo. Gene Ther Appl 22:343–356. https://doi.org/10.1089/HUM.2010.060
Miki H, Takagi M (2015) Design of serum-free medium for suspension culture of CHO cells on the basis of general commercial media. Cytotechnology 67:689. https://doi.org/10.1007/S10616-014-9778-0
Mueller C, Ratner D, Zhong L et al (2012) Production and discovery of novel recombinant adeno-associated viral vectors. Curr Protoc Microbiol. https://doi.org/10.1002/9780471729259.mc14d01s
Naso MF, Tomkowicz B, Perry WL, Strohl WR (2017) Adeno-associated virus (AAV) as a vector for gene therapy. BioDrugs 31:317–334. https://doi.org/10.1007/S40259-017-0234-5
Nguyen TNT, Sha S, Hong MS et al (2021) Mechanistic model for production of recombinant adeno-associated virus via triple transfection of HEK293 cells. Mol Ther Methods Clin Dev 21:642–655. https://doi.org/10.1016/J.OMTM.2021.04.006
O’Doherty U, Swiggard WJ, Malim MH (2000) Human immunodeficiency virus type 1 spinoculation enhances infection through virus binding. J Virol 74:10074. https://doi.org/10.1128/JVI.74.21.10074-10080.2000
Olsen JC, Sechelski J (2008) Use of sodium butyrate to enhance production of retroviral vectors expressing CFTR cDNA. Hum Gene Ther 6:1195–1202. https://doi.org/10.1089/HUM.1995.6.9-1195
Pang Y, Song H, Cheng W (2016) Using optical trap to measure the refractive index of a single animal virus in culture fluid with high precision. Biomed Opt Express. https://doi.org/10.1364/boe.7.001672
Perevyazko IY, Vollrath A, Pietsch C et al (2012) Nanoprecipitation of poly(methyl methacrylate)-based nanoparticles: effect of the molar mass and polymer behavior. J Polym Sci Part A Polym Chem 50:2906–2913. https://doi.org/10.1002/POLA.26071
Perry C, Rayat ACME (2021) Lentiviral vector bioprocessing. Viruses 13:268. https://doi.org/10.3390/V13020268
Pezzoli D, Giupponi E, Mantovani D, Candiani G (2017) Size matters for in vitro gene delivery: investigating the relationships among complexation protocol, transfection medium, size and sedimentation. Sci Rep. https://doi.org/10.1038/SREP44134
Piovan C, Marin V, Scavullo C et al (2017) Vectofusin-1 promotes RD114-TR-pseudotyped lentiviral vector transduction of human HSPCs and T lymphocytes. Mol Ther Methods Clin Dev 5:22–30. https://doi.org/10.1016/J.OMTM.2017.02.003
Rajendra Y, Balasubramanian S, Hacker DL (2017) Large-scale transient transfection of chinese hamster ovary cells in suspension. Methods Mol Biol 1603:45–55. https://doi.org/10.1007/978-1-4939-6972-2_3
Raymond C, Tom R, Perret S et al (2011) A simplified polyethylenimine-mediated transfection process for large-scale and high-throughput applications. Methods 55:44–51. https://doi.org/10.1016/J.YMETH.2011.04.002
Reed SE, Staley EM, Mayginnes JP et al (2006) Transfection of mammalian cells using linear polyethylenimine is a simple and effective means of producing recombinant adeno-associated virus vectors. J Virol Methods 138:85–98. https://doi.org/10.1016/J.JVIROMET.2006.07.024
Remley VA, Jin J, Sarkar S et al (2021) High efficiency closed-system gene transfer using automated spinoculation. J Transl Med 19:1–15. https://doi.org/10.1186/S12967-021-03126-4/FIGURES/6
Rubin JD, Nguyen TV, Allen KL et al (2019) Comparison of gene delivery to the kidney by adenovirus, adeno-associated virus, and lentiviral vectors after intravenous and direct kidney injections. Hum Gene Ther 30:1559. https://doi.org/10.1089/HUM.2019.127
Sanber KS, Knight SB, Stephen SL et al (2015) Construction of stable packaging cell lines for clinical lentiviral vector production. Sci Rep. https://doi.org/10.1038/SREP09021
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 2012 97:671–675. https://doi.org/10.1038/nmeth.2089
Subedi GP, Johnson RW, Moniz HA et al (2015) High yield expression of recombinant human proteins with the transient transfection of HEK293 cells in suspension. J Vis Exp. https://doi.org/10.3791/53568
Sung YK, Kim SW (2019) Recent advances in the development of gene delivery systems. Biomater Res 2019 231 23:1–7. https://doi.org/10.1186/S40824-019-0156-Z
Tang Y, Garson K, Li L, Vanderhyden BC (2015) Optimization of lentiviral vector production using polyethylenimine-mediated transfection. Oncol Lett 9:55. https://doi.org/10.3892/OL.2014.2684
Tomás HA, Rodrigues AF, Carrondo MJT, Coroadinha AS (2018) LentiPro26: novel stable cell lines for constitutive lentiviral vector production. Sci Rep. https://doi.org/10.1038/S41598-018-23593-Y
Valkama AJ, Leinonen HM, Lipponen EM et al (2017) Optimization of lentiviral vector production for scale-up in fixed-bed bioreactor. Gene Ther 2018 251 25:39–46. https://doi.org/10.1038/gt.2017.91
Valkama AJ, Oruetxebarria I, Lipponen EM et al (2020) Development of large-scale downstream processing for lentiviral vectors. Mol Ther Methods Clin Dev. https://doi.org/10.1016/j.omtm.2020.03.025
Wang M, Chai L, Liang S et al (2020) Fetal calf serum exerts an inhibitory effect on replication of duck hepatitis A virus genotype 1 in duck embryo fibroblast cells. Viruses 12:1–14. https://doi.org/10.3390/v12010080
Wang Z, Zhu T, Qiao C et al (2005) Adeno-associated virus serotype 8 efficiently delivers genes to muscle and heart. Nat Biotechnol 23:321–328. https://doi.org/10.1038/NBT1073
Wright JF (2009) Transient transfection methods for clinical adeno-associated viral vector production. Hum Gene Ther 20:698. https://doi.org/10.1089/HUM.2009.064
Xie Q, Xinyong G, Xianjin C, Yayu W (2013) PEI/DNA formation affects transient gene expression in suspension Chinese hamster ovary cells via a one-step transfection process. Cytotechnology 65:263–271. https://doi.org/10.1007/s10616-012-9483-9
Zhao H, Lee KJ, Daris M et al (2020) Creation of a high-yield AAV vector production platform in suspension cells using a design-of-experiment approach. Mol Ther Methods Clin Dev 18:312–320. https://doi.org/10.1016/J.OMTM.2020.06.004
Acknowledgements
This project is financially supported by Sabanci University Nanotechnology Research and Application Center (SUNUM).
Author information
Authors and Affiliations
Contributions
G.C.T. and A.A.Y. conducted the experiments, performed the statistical analysis, wrote, and edited the original draft. C.E., A.C., O.K., and S.C. conceived and participated in the study design. O.K. and S.C. reviewed the manuscript. All authors contributed to and have approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Celebi Torabfam, G., Yetisgin, A.A., Erdem, C. et al. A feasibility study of different commercially available serum-free mediums to enhance lentivirus and adeno-associated virus production in HEK 293 suspension cells. Cytotechnology 74, 635–655 (2022). https://doi.org/10.1007/s10616-022-00551-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-022-00551-1