Abstract
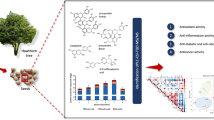
Perilla frutescens (L.) Britton var. frutescens (egoma in Japan) is a traditional oilseed that has several varieties with different photoperiod responses. Although egoma pomace, industrial waste produced during oil extraction, is a rich source of macro- and micro-nutrients such as protein, fiber, minerals, and polyphenols, it has not yet been used for purposes other than livestock feeding. To find out a better use of perilla pomace and its function, we selected four varieties of egoma originating from different regions with different photoperiod responses: two varieties were from Japan, which are broadly cultivated for oilseed and are highly sensitive to light and temperature. The other two varieties from Nepal, which are tolerant to low light and low temperature. Rosmarinic acid-3-O-glucoside, rosmarinic acid, and apigenin-7-O-glucoside were detected as the main polyphenolic constituents in every variety, while apigenin and luteolin were present only in perilla pomace from Japan. In IgE-sensitized RBL-2H3 cells, polyphenols derived from two varieties of Japan suppressed degranulation of mast cells, but those derived from the two varieties of Nepal did not, indicating that apigenin and luteolin may be in part responsible for the anti-allergic response. In addition, it was found that proteins involved in the degranulation signaling pathway, such as PLCγ2, Syk, and Akt, were less phosphorylated in cells treated with the egoma pomace extracts of Japanese origin. Taken together, pomace from egoma varieties derived from different regions may differently modulate allergic response in part due to the difference in polyphenol composition and may be applied to develop nutraceuticals and functional foods fortified with anti-allergic properties.



Similar content being viewed by others
References
Ainsworth EK, Gillespie KM (2007) Estimation of total phenolic and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat Protoc 2:875–877. https://doi.org/10.1038/nprot.2007.102
Chang CC, Yang MH, Wen HM, Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal 10:178–182
Dhyani A, Chopra R, Garg M (2019) A review on nutritional value, functional properties and pharmacological application of Perilla (Perilla frutescens L.). Biomed Pharmacol J 12:649–660
Hu Y, Sun LW, Neo MC, Zhang YX, Wen CX, Xie XL, Liu YJ (2010) Primary identifications and palynological observations of Perilla in China. J Syst Evol 48:133–145
Hwang K, Hwang Y-J, Song J (2018) Anti-allergic effect of Aster yomena on ovalbumin-sensitized mouse RHL-2H3 cells via Th1/Th2 cytokine balance. J Funct Foods 44:1–8
Iwamoto A, Hamajima H, Tsuge K, Tsuruta Y, Nagata Y, Yotsumoto H, Yanagita Y (2020) Inhibitory effects of green asparagus extract, especially phospholipids, on allergic response in vitro and in vivo. J Agric Food Chem 68:15199–15207
Khanam UKS, Oba S (2013) Bioactive substances in leaves of two amaranth species, Amaranthus tricolor and A. hypochondriacus. Can J Plant Sci 93:47–58
Lee AY, Choi JM, Lee MH, Lee J, Lee S, Cho EJ (2018) Protective effects of perilla oil and alpha linolenic acid on SH-SY5Y neuronal cell death induced by hydrogen peroxide. Nutr Res Prac 12:93–100. https://doi.org/10.4162/nrp.2018.12.2.93
Makino T, Furuta Y, Wakushima H, Fujii H, Saito K, Kano Y (2003) Anti-allergic effect of Perilla frutescens and its active constituents. Phytother Res 17:240–243
Matsuda H, Nakamura S, Yoshikawa M (2016) Degranulation inhibitors from medicinal plants in antigen-stimulated rat basophilic leukemia (RBL-2H3) cells. Chem Pharm Bull 64:96–103
Mlcek J, Jurikova T, Skrovankova S, Sochor J (2016) Quercetin and Its anti-allergic immune response. Molecules 21:623. https://doi.org/10.3390/molecules21050623
Mwakalukwa R, Ashour A, Amen Y, Niwa Y, Tamrakar S, Miyamoto T, Shimizu K (2019) Anti-allergic activity of polyphenolic compounds isolated from olive mill wastes. J Funct Foods 58:207–217
Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxi Med Cell Long 2:270–278. https://doi.org/10.4161/oxim.2.5.9498
Park C-H, Min S-Y, Yu H-W, Kim K, Kim S, Lee H-J, Kim J-H, Park Y-J (2020) Effects of apigenin on RBL-2H3, RAW264.7, and HaCat cells: anti-allergic, anti-inflammatory, and skin-protective activities. Int J Mol Sci 21:4620–4636
Shimoda H, Tanaka J, Yamada E, Morikawa T, Kasajima N, Yoshikawa M (2006) Anti type I allergic property of Japanese butterbur extract and its mast cell degranulation inhibitor ingredients. J Agric Food Chem 54:2915–2920
Takano H, Osakabe N, Sanbongi C, Yanagisawa R, Inoue K, Yasuda A, Natsume M, Baba S, Ichiishi E, Yoshikawa T (2004) Extract of Perilla frutescens enriched for rosmarinic acid, a polyphenolic phytochemical, inhibits seasonal allergic rhinoconjunctivitis in humans. Exp Biol Med 229:247–254
Tian Y, Wang H, Yuan F, Li N, Huang Q, He L, Wang L, Liu Z (2016) Perilla oil has similar protective effects of fish oil on high-fat diet-Induced nonalcoholic fatty liver disease and gut dysbiosis. BioMed Res Int 2016:1–11. https://doi.org/10.1155/2016/9462571
Touaibia M, Jean-Francois J, Doiron J (2011) Caffeic acid, a versatile pharmacophore: an overview. Mini Review in Medicinal Chemistry 11:695–713
Tsuruta Y, Nagao K, Kai S, Tsuge K, Yoshimura T, Koganemaru K, Yanagita T (2011) polyphenolic extract of lotus root (edible rhizome of Nelumbo nucifera) alleviates hepatic steatosis in obese diabetic db/db mice. Lipid Health Dis 10:202–209
Ueda H, Yamazaki C, Yamazaki M (2002) Luteolin as an anti-inflammatory and anti-allergic constituent of Perilla frutescens. Biol Pharm Bull 25:1197–1202
Wang M, Firrman J, Liu L, Yam K (2019) A review on flavonoid apigenin: dietary intake, ADME, antimicrobial effects, and interactions with human gut microbiota. BioMed Res Int 2019:7010467
Yan X, Qi M, Li P, Zhan Y, Shao H (2017) Apigenin in cancer therapy: anti-cancer effects and mechanisms of action. Cell Biosci 7:50
Yoshioka H, Kamata A, Konishi T, Takahashi J, Oda H, Tamai T, Toyohara H, Sugahara T (2013) Inhibitory effect of chlorophyll c2 from brown algae, Sargassum horneri, on degranulation of RBL-2H3 cells. J Funct Foods 5:204–210
Zhou X-J, Yan L-L, Yin P-P, Shi L-L, Zhang J-H, Liu Y-J, Ma C (2014) Structural characterisation and antioxidant activity evaluation of phenolic compounds from cold-pressed Perilla frutescens var. arguta seed flour. Food Chem 164:150–157
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
YRG designed, executed the study and edited the manuscript. AI, KT, SO, and HH helped in the analysis. YN and TY supervised the study and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gaihre, Y.R., Iwamoto, A., Oogai, S. et al. Perilla pomace obtained from four different varieties have different levels and types of polyphenols and anti-allergic activity. Cytotechnology 74, 341–349 (2022). https://doi.org/10.1007/s10616-022-00522-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-022-00522-6