Abstract
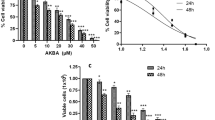
In prostate cancer, flavonoids possess a wide variety of anticancer effects, focused on the antioxidant/pro-oxidant activity, inactivation of the androgen receptor, cell cycle arrest, apoptosis induction, metastasis inhibition, among others. This current research investigated the antitumoral in vitro activity of Brachydin A (BrA), a dimeric flavonoid isolated from Fridericia platyphylla, in human castration-resistant prostate cancer DU145. It was compared BrA selective effects in tumor prostate DU145 cells with non-tumor prostate epithelial PNT2 cells. Cell viability experiments (resazurin, neutral red, MTT, and LDH release assays) showed that BrA was sevenfold more cytotoxic to tumor cells than non-tumor prostate cells, with IC50 values of 77.7 µM and 10.7 µM for PNT2 and DU145 cells, respectively. Furthermore, BrA induced necrosis and apoptosis (triple fluorescence staining assay) without interfering with oxidative stress (CM-H2DCFDA) in DU145 cells. Also, BrA (15.36 µM) reduced cell proliferation on clonogenic assay (DU145 cells) but no change in cell number and protein content was observed when cell growth curve assay was used. Wound healing and transwell assays were used for checking the effects of BrA on cell migration and invasion, and BrA impaired these processes in PNT2 (wound healing) and DU145 cells (transwell). Our results inspire further studies to test BrA as a novel chemotherapeutic drug and to evaluate its effects on drug-resistant metastatic cancer cells.
Graphic abstract







Similar content being viewed by others
References
Bonta RK (2020) Dietary phenolic acids and flavonoids as potential anti-cancer agents: current state of the art and future perspectives. Anticancer Agents Med Chem 20:29–48. https://doi.org/10.2174/1871520619666191019112712
Choudhari AS, Mandave PC, Deshpande M et al (2020) Phytochemicals in cancer treatment: from preclinical studies to clinical practice. Front Pharmacol. https://doi.org/10.3389/fphar.2019.01614
Costea T, Nagy P, Ganea C et al (2019) Molecular mechanisms and bioavailability of polyphenols in prostate cancer. Int J Mol Sci. https://doi.org/10.3390/ijms20051062
Costea T, Vlad OC, Miclea L-C et al (2020) Alleviation of multidrug resistance by flavonoid and non-flavonoid compounds in breast, lung, colorectal and prostate cancer. Int J Mol Sci. https://doi.org/10.3390/ijms21020401
da Costa Lopes L, Albano F, Augusto Travassos Laranja G et al (2000) Toxicological evaluation by in vitro and in vivo assays of an aqueous extract prepared from Echinodorus macrophyllus leaves. Toxicol Lett 116:189–198. https://doi.org/10.1016/s0378-4274(00)00220-4
da Rocha CQ, Queiroz EF, Meira CS et al (2014) Dimeric flavonoids from Arrabidaea brachypoda and assessment of their anti-trypanosoma cruzi activity. J Nat Prod 77:1345–1350. https://doi.org/10.1021/np401060j
da Rocha CQ, de-Faria FM, Marcourt L et al (2017) Gastroprotective effects of hydroethanolic root extract of Arrabidaea brachypoda: evidences of cytoprotection and isolation of unusual glycosylated polyphenols. Phytochemistry 135:93–105. https://doi.org/10.1016/j.phytochem.2016.12.002
de Sousa Andrade LM, de Oliveira ABM, Leal ALAB et al (2020) Antimicrobial activity and inhibition of the NorA efflux pump of Staphylococcus aureus by extract and isolated compounds from Arrabidaea brachypoda. Microb Pathog 140:103935. https://doi.org/10.1016/j.micpath.2019.103935
do Nascimento JR, Ribeiro GAC, Serrano SHP et al (2021) Electrochemical behavior of unusual dimeric flavonoids isolated from Fridericia platyphylla. J Braz Chem Soc 32:1137
Dyshlovoy SA, Tarbeeva D, Fedoreyev S et al (2020) Polyphenolic compounds from lespedeza bicolor root bark inhibit progression of human prostate cancer cells via induction of apoptosis and cell cycle arrest. Biomolecules. https://doi.org/10.3390/biom10030451
Eilenberger C, Kratz SRA, Rothbauer M et al (2018) Optimized alamarBlue assay protocol for drug dose-response determination of 3D tumor spheroids. MethodsX 5:781–787. https://doi.org/10.1016/j.mex.2018.07.011
Franken NAP, Rodermond HM, Stap J et al (2006) Clonogenic assay of cells in vitro. Nat Protoc 1:2315–2319. https://doi.org/10.1038/nprot.2006.339
Hazafa A, Rehman K-U-, Jahan N, Jabeen Z (2020) The role of polyphenol (flavonoids) compounds in the treatment of cancer cells. Nutr Cancer 72:386–397. https://doi.org/10.1080/01635581.2019.1637006
Imran M, Rauf A, Abu-Izneid T et al (2019) Luteolin, a flavonoid, as an anticancer agent: a review. Biomed Pharmacother 112:108612. https://doi.org/10.1016/j.biopha.2019.108612
Izzo S, Naponelli V, Bettuzzi S (2020) Flavonoids as epigenetic modulators for prostate cancer prevention. Nutrients 12:10. https://doi.org/10.3390/nu12041010
Jiménez-Vacas JM, Herrero-Aguayo V, Montero-Hidalgo AJ et al (2020) Dysregulation of the splicing machinery is directly associated to aggressiveness of prostate cancer. EBioMedicine 51:102547. https://doi.org/10.1016/j.ebiom.2019.11.008
Johnson IR, Parkinson-Lawrence EJ, Butler LM, Brooks DA (2014) Prostate cell lines as models for biomarker discovery: performance of current markers and the search for new biomarkers. Prostate 74:547–560. https://doi.org/10.1002/pros.22777
Justus CR, Leffler N, Ruiz-Echevarria M, Yang LV (2014) In vitro cell migration and invasion assays. J Vis Exp JoVE. https://doi.org/10.3791/51046
Kopustinskiene DM, Jakstas V, Savickas A, Bernatoniene J (2020) Flavonoids as anticancer agents. Nutrients. https://doi.org/10.3390/nu12020457
Kvízová J, Pavlíčková V, Kmoníčková E et al (2021) Quo Vadis advanced prostate cancer therapy? Novel treatment perspectives and possible future directions. Mol Basel Switz 26:10. https://doi.org/10.3390/molecules26082228
Liang C-C, Park AY, Guan J-L (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333. https://doi.org/10.1038/nprot.2007.30
Martinotti S, Ranzato E (2020) Scratch wound healing assay. Methods Mol Biol Clifton NJ 2109:225–229. https://doi.org/10.1007/7651_2019_259
Mirzayans R, Andrais B, Murray D (2018) Viability assessment following anticancer treatment requires single-cell visualization. Cancers. https://doi.org/10.3390/cancers10080255
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Namekawa T, Ikeda K, Horie-Inoue K, Inoue S (2019) Application of prostate cancer models for preclinical study: advantages and limitations of cell lines, patient-derived xenografts, and three-dimensional culture of patient-derived cells. Cells 8:10. https://doi.org/10.3390/cells8010074
Nunes HL, Tuttis K, Serpeloni JM et al (2020) Characterization of the invitro cytotoxic effects of brachydins isolated from Fridericia platyphylla in a prostate cancer cell line. J Toxicol Environ Health A 83:547–558. https://doi.org/10.1080/15287394.2020.1784339
Pereira TF, Levin G, DeOcesano-Pereira C et al (2020) Fluorescence-based method is more accurate than counting-based methods for plotting growth curves of adherent cells. BMC Res Notes 13:57. https://doi.org/10.1186/s13104-020-4914-8
Präbst K, Engelhardt H, Ringgeler S, Hübner H (2017) Basic colorimetric proliferation assays: MTT, WST, and resazurin. In: Methods in Molecular Biology. pp 1–17
Proietti De Santis L, Balajee AS, Lorenti Garcia C et al (2003) Inhibition of p53, p21 and Bax by pifithrin-alpha does not affect UV induced apoptotic response in CS-B cells. DNA Repair 2:891–900. https://doi.org/10.1016/s1568-7864(03)00088-0
Rampersad SN (2012) Multiple applications of alamar blue as an indicator of metabolic function and cellular health in cell viability bioassays. Sens Switz 12:12347–12360. https://doi.org/10.3390/s120912347
Repetto G, del Peso A, Zurita JL (2008) Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat Protoc 3:1125–1131. https://doi.org/10.1038/nprot.2008.75
Riss TL, Moravec RA, Niles AL et al (2004) Cell viability assays. In: Markossian S, Sittampalam GS, Grossman A et al (eds) Assay guidance manual. Eli Lilly & Company and the National Center for Advancing Translational Sciences, Bethesda
Salgado C, Morin H, Coriolano de Aquino N et al (2020) In vitro anti-inflammatory activity in arthritic synoviocytes of A. brachypoda root extracts and its unusual dimeric flavonoids. Mol Basel Switz 25:10. https://doi.org/10.3390/molecules25215219
Serpeloni JM, Specian AFL, Ribeiro DL et al (2015) Antimutagenicity and induction of antioxidant defense by flavonoid rich extract of Myrcia bella Cambess. in normal and tumor gastric cells. J Ethnopharmacol 176:345–355. https://doi.org/10.1016/j.jep.2015.11.003
Serpeloni JM, Specian A, Ribeiro DL et al (2020) Fridericia platyphylla (Cham.) L.G. Lohmann root extract exerts cytotoxic and antiproliferative effects on gastric tumor cells and downregulates BCL-XL, BIRC5, and MET genes. Hum Exp Toxicol 39:338–354. https://doi.org/10.1177/0960327119888261
Shenoy N, Stenson M, Lawson J et al (2017) Drugs with anti-oxidant properties can interfere with cell viability measurements by assays that rely on the reducing property of viable cells. Lab Invest 97:494–497. https://doi.org/10.1038/labinvest.2017.18
Specian AFL, Serpeloni JM, Tuttis K et al (2016) LDH, proliferation curves and cell cycle analysis are the most suitable assays to identify and characterize new phytotherapeutic compounds. Cytotechnology 68:2729–2744. https://doi.org/10.1007/s10616-016-9998-6
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. https://doi.org/10.3322/caac.21660
Van Tonder A, Joubert AM, Cromarty AD (2015) Limitations of the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay when compared to three commonly used cell enumeration assays. BMC Res Notes 8:47. https://doi.org/10.1186/s13104-015-1000-8
Vinci M, Box C, Zimmermann M, Eccles SA (2013) Tumor spheroid-based migration assays for evaluation of therapeutic agents. Methods Mol Biol Clifton NJ 986:253–266. https://doi.org/10.1007/978-1-62703-311-4_16
Yang C, Zhang W, Wang J et al (2019) Effect of docetaxel on the regulation of proliferation and apoptosis of human prostate cancer cells. Mol Med Rep 19:3864–3870. https://doi.org/10.3892/mmr.2019.9998
Zeng K, Xie W, Huang J et al (2020) PAX3 silencing inhibits prostate cancer progression through the suppression of the TGF-β/Smad signaling axis. Cell Biol Int 44:2131–2139. https://doi.org/10.1002/cbin.11421
Funding
This work was supported by Brazilian National Council for Scientific and Technological Development (CNPq: Grants 426246/2018-7 and 401516/2016-4) and Coordination for the Improvement of Higher Education Personnel - [Finnance Code 01].
Author information
Authors and Affiliations
Contributions
Conceptualization and design: JMS; IMSC; LCBO; Provision of study materials and funding acquisition: JMS; CQR; DLR; Collection and assembly of data: LCBO; JRN. Data analysis and interpretation: LCBO; JMS; IMSC. Writing—original draft and editing: LCBŌ; DLR; IMSC; JMS. Supervision and project administration: DLR; IMSC; JMS.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to report.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Oliveira, L.C.B., Nunes, H.L., Ribeiro, D.L. et al. Aglycone flavonoid brachydin A shows selective cytotoxicity and antitumoral activity in human metastatic prostate (DU145) cancer cells. Cytotechnology 73, 761–774 (2021). https://doi.org/10.1007/s10616-021-00495-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-021-00495-y