Abstract
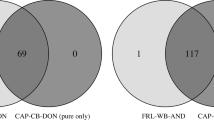
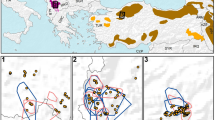
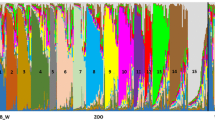
The main aim of ex situ programmes in conservation is to provide a suitable source of individuals for future reintroductions or reinforcement of existing populations. A fundamental prerequisite is creating and maintaining healthy and sustainable captive populations that show high levels of phenotypic and genetic similarity to their wild counterparts. The Eurasian lynx (Lynx lynx) is a model of a locally extinct species that has been subject to long-term captive breeding and of past and ongoing reintroduction efforts. To test for genetic suitability of ex situ population, a comparative genetic evaluation including in situ populations was undertaken. The assignment analysis of 97 captive lynx from 45 European zoos, wildlife parks and private breeds was performed using 124 lynx from different wild Eurasian populations belonging to three evolutionary lineages: the Carpathian, the Northern, and the Siberian lynx. The results showed a high proportion of Siberian lynx (51%) in the European captive lynx population. Remaining captive animals were assigned to either the Carpathian (28%), or the Northern lynx lineage (13%). Admixture between lineages was rather low (8%). Notably, no or very low difference in genetic diversity was detected between the wild and captive lynx populations. Our results support the potential of the captive population to provide genetically suitable individuals for genetic rescue programmes. The transfer of genes between isolated populations, including those in captivity, should become an important management tool to preserve genetic variability and prevent inbreeding depression in native and reintroduced populations of this iconic predator.




Similar content being viewed by others
Data availability
Individuals genotypes associated with this study are available within the supplementary material of this article.
References
Adams JR, Waits LP (2007) An efficient method for screening faecal DNA genotypes and detecting new individuals and hybrids in the red wolf (Canis rufus) experimental population area. Conserv Genet 8:123–131. https://doi.org/10.1007/s10592-006-9154-5
Ballou JD (1997) Ancestral inbreeding only minimally affects inbreeding depression in mammalian populations. J Hered 88:169–178. https://doi.org/10.1093/oxfordjournals.jhered.a023085
Belkhir K, Borsa P, Chikhi L, Raufaste N, Bonhomme F (1996–2004) GENETIX 405, logiciel sous Windows TM pour la génétique des populations. Laboratoire Genome, Populations, Interactions, CNRS UMR 5000. Universite de Montpellier II, Montpellier
Boakes EH, Wang JL, Amos W (2007) An investigation of inbreeding depression and purging in captive pedigreed populations. Heredity 98:172–182. doi: https://doi.org/10.1038/sj.hdy.6800923
Bonn Lynx Expert Group (2021) Recommendations for the conservation of the eurasian lynx in Western and Central Europe. Conclusions from the workshop of the “Bonn Lynx Expert Group” in Bonn, Germany, 16–19 June 2019. Cat News Special Issue 14:78–86
Bouman J (1977) The future of Przewalski horses (Equus przewalskii) in captivity. Int Zoo Yb 17:62–68
Bowling AT, Zimmermann W, Ryder O, Penado C, Peto S, Chemnick L, Yasinetskaya N, Zharkikh T (2003) Genetic variation in Przewalski’s horses, with special focus on the last wild caught mare, 231 Orlitza III. Cytogenet Genome Res 102:226–234
Breitenmoser-Würsten Ch, Obexer-Ruff G (2003) Population and conservation genetics of two re-introduced lynx (Lynx lynx) populations in Switzerland—a molecular evaluation 30 years after translocation. In: Proceedings of the 2nd Conference on the Status and Conservation of the Alpine Lynx Population (SCALP):7–9
Bull JK, Heurich M, Saveljev AP, Schmidt K, Fickel J, Förster DW (2016) The effect of reintroductions on the genetic variability in eurasian lynx populations: the cases of bohemian–bavarian and Vosges–Palatinian populations. Conserv Genet 17:1229–1234. https://doi.org/10.1007/s10592-016-0839-0
Caniglia R, Galaverni M, Velli E et al (2020) A standardized approach to empirically define reliable assignment thresholds and appropriate management categories in deeply introgressed populations. Sci Rep 10:2862. https://doi.org/10.1038/s41598-020-59521-2
Chen KY, Marschall EA, Sovic MG, Fries AC, Gibbs HL, Ludsin SA (2018) assignPOP: an R package for population assignment using genetic, non-genetic, or integrated data in a machine learning framework. Methods Ecol Evol 9:439–446. https://doi.org/10.1111/2041-210X.12897
Do C, Waples RS, Peel D, Macbeth GM, Tillett BJ, Ovenden JR (2014) NeEstimator v2: re-implementation of software for the estimation of contemporary effective population size (ne) from genetic data. Mol Ecol Resour 14:209–214. doi: https://doi.org/10.1111/1755-0998.12157
Dziech A (2021) Identification of Wolf-Dog Hybrids in Europe – An Overview of Genetic Studies. Front Ecol Evol 9:760160. https://doi.org/10.3389/fevo.2021.760160
Ebenhard T (1995) Conservation breeding as a tool for saving animal species from extinction. Trends Ecol Evol 10:438–443. https://doi.org/10.1016/S0169-5347(00)89176-4
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. DOI: https://doi.org/10.1111/j.1365-294X.2005.02553.x
Frankham R (2008) Genetic adaptation to captivity in species conservation programs. Mol Ecol 17:325–333. DOI: https://doi.org/10.1111/j.1365-294X.2007.03399.x
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to Conservation Genetics. Cambridge University Press, Cambridge
Gajdárová B, Belotti E, Bufka L, Duľa M, Kleven O, Kutal M, Ozoliņš J, Nowak C, Reiners TE, Tám B, Volfová J, Krojerová-Prokešová J (2021) Long-distance eurasian lynx dispersal – a prospect for connecting native and reintroduced populations in Central Europe. Conserv Genet. https://doi.org/10.1007/s10592-021-01363-0
Gautschi B, Müller JP, Schmid B, Shykoff JA (2003) Effective number of breeders and maintenance of genetic diversity in the captive bearded vulture population. Heredity 91:9–16. https://doi.org/10.1038/sj.hdy.6800278
Gilligan DM, Frankham R (2003) Dynamics of genetic adaptation to captivity. Conserv Genet 4:189–197. Doi: https://doi.org/10.1023/A:1023391905158
Goncalves da Silva A, Lalonde DR, Quse V, Shoemaker A, Russello MA (2010) Genetic approaches refine ex situ Lowland Tapir (Tapirus terrestris) conservation. J Hered 101:581–590. https://doi.org/10.1093/jhered/esq055
Goudet J (2005) Hierfstat, a package for R to compute and test hierarchical F-statistics. Mol Ecol Notes 5:184–186. https://doi.org/10.1111/j.1471-8286.2004.00828.x. Latest version available at github
Goudet J (2001) FSTAT, a program to estimate and test gene diversities and fixation indices (version 2.9.3). Updated from Goudet 1995
Gugolz D, Bernasconi MV, Breitenmoser-Würsten Ch, Wandeler P (2008) Historical DNA reveals the phylogenetic position of the extinct Alpine lynx. J Zool 275:201–208. https://doi.org/10.1111/j.1469-7998.2008.00428.x
Hedrick PW (1999) Perspective: highly variable loci and their interpretation in evolution and conservation. Evolution 53:313–318. DOI: https://doi.org/10.1111/j.1558-5646.1999.tb03767.x
Hedrick PW (2005) Genetics of populations. Jones and Bartlett Publishers, Sudbury
IUCN, the Commission on Environmental Policy (1987) The IUCN Position Statement on Translocation of living organisms. Introductions, Re-introductions and Re-stocking. Prepared by the Species Survival Commission in collaboration with the Commission on Ecology, and, Law and Administration. As approved by the 22nd Meeting of the IUCN Council, Gland, Switzerland, 4 September 1987, pp. 1–13
IUCN (1998) Guidelines for re-introductions. Prepared by the IUCN/SSC re-introduction specialist Group. IUCN, Gland, Switzerland and Cambridge, UK, pp 1–10
IUCN Red List Mapping 2012–2016 for the regional assessment of the Eurasian Lynx (Lynx lynx) in Europe. Large Carnivore Initiative for Europe IUCN/SSC Specialist Group et al (2020) shapefile corrected in May 2020
IUCN (2013) Guidelines for reintroductions and other Conservation Translocations. Version 1.0. Gland. IUCN Species Survival Commission, viiii + 57 pp, Switzerland
Jule KR, Leaver LA, Lea SEG (2008) The effects of captive experience on reintroduction survival in carnivores: a review and analysis. Biol Conserv 141:355–363. https://doi.org/10.1016/j.biocon.2007.11.007
Kaczensky P, Chapron G, von Arx M, Huber D, Andrén H, Linnell J (2013) Status, management and distribution of large carnivores – bear, lynx, wolf and wolverine – in Europe. Report to the EU Commission, Part 1 and Part 2
Keller LF, Waller DM (2002) Inbreeding effects in wild populations. Trends Ecol Evol 17:230–241
Kitchener AC, Breitenmoser-Würsten Ch, Eizirik E, Gentry A, Werdelin L, Wilting A, Yamaguchi N, Abramov AV, Christiansen P, Driscoll C, Duckworth JW, Johnson W, Luo S-J, Meijaard E, O’Donoghue P, Sanderson J, Seymour K, Bruford M, Groves C, Hoffmann M, Nowell K, Timmons Z, Tobe S (2017) A revised taxonomy of the Felidae. The final report of the cat classification Task Force of the IUCN/ SSC cat specialist Group. Cat News Special Issue 11:80
Kopelman NM, Mayzel J, Jakobsson M, Rosenberg NA, Mayrose I (2015) CLUMPAK: a program for identifying clustering modes and packaging population structure inferences across K. Mol Ecol Resour 15(5):1179–1191. https://doi.org/10.1111/1755-0998.12387
Kraaijeveld-Smit FJL, Griffiths RA, Moore RD, Beebee TJC (2006) Captive breeding and the fitness of reintroduced species: a test of the responses to predators in a threatened amphibian. J Appl Ecol 43:360–365. https://doi.org/10.1111/j.1365-2664.2006.01137.x
Kratochvíl J (1968) Recent distribution of the lynx in Europe. Acta Scientarum Naturalium Academiae Scientiarum Bohemicae Brno 5/6:1–74
Krojerová-Prokešová J, Turbaková B, Jelenčič M, Bojda M, Kutal M, Skrbinšek T, Koubek P, Bryja J (2019) Genetic constraints of population expansion of the Carpathian lynx at the western edge of its native distribution range in Central Europe. Heredity 122(6):785–799. https://doi.org/10.1038/s41437-018-0167-x
Kutal M, Duľa M, Krojerová-Prokešová J, Belotti E, Volfová J, Bufka L (2021) Uncoordinated reintroductions of eurasian lynx might be a threat for the species recovery in Central Europe. Biodivers Conserv 30:3737–3740
Lacy RC (1993) Impacts of inbreeding in natural and captive populations of vertebrates: implications for conservation. Perspect Biol Med 36:480–496. Doi: https://doi.org/10.1353/pbm.1993.0081
Leimu R, Fischer M (2010) Between-population outbreeding affects plant defence. PLoS ONE 5(9):e12614. https://doi.org/10.1371/journal.pone.0012614
Li YL, Liu JX (2018) StructureSelector: a web-based software to select and visualize the optimal number of clusters using multiple methods. Mol Ecol Resour 18:176–177. https://doi.org/10.1111/1755-0998.12719
Lucena-Perez M, Marmesat E, Kleinman-Ruiz D, Martínez-Cruz B, Węcek K, Saveljev AP, Seryodkin IV, Okhlopkov I, Dvornikov MG, Ozolins J, Galsandorj N, Paunovic M, Ratkiewicz M, Schmidt K, Godoy JA (2020) Genomic patterns in the widespread eurasian lynx shaped by late quaternary climatic fluctuations and anthropogenic impacts. Mol Ecol 29:812–828. https://doi.org/10.1111/mec.15366
Marshall TC, Sunnucks P, Spalton JA, Greth A, Pemberton J (1999) Use of genetic data for conservation management: the case of the arabian oryx. Anim Conserv 2:269–278. https://doi.org/10.1111/j.1469-1795.1999.tb00073.x
Mengüllüoğlu D, Ambarlı H, Barlow A, Paijmans JLA, Sayar AO, Emir H, Kandemir İ, Hofer H, Fickel J, Förster DW (2021) Mitogenome phylogeny including data from additional subspecies provides new insights into the historical biogeography of the eurasian lynx Lynx lynx. Genes 12:1216. DOI: https://doi.org/10.3390/genes12081216
Mueller SA, Reiners TE, Middelhoff TL, Anders O, Kasperkiewicz A, Nowak C (2020) The rise of a large carnivore population in Central Europe: genetic evaluation of lynx reintroduction in the Harz Mountains. Conserv Genet 21:577–587. https://doi.org/10.1007/s10592-020-01270-w
Mueller SA, Prost S, Anders O, Breitenmoser-Würsten C, Kleven O, Klinga P, Konec M, Kopatz A, Krojerová-Prokešová J, Middelhoff TL, Obexer-Ruff G, Reiners TE, Schmidt K, Sindičič M, Skrbinšek T, Tám B, Saveljev AP, Naranbaatar G, Nowak C (2022) Genome-wide diversity loss in reintroduced eurasian lynx populations urges immediate conservation management. Biol Conserv 266:109442. https://doi.org/10.1016/j.biocon.2021.109442
Newman D, Pilson D (1997) Increased probability of extinction due to decreased genetic effective population size: experimental populations of Clarkia pulchella. Evolution 51(2):354–362. https://doi.org/10.1111/j.1558-5646.1997.tb02422.x
Nowell K, Jackson P (1996) Wild cats. Status survey and conservation action plan. IUCN, Gland
O’Brien J (2007) Animal Conservation Genetics – an overview with relevance to captive breeding programmes. EAZA News 57:26–35
Paetkau D, Calvert W, Stirling I, Strobeck C (1995) Microsatellite analysis of population structure in canadian polar bears. Mol Ecol 4:347–354. https://doi.org/10.1111/j.1365-294X.1995.tb00227.x
Paetkau D, Slade R, Burden M, Estoup A (2004) Direct, real-time estimation of migration rate using assignment methods: a simulation-based exploration of accuracy and power. Mol Ecol 13:55–65. https://doi.org/10.1046/j.1365-294X.2004.02008.x
Park SDE (2001) The excel microsatellite toolkit (version 3.1) Animal Genomics Laboratory, University College, Dublin
Pelletier F, Reale D, Watters J, Boakes EH, Garant D (2009) Value of captive populations for quantitative genetics research. Trends Ecol Evol 24:263–270. https://doi.org/10.1016/j.tree.2008.11.013
Piry S, Alapetite A, Cornuet J-M, Paetkau D, Baudouin L, Estoup A (2004) GeneClass2: a Software for genetic assignment and first-generation migrant detection. J Hered 95:536–539. https://doi.org/10.1093/jhered/esh074
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959. doi: https://doi.org/10.1093/genetics/155.2.945
Puechmaille SJ (2016) The program structure does not reliably recover the correct population structure when sampling is uneven: subsampling and new estimators alleviate the problem. Mol Ecol Resour 16:608–627. https://doi.org/10.1111/1755-0998.12512
Quattro JM, Vrijenhoek RC (1989) Fitness differences in remnant populations of the endangered Sonoran topmin-now. Science 245:976–978. doi: https://doi.org/10.1126/science.2772650
R Core Team (2020) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ralls K, Brugger K, Ballou JD (1979) Inbreeding and juvenile mortality in small populations of ungulates. Science 206:1101–1103. DOI: https://doi.org/10.1126/science.493997
Ralls K, Ballou JD, Templeton AR (1988) Estimates of lethal equivalents and the cost of inbreeding in mammals. Conserv Biol 2:185–193. DOI:https://doi.org/10.1111/J.1523-1739.1988.TB00169.X
Ratkiewicz M, Matosiuk M, Saveljev AP, Sidorovich V, Ozolins J, Männil P, Balciauskas L, Kojola I, Okarma H, Kowalczyk R, Schmidt K (2014) Long-range gene flow and the effects of climatic and ecological factors on genetic structuring in a large, solitary carnivore: the eurasian lynx. PLoS ONE 9(12):e115160. https://doi.org/10.1371/journal.pone.0115160
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249. https://doi.org/10.1093/oxfordjournals.jhered.a111573
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225. https://doi.org/10.2307/2409177
Richards CM (2000) Inbreeding depression and genetic rescue in a plant metapopulation. Am Nat 155:383–394. DOI: https://doi.org/10.1086/303324
Robert A (2009) Captive breeding genetics and reintroduction success. Biol Conserv 142:2915–2922. https://doi.org/10.1016/j.biocon.2009.07.016
Rueness EK, Naidenko S, Trosvik P, Stenseth NC (2014) Large-scale genetic structuring of a widely distributed carnivore – the eurasian lynx (Lynx lynx). PLoS ONE 9(4):e93675. https://doi.org/10.1371/journal.pone.0093675
Ruiz-López MJ, Roldán ERS, Espeso G, Gomendio M (2009) Pedigrees and microsatellites among endangered ungulates: what do they tell us? Mol Ecol 18:1352–1364. DOI: https://doi.org/10.1111/j.1365-294X.2009.04121.x
Ryser-Degiorgis M-P, Ryser A, Obexer-Ruff G, Breitenmoser-Würsten Ch, Breitenmoser U, Lang J (2004) Emergence of congenital malformations in free-ranging Lynx from Switzerland: first evidence of inbreeding depression? European Association of Zoo- and Wildlife Veterinarians. EAZWY:307–311
Saccheri I, Kuussaari M, Kankare M, Vikman P, Fortelius W, Hanski I (1998) Inbreeding and extinction in a butterfly metapopulation. Nature 392:491–494. https://doi.org/10.1038/33136
Säisä M, Koljonen M-L, Tähtinen J (2003) Genetic changes in Atlantic salmon stocks since historical times and the effective population size of a long-term captive breeding programme. Conserv Genet 4:613–627. DOI: https://doi.org/10.1023/A:1025680002296
Signer EN, Schmidt CR, Jeffreys AJ (1994) DNA variability and parentage testing in captive Waldrapp ibises. Mol Ecol 3:291–300. doi: https://doi.org/10.1111/j.1365-294x.1994.tb00069.x
Sindičić M, Polanc P, Gomerčić T, Jelenčič M, Huber Đ, Trontelj P, Skrbinšek T (2013) Genetic data confirm critical status of the reintroduced dinaric population of eurasian lynx. Conserv Genet 14:1009–1018. DOI: https://doi.org/10.1007/s10592-013-0491-x
Skrbinšek T, Boljte B, Jelenčič M, Sindičić M, Paule L, Promberger B, Rigg R, Bartol M, Pop M, Kubala J, Slijepčević V, Molinari P, Molinari-Jobin A, Krofel M, Bizjan P, Hočevar L, Oliveira T, Potočnik H, Kos I, Kljun F, Stronen AV, Tám B, Črtalič J, Konec M (2019) Baseline (pre-reinforcement) genetic status of SE Alpine and Dinaric lynx population. Report of LIFE Lynx, p 24
Soorae PS, Stanley Price MR(1997) Successful re-introductions of large carnivores- what are the secrets? Paper presented at the 11th International Conference on Bear Research and Management, Graz, Austria
39th Standing Committee meeting of Bern Convention, file no, T-PVS (2019) 7 – Meeting conclusions of the Expert conference on the conservation of the lynx in West and Central Europe, Strassbourg, https://www.coe.int/en/web/bern-convention/-/39th-standing-committee-meeting
Steyer K, Tiesmeyer A, Muñoz-Fuentes V, Nowak C (2018) Low rates of hybridization between european wildcats and domestic cats in a human-dominated landscape. Ecol Evol 8:2290–2304. https://doi.org/10.1002/ece3.3650
Taberlet P, Griffin S, Goossens B, Questiau S, Manceau V, Escaravage N, Waits LP, Bouvet J (1996) Reliable genotyping of samples with very low DNA quantities using PCR. Nucleic Acids Res 26:3189–3194. doi: https://doi.org/10.1093/nar/24.16.3189
Turček FJ (1951) Effect of introductions on two game populations in Czechoslovakia. J Wildl Manage 15:113–114)
Versteege L, Lengger J, Fienieg E, Schad K (2017) Long-term management plan for the eurasian lynx (Lynx lynx) european studbook (ESB). Safaripark Beekse Bergen, Hilvarenbeek, the Netherlands
von Arx M, Breitenmoser-Würsten Ch, Zimmermann F, Breitenmoser U(2004) Status and conservation of the Eurasian lynx (Lynx lynx) in Europe in 2001. KORA Bericht Nr. 19e. KORA, Muri, Switzerland
von Arx M, Breitenmoser-Würsten C, Breitenmoser U (2009) Lessons from the reintroduction of the eurasian lynx in Central and West Europe. In: Vargas A, Breitenmoser-Würsten C, Breitenmoser U (eds) Iberian Lynx ex situ conservation: an interdisciplinary approach. Fundación Biodiversidad in collaboration with. IUCN Cat Specialist Group, Madrid, pp 403–409
Vrijenhoek RC (1994) Genetic diversity and fitness in small populations. In: Loescheke V, Tomiuk J, Jain SK (eds) Conservation Genetics. Birkhauser-Verlag, Switzerland, pp 37–53
Wang J (2007) Triadic IBD coefficients and applications to estimating pairwise relatedness. Genet Res (Camb) 89:135–153. doi:https://doi.org/10.1017/S0016672307008798
Wang J (2011) Coancestry: a program for simulating, estimating and analysing relatedness and inbreeding coefficients. Mol Ecol Resour 11:141–145. doi:https://doi.org/10.1111/j.1755-0998.2010.02885.x
Wang J, Santiago E, Caballero A (2016) Prediction and estimation of effective population size. Heredity 117:193–206. https://doi.org/10.1038/hdy.2016.43
Waples RS (1998) Separating the wheat from the chaff: patterns of genetic differentiation in high gene flow species. J Hered 89:438–450. https://doi.org/10.1093/jhered/89.5.438
Waples RS (2006) A bias correction for estimates of effective population size based on linkage disequilibrium at unlinked gene loci. Conserv Genet 7:167–184. https://doi.org/10.1007/s10592-005-9100-y
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370. https://doi.org/10.2307/2408641
Wickham H(2016) ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. ISBN 978-3-319-24277-4, https://ggplot2.tidyverse.org
Witzenberger KA, Hochkirch A (2011) Ex situ conservation genetics: a review of molecular studies on the genetic consequences of captive breeding programmes for endangered animal species. Biodivers Conserv 20:1843–1861. https://doi.org/10.1007/s10531-011-0074-4
Acknowledgements
The study was financially supported by the Ministry of the Environment of the Czech Republic (projects of Liberec Zoo: 60453/ENV/12-2239/630/12 and 49730/ENV/13-2154/630/13), by INTER-EXCELLENCE - INTER-COST (LTC20021), by EEA grants (project MGSII-40), and by Institutional Research Plan (RVO: 68081766). We would like to thank previous ESB keeper Lars Versteege and zoo’s and other breeding facilities, who participated in this study, for providing samples of lynx in their care. Further, we thank to PLA Administration Beskydy, ČSOP Salamandr, Friends of the Earth Czech Republic, Ole Anders, Beňadik Machciník, Lilli Middelhoff, and Jerguš Tesák for their valuable help with sample collection, and to Anna Bryjová for technical support related to fragmentation analysis. Also, we would like to thank to Rory Putman for revision of English and to the editor and anonymous reviewers for valuable comments and suggestions to the manuscript.
Author information
Authors and Affiliations
Contributions
JK-P conducted the statistical analysis and prepared the manuscript. JK-P with the help of PV and PB designed the study. All co-authors contributed samples, JK-P with the help of BG performed laboratory analyses. All authors were involved in revision and editing the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Krojerová-Prokešová, J., Gajdárová, B., Reiners, T.E. et al. Ex situ versus in situ Eurasian lynx populations: implications for successful breeding and genetic rescue. Conserv Genet 24, 203–217 (2023). https://doi.org/10.1007/s10592-022-01494-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-022-01494-y