Abstract
Climate change is a multidimensional phenomenon. As such, no single metric can capture all trajectories of change and associated impacts. While numerous metrics exist to measure climate change, they tend to focus on central tendencies and neglect the multidimensionality of extreme weather events (EWEs). EWEs differ in their frequency, duration, and intensity, and can be described for temperature, precipitation, and wind speed, while considering different thresholds defining “extremeness.” We review existing EWE metrics and outline a framework for classifying and interpreting them in light of their foreseeable impacts on biodiversity. Using an example drawn from the Caribbean and Central America, we show that metrics reflect unequal spatial patterns of exposure across the region. Based on available evidence, we discuss how such patterns relate to threats to biological populations, empirically demonstrating how ecologically informed metrics can help relate EWEs to biological processes such as mangrove recovery. Unveiling the complexity of EWE trajectories affecting biodiversity is only possible through mobilisation of a plethora of climate change metrics. The proposed framework represents a step forward over assessments using single dimensions or averages of highly variable time series.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
One defining feature of contemporary climate change is the increasing frequency, intensity and duration of extreme weather events (EWEs) (Lange et al. 2020; Laufkötter et al. 2020). EWEs—including unusual and severe events (Seneviratne et al. 2012)—present a major challenge to human society and to the long-term persistence of biodiversity, as we know it today (Lange et al. 2020; Smith 2011; Thompson et al. 2023). Extreme weather events can affect different levels of biological organisation, ultimately altering ecological and evolutionary processes. Among their known impacts, EWEs can exert strong selection pressures on organisms (e.g., Grant et al. 2017; Stroud et al. 2020), change individuals’ behaviour (e.g., Cohen et al. 2021), trigger phenological shifts (e.g., Butt et al. 2015; Jentsch et al. 2009), cause local population collapses (e.g., Frederiksen et al. 2008; Smale and Wernberg 2013), and destabilise community and ecosystem organisation when tipping points are overcome (e.g., Armstrong McKay et al. 2022; Heinze et al. 2021; Kreyling et al. 2014). Yet to date vulnerability assessments for climate change have primarily focused on how changes in central tendencies of climate change are influencing biodiversity (Chapman et al. 2014; Jones et al. 2016), with only limited attention being paid to the effects of EWEs on biota (Harris et al. 2018; Sabater et al. 2022; Wethey et al. 2011).
While detection and attribution of climate change effects on biodiversity is challenging (Gonzalez et al. 2023; Taheri et al. 2021b), with concurrent environmental drivers affecting species and communities differently across sections of their geographical distributions (Taheri et al. 2021a), the issue is further complicated when dealing with highly dimensional phenomena such as EWEs. An extreme weather event can be defined according to its unusual frequency, intensity and/or duration, and measured from different sets of variables (e.g., temperature, precipitation, wind speed), while using different thresholds to determine its “extremeness” (McPhillips et al. 2018; Stephenson 2008). The resulting combination of EWE measurements can be massive and can often lead to results that are difficult to interpret.
A plethora of metrics exist to quantify one or more dimensions of extreme climate change (see, for instance, Zhang et al. 2011; Sillmann et al. 2013; Donat et al. 2013; Perkins and Alexander 2013; Garcia et al. 2014; Buckley and Huey 2016; Xu et al. 2019; McClanahan 2022), but much indeterminacy exists regarding their information content (Latimer and Zuckerberg 2019; McClanahan 2022): what climate change properties do different metrics convey? How are different metrics linked to biodiversity’s vulnerability? Answering these questions is important for anticipating threats to biodiversity. Here, we first review existing metrics describing EWEs and then develop a conceptual framework for classifying and interpreting the different metrics of EWEs considering their foreseeable impacts on biodiversity. We then establish the connection between EWE metrics and biodiversity threats by drawing on an example from the Caribbean and Central America, two regions of the world with the highest exposure to extreme climate change (Castellanos et al. 2022). We explore the patterns of covariation among 89 metrics calculated for the region and over a 70-year period. We then link the areal exposure to extreme weather events and the potential effects on biodiversity. Finally, we conduct a case study to test the usefulness of ecologically relevant EWE metrics in assessing mangrove’s vulnerability to heat waves, droughts, and heavy rainfall events in the Caribbean and Central America.
2 Metrics describing extreme weather events
Climate is a complicated phenomenon, and no single measurement can describe all its multiple dimensions. While the climate of a region represents “the finite distribution of climate variables over time relative to a regime of varying external conditions” (Werndl 2016), there are several approaches to measuring changes in climate over space and time. These approaches, referred to as “climate change metrics,” can be used to summarise the central tendencies or the variability of extreme values in meteorological time series (Buenafe et al. 2023; Garcia et al. 2014). Our review of the literature enables identification of several metrics commonly used by researchers to characterise EWEs (Table 1). Although under different names, all existing metrics represent a measurement of one or two dimensions of extreme weather events using different climate parameters and thresholds. As a first step towards understanding the metrics’ properties, we propose categorising EWE metrics hierarchically according to their dimensions, variables, and types of thresholds considered.
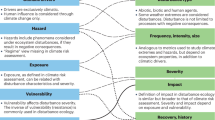
The first hierarchical level describes the dimensions of change captured by the metrics. These can measure the “Intensity” of the EWE, i.e., the excess over the threshold; the “Frequency,” i.e., the number of times that the threshold is exceeded in a period, often referred as probability or empirical probability of an extreme event; or the “Duration,” i.e., the time span between the start and end of the extreme weather event. It is important to acknowledge that metrics typically characterise one or two dimensions of climate change but rarely, if ever, characterise three or more dimensions (by more than three, we mean dimensions resulting from interactions among the three main dimensions described earlier) (see Fig. 1).
A conceptual classification of metrics according to their ability to capture one or more dimensions of EWEs (see Table 1 for a description of the metrics). neSA - Number of extreme standardised anomalies; MSA - Mean standardised anomalies; HS Anomalies above the seasonal mean; dV – Daily variability; D - Intraseasonal difference in the most extreme values; EWR - Inter-seasonal extreme weather range; DHW or DHM – Heating degree weeks (or months); MCIpe - Cumulative intensity of persistent climate extremes; RT - Extreme weather residence time; MRTpe or LRT - Duration of persistent climate extremes; CTpe - Change in timing of persistent climate extremes; FEE - Frequency of climate extremes; REE - Recurrence of climate extremes; FEpe - Frequency of persistent climate extremes; REpe - Recurrence of persistent climate extremes
The second and third levels of the categorisation of EWE are the variables and thresholds utilised to determine “extremeness,” respectively. Any calculation of EWE metrics requires a time series of meteorological variables so that excesses beyond an established threshold can be quantified. For example, among the most widespread metrics, we found those describing heat waves and warm spells, which are metrics that capture the intensity and duration of EWEs by using a combination of maximum temperatures and upper percentiles of the distribution as thresholds (McClanahan 2022; Zhang et al. 2011). However, metrics can be computed using other variables, such as precipitation or wind speed, as well as different types of thresholds (see Table 1). Different approaches to establishing thresholds exist, some of which focus on the statistical descriptions of the climatic variables of interest (henceforth termed statistically-derived threshold), while others focus on the thresholds known to trigger biological responses to climate changes (henceforth termed mechanistically-informed threshold) (see, for instance, van de Pol et al. 2017).
Thresholds based on statistical analysis of climatic drivers over time are established under the principle that extreme events are those events occurring in the tails of a frequency distribution of meteorological data values (Davison and Huser 2015; Stephenson 2008). Distributions are usually built using the maximum or minimum values of a climate variable per unit of time (e.g., per day, month, year) in a time series encompassing at least 30 years (as recommended by the World Meteorological Organisation). The lower (1st, 5th or 10th) or upper (90th, 95th or 99th) percentiles of the distribution are computed to determine the size of the tail. In turn, mechanistically informed thresholds are established based on prior knowledge of the value, or range of values, at which an event can qualitatively alter the state or development of the biological system of interest (Lenton et al. 2008). For example, wind speeds above 100km/h represent a mechanistically informed threshold that would likely lead to mangrove die-off (Amaral et al. 2023; Servino et al. 2018). Critical temperatures over 45–47°C are deadly for most eukaryotes and plants (Araújo et al. 2013), although complex interactions with water availability in terrestrial systems can alter perceived heat stress (Herrando-Pérez et al. 2020). Likewise, the occurrence of a high number of days with 1 or 2°C above the Monthly Maximum Mean (MMM) or climatological mean of the warmest month (McClanahan 2022) likely leads to mass mortality events in Corals due to high physiological stress (e.g., coral bleaching, quenching reaction, diseases) (Baker et al. 2008; Randall and van Woesik 2017).
3 Linking dimensions of change to threats to biodiversity
The best use of EWE metrics is achieved when their information content is analysed in light of the expected biological responses they are likely to trigger. Yet linking multidimensional patterns of climate change with biological responses is not straightforward since organisms’ sensitivity to climate dynamics (whether species, populations, communities, ecosystems, or biomes) involves responses that can be sometimes reported as individualistic (Baselga and Araújo 2009; Mcgeoch et al. 2006), while on other occasions they may be qualified as collective (Gilman et al. 2010; Mendoza and Araújo 2019). Individualistic responses are interpreted as the responses of the constituent parts of a system and are rooted in the view that organisms seek to maximise their own fitness, hence responding individually to external environmental pressures (Dawson et al. 2011). In contrast, collective biological responses are system-wide responses affecting the constituent parts of the system, and are easier to forecast once the rules of the system are understood (Gilman et al. 2010; Mestre et al. 2022). A comprehensive account of both, organismal or system-wide, responses before, during, and after EWEs is thus needed to gain understanding on the implications of extreme climate change on biodiversity. However, to date, empirical and quantitative evidence of such interactions is rather limited, requiring a number of simplifying assumptions linking measurements of climate change and biodiversity change (Buenafe et al. 2023; Garcia et al. 2016).
To begin structuring thinking about the links between extreme weather events and biological impacts, we classify impacts into critical dynamic population features, such as birth rates, growth rates, and mortality rates (Table 2). In contrast with gradual changes in climate, an increase in one or more dimensions of EWEs can cause abrupt demographic collapses and rapid disassembling of community dynamics, while preventing species adaptation as might be expected with gradual tracking of climate conditions (Hof et al. 2011; Hughes et al. 2018). We prioritise the demographic aspects of populations as their response encompasses both the direct impacts of EWEs on physiology, behaviour, and life history traits of organisms (Cerini et al. 2023; Sergio et al. 2018), as well as the indirect impacts caused by changes in biological interactions (Haberstroh and Werner 2022). Hence, all else being equal, increasing exposure to EWEs is likely to decrease the fitness of local populations, particularly when (i) organisms cannot tolerate changes in any dimension of extreme weather events exceeding known historical baselines (Allen et al. 2021; Hansen et al. 2022) or physiological limits (Jiguet et al. 2006; Valladares et al. 2014); (ii) individuals’ adaptive capacity is surpassed by compounded EWEs that shorten recovery times (Artigas et al. 2013; Fivash et al. 2021); (iii) biological populations have lower growth rates and narrower distributional ranges (Boyce et al. 2022); and (iv) other highly competitive or pathogenic species increase their abundances owing to EWEs (Haberstroh and Werner 2022; Morley and Lewis 2014).
It is also important to recognise that biological responses to extreme weather events depend on the specific dimension of change involved. Different threats can be inferred from the individual or joint assessment of the dimensions of change (see Table 2). Events characterised by extreme intensity, for instance, are likely to increase mortality rates across all the developmental stages of a population, triggering mass mortality events and decreasing post-disturbance survival rates (Buckley and Huey 2016; Frederiksen et al. 2008; Smale and Wernberg 2013). Events involving an increase of frequency are likely to impact population dynamics in the long-term by reducing recruitment and impacting growth rates, such that has been consistently observed across populations of plants, corals, insects and reptiles (Enright et al. 2015; Hughes et al. 2018; Maxwell et al. 2019; Neilson et al. 2020; Yu et al. 2022). Finally, events of increasing duration are likely to impact population dynamics by gradually altering the ratio between birth and mortality rates. For example, prolonged, but not necessarily more intense, EWEs have been shown to affect the recruitment process in corals and their symbionts by increasing mortality rates and decreasing birth rates as long as the event endures (Baker et al. 2008; Glynn 1996).
Interactions among multiple dimensions and variables can magnify the impact over the different features of population dynamics. Evidence from marine systems shows that prolonged ocean heat waves decrease the size of populations and make them more vulnerable to events of greater intensity in temperature or other climatic variables (Hughes et al. 2019; Kendrick et al. 2019). Likewise, evidence from terrestrial systems shows that the frequency and duration of droughts and heat waves interact to trigger population-level die-offs or magnify the impacts on the reproductive phenology of trees (Breshears et al. 2021). In the Tropics, such an impact on trees, together with water- and temperature-related stress, have been shown to change the population demography of mammals that rely on fruits and seeds (Butt et al. 2015; Campos et al. 2020; Marcelino et al. 2020).
4 Characterising multiple dimensions of EWEs across Central America and the Caribbean region
Central America and the Caribbean have been dubbed a “miner’s canary” of climate change due to the marked increase in extreme weather events. Nowhere in the world coincide so many climate hazards over a global hotspot of biodiversity, within a background of marked human socioeconomic vulnerability (Castellanos et al. 2022; Gould et al. 2020; Reyer et al. 2017). The region is known for its hurricanes and cyclones but droughts and heat waves are not unusual (Cook et al. 2022; Taylor et al. 2012). Documented impacts of such extreme events include direct economic losses (Devis-Morales et al. 2017; Lewsey et al. 2004), and erosion of human health (Di Napoli et al. 2022) and biodiversity (Amaral et al. 2023; Muñiz-Castillo et al. 2019). Yet a comprehensive assessment of EWEs, across their multiple dimensions and climatic variables, has not yet been undertaken.
To begin addressing this gap, we implement 89 EWE metrics, using ERA5-Land climatological time series (daily records at 0.1-degree spatial resolution, Hersbach et al. 2020), across the Caribbean and Central America over the last 70 years (1950–2020). ERA5 is a reanalysis product that effectively combines model data with global observations using the laws of physics to create a comprehensive gridded dataset spanning the entire globe. Despite the high uncertainty in some estimates for the Tropics, due to limited data availability (Hersbach et al. 2020), recent studies have demonstrated the ability of ERA5 to accurately capture spatiotemporal changes in climatic variables for Central and South America (Balmaceda‐Huarte et al. 2021; Gouveia et al. 2022), including changes in extreme values (Avila-Diaz et al. 2023; Bian et al. 2021). The ERA5 dataset is thus a valuable resource for investigating regional patterns of exposure, and spatiotemporal concordance between different types of EWEs. Nevertheless, it should be noted that achieving more accurate assessments may require a systematic quantification of the uncertainty associated with every metric, particularly in those capturing the intensity since reanalysis data might underestimate the actual daily value (Tan et al. 2023).
The number of selected metrics (n=89) results from combining multiple dimensions (intensity, frequency, and duration) and variables (wind speed, precipitation, and temperature), as well as interactions amongst such combinations (see Figure SM1.1 and Table SM1.1). Interactions were described by counting the number and duration of events of different nature (i.e., distinct climatic parameters) occurring within the same season or year. Metrics were computed separately for 0.1 × 0.1° grid cells using daily values of wind speed, precipitation and temperature for the dry season (DJFMA, i.e., December to April) and the wet season (JJASO, i.e., June to October), using as climatic thresholds the 5th (or 95th) percentiles for the daily minimum or maximum values obtained in every grid cell during the baseline period (1951–1981). EWE patterns for the last 40 years—excluding the 30-years baseline—were characterised by computing the average, frequency, or maximum value per grid cell (see Table 3). In an exploratory analysis, we found that a considerable proportion of these metrics showed low (54.4% with Spearman rho < 0.03) and/or non-significant correlation (24.5% with p < 0.05) with other metrics (Figure SM1.2). An extended version of the methods can be found as Supplementary Material (SM1). Data and code, as well as a dynamic overview of the spatiotemporal of EWE in the region is available in a GitHub repository (https://github.com/jdgonzalezt/multipleDimensionsExtremeClimateChange).
To understand the internal variability of EWE, as measured with the 89 metrics, we employed a Principal Components Analysis (PCA) on the standardised outputs of all metrics (with mean 0 and standard deviation 1). We then use PCA results to group metrics and grid cells sharing a similar EWE profile, explore the geographical patterns displayed by EWE profiles, and hypothesise links between the distribution of EWEs and biodiversity based on the conjectures developed in the conceptual framework (Section 4).
The PCA revealed exceptionally high variability among EWE metrics calculated for the Caribbean and Central America in the past 40 years. Specifically, the first Principal Component (PC) captured just 19% of the total variation among metrics, with 21 additional principal components being needed to account for ca. 80% of the variance (Figure SM1.3 and Table SM1.2). Should metrics display a greater level of redundancy, as one would expect in low-dimensional phenomena, the PCA would reduce metrics variability into a few components. In several ecological studies examining patterns of covariation among climate variables, two to three components are typically identified (Araújo et al. 2001; Petitpierre et al. 2017) with the first component accounting for a disproportionately high proportion of the total variation.
To identify areas where biodiversity is likely at risk due to increasing trends of EWE occurrence, and for simplicity, we focus on describing the spatial patterns captured by the first four principal components (Fig. 2, amounting ca. 43% of the total variance). The geographical patterns of the metrics with the highest correlation to these four principal components are included in the supplementary material (Figures SM1.3-6). Note that PCA groups together variables that have a similar relationship with each other. It then calculates a linear function that summarises the trends shared by the variables within each group, or component. This procedure ensures that the components have low correlation among themselves (also known as "quasi-orthogonal," Jolliffe and Cadima 2016) and, as a result, describe distinct types of EWEs.
Spatial variability across the multiple EWE metrics as summarised by Principal Component Analysis (PCA). To facilitate visualisation, PCA scores and loadings are represented individually on the left and central sides of the figure. Loadings describe the correlation of the metrics with every PC, where metrics with the highest loadings exhibit similar variability across space. Scores describe the position of grid cells along the principal components. Colours represent their association with the group of metrics correlated with each PC. To simplify interpretation, only the six metrics exhibiting the highest negative (red) and positive (grey) loadings are shown (names and correlation values are included in the tables on the right). Radar plots help identify the number, dimension, and climatic parameter (T - temperature, P - precipitation, and W - wind) of the 12 metrics with the highest loadings in each PC. Compounded metrics are represented with a circle. Full names of metrics can be found in Table 1
Geographically, the first component specifies areas of Central America and the Southern Caribbean that were highly exposed to more frequent and intense warm days during the historical baseline period studied (cells in red in PC-1 of Fig. 2), and areas highly exposed to heavy precipitation and cold days towards the north of the region (cells in grey in PC-1 Fig. 2). Heat waves and cold spells can compromise population persistence by increasing mortality, reducing birth rates, and altering sex ratios (Mazzotti et al. 2016; Wiederholt and Post 2010; Wright et al. 2015). Under limited dispersal conditions, the threat might be greatest for those small-sized populations at the edge of their physiological limits or distributional ranges (DeCarlo et al. 2017; Gutschick and BassiriRad 2003). As described by the second principal component, the threat for populations with such characteristics can be further exacerbated around the margins of Central and North America, where compounded events were detected (i.e., extreme wind, temperature, or precipitation occurring at the same location within the same year or season, cells in red in PC-2 of Fig. 2). Recent evidence indicates that the synergistic interaction between heat waves and meteorological droughts, for example, will result in more lethal events, which jeopardise the long-term survival of several species across the globe (Breshears et al. 2021; Sheppard et al. 2020).
The third and fourth principal component show that areas in the Caribbean islands and Central American corridor have been highly exposed to extreme winds and temperatures (red cells in PC-3 and PC-4 of Fig. 2). Hurricanes, cyclones and heat waves have immediate and lagged effects on population growth by simultaneously decreasing survival and fecundity (Morcillo et al. 2020; van den Burg et al. 2022). Given the available evidence, the increased exposure to more intense and frequent extreme winds, and prolonged high-temperature events, can potentially threaten species with lower fecundity, smaller population sizes, and narrow distributional ranges (Maxwell et al. 2019). In the case that dispersal of individuals from the mainland is restricted, the population size of such species would be reduced and likely lead to local extinctions across the islands and across fragments with low connectivity in the corridor. A similar process can occur in montane systems, in which extreme (and novel) climatic conditions may increase local extinction risks for high-altitude populations by exceeding the tolerances of individuals and increasing climatic barriers to their dispersal (Kerr 2020).
The fourth principal component also reveals that a wide extension of Central America and the Southern Caribbean has been exposed to sharp deficits of precipitation (grey cells in PC-3 of Fig. 2). While droughts and megadroughts are common to the American continent (Cook et al. 2022), and tropical species exhibit different adaptations to water deficit (Oliveira et al. 2021), there are different groups of species that can be at risk due to direct and indirect drought-induced effects. Population size of fast-growing plant species, for example, is directly affected by droughts because they do not invest resources in forming hydraulically-safe tissues that protect their structures from coping with water constraints (González-M et al. 2021; Guillemot et al. 2022). Likewise, population growth rate of frugivores, such as small primates, can be directly affected by water deficits and higher temperatures, or indirectly by food deficits resulting from tree die-off (Campos et al. 2020).
5 Linking multiple dimensions of EWEs to mangrove recovery in the Caribbean and Central America
The high dimensionality of the EWE metrics, revealed by 21 principal components needed to summarise 80% of the data, demonstrates that extreme weather events cannot be fully characterised unless a multidimensional analysis is performed. However, such high dimensionality further complicates the establishment of links between metrics and biodiversity. Instead of using “brute force” to navigate the “jungle” of metrics, i.e., computing several metrics before undertaking a ‘post hoc’ evaluation of their patterns of covariation to identify non-redundant patterns, an alternative is to select a set of metrics based on a priori knowledge of their ecological relevance.
To illustrate the principle, we select a subset of metrics to evaluate how greater exposure to heatwaves, droughts, and unusual rainfalls in the dry season (December to May) might have interfered with the post-hurricane recovery of mangrove populations in the North Atlantic Basin (NAB). Available evidence suggests that increased exposure to the intensity, frequency, and duration of such EWEs could be preventing mangrove recovery via direct and indirect effects on tree growth, resprouting, and seedling production (Amaral et al. 2023; Harris et al. 2010; Jimenez et al. 1985; Lagomasino et al. 2021). Decreasing individuals’ fitness is a direct effect of extremely intense temperatures and prolonged droughts, particularly for species such as Avicennia germinans or Laguncularia racemosa, or in poorly drained areas with lower fertility, such as in the Yucatán peninsula (Harris et al. 2010; Imbert 2018; Lagomasino et al. 2021; Vogt et al. 2012). In addition, both heat waves and droughts can induce indirect changes to biotic and abiotic conditions for biodiversity, via both natural and human induced processes. Natural processes include changes in the occurrence of early-successional plant species, which facilitate the establishment of mangrove propagules after disturbances, especially in the case of Rhizophora mangle (Donnelly and Walters 2014).
We asked whether the exposure to such EWEs in the dry season was lower in grid cells that exhibited some degree of recovery in mangrove forest coverage than in cells where no signs of recovery were evidenced. To assess exposure differences, we first identified grid cells with hurricane-damaged and recovered mangrove forests between 1998 and 2018. The geographic positions and status (damaged or recovered) of grid cells were retrieved from Amaral et al.’s (2023) study on the drivers of mangrove vulnerability and resilience to tropical cyclones in the NAB. For damage estimation, Amaral and colleagues traced changes in the Normalized Difference Vegetation Index (NDVI) over time. Specifically, they classified grid cells as ‘damaged’ if the mean ex-ante NDVI value (2 years before disturbance) differed from the mean ex-post NDVI value (after the disturbance) by more than -0.2, a threshold that had been field-validated for tree death (Lagomasino et al. 2021). For recovery estimation, on the other hand, they used the ex-post NDVI slope trend 1 year after the disturbance. Specifically, they classified cells with negative or zero NDVI trends as “not recovered,” while those with positive trends were classified as “recovered.” For further details on the NDVI-based analysis, including image pre-processing for water reflectance, refer to Taillie et al. (2020), Lagomasino et al. (2021) and Amaral et al. (2023).
We then estimated the degree of exposure to droughts, heat waves and heavy rainfall in both recovered and non-recovered grid cells. As proxies of such EWEs, we computed a set of metrics capturing the intensity (MCIpe), duration (RTpe) and frequency (FEpe) using two climate variables (temperature and precipitation) and two climatic thresholds (percentiles 95th and 5th of the baseline distribution: 1951 to 1981) (see Table SM1.3). We also quantified the occurrence of multiple events at the same location and year (Cpe). To determine the statistical difference between the two groups of grid cells (recovered vs. non-recovered), we used Wilcoxon signed-rank tests, and the Cliff’s delta (Cliff 1993) effect size to assess the magnitude and direction of the differences. Cliff’s delta estimates the probability that a value selected from one of the groups being compared is greater than a value selected from the other group. It varies from −1 to +1, with values farther from zero indicating the absence of overlap between the two groups. In our analysis, negative values indicate that non-recovered cells had greater exposure to extreme climate than recovered cells, whereas positive values indicated the opposite.
In the last two decades, mangrove forests exhibited low short-term resilience in areas that were damaged by hurricanes, with half of the affected areas (48%) showing no recovery signs 12 months after hurricane made landfall (Amaral et al. 2023). EWEs such as droughts, heat waves and intense rainfalls have been suggested as major factors delaying forest recovery, but a formal quantification of exposure has not been performed to date. By using a meaningful combination of climate change metrics, we show that exposure to EWEs during the dry season was significantly higher in grid cells with less mangrove recovery (Fig. 3a). We also identified the geographical areas where increased exposure has coincided with a lack of recovery (Fig. 3b–f). Such results suggest that post-cyclone recovery in mangrove forests can be reduced by the occurrence of individual or compounded events of extreme temperature and lack of precipitation in the dry season.
Comparison of exposure to the multiple dimensions of extreme climate change between grid cells with recovered and non-recovered mangrove forests from 1998 to 2018. (A) The effect size estimations using Cliff’s delta are shown here for metrics describing the intensity, duration, frequency, and compounded occurrence of EWEs. Lines to the left and right of the circles indicate the 95% confidence intervals of the calculated Cliff’s delta. Asterisks indicate significant differences between groups using the Wilcoxon signed-rank test (alpha < 0.05). (B-F) Levels of exposure to different dimensions and variables in grid cells where mangrove forests showed no recovery signs after cyclones made landfall. ‘Intensity’ is described as the cumulative intensity of five (or more) consecutive days with values exceeding the established threshold (MCIpe); “Duration” as the number of consecutive days with values exceeding the established threshold (RTpe); and “Frequency” as the maximum number of persistent events (five or more days exceeding the threshold) that occurred (FEpe). Thresholds were established as the percentiles 95th and 5th of maximum and minimum values from 1951 to 1981
However, it is important to note that the assessment provided represents only an approximation of the effect of increased climate exposure. For more robust estimates, it is essential to consider other environmental and biological factors alongside extreme climate. Key environmental factors, including land use, slope, distance from the coast, and soil depth, play a crucial role in determining the magnitude of hurricane damage and the post-landfall recovery process (Amaral et al. 2023). Moreover, greater exposure does not necessarily indicate greater risk, as species with similar levels of exposure might experience greater or lesser impacts due to species’ intrinsic sensitivity and adaptive capacity (e.g., Bailey et al. 2019; Valladares et al. 2014). Future studies should explore how adaptive responses (i.e., tolerance, migration, or evolutionary adaptation) can modulate mangrove vulnerability to the multiple dimensions of EWEs. The results presented herein can guide the establishment of a network of distributed experiments to assess such responses of mangroves across sites exposed to different stressors, including EWEs. They also provide a glimpse on the variables and dimensions that need to be accounted for when assessing the impacts of extreme climate change in the Caribbean and Central America regions.
6 Concluding remarks and outlook
Climate change is affecting living systems worldwide and we lack the models to appropriately characterise and forecast such changes. Weaknesses are particularly striking when it comes to modelling the effects of extreme events on biological systems. In the absence of comprehensive models that mechanistically link extreme climate dynamics to biodiversity dynamics, a first approach is to conduct a comprehensive analysis of extreme weather event patterns, while conceptually linking them with biodiversity threats.
Numerous climate change metrics exist to conduct such pattern analysis. Our review focused on metrics capturing different dimensions of extreme climate change (see Table 1). Using an example drawn from the Caribbean and Central American regions, we show that existing climate change metrics reveal extremely high dimensionality, meaning that no reduced set of metrics can describe the multiple patterns of change that follow from extreme weather events. Metrics capturing different dimensions (intensity, duration, and frequency), considering different variables (wind speed, temperature, and precipitation) and using distinct thresholds for defining “extremeness,” often lead to dissimilar assessments of exposure to EWE across different regions.
Although empirical evidence is patchy and vastly incomplete, we provide a coherent framework linking critical population dynamic features (e.g., birth, growth, and mortality rates) to the multiple dimensions of extreme climate change, thus overcoming some of the limitations of sparse observational data for predicting future threats to biodiversity. In addition, when more ecological information is available, the framework can help guide assessments of the underlying threats to species persistence under different scenarios of future climate change. As shown in our case study, interpreting exposure patterns in the context of EWE dimension, variable and threshold provide insight into factors impacting the recovery of mangrove populations after hurricanes made landfall. While connections have been established at the population level, achieving a comprehensive understanding requires systematic establishment of links at the community and ecosystem level.
Our review provides evidence supporting the usefulness of climate change metrics for studying environmental risks associated with EWEs. It also reinforces the view that the metrics provide an exploratory tool to examine past, current, and future consequences of EWE. The proposed framework can also guide the selection of metrics in empirical studies addressing the impacts of the multiple dimensions of extreme weather events on biological systems. As we move forward, it is essential for future studies to address potential biases and uncertainties specific to each metric, particularly concerning climatic data. In regions with limited long-term observations, the representation of extreme climatic values in datasets from reanalyses and model projections (both CMIP5 and CMIP6) may be inaccurate in space and time (Avila-Diaz et al. 2023; Gouveia et al. 2022; Ortega et al. 2021), leading to potential over- or underestimation of the intensity, frequency, and duration of EWEs. By undertaking sensitivity analyses, researchers can gain insights into the reliability and accuracy of these metrics in assessing the potential threats to biodiversity (see, for instance, Buenafe et al. 2023). Such analyses can provide valuable information for making more informed decisions and developing robust strategies for conservation and mitigation efforts, ensuring the protection and preservation of biodiversity in the face of climate change challenges. By addressing uncertainties in climate change forecasts, we can enhance the applicability and reliability of climate change metrics, strengthening our ability to effectively manage and respond to environmental risks associated with extreme weather events.
Data availability
Era5 Climate data is readily available at https://cds.climate.copernicus.eu/cdsapp#!/dataset/reanalysis-era5-land?tab=overview. Scripts, as well as a dynamic overview of the spatiotemporal of EWE in the region are available in https://github.com/jdgonzalezt/multipleDimensionsExtremeClimateChange.
References
Allen KJ, Verdon-Kidd DC, Sippo JZ, Baker PJ (2021) Compound climate extremes driving recent sub-continental tree mortality in northern Australia have no precedent in recent centuries. Sci Rep 11:18337. https://doi.org/10.1038/s41598-021-97762-x
Amaral C, Poulter B, Lagomasino D et al (2023) Drivers of mangrove vulnerability and resilience to tropical cyclones in the North Atlantic Basin. Sci Total Environ 898:165413. https://doi.org/10.1016/j.scitotenv.2023.165413
Araújo MB, Ferri‐Yáñez F, Bozinovic F et al (2013) Heat freezes niche evolution. Ecol Lett 16:1206–1219. https://doi.org/10.1111/ele.12155
Araújo MB, Humphries CJ, Densham PJ et al (2001) Would environmental diversity be a good surrogate for species diversity? Ecography 24:103–110. https://doi.org/10.1034/j.1600-0587.2001.240112.x
Armstrong McKay DI, Staal A, Abrams JF et al (2022) Exceeding 1.5°C global warming could trigger multiple climate tipping points. Science 377:eabn7950. https://doi.org/10.1126/science.abn7950
Artigas J, García-Berthou E, Bauer DE et al (2013) Global pressures, specific responses: effects of nutrient enrichment in streams from different biomes. Environ Res Lett 8:014002. https://doi.org/10.1088/1748-9326/8/1/014002
Ashcroft MB, Gollan JR, Warton DI, Ramp D (2012) A novel approach to quantify and locate potential microrefugia using topoclimate, climate stability, and isolation from the matrix. Glob Chang Biol 18:1866–1879. https://doi.org/10.1111/j.1365-2486.2012.02661.x
Avila-Diaz A, Torres RR, Zuluaga CF et al (2023) Current and Future Climate Extremes Over Latin America and Caribbean: Assessing Earth System Models from High Resolution Model Intercomparison Project (HighResMIP). Earth Syst Environ 7:99–130. https://doi.org/10.1007/s41748-022-00337-7
Bailey LD, Ens BJ, Both C et al (2019) Habitat selection can reduce effects of extreme climatic events in a long‐lived shorebird. J Anim Ecol 88:1474–1485. https://doi.org/10.1111/1365-2656.13041
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:435–471. https://doi.org/10.1016/j.ecss.2008.09.003
Balmaceda‐Huarte R, Olmo ME, Bettolli ML, Poggi MM (2021) Evaluation of multiple reanalyses in reproducing the spatio‐temporal variability of temperature and precipitation indices over southern South America. Int J Climatol 41:5572–5595. https://doi.org/10.1002/joc.7142
Baselga A, Araújo MB (2009) Individualistic vs community modelling of species distributions under climate change. Ecography 32:55–65. https://doi.org/10.1111/j.1600-0587.2009.05856.x
Bian G-F, Nie G-Z, Qiu X (2021) How well is outer tropical cyclone size represented in the ERA5 reanalysis dataset? Atmos Res 249:105339. https://doi.org/10.1016/j.atmosres.2020.105339
Boyce DG, Tittensor DP, Garilao C et al (2022) A climate risk index for marine life. Nat Clim Chang. https://doi.org/10.1038/s41558-022-01437-y
Breshears DD, Fontaine JB, Ruthrof KX et al (2021) Underappreciated plant vulnerabilities to heat waves. New Phytol 231:32–39. https://doi.org/10.1111/nph.17348
Buckley LB, Huey RB (2016) Temperature extremes: geographic patterns, recent changes, and implications for organismal vulnerabilities. Glob Chang Biol 22:3829–3842. https://doi.org/10.1111/gcb.13313
Buenafe KCV, Dunn DC, Everett JD et al (2023) A metric‐based framework for climate‐smart conservation planning. Ecol Appl 33:e2852. https://doi.org/10.1002/eap.2852
Burrows MT, Schoeman DS, Buckley LB et al (2011) The pace of shifting climate in marine and terrestrial ecosystems. Science 334:652–655. https://doi.org/10.1126/science.1210288
Butt N, Seabrook L, Maron M et al (2015) Cascading effects of climate extremes on vertebrate fauna through changes to low-latitude tree flowering and fruiting phenology. Glob Chang Biol 21:3267–3277. https://doi.org/10.1111/gcb.12869
Campos FA, Kalbitzer U, Melin AD et al (2020) Differential impact of severe drought on infant mortality in two sympatric neotropical primates. R Soc Open Sci 7:200302. https://doi.org/10.1098/rsos.200302
Cardil A, Molina DM, Kobziar LN (2014) Extreme temperature days and their potential impacts on southern Europe. Nat Hazards Earth Syst Sci 14:3005–3014. https://doi.org/10.5194/nhess-14-3005-2014
Castellanos E, Lemos M, Astigarraga L et al (2022) Central and South America. In: Climate Change 2022: Impacts, Adaptation and Vulnerability. In: Pörtner H-O, Roberts DC, Tignor M, Poloczanska ES, Mintenbeck K, Alegría A, Craig M, Langsdorf S, Löschke S, Möller V, Okem A, Rama B (eds) Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK and New York, NY, USA, pp 1689–1816
Cerini F, Childs DZ, Clements CF (2023) A predictive timeline of wildlife population collapse. Nat Ecol Evol 7:320–331. https://doi.org/10.1038/s41559-023-01985-2
Chapman S, Mustin K, Renwick AR et al (2014) Publishing trends on climate change vulnerability in the conservation literature reveal a predominant focus on direct impacts and long time‐scales. Divers Distrib 20:1221–1228. https://doi.org/10.1111/ddi.12234
Cliff N (1993) Dominance statistics: Ordinal analyses to answer ordinal questions. Psychol Bull 114:494–509. https://doi.org/10.1037/0033-2909.114.3.494
Cohen JM, Fink D, Zuckerberg B (2021) Extreme winter weather disrupts bird occurrence and abundance patterns at geographic scales. Ecography 44:1143–1155. https://doi.org/10.1111/ecog.05495
Cook BI, Smerdon JE, Cook ER et al (2022) Megadroughts in the Common Era and the Anthropocene. Nat Rev Earth Environ 3:741–757. https://doi.org/10.1038/s43017-022-00329-1
Cunningham SJ, Martin RO, Hojem CL, Hockey PAR (2013) Temperatures in Excess of Critical Thresholds Threaten Nestling Growth and Survival in A Rapidly-Warming Arid Savanna: A Study of Common Fiscals. PLoS One 8:e74613. https://doi.org/10.1371/journal.pone.0074613
Davison AC, Huser R (2015) Statistics of Extremes. Annu Rev Stat Appl 2:203–235. https://doi.org/10.1146/annurev-statistics-010814-020133
Dawson TP, Jackson ST, House JI et al (2011) Beyond Predictions: Biodiversity Conservation in a Changing Climate. Science 332:53–58. https://doi.org/10.1126/science.1200303
DeCarlo TM, Cohen AL, Wong GTF et al (2017) Mass coral mortality under local amplification of 2 °C ocean warming. Sci Rep 7:44586. https://doi.org/10.1038/srep44586
Devis-Morales A, Montoya-Sánchez RA, Bernal G, Osorio AF (2017) Assessment of extreme wind and waves in the Colombian Caribbean Sea for offshore applications. Appl Ocean Res 69:10–26. https://doi.org/10.1016/j.apor.2017.09.012
Di Napoli C, Allen T, Méndez‐Lázaro PA, Pappenberger F (2022) Heat stress in the Caribbean: Climatology, drivers, and trends of human biometeorology indices. Intl J Climatol 7774. https://doi.org/10.1002/joc.7774
Donat MG, Alexander LV, Yang H et al (2013) Updated analyses of temperature and precipitation extreme indices since the beginning of the twentieth century: The HadEX2 dataset: HADEX2-GLOBAL GRIDDED CLIMATE EXTREMES. J Geophys Res Atmos 118:2098–2118. https://doi.org/10.1002/jgrd.50150
Donnelly M, Walters L (2014) Trapping of Rhizophora mangle Propagules by Coexisting Early Successional Species. Estuar Coasts 37:1562–1571. https://doi.org/10.1007/s12237-014-9789-2
Enright NJ, Fontaine JB, Bowman DM et al (2015) Interval squeeze: altered fire regimes and demographic responses interact to threaten woody species persistence as climate changes. Front Ecol Environ 13:265–272. https://doi.org/10.1890/140231
Fivash GS, Temmink RJM, D’Angelo M et al (2021) Restoration of biogeomorphic systems by creating windows of opportunity to support natural establishment processes. Ecol Appl 31. https://doi.org/10.1002/eap.2333
Frederiksen M, Daunt F, Harris MP, Wanless S (2008) The demographic impact of extreme events: stochastic weather drives survival and population dynamics in a long-lived seabird. J Anim Ecol 77:1020–1029. https://doi.org/10.1111/j.1365-2656.2008.01422.x
Frich P, Alexander L, Della-Marta P et al (2002) Observed coherent changes in climatic extremes during the second half of the twentieth century. Clim Res 19:193–212. https://doi.org/10.3354/cr019193
Garcia RA, Cabeza M, Altwegg R, Araújo MB (2016) Do projections from bioclimatic envelope models and climate change metrics match?: Bioclimatic models versus climate change metrics. Glob Ecol Biogeogr 25:65–74. https://doi.org/10.1111/geb.12386
Garcia RA, Cabeza M, Rahbek C, Araújo MB (2014) Multiple Dimensions of Climate Change and Their Implications for Biodiversity. Science 344:1247579. https://doi.org/10.1126/science.1247579
Gilman SE, Urban MC, Tewksbury J et al (2010) A framework for community interactions under climate change. Trends Ecol Evol 25:325–331. https://doi.org/10.1016/j.tree.2010.03.002
Glynn PW (1996) Coral reef bleaching: facts, hypotheses and implications. Glob Chang Biol 2:495–509. https://doi.org/10.1111/j.1365-2486.1996.tb00063.x
Gonzalez A, Chase JM, O’Connor MI (2023) A framework for the detection and attribution of biodiversity change. Philos Trans R Soc B 378:20220182. https://doi.org/10.1098/rstb.2022.0182
González-M R, Posada JM, Carmona CP et al (2021) Diverging functional strategies but high sensitivity to an extreme drought in tropical dry forests. Ecol Lett 24:451–463. https://doi.org/10.1111/ele.13659
Goreau TJ, Hayes RL (1994) Coral Bleaching and Ocean “Hot Spots.”. Ambio 23:176–180
Gould WA, Castro-Prieto J, Álvarez-Berríos NL (2020) Climate Change and Biodiversity Conservation in the Caribbean Islands. In: Encyclopedia of the World’s Biomes. Elsevier, pp 114–125
Gouveia CD, Rodrigues Torres R, Marengo JA, Avila‐Diaz A (2022) Uncertainties in projections of climate extremes indices in South America via Bayesian inference. Int J Climatol 42:7362–7382. https://doi.org/10.1002/joc.7650
Grant PR, Grant BR, Huey RB et al (2017) Evolution caused by extreme events. Philos Trans R Soc B 372:20160146. https://doi.org/10.1098/rstb.2016.0146
Guillemot J, Martin‐StPaul NK, Bulascoschi L et al (2022) Small and slow is safe: On the drought tolerance of tropical tree species. Glob Chang Biol 28:2622–2638. https://doi.org/10.1111/gcb.16082
Gutschick VP, BassiriRad H (2003) Extreme events as shaping physiology, ecology, and evolution of plants: toward a unified definition and evaluation of their consequences. New Phytol 160:21–42. https://doi.org/10.1046/j.1469-8137.2003.00866.x
Haberstroh S, Werner C (2022) The role of species interactions for forest resilience to drought. Plant Biol J 24:1098–1107. https://doi.org/10.1111/plb.13415
Hansen WD, Schwartz NB, Williams AP et al (2022) Global forests are influenced by the legacies of past inter-annual temperature variability. Environ Res: Ecology 1:011001. https://doi.org/10.1088/2752-664X/ac6e4a
Harris RJ, Milbrandt EC, Everham EM, Bovard BD (2010) The Effects of Reduced Tidal Flushing on Mangrove Structure and Function Across a Disturbance Gradient. Estuar Coasts 33:1176–1185. https://doi.org/10.1007/s12237-010-9293-2
Harris RMB, Beaumont LJ, Vance TR et al (2018) Biological responses to the press and pulse of climate trends and extreme events. Nat Clim Chang 8:579–587. https://doi.org/10.1038/s41558-018-0187-9
Heinze C, Blenckner T, Martins H et al (2021) The quiet crossing of ocean tipping points. Proc Natl Acad Sci USA 118:e2008478118. https://doi.org/10.1073/pnas.2008478118
Herrando-Pérez S, Belliure J, Ferri-Yáñez F et al (2020) Water deprivation drives intraspecific variability in lizard heat tolerance. Basic Appl Ecol 48:37–51. https://doi.org/10.1016/j.baae.2020.08.001
Hersbach H, Bell B, Berrisford P et al (2020) The ERA5 global reanalysis. QJR Meteorol Soc 146:1999–2049. https://doi.org/10.1002/qj.3803
Hobday A, Oliver E, Sen Gupta A et al (2018) Categorizing and Naming Marine Heatwaves. Oceanog 31. https://doi.org/10.5670/oceanog.2018.205
Hobday AJ, Alexander LV, Perkins SE et al (2016) A hierarchical approach to defining marine heatwaves. Prog Oceanogr 141:227–238. https://doi.org/10.1016/j.pocean.2015.12.014
Hof C, Levinsky I, Araújo MB, Rahbek C (2011) Rethinking species’ ability to cope with rapid climate change: Biodiversity and rapid climate change. Glob Chang Biol 17:2987–2990. https://doi.org/10.1111/j.1365-2486.2011.02418.x
Hughes TP, Kerry JT, Baird AH et al (2018) Global warming transforms coral reef assemblages. Nature 556:492–496. https://doi.org/10.1038/s41586-018-0041-2
Hughes TP, Kerry JT, Connolly SR et al (2019) Ecological memory modifies the cumulative impact of recurrent climate extremes. Nat Clim Chang 9:40–43. https://doi.org/10.1038/s41558-018-0351-2
Imbert D (2018) Hurricane disturbance and forest dynamics in east Caribbean mangroves. Ecosphere 9:e02231. https://doi.org/10.1002/ecs2.2231
Jentsch A, Kreyling J, Boettcher-Treschkow J, Beierkuhnlein C (2009) Beyond gradual warming: extreme weather events alter flower phenology of European grassland and heath species. Glob Chang Biol 15:837–849. https://doi.org/10.1111/j.1365-2486.2008.01690.x
Jiguet F, Julliard R, Thomas CD et al (2006) Thermal range predicts bird population resilience to extreme high temperatures. Ecol Lett 9:1321–1330. https://doi.org/10.1111/j.1461-0248.2006.00986.x
Jimenez JA, Lugo AE, Cintron G (1985) Tree Mortality in Mangrove Forests. Biotropica 17:177. https://doi.org/10.2307/2388214
Jolliffe IT, Cadima J (2016) Principal component analysis: a review and recent developments. Phil Trans R Soc A 374:20150202. https://doi.org/10.1098/rsta.2015.0202
Jones KR, Watson JEM, Possingham HP, Klein CJ (2016) Incorporating climate change into spatial conservation prioritisation: A review. Biol Conserv 194:121–130. https://doi.org/10.1016/j.biocon.2015.12.008
Kendrick GA, Nowicki RJ, Olsen YS et al (2019) A Systematic Review of How Multiple Stressors From an Extreme Event Drove Ecosystem-Wide Loss of Resilience in an Iconic Seagrass Community. Front Mar Sci 6:455. https://doi.org/10.3389/fmars.2019.00455
Kerr JT (2020) Racing against change: understanding dispersal and persistence to improve species’ conservation prospects. Proc R Soc B 287:20202061. https://doi.org/10.1098/rspb.2020.2061
Kreyling J, Jentsch A, Beier C (2014) Beyond realism in climate change experiments: gradient approaches identify thresholds and tipping points. Ecol Lett 17:125–1e1. https://doi.org/10.1111/ele.12193
Lagomasino D, Fatoyinbo T, Castañeda-Moya E et al (2021) Storm surge and ponding explain mangrove dieback in southwest Florida following Hurricane Irma. Nat Commun 12:4003. https://doi.org/10.1038/s41467-021-24253-y
Lange S, Volkholz J, Geiger T et al (2020) Projecting Exposure to Extreme Climate Impact Events Across Six Event Categories and Three Spatial Scales. Earth’s Future 8. https://doi.org/10.1029/2020EF001616
Latimer CE, Zuckerberg B (2019) How extreme is extreme? Demographic approaches inform the occurrence and ecological relevance of extreme events. Ecol Monogr 89. https://doi.org/10.1002/ecm.1385
Laufkötter C, Zscheischler J, Frölicher TL (2020) High-impact marine heatwaves attributable to human-induced global warming. Science 369:1621–1625. https://doi.org/10.1126/science.aba0690
Lenton TM, Held H, Kriegler E et al (2008) Tipping elements in the Earth’s climate system. Proc Natl Acad Sci USA 105:1786–1793. https://doi.org/10.1073/pnas.0705414105
Lewsey C, Cid G, Kruse E (2004) Assessing climate change impacts on coastal infrastructure in the Eastern Caribbean. Mar Policy 28:393–409. https://doi.org/10.1016/j.marpol.2003.10.016
Marcelino J, Silva JP, Gameiro J et al (2020) Extreme events are more likely to affect the breeding success of lesser kestrels than average climate change. Sci Rep 10:7207. https://doi.org/10.1038/s41598-020-64087-0
Maxwell SL, Butt N, Maron M et al (2019) Conservation implications of ecological responses to extreme weather and climate events. Divers Distrib 25:613–625. https://doi.org/10.1111/ddi.12878
Mazzotti FJ, Cherkiss MS, Parry M et al (2016) Large reptiles and cold temperatures: Do extreme cold spells set distributional limits for tropical reptiles in Florida? Ecosphere 7. https://doi.org/10.1002/ecs2.1439
McClanahan TR (2022) Coral responses to climate change exposure. Environ Res Lett 17:073001. https://doi.org/10.1088/1748-9326/ac7478
McClanahan TR, Azali MK (2021) Environmental Variability and Threshold Model’s Predictions for Coral Reefs. Front Mar Sci 8:778121. https://doi.org/10.3389/fmars.2021.778121
Mcgeoch MA, Le Roux PC, Hugo EA, Chown SL (2006) Species and community responses to short-term climate manipulation: Microarthropods in the sub-Antarctic. Austral Ecol 31:719–731. https://doi.org/10.1111/j.1442-9993.2006.01614.x
McKechnie AE, Wolf BO (2010) Climate change increases the likelihood of catastrophic avian mortality events during extreme heat waves. Biol Lett 6:253–256. https://doi.org/10.1098/rsbl.2009.0702
McPhillips LE, Chang H, Chester MV et al (2018) Defining Extreme Events: A Cross‐Disciplinary Review. Earth’s Future 6:441–455. https://doi.org/10.1002/2017EF000686
Mendoza M, Araújo MB (2019) Climate shapes mammal community trophic structures and humans simplify them. Nat Commun 10:5197. https://doi.org/10.1038/s41467-019-12995-9
Mestre F, Rozenfeld A, Araújo MB (2022) Human disturbances affect the topology of food webs. Ecol Lett 25:2476–2488. https://doi.org/10.1111/ele.14107
Morán-Ordóñez A, Briscoe NJ, Wintle BA (2018) Modelling species responses to extreme weather provides new insights into constraints on range and likely climate change impacts for Australian mammals. Ecography 41:308–320. https://doi.org/10.1111/ecog.02850
Morcillo DO, Steiner UK, Grayson KL et al (2020) Hurricane-induced demographic changes in a non-human primate population. R Soc Open Sci 7:200173. https://doi.org/10.1098/rsos.200173
Morley NJ, Lewis JW (2014) Extreme climatic events and host–pathogen interactions: The impact of the 1976 drought in the UK. Ecol Complex 17:1–19. https://doi.org/10.1016/j.ecocom.2013.12.001
Muñiz-Castillo AI, Rivera-Sosa A, Chollett I et al (2019) Three decades of heat stress exposure in Caribbean coral reefs: a new regional delineation to enhance conservation. Sci Rep 9:11013. https://doi.org/10.1038/s41598-019-47307-0
Neilson EW, Lamb CT, Konkolics SM et al (2020) There’s a storm a‐coming: Ecological resilience and resistance to extreme weather events. Ecology and Evolution 10:12147–12156. https://doi.org/10.1002/ece3.6842
Oliveira RS, Eller CB, Barros FD et al (2021) Linking plant hydraulics and the fast–slow continuum to understand resilience to drought in tropical ecosystems. New Phytol 230:904–923. https://doi.org/10.1111/nph.17266
Orlowsky B, Seneviratne SI (2012) Global changes in extreme events: regional and seasonal dimension. Clim Chang 110:669–696. https://doi.org/10.1007/s10584-011-0122-9
Ortega G, Arias PA, Villegas JC et al (2021) Present‐day and future climate over central and South America according to CMIP5 / CMIP6 models. Int J Climatol 41:6713–6735. https://doi.org/10.1002/joc.7221
Pearson RG, Stanton JC, Shoemaker KT et al (2014) Life history and spatial traits predict extinction risk due to climate change. Nat Clim Chang 4:217–221. https://doi.org/10.1038/nclimate2113
Perkins SE, Alexander LV (2013) On the Measurement of Heat Waves. J Clim 26:4500–4517. https://doi.org/10.1175/JCLI-D-12-00383.1
Perkins-Kirkpatrick SE, Lewis SC (2020) Increasing trends in regional heatwaves. Nat Commun 11:3357. https://doi.org/10.1038/s41467-020-16970-7
Petitpierre B, Broennimann O, Kueffer C et al (2017) Selecting predictors to maximize the transferability of species distribution models: lessons from cross-continental plant invasions: Which predictors increase the transferability of SDMs? Glob Ecol Biogeogr 26:275–287. https://doi.org/10.1111/geb.12530
Randall CJ, van Woesik R (2017) Some coral diseases track climate oscillations in the Caribbean. Sci Rep 7:5719. https://doi.org/10.1038/s41598-017-05763-6
Reyer CPO, Adams S, Albrecht T et al (2017) Climate change impacts in Latin America and the Caribbean and their implications for development. Reg Environ Chang 17:1601–1621. https://doi.org/10.1007/s10113-015-0854-6
Sabater S, Freixa A, Jiménez L et al (2022) Extreme weather events threaten biodiversity and functions of river ecosystems: evidence from a meta‐analysis. Biol Rev:brv12914. https://doi.org/10.1111/brv.12914
Schlegel RW, Smit AJ (2018) heatwaveR: A central algorithm for the detection of heatwaves and cold-spells. JOSS 3:821. https://doi.org/10.21105/joss.00821
Seneviratne S, Nicholls N, Easterling D et al (2012) Changes in climate extremes and their impacts on the natural physical environment. https://doi.org/10.7916/D8-6NBT-S431
Sergio F, Blas J, Hiraldo F (2018) Animal responses to natural disturbance and climate extremes: a review. Glob Planet Chang 161:28–40. https://doi.org/10.1016/j.gloplacha.2017.10.009
Servino RN, de Oliveira Gomes LE, Bernardino AF (2018) Extreme weather impacts on tropical mangrove forests in the Eastern Brazil Marine Ecoregion. Sci Total Environ 628–629:233–240. https://doi.org/10.1016/j.scitotenv.2018.02.068
Sheppard C, Sheppard A, Fenner D (2020) Coral mass mortalities in the Chagos Archipelago over 40 years: Regional species and assemblage extinctions and indications of positive feedbacks. Mar Pollut Bull 154:111075. https://doi.org/10.1016/j.marpolbul.2020.111075
Sillmann J, Kharin VV, Zhang X et al (2013) Climate extremes indices in the CMIP5 multimodel ensemble: Part 1. Model evaluation in the present climate. J Geophys Res Atmos 118:1716–1733. https://doi.org/10.1002/jgrd.50203
Smale DA, Wernberg T (2013) Extreme climatic event drives range contraction of a habitat-forming species. Proc R Soc B 280:20122829. https://doi.org/10.1098/rspb.2012.2829
Smith MD (2011) An ecological perspective on extreme climatic events: a synthetic definition and framework to guide future research: Defining extreme climate events. J Ecol 99:656–663. https://doi.org/10.1111/j.1365-2745.2011.01798.x
Stephenson DB (2008) Definition, diagnosis, and origin of extreme weather and climate events. In: Diaz HF, Murnane RJ (eds) Climate Extremes and Society, 1st edn. Cambridge University Press, pp 11–23
Stroud JT, Mothes CC, Beckles W et al (2020) An extreme cold event leads to community-wide convergence in lower temperature tolerance in a lizard community. Biol Lett 16:20200625. https://doi.org/10.1098/rsbl.2020.0625
Sully S, Burkepile DE, Donovan MK et al (2019) A global analysis of coral bleaching over the past two decades. Nat Commun 10:1264. https://doi.org/10.1038/s41467-019-09238-2
Taheri S, García‐Callejas D, Araújo MB (2021a) Discriminating climate, land‐cover and random effects on species range dynamics. Glob Chang Biol 27:1309–1317. https://doi.org/10.1111/gcb.15483
Taheri S, Naimi B, Rahbek C, Araújo MB (2021b) Improvements in reports of species redistribution under climate change are required. Sci Adv 7:eabe1110. https://doi.org/10.1126/sciadv.abe1110
Taillie PJ, Roman-Cuesta R, Lagomasino D et al (2020) Widespread mangrove damage resulting from the 2017 Atlantic mega hurricane season. Environ Res Lett 15:064010. https://doi.org/10.1088/1748-9326/ab82cf
Tan ML, Armanuos AM, Ahmadianfar I et al (2023) Evaluation of NASA POWER and ERA5-Land for estimating tropical precipitation and temperature extremes. J Hydrol 624:129940. https://doi.org/10.1016/j.jhydrol.2023.129940
Taylor MA, Stephenson TS, Chen AA, Stephenson KA (2012) Climate Change and the Caribbean: Review and Response. Caribb Stud 40:169–200
Thompson V, Mitchell D, Hegerl GC et al (2023) The most at-risk regions in the world for high-impact heatwaves. Nat Commun 14:2152. https://doi.org/10.1038/s41467-023-37554-1
Valladares F, Matesanz S, Guilhaumon F et al (2014) The effects of phenotypic plasticity and local adaptation on forecasts of species range shifts under climate change. Ecol Lett 17:1351–1364. https://doi.org/10.1111/ele.12348
van de Pol M, Jenouvrier S, Cornelissen JHC, Visser ME (2017) Behavioural, ecological and evolutionary responses to extreme climatic events: challenges and directions. Philos Trans R Soc B 372:20160134. https://doi.org/10.1098/rstb.2016.0134
van den Burg MP, Madden H, van Wagensveld TP, Boman E (2022) Hurricane‐associated population decrease in a critically endangered long‐lived reptile. Biotropica 54:708–720. https://doi.org/10.1111/btp.13087
Vogt J, Skóra A, Feller IC et al (2012) Investigating the role of impoundment and forest structure on the resistance and resilience of mangrove forests to hurricanes. Aquat Bot 97:24–29. https://doi.org/10.1016/j.aquabot.2011.10.006
Werndl C (2016) On Defining Climate and Climate Change. Br J Philos Sci 67:337–364. https://doi.org/10.1093/bjps/axu048
Wethey DS, Woodin SA, Hilbish TJ et al (2011) Response of intertidal populations to climate: Effects of extreme events versus long term change. J Exp Mar Biol Ecol 400:132–144. https://doi.org/10.1016/j.jembe.2011.02.008
Wiederholt R, Post E (2010) Tropical warming and the dynamics of endangered primates. Biol Lett 6:257–260. https://doi.org/10.1098/rsbl.2009.0710
Winter A, Appeldoorn RS, Bruckner A et al (1998) Sea surface temperatures and coral reef bleaching off La Parguera, Puerto Rico (northeastern Caribbean Sea). Coral Reefs 17:377–382. https://doi.org/10.1007/s003380050143
Wright DR, Underhill LG, Keene M, Knight AT (2015) Understanding the Motivations and Satisfactions of Volunteers to Improve the Effectiveness of Citizen Science Programs. Soc Nat Resour 28:1013–1029. https://doi.org/10.1080/08941920.2015.1054976
Xu L, Wang A, Wang D, Wang H (2019) Hot Spots of Climate Extremes in the Future. J Geophys Res Atmos 124:3035–3049. https://doi.org/10.1029/2018JD029980
Yu X-L, Li J-Y, Zhou Y-T et al (2022) Simulated extreme high temperatures alter the demographic parameters of Aphelinus asychis and diminish parasitoid fitness. Biol Control 174:105028. https://doi.org/10.1016/j.biocontrol.2022.105028
Zhang X, Alexander L, Hegerl GC et al (2011) Indices for monitoring changes in extremes based on daily temperature and precipitation data. Wiley Interdiscip Rev: Clim Change 2:851–870. https://doi.org/10.1002/wcc.147
Zscheischler J, Martius O, Westra S et al (2020) A typology of compound weather and climate events. Nat Rev Earth Environ 1:333–347. https://doi.org/10.1038/s43017-020-0060-z
Acknowledgements
The authors acknowledge support from the BNP-PARIBAS Foundation on their 2019 Climate and Biodiversity Initiative call, through the CORESCAM project (“Coastal Biodiversity Resilience to Increasing Extreme Events in Central America”). RMR-C also acknowledges USAID funding to the SWAMP II Programme (Sustainable Wetland Adaptation and Mitigation Programme). Comments from Raquel A. García and Vinicius A. Bastazini improved our manuscript.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work was funded by BNP-PARIBAS Foundation on their 2019 Climate and Biodiversity Initiative call, through the CORESCAM project (“Coastal Biodiversity Resilience to Increasing Extreme Events in Central America”).
Author information
Authors and Affiliations
Contributions
JDGT and MBA conceived the study. JDGT curated the data collection, built the models, and performed the analyses. JDGT, RMR-C and MBA discussed the results. JDGT and MBA wrote the manuscript with contributions from all co-authors.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 7724 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
González-Trujillo, J.D., Román-Cuesta, R.M., Muñiz-Castillo, A.I. et al. Multiple dimensions of extreme weather events and their impacts on biodiversity. Climatic Change 176, 155 (2023). https://doi.org/10.1007/s10584-023-03622-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10584-023-03622-0