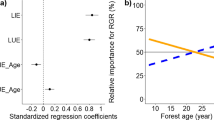
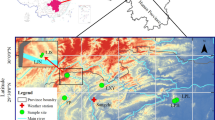
Abstract
We hypothesized that yearly response of plant growth to seasonal weather conditions would provide insight into the biogeographic status of three closely related and sympatric Dryopteris fern species and elucidate their potential future performance in conditions of climate change. In a 9-year experiment, we surveyed the yearly performance of 108 ex situ pre-grown sporophytes of D. carthusiana, D. dilatata, and D. expansa in six forests in Estonia. We used multi-factorial models to assess species-specific responses of frond traits and population parameters to seasonal temperature, precipitation, and snow cover, and also considered the effects of continentality and overstory density. Frond length was highly limited by plant age, and even the 11-year-old plants did not reach their mature size. The number of fronds stabilized already in the first few years. Climate effects on leaf traits accumulated over 2 years, as their growth benefited from the abundant precipitation during two growing seasons and the presence of dry cold winters. Milder climate near the coast and semi-open overstory provided some extra support for the growth. Species-specific effects were rare. The species’ responses to habitat and climate effects only partly mirrored their present biogeographic distribution. Evidently, possible effects of climate change might go unnoticed for decades in these ferns since it would be mitigated by (1) their slow ontogenesis, (2) organ preformation during preceding year(s), and (3) region-specific and habitat-specific effects of environmental conditions.





Similar content being viewed by others
References
Anderson MC (1964) Studies of the woodland light climate: I. The photographic computation of light conditions J Ecol 52:27–41. https://doi.org/10.2307/2257780
Atlas of the Estonian Flora. https://elurikkus.ee/en/plant-atlas. Accessed 15 December 2018
Bliss LC (1971) Arctic and alpine plant life cycles. Annu Rev Ecol Syst 2:405–438. https://doi.org/10.1146/annurev.es.02.110171.002201
Bremer P, Jongejans E (2010) Frost and forest stand effects on the population dynamics of Asplenium scolopendrium. Popul Ecol 52:211. https://doi.org/10.1007/s10144-009-0143-7
Campbell JL, Mitchell MJ, Groffman PM et al (2005) Winter in northeastern North America: a critical period for ecological processes. Front Ecol Environ 3:314–322. https://doi.org/10.1890/1540-9295(2005)003[0314:WINNAA]2.0.CO;2
Chapin FS, Bloom AJ, Field CB, Waring RH (1987) Plant responses to multiple environmental factors. BioScience 37:49–57. https://doi.org/10.2307/1310177
Chen I-C, Hill JK, Ohlemüller R et al (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024. https://doi.org/10.1126/science.1206432
Christensen JH, Christensen OB (2007) A summary of the PRUDENCE model projections of changes in European climate by the end of this century. Clim Chang 81:7–30. https://doi.org/10.1007/s10584-006-9210-7
Collins M, Knutti R, Arblaster J et al (2013) Long-term climate change: projections, commitments and irreversibility. In: Stocker TF, Qin D, Plattner GK et al (eds) Climate change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, pp 1029–1136 https://www.ipcc.ch/report/ar5/wg1/
Crimmins SM, Dobrowski SZ, Greenberg JA et al (2011) Changes in climatic water balance drive downhill shifts in plant species' optimum elevations. Science 331:324. https://doi.org/10.1126/science.1199040
Daryanto S, Wang L, Jacinthe P-A (2017) Global synthesis of drought effects on cereal, legume, tuber and root crops production: a review. Agric Water Manag 179:18–33. https://doi.org/10.1016/j.agwat.2016.04.022
De Frenne P, Rodríguez-Sánchez F, Coomes DA et al (2013) Microclimate moderates plant responses to macroclimate warming. Proc Natl Acad Sci U S A 110:18561. https://doi.org/10.1073/pnas.1311190110
De Groot GA, Verduyn B, Wubs ERJ et al (2012) Inter- and intraspecific variation in fern mating systems after long-distance colonization: the importance of selfing. BMC Plant Biol 12:3. https://doi.org/10.1186/1471-2229-12-3
Dostál J, Fraser-Jenkins CR, Reichstein T (1984) Dryopteris. In: Kramer KU, Dostál J, Reichstein T et al (eds) Illustrierte Flora von Mitteleuropa. Band I. Pteridophyta. Teil I. Verlag Paul Parey, Berlin, pp 137–169
Ekrt L, Holubová R, Trávnícek P et al (2010) Species boundaries and frequency of hybridization in the Dryopteris carthusiana (Dryopteridaceae) complex: a taxonomic puzzle resolved using genome size data. Am J Bot 97:1208–1219. https://doi.org/10.3732/ajb.0900206
Espinar JL, Díaz-Delgado R, Bravo-Utrera MA et al (2015) Linking Azolla filiculoides invasion to increased winter temperatures in the Doñana marshland (SW Spain). Aquat Invasions 10:17–24. https://doi.org/10.3391/ai.2015.10.1.02
Flinn K (2006) Reproductive biology of three fern species may contribute to differential colonization success in post-agricultural forests. Am J Bot 93:1289–1294 http://www.jstor.org/stable/4122768
Fraser-Jenkins CR (1993) Dryopteris adanson. In: Tutin TG, Heywood VH, Burges NA et al (eds) Flora Europea. Vol. I, 2nd edn. Cambridge University Press, Cambridge, pp 27–30
Gallinat AS, Primack RB, Wagner DL (2015) Autumn, the neglected season in climate change research. Trends Ecol Evol 30:169–176. https://doi.org/10.1016/j.tree.2015.01.004
Gaudet DA, Laroche A (1999) Snow mold-crop-environment interactions. In: Margesin R, Schinner F (eds) Biotechnological applications of cold-adapted organisms. Springer, Berlin, pp 191–202
Geber AM, Watson MA, de Kroon H (1997) Organ preformation, development, and resource allocation in perennials. In: Bazzaz FA, Grace J (eds) Plant resource allocation. Academic Press, San Diego, pp 113–141
Gibby M, Walker S (1977) Further cytogenetic studies and a reappraisal of the diploid ancestry in the Dryopteris carthusiana complex. Fern Gazette 11:315–324
Gilliam FS (2007) The ecological significance of the herbaceous layer in temperate forest ecosystems. BioScience 57:845–858. https://doi.org/10.3159/10.1641/B571007
Goldblum D, Kwit MC (2012) The relative photosynthetic contribution of old and new fronds of the wintergreen fern Dryopteris carthusiana, Ontario, Canada. J Torrey Bot Soc 139:270–282. https://doi.org/10.3159/TORREY-D-11-00032.1
Hari P, Heliövaara K, Kulmala L (eds) (2013) Physical and physiological forest ecology. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-5603-8
Hedenås H, Christensen P, Svensson J (2016) Changes in vegetation cover and composition in the Swedish mountain region. Environ Monit Assess 188:452. https://doi.org/10.1007/s10661-016-5457-2
Henry HAL, Hutchison JS, Kim MK et al (2015) Context matters for warming: interannual variation in grass biomass responses to 7 years of warming and N addition. Ecosystems 18:103–114. https://doi.org/10.1007/s10021-014-9816-y
Hopkins AD (1938) Bioclimatics: a science of life and climate relations. Department of Agriculture, Washington
Hornych O, Ekrt L (2017) Spore abortion index (SAI) as a promising tool of evaluation of spore fitness in ferns: an insight into sexual and apomictic species. Plant System Evol 303:497–507. https://doi.org/10.1007/s00606-016-1386-3
Hultén E, Fries M (1986) Atlas of north European vascular plants north of the tropic of cancer I. Koeltz Scientific Books, Königstein
Inouye DW (2000) The ecological and evolutionary significance of frost in the context of climate change. Ecol Lett 3:457–463. https://doi.org/10.1046/j.1461-0248.2000.00165.x
Ishioka R, Muller O, Hiura T et al (2013) Responses of leafing phenology and photosynthesis to soil warming in forest-floor plants. Acta Oecol 51:34–41. https://doi.org/10.1016/j.actao.2013.05.011
Jaagus J (1997) The impact of climate change on the snow cover pattern in Estonia. Clim Chang 36:65–77. https://doi.org/10.1023/A:1005304720412
Jaagus J, Ahas R (2000) Space-time variations of climatic seasons and their correlation with the phenological development of nature in Estonia. Clim Res 15:207–219. https://doi.org/10.3354/cr015207
Jaagus J, Mändla K (2014) Climate change scenarios for Estonia based on climate models from the IPCC Fourth Assessment Report. Estonian J Earth Sci 63:166–180. https://doi.org/10.3176/earth.2014.15
Juslén A, Väre H, Wikström N (2011) Relationships and evolutionary origins of polyploid Dryopteris (Dryopteridaceae) from Europe inferred using nuclear pgiC and plastid trnL-F sequence data. Taxon 60:1284–1294. https://doi.org/10.1002/tax.605005
Karst J, Gilbert B, Lechowicz MJ (2005) Fern community assembly: the roles of chance and the environment at local and intermediate scales. Ecology 86:2473–2486. https://doi.org/10.1890/04-1420
Kessler M (2010) Biogeography of ferns. In: Mehltreter K, Walker LR, Sharpe JM (eds) Fern ecology. Cambridge University Press, Cambridge, pp 23–60
Kovács B, Tinya F, Ódor P (2017) Stand structural drivers of microclimate in mature temperate mixed forests. Agric For Meteorol 234-235:11–21. https://doi.org/10.1016/j.agrformet.2016.11.268
Kreyling J, Haei M, Laudon H (2012) Absence of snow cover reduces understory plant cover and alters plant community composition in boreal forests. Oecologia 168:577–587. https://doi.org/10.1007/s00442-011-2092-z
Kukk T, Kull T (eds) (2005) Atlas of the Estonian Flora. Institute of Agricultural and Environmental Sciences of the Estonian University of Life Sciences, Tartu
Larsson D (2017) The allopolyploid origin of Dryopteris dilatata (Hoffm.) A. Gray in Europe. Masters Thesis, Institutionen för biologisk grundutbildning, Stockholms universitet, Stockholm
Lee PH, Huang YM, Chiou WL (2018) Fern phenology. In: Fernández H (ed) Current advances in fern research. Springer, Cham. https://doi.org/10.1007/978-3-319-75103-0_18
Lemke IH, Kolb A, Graae BJ et al (2015) Patterns of phenotypic trait variation in two temperate forest herbs along a broad climatic gradient. Plant Ecol 216:1523–1536. https://doi.org/10.1007/s11258-015-0534-0
Liang L (2016) Beyond the bioclimatic law: geographic adaptation patterns of temperate plant phenology. Prog Phys Geogr 40:811–834. https://doi.org/10.1177/0309133316656558
Liira J, Sepp T, Kohv K (2012) The ecology of tree regeneration in mature and old forests: combined knowledge for sustainable forest management. J For Res 16:184–193. https://doi.org/10.1007/s10310-011-0257-6
Littell RC, Milliken GA, Stroup WW et al (1996) SAS® system for mixed models. SAS Institute Inc., Cary
Manton I (1950) Problems of cytology and evolution in the Pteridophyta. Cambridge University Press, Cambridge
Manton I, Walker S (1954) Induced apogamy in Dryopteris dilatata (Hoffm.) A. Gray and D. filix-mas (L.) Schott emend. and its significance for the interpretation of the two species. Ann Bot 18:377–383. https://doi.org/10.1093/oxfordjournals.aob.a083402
Mehltreter K, Walker LR, Sharpe JM (2010) Fern Ecology. Cambridge University Press, Cambridge
Minoletti ML, Boerner REJ (1993) Seasonal photosynthesis, nitrogen and phosphorus dynamics, and resorption in the wintergreen fern Polystichum acrostichoides (Michx.) Schott. Bull Torrey Bot Club 120:397–404. https://doi.org/10.2307/2996743
Morgan JA, Parton W, Derner JD et al (2016) Importance of early season conditions and grazing on carbon dioxide fluxes in Colorado shortgrass steppe. Rangel Ecol Manag 69:342–350. https://doi.org/10.1016/j.rama.2016.05.002
Moser B, Walthert L, Metslaid M et al (2017) Spring water deficit and soil conditions matter more than seed origin and summer drought for the establishment of temperate conifers. Oecologia 183:519–530. https://doi.org/10.1007/s00442-016-3766-3
Neufeld HS, Young DR (2003) Ecophysiology of the herbaceous layer in temperate deciduous forests. In: Gilliam FS, Roberts MR (eds) The herbaceous layer in forests of eastern North America. Oxford University Press, New York, pp 38–90. https://doi.org/10.1093/acprof:osobl/9780199837656.001.0001
Nilsson MC, Wardle DA (2005) Understory vegetation as a forest ecosystem driver: evidence from the northern Swedish boreal forest. Front Ecol Environ 3:421–428. https://doi.org/10.1007/10.2307/3868658
Paal T, Kütt L, Lõhmus K et al (2017) Both spatiotemporal connectivity and habitat quality limit the immigration of forest plants into wooded corridors. Plant Ecol 218:417–431. https://doi.org/10.1007/s11258-017-0700-7
Paal T, Zobel K, Liira J (2019) Standardized response signatures of functional traits pinpoint limiting ecological filters during the migration of forest plant species into wooded corridors. Ecol Indic. In press
Page CN (1997) The ferns of Britain and Ireland. Cambridge University Press, Cambridge
Page CN (2002) Ecological strategies in fern evolution: a neopteridological overview. Rev Palaeobot Palynology 119:1–33. https://doi.org/10.1016/S0034-6667(01)00127-0
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42. https://doi.org/10.1038/nature01286
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Glob Ecol Biogeogr 12:361–371. https://doi.org/10.1046/j.1466-822X.2003.00042.x
Peck J, Peck C, Farrar D (1990) Influences of life history attributes on formation of local and distant fern populations. Am Fern J 80:126–142. https://doi.org/10.2307/1547200
Rapacz M, Ergon Å, Höglind M et al (2014) Overwintering of herbaceous plants in a changing climate. Still more questions than answers. Plant Sci 225:34–44. https://doi.org/10.1016/j.plantsci.2014.05.009
Rasmus S, Lundell R, Saarinen T (2011) Interactions between snow, canopy, and vegetation in a boreal coniferous forest. Plant Ecol Divers 4:55–65. https://doi.org/10.1080/17550874.2011.558126
Rünk K (2002) Initial survey of the Dryopteris carthusiana complex in Estonia. Fern Gazette 16:450
Rünk K, Zobel K (2007) Phenotypic plasticity and biomass allocation pattern in three Dryopteris (Dryopteridaceae) species on an experimental light-availability gradient. Plant Ecol 193:85–99. https://doi.org/10.1007/s11258-006-9250-0
Rünk K, Zobel M (2009) Differences in post-emergence growth of three fern species could help explain their varying local abundance. Am Fern J 99:307–322. https://doi.org/10.1640/0002-8444-99.4.307
Rünk K, Moora M, Zobel M (2004) Do different competitive abilities of three fern species explain their different regional abundances? J Veg Sci 15:351–356. https://doi.org/10.1111/j.1654-1103.2004.tb02271.x
Rünk K, Moora M, Zobel M (2006) Population stage structure of three congeneric Dryopteris species in Estonia. Proc Estonian Acad Sci Biol Ecol 55:15–30
Rünk K, Zobel M, Zobel K (2010) Different factors govern the performance of three closely related and ecologically similar Dryopteris species with contrastingly different abundance in a transplant experiment. Botany 88:961–969. https://doi.org/10.1139/B10-066
Sarvela J (2000) Dryopteris. In: Jonsell B (ed) Flora Nordica. Vol. 1: Lycopodiaceae to Polygonaceae. The Bergius Foundation. The Royal Swedish Academy of Sciences, Stockholm, pp 74–84
Sato T (1982) Phenology and wintering capacity of sporophytes and gametophytes of ferns native to northern Japan. Oecologia 55:53–61 http://www.jstor.org/stable/4216791
Sato T (1990) Estimation of chronological age for sporophyte maturation in three semi-evergreen ferns in Hokkaido. Ecol Res 5:55–62. https://doi.org/10.1007/BF02348463
Saxe H, Cannell MGR, Johnsen Ø et al (2001) Tree and forest functioning in response to global warming. New Phytol 149:369–399. https://doi.org/10.1046/j.1469-8137.2001.00057.x
Schwerbrock R, Leuschner C (2016) Air humidity as key determinant of morphogenesis and productivity of the rare temperate woodland fern Polystichum braunii. Plant Biol 18:649–657. https://doi.org/10.1111/plb.12444
Seifert M (1992) Populationsbiologie und Aspekte der Morphologie zweier Wurmfarne, Dryopteris carthusiana und Dryopteris dilatata. PhD Thesis, Universität Zürich, Zürich
Sepp M, Tamm T, Sagris V (2018) The future climate regions in Estonia. Estonian J Earth Sci 68:259–268. https://doi.org/10.3176/earth.2018.19
Sessa EB, Zimmer EA, Givnish TJ (2012) Unraveling reticulate evolution in North American Dryopteris (Dryopteridaceae). BMC Evol Biol 12:104. https://doi.org/10.1186/1471-2148-12-104
Sessa EB, Testo WL, Watkins JE Jr (2016) On the widespread capacity for, and functional significance of, extreme inbreeding in ferns. New Phytol 211:1108–1119. https://doi.org/10.1111/nph.13985
Sharpe JM, Mehltreter K, Walker LR (2010) Ecological importance of ferns. In: Mehltreter K, Walker LR, Sharpe JM (eds) Fern ecology. Cambridge University Press, Cambridge, pp 1–21
Simons AM, Goulet JM, Bellehumeur KF (2010) The effect of snow depth on overwinter survival in Lobelia inflata. Oikos 119:1685–1689. https://doi.org/10.1111/j.1600-0706.2010.18515.x
Spinoni J, Vogt JV, Naumann G, Barbosa P, Dosio A (2018) Will drought events become more frequent and severe in Europe? Int J Climatol 38:1718–1736. https://doi.org/10.1002/joc.5291
Starr G, Oberbauer SF (2003) Photosynthesis of arctic evergreens under snow: implications for tundra ecosystem carbon balance. Ecology 84:1415–1420. https://doi.org/10.1890/02-3154
Stein DB, Hutton C, Conant DS et al (2010) Reconstructing Dryopteris “semicristata” (Dryopteridaceae): molecular profiles of tetraploids verify their undiscovered diploid ancestor. Am J Bot 97:998–1004. https://doi.org/10.3732/ajb.0900355
Stephenson N (1998) Actual evapotranspiration and deficit: biologically meaningful correlates of vegetation distribution across spatial scales. J Biogeogr 25:855–870. https://doi.org/10.1046/j.1365-2699.1998.00233.x
Tammets T (2012) Eesti ilma riskid (in Estonian). Eesti Meteoroloogia ja Hüdroloogia Instituut, Tallinn http://www.ilmateenistus.ee/ilmatarkus/publikatsioonid/eesti-ilma-riskid/
Teskey R, Wertin T, Bauweraerts I et al (2015) Responses of tree species to heat waves and extreme heat events. Plant Cell Environ 38:1699–1712. https://doi.org/10.1111/pce.12417
Tessier JT (2001) Vernal photosynthesis and nutrient retranslocation in Dryopteris intermedia. Am Fern J 91:187–197. https://doi.org/10.1640/0002-8444(2001)091[0187:VPANRI]2.0.CO;2
Tessier JT (2014) Reduced winter snowfall damages the structure and function of wintergreen ferns. Am J Bot 101:965–969. https://doi.org/10.3732/ajb.1400181
Tessier JT (2018) Upright fronds of Dryopteris intermedia suffer frost damage and breakage during winter. Am Fern J 108:19–26. https://doi.org/10.1640/0002-8444-108.1.19
Tessier JT, Bornn MP (2007) Old fronds serve as a vernal carbon source in the wintergreen fern Dryopteris intermedia (Aspleniaceae). Am J Bot 94:25–28. https://doi.org/10.3732/ajb.94.1.25
Trivedi MR, Morecroft MD, Berry PM et al (2008) Potential effects of climate change on plant communities in three montane nature reserves in Scotland, UK. Biol Conserv 141:1665–1675. https://doi.org/10.1016/j.biocon.2008.04.008
Valdés A, Lenoir J, Gallet-Moron E et al (2015) The contribution of patch-scale conditions is greater than that of macroclimate in explaining local plant diversity in fragmented forests across Europe. Glob Ecol Biogeogr 24:1094–1105. https://doi.org/10.1111/geb.12345
Van Buskirk J, Edwards J (1995) Contribution of wintergreen leaves to early spring growth in the wood fern Dryopteris intermedia. Am Fern J 85:54–57. https://doi.org/10.2307/1547466
Van Couwenberghe R, Collet C, Lacombe E et al (2011) Abundance response of western European forest species along canopy openness and soil pH gradients. For Ecol and Manag 262:1483–1490. https://doi.org/10.1016/j.foreco.2011.06.049
Vanneste T, Govaert S, Spicher F et al (2020) Contrasting microclimates among hedgerows and woodlands across temperate Europe. Agric For Meteorol 281:107818. https://doi.org/10.1016/j.agrformet.2019.107818
von Arx G, Dobbertin M, Rebete M (2012) Spatio-temporal effects of forest canopy on understory microclimate in a long-term experiment in Switzerland. Agric For Meteorol 166-167:144–155. https://doi.org/10.1016/j.agrformet.2012.07.018
Wagner WH, Chen KL (1965) Abortion of spores and sporangia as a tool in the detection of Dryopteris hybrids. Am Fern J 55:9–29. https://doi.org/10.2307/1546429
Walker S (1955) Cytogenetic studies in the Dryopteris spinulosa complex - I. Watsonia 3:193–209
Wardlaw CW, Sharma DN (1963) Experimental and analytical studies of Pteridophytes: XL. Factors in the formation and distribution of sori in leptosporanglate ferns. Ann Bot 27:101–121. https://doi.org/10.1093/oxfordjournals.aob.a083826
Wasof S, Lenoir J, Gallet-Moron E et al (2013) Ecological niche shifts of understorey plants along a latitudinal gradient of temperate forests in north-western Europe. Glob Ecol Biogeogr 22:1130–1140. https://doi.org/10.1111/geb.12073
Woodward F (1996) Climate and plant distribution. Cambridge University Press, Cambridge
Wu C, Gough CM, Chen JM, Gonsamo A (2013) Evidence of autumn phenology control on annual net ecosystem productivity in two temperate deciduous forests. Ecol Eng 60:88–95. https://doi.org/10.1016/j.ecoleng.2013.07.019
Yoshie F, Yoshida S (1989) Wintering forms of perennial herbs in the cool temperate regions of Japan. Can J Bot 67:3563–3569. https://doi.org/10.1139/b89-438
Acknowledgments
We are very grateful to Ülle Jõgar and Elgi Brand for their help and support during the fieldwork. This project was supported by the Estonian Research Council projects IUT 20–31 and PRG-1223, the EU through the European Regional Development Fund (the Centre of Excellence, EcolChange), and the EU Horizon2020 funding (EFFECT). We are grateful to the Estonian Weather Service for the climate data and to Robert Szava-Kovats for the language editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 1167 kb)
Rights and permissions
About this article
Cite this article
Rünk, K., Zobel, K. & Liira, J. Long-term growth of three sympatric Dryopteris fern species shows the accumulation of climatic effects over 2 years because of organ preformation. Climatic Change 164, 16 (2021). https://doi.org/10.1007/s10584-021-02967-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s10584-021-02967-8