Abstract
Biopolymers that mimic the extracellular matrix are favourable in tissue engineering. However, the rapid degradation and the lack of mechanical and enzymatic stabilities of these biopolymers prompt researchers to composite different biopolymers. In this study, we aim to develop an acellular gelatin-chitosan-cellulose nanocrystal (GCCNC) scaffold as a potential wound dressing. The GCCNC mixture was homogenised via ultrasonication and the genipin crosslinking was performed by magnetic stirring. The mixture was then frozen at − 80 °C for 6 h and freeze-dried. The effects of different ratios of gelatin and chitosan with cellulose nanocrystals on the physiochemical properties, mechanical properties, and cellular biocompatibility were studied. Our results herein showed that G3C7CNC demonstrated a homogenous interconnected porous structure with a good porosity (67.37 ± 9.09%) and pore size (148.46 ± 48.68 µm), acceptable swelling ratio (1071.11 ± 140.26%), adequate water vapour transmission rate (315.59 ± 25.27 g/m2/day), low contact angle (70.21 ± 6.79°), and sufficient mechanical strength (modulus of 64.67 ± 12.42 MPa). The lower biodegradation rate in the G3C7CNC (0.06 ± 0.01 mg/hr) compared to G10CNC (0.48 ± 0.07 mg/hr) together with the absence of glass transition phenomenon indicated an increase in both enzymatic and thermal stabilities. Furthermore, G3C7CNC was non-cytotoxic and biocompatible with human epidermal keratinocytes (HEKs) and human dermal fibroblasts (HDFs). The presence of collagen type I and α-smooth muscle actin expression in HDFs, together with the expression of cytokeratin-14 in HEKs, demonstrated our scaffold’s ability to maintain normal skin physiological functions. Therefore, this study proposes that the fabricated GCCNC scaffold could serve as a potential acellular skin substitute in managing chronic wounds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the largest organ in the human body, the skin accounts for approximately 15% of the total body weight and serves as a protective barrier against all varieties of external environmental insults (Ashrafi et al. 2017). A disruption in the continuity of the skin structure and underlying soft tissues leads to wound formation in which the recovery follows by haemostasis, inflammatory, proliferative, and remodelling phases during wound healing (Cheah et al. 2021; Phang et al. 2021). Despite the availability of invasive skin grafting that carries the risk of graft failure and donor site infection (Guogien et al. 2018; Turissini et al. 2019), wound management remains a significant challenge in the twenty first century (Snyder et al. 2020). Furthermore, evidence that demonstrates the efficacy of these high-cost skin substitutes is still lacking (Jones et al. 2018). Therefore, an ideal skin scaffold should carry a 3D structure that mimics the natural skin’s extracellular matrix (ECM). Besides, the abilities to promote cell-biomaterial intercell adhesion and ECM deposition, exchange of nutrients and waste products while being non-cytotoxic and biocompatible with a proper biodegradation rate are also necessary for a skin scaffold to promote tissue regeneration (Lanza et al. 2007).
Collagen is the main component of the skin ECM. In addition to maintaining the skin’s structural integrity, collagen plays an important role in regulating the skin cells' attachment, migration, proliferation, and differentiation (Chowdhury et al. 2018; Phang et al. 2022). However, extracting pure collagen is tedious in the prevention of denaturation and necessitates extra precautions, which indirectly translates into high production costs (Rýglová et al. 2017). Although collagen is only weakly immunogenic, the inherent immunity towards bovine collagen poses a significant obstacle in future clinical applications. On the other hand, gelatin, a denatured form of collagen, is non-antigenic due to the absence of aromatic groups as gelatin is deficient in tyrosine and tryptophan with a very trace amount of phenylalanine. Other advantages of gelatin are its low cost, availability and processability, vast functional groups, and preservation of the Arg-Gly-Asp (RGD) motif, which are all crucial for cell attachment (Nandan et al. 1988; Su and Wang 2015). However, poor mechanical strength and the rapid degradation of gelatin present a challenging task to be solved (Afewerki et al. 2019).
Chitosan is a polysaccharide derived from the deacetylation of chitin. Chitin is typically found in the exoskeleton of crustaceans such as shrimps and crabs. The polycationic nature of chitosan allows it to interact with negatively-charged bacteria, thus causing cell wall disruption and intracellular leakage (Tamara et al. 2018). Besides their antimicrobial, analgesic, and haemostatic properties, chitosan has been demonstrated to be biodegradable, biocompatible, and non-cytotoxic towards human skin cells (Malette et al. 1983; Okamoto et al. 2002; Lan et al. 2015; Lu et al. 2016). Nonetheless, like gelatin, a scaffold made with pure chitosan is fragile to handle and has poor mechanical strength (Samimi Gharaie et al. 2018).
Nanocellulose is a cellulosic material that falls within the nanometre scale in at least one of the three dimensions (length, width, and diameter). Nanocellulose can be categorised into bacterial nanocellulose, cellulose nanocrystals (CNCs), and cellulose nanofibrils (CNFs) based on the source and preparation (Thomas et al. 2020). Among all, plant-derived CNCs are gaining more attention due to their outstanding rigidity post-removal of the amorphous region by acid hydrolysis, which can improve the mechanical strength of a scaffold while being non-animal derived (Rasli et al. 2017; Xue et al. 2017; Ilyas et al. 2018; Zinge and Kandasubramanian 2020). Furthermore, the numerous hydroxyl groups give CNCs their hydrophilic nature and these functional groups can be modified (Daud and Lee 2017). Unlike bacterial nanocellulose, which can stand its ground as a sole polymer in biomaterial fabrication, CNCs are preferable as lightweight reinforcing agents with their low density and high elastic modulus to enhance the mechanical, thermal, and barrier strength of other polymers (Chu et al. 2020; Peng et al. 2020). Furthermore, CNCs have been found to provide a sustained release of platelet lysate in a chitosan-cellulose nanocrystal hydrogel, which in turn enhances the proliferation of fibroblasts and wound closure in vitro (Bhatnagar et al. 2021).
Given the poor stability of pure biopolymers, compositing multiple biopolymers have been proposed as a better option during scaffold biofabrication. In skin tissue engineering, biopolymers are preferred as they mimic the ECM and hence promote wound healing (Park et al. 2010). A study on the dialdehyde chitosan-hyaluronic acid scaffolds found that the human skin cell viabilities and thermal plus mechanical properties were affected when the ratio of the polymers was manipulated (Grabska-Zielińska et al. 2021). The collagen–gelatin hydrogel was also found to demonstrate higher human apical papilla cell viability, adhesion, and spreading compared to pure collagen hydrogel (Leite et al. 2021). Meanwhile, a study on gelatin-chitosan scaffolds reported an increased porosity of the composite scaffold upon the addition of viscous chitosan (Han et al. 2014). It was also revealed that the addition of palm oil-derived nanocellulose increased the mechanical property and crystallinity of the final fabricated collagen-nanocellulose scaffolds (Ooi et al. 2020). Taken together, the combination of different polymers within a composite scaffold can confer additional benefits while eliminating their respective limitations.
There are numerous types of scaffolds available in tissue engineering including sponges, hydrogels, films, and electrospinning membranes. Sponges are well-known for their high porosity with high exudate absorption capacity, which makes them suitable to be administered in a highly exudative wound. In contrast, hydrogels are scaffolds with a highly hydrated polymeric network but are often criticised for their weak mechanical property and rapid degradation (Graça et al. 2020). Zhang et al. found that chondrocytes proliferated faster with increased collagen type I (COL-I) expression in the chitosan sponges compared to chitosan hydrogels. Furthermore, the chondrocytes displayed a spindle-shaped morphology in the sponges, which closely mimics the native environment compared to the spherical morphology observed in the hydrogels (Zhang et al. 2013).
In our current study, we aim to investigate the effect of different ratios of gelatin:chitosan in the physicochemical, mechanical, and cellular biocompatibility properties. In doing so, a porous scaffold based on the biopolymers and crosslinking agents via freeze-drying was fabricated as an acellular skin substitute for the application in highly exudative chronic wounds.
Materials and methods
This research was approved by the Universiti Kebangsaan Malaysia (UKM) Research Ethics Committee (JEP-2021–076) under FRGS/1/2020/SKK0/UKM/02/6. All studies were performed in controlled facilities under the ISO9001:2015 management system.
Source and preparation of the polymers
Gelatin was obtained from Nitta-Gelatin Ltd. (Osaka, Japan) whereby the gelatin was extracted from buffalo raw bone materials containing a low endotoxin unit of < 3000 to prevent immune rejection. Low molecular weight chitosan powder of 50–190 kDa with a deacetylation degree of 75–85% was purchased from Sigma Aldrich (Dublin, Ireland). Rice husk supplied by Bernas Malaysia Sdn. Bhd was used as the source of nanocellulose. The preparation of CNCs was done by the Faculty of Science and Technology, Universiti Kebangsaan Malaysia based on previous publications (Johar et al. 2012; Zahari et al. 2018). Rice husk was grounded to obtain a rice husk powder before it was treated with 4% w/v sodium hydroxide at a reflux temperature for 2 h to remove lignin and hemicellulose. Subsequently, the alkali-treated fibres were bleached with a combination of acetic acid, aqueous chlorite (1.7 wt%), and distilled water at reflux (using a silicon oil bath at 100–130 °C) for 4 h to further purify the cellulose. Then, acid hydrolysis was conducted by using 10.0 mol L−1 sulphuric acid at 50 °C for 40 min under continuous stirring. The resulting suspension was centrifuged for 10 min at 10 °C, dialysed against distilled water for several days until a constant pH in the range of 5–6 was achieved, and ultrasonicated to disentangle the nanocrystals. Finally, the final product was freeze-dried to obtain a white powder and stored at 4 °C until further use.
Fabrication of gelatin-chitosan-cellulose nanocrystal (GCCNC) scaffold
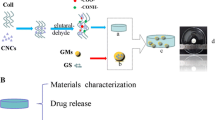
The schematic diagram of the fabrication of GCCNC is shown in Fig. 1. Gelatin stock (5% w/v) was prepared by dissolving gelatin in distilled water at 37 °C. Chitosan stock (2% w/v) was prepared by dissolving the low molecular weight chitosan in 1% v/v acetic acid (Merck, Darmstadt, Germany) at room temperature. The final volume was fixed at 10 ml with the ratio of gelatin:chitosan at 10:0, 7:3, 5:5, 3:7, and 0:10, and 50 mg of CNCs were added to all composites to obtain a final concentration of 0.5% w/v CNCs. The composite mixtures were then ultrasonicated at an amplitude of 50% for 5 min to homogenise the mixtures. Genipin powder (Fujifilm Wako Pure Chemical Corporation, Osaka, Japan) was dissolved using 70% ethanol (EtOH; MERCK, Darmstadt, Germany) before adding to the composite mixtures to achieve the final genipin concentration at 0.1% w/v and the mixtures were magnetically stirred at 300 rpm for 30 min. Subsequently, the mixtures were transferred to silicon moulds, frozen at -80 °C for 6 h, and then freeze-dried at 5 mTor and -55 °C for 24–48 h. A total of 3 batches of scaffolds (N = 3) were fabricated for all the downstream experimental studies to address the batch-to-batch variation, whereas 3 scaffolds within the same batch (n = 3) were used in each experiment as technical replicates.
Gross appearance
The gross appearance of the scaffolds was captured using a digital camera (Nikon, Tokyo, Japan).
3D microstructural analysis
The surface topography and cross-sectional microstructure of the scaffolds were studied using scanning electron microscopy (SEM) at 10 kV. The scaffolds were cut into the size of 1 cm × 1 cm and mounted on an aluminium stub using carbon tape. The scaffolds (N = 1) were coated with gold before viewing them under the SEM (LEO-1450 VP). The SEM images were acquired from 3 randomly selected points for each scaffold and a representative image was chosen for each experimental group.
The pore size distribution was measured using ImageJ software (Version 1.53; National Institutes of Health, Bethesda, MD, USA) by taking 50 pores randomly from the cross-sectional SEM images for each scaffold. The porosity test was done according to the liquid displacement method using absolute ethanol (EtOH; MERCK, Darmstadt, Germany). All scaffolds (N = 3, n = 3) were weighed pre and post-immersion in absolute ethanol for 18 h and the data were analysed using the formula below:
Water vapour transmission rate (WVTR)
The scaffolds (N = 3, n = 3) were cut into a disc-shaped of 1.1 cm in diameter and placed on the mouth of the glass vials containing 10 mL of distilled water. The glass vials containing scaffolds were then placed in an incubator at 37 °C and 5% CO2. Evaporation of water through the tested scaffold was monitored by weighing the vials at a specific time interval. The results were recorded and analysed using the formula below:
Water absorption capacity
The water absorption capacity of the scaffold was determined by immersing the scaffolds in Dulbecco’s Phosphate Buffered Saline (DPBS; Sigma-Aldrich, St. Louis, MO, USA) for 24 h at room temperature. The excess DPBS was removed using filter paper and the wet weight of the scaffold (N = 3, n = 3) was measured. The swelling ratio was calculated using the formula below:
Contact angle
The contact angle between the surface of the scaffold and the water droplet was measured to determine the surface wettability (Law 2014), which indicated its hydrophilicity. A total of 10 μL of distilled water was dropped on the surface of the scaffold (N = 3, n = 3) and the images were captured using a digital camera. The result was analysed using ImageJ software (Version 1.53; National Institutes of Health, Bethesda, MD, USA).
In vitro biodegradation rate
The initial weight (Wi) of the scaffold was measured and subsequently immersed in 0.0006% w/v of collagenase type I (Worthington, Columbus, OH, USA) at 37 °C for 24 h. Subsequently, the scaffold was rinsed with distilled water thrice followed by a freeze-drying process for 24 h. The weight of the scaffold (N = 3, n = 3) was then re-measured and the enzymatic biodegradation rate was calculated using the formula below:
Mechanical tensile properties
The mechanical strength of the scaffolds was carried out in a dry condition at room temperature using an Instron 8874 universal testing machine fitted with a 5 kN of load transducer at a crosshead velocity of 10 mm min−1. Three scaffolds of 3 cm2 from each group (N = 3, n = 3) were tested for evaluation on Young’s modulus and tensile strain (elongation at break).
Energy dispersive X-ray (EDX)
Energy Dispersive X-Ray (EDX) analysis was performed using a Phenom Pro X SEM_EDX microscope (Phenom, Eindhoven, The Netherlands) to analyse the elemental content on the surface of each scaffold together with their distribution (N = 1).
Fourier transform infrared spectroscopy (FTIR)
The chemical characterisation of the scaffolds was performed using Fourier Transform Infrared Spectroscopy (FTIR). One mm3 of each of the scaffolds (N = 1) was analysed, and the spectral data were recorded using a PE Spectrum 100 FTIR spectrometer (PE, Waltham, MA, USA) at a wavelength range of 700–4000 cm−1. The absorbance peaks were analysed to identify the chemical structure and changes resulting from the fabrication and crosslinking.
X-Ray diffraction study (XRD)
The X-Ray Diffraction (XRD) characterisation of the scaffold (N = 1) was performed using Bruker D8 Advance (Bruker, Hamburg, Germany) at room temperature with CuK radiation (λ = 0.154 3 nm) at 40 kV and 40 mA. A 2θ (degree) vs. intensity (a.u.) chart was used to present the data.
Thermogravimetric analysis (TGA)
Thermal stability evaluation was measured by thermogravimetric analysis (TGA), which allows the measurement of the mass change of a sample in a nitrogen-contained environment at a heating rate of 20 °C min−1 between 30 and 800 °C. The initial weight of each selected sample was 1–5 mg (N = 1).
Cell Isolation and culture
Redundant skin samples were obtained from consented healthy patients undergoing circumcision or abdominoplasty. A 3 cm2 skin sample was cleaned from unwanted fragments such as fat, hair, and debris before being minced into small pieces of approximately 1 mm2 each. Subsequently, the minced skin was digested with 10 ml of 0.6% collagenase type I (Worthington, Lakewood, NJ, USA) for 4 h in a 37 °C shaker incubator followed by cell dissociation using 10 ml of 0.05% trypsin–EDTA (Gibco, Carlsbad, CA, USA) for 10 min. The inhibition of cell dissociation was done by the addition of 10 ml human dermal fibroblast (HDF) growth medium; F12: Dulbecco’s Modified Eagle Medium (Gibco, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Biowest, Riverside, MO, USA). The digested and dissociated skin cell suspension containing both HEKs and HDFs was re-suspended in a co-culture medium at a 1:1 ratio (a mixture of HEKs growth medium; EpiLife® (Gibco/BRL, Grand Island, NY, USA), and HDFs growth medium) before seeding them into three wells (surface area of 9.6 cm2/well) of a 6-well culture plate (Greiner Bio-One, Monroe, NC, USA) and incubated at 37 °C with 5% CO2. The culture medium was changed every 2–3 days. When the cells reached 70–80% confluency, differential trypsinisation was performed using 0.05% Trypsin–EDTA for 5 min at 37 °C to dissociate the HDFs. The HDFs were collected and cultured in 75 cm2 flasks with F12: DMEM medium containing 10% FBS, whereas HEKs were cultured in 6-well plates with EpiLife®. A total of 3 batches of cells isolated from different donors (N = 3) were used in the experiments to overcome interindividual variation, whereas the cells from each donor were seeded onto 3 separate scaffolds (n = 3) during experimentation to address the technical variation. Both HEKs and HDFs at passages 2–3 were seeded on the scaffolds at a density of 6 × 104 cells cm−3 and 4 × 104 cells cm−3, respectively.
Cell attachment
For cell attachment analysis, the unattached cells in the culture medium were quantified at 24 h after seeding using a trypan blue exclusion test. The percentage of cell attachment on the scaffolds (N = 3, n = 3) was measured using the following equation:
Cell cytotoxicity and viability
The LIVE/DEAD® Cell Viability Assay (Life Technologies, Carlsbad, CA, USA) was used to analyse the cytotoxic and viability effects of the scaffolds on HEKs and HDFs qualitatively according to the manufacturer’s protocol. HEKs and HDFs (both N = 3, n = 3) cultured on the scaffolds were incubated with 2 mM calcein AM and 4 mM EthD-1 in DPBS for 30 min and washed with DPBS before observation using Nikon A1R-A1 confocal laser scanning microscopy (CLSM; Nikon, Tokyo, Japan).
The quantitative effect of cytotoxicity and viability of both HEKs and HDFs on the scaffolds was analysed using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay kit (Thermo Fisher Scientific, Waltham, MA, USA). The HEKs and HDFs were cultured on the scaffolds with a density of 6 × 104 cells cm−3 and 4 × 104 cells cm−3, respectively in 48 well plates. MTT assay (N = 3, n = 3) was performed according to the manufacturer’s recommendations on days 1, 3, and 7 to evaluate the viability. The culture medium was changed to 120 µL of fresh medium for each well with 12 µL of 12 mM MTT reagent (prepared with 1 mL of sterile DPBS to 5 mg of MTT) added to the wells and incubated for 4 h at 37 °C for the formation of formazan crystals. Subsequently, 90 µl of the media was removed with the incubation with 100 µl of DMSO (Sigma-Aldrich, St. Louis, MO, USA) to each well at 37 °C for 10 min. The absorbance was measured using a spectrophotometer at 540 nm wavelength.
Immunofluorescence imaging
After 1 day of seeding, the cell-seeded scaffolds were rinsed with DPBS before fixation with 4% paraformaldehyde (Sigma-Aldrich, USA) for 15 min followed by permeabilization with 0.5% Triton X-100 solution (Sigma-Aldrich) for 20 min at room temperature and then blocking with 10% normal goat serum (Agilent, USA) for 1 h at 37 °C. Subsequently, the cell-seeded scaffolds were incubated overnight at 4 °C with Rabbit Anti-Human Collagen I (Abcam, Cambridge, MA, USA) and Mouse Anti-Human alpha-smooth muscle actin (Abcam, Cambridge, MA, USA) for HDFs, whereas Rabbit Anti-human vinculin (Abcam, Cambridge, MA, USA) and Mouse Anti-Human Cytokeratin-14 (Abcam, Cambridge, MA, USA) for HEKs. Then, the cell-seeded scaffolds (N = 3, n = 3) were rinsed with DPBS followed by incubation in dark with the secondary antibodies of Anti-mouse Alexa Fluor® 488 (Abcam, Cambridge, MA, USA) and Anti-rabbit Alexa Fluor® 594 (Abcam, Cambridge, MA, USA) for 2 h at 37 °C and counterstained with 300 nM of DAPI (Sigma-Aldrich, USA) for 20 min at room temperature. Images were captured using CLSM (Nikon). All antibodies were prepared at a dilution factor of 1:1000.
Statistical analysis
All data were analysed using GraphPad Prism version 8.0 (GraphPad Software, Inc., San Diego, CA, USA). The data collected from various quantitative parameters were presented in the form of mean ± standard deviation (SD) of the sample size. One-way and two-way analysis of variance (ANOVA) was applied to compare the control with multiple groups. The result was expressed as mean and standard deviation. A significant difference was considered when the p value is < 0.05.
Results and discussion
Gross appearance
The gross appearance of the GCCNC scaffolds is illustrated in Fig. 2a. G10CNC and G7C3CNC appeared grey in colour, whereas G5C5CNC, G3C7CNC, and C10CNC appeared greenish-grey in colour despite the similar concentration of genipin used during crosslinking.
The effect of different ratios of gelatin: chitosan in the GCCNC scaffolds. a The gross appearance of the GCCNC scaffolds together with the SEM cross-sectional views and the mean pore size. b The pore size distribution. c Porosity. d WVTR. e Swelling ratio. f Contact angle. g In-vitro biodegradation rate. h Young’s modulus. i Tensile strain. The red scale bars represent 1 cm whereas the yellow scale bars represent 100 µm. N = 3, n = 3, * p < 0.05
3D microstructural analysis
A porous structure is crucial in biomaterial design for the exchange of nutrients and toxic by-products in addition to the cellular attachment, migration, and proliferation properties (Lanza et al. 2007). The pore characteristics such as the size, porosity, and interconnectivity have been shown to influence water absorption capacity, WVTR, enzymatic, thermal, and mechanical stabilities as well as cellular attachment and biocompatibility. The microstructural cross-sectional views are shown in Fig. 2a. In this study, a porous skin scaffold was produced using freeze-drying. Freeze-drying is a well-established and simple method, which applies the principle of rapidly freezing liquid into an ice form and sublimating the ice crystals away leaving behind the porous solid structure (Collins et al. 2021).
Generally, G10CNC showed round-shaped pores, whereas C10CNC showed columnar porous structures. Meanwhile, G7C3CNC and G5C5CNC showed similar round-shaped pores as G10CNC, whereas G3C7CNC showed an intermediate feature between G10CNC and C10CNC with columnar pores and smaller pore size compared to the others (Alizadeh et al. 2013; Lu et al. 2016). The microscopic top views (Fig. 3) revealed that the surface of all the scaffolds was rough. However, G10CNC, G7C3CNC, and G5C5CNC showed porous surfaces as compared to the G3C7CNC and C10CNC.
In terms of pore size distribution, G3C7CNC and C10CNC showed the highest average pore size distribution between 100–199 µm when compared to the other three groups (Fig. 2b). There were no pores bigger than 300 µm. The mean pore size for G3C7CNC and C10CNC was 148.46 ± 48.68 µm and 149.73 ± 68.86 µm, respectively which were smaller compared to G10CNC, G7C3CNC, and G5C5CNC with mean values of 218.50 ± 74.45 µm, 213.73 ± 82.94 µm and 230.83 ± 80.36 µm, respectively. All scaffolds achieved a pore size within the range of 148–230 µm, which is suitable for skin cell proliferation (Loh and Choong 2013; Wang et al. 2013).
The porosity of the GCCNC scaffolds was measured by the ethanol displacement method (Fig. 2c). G10CNC exhibited the lowest porosity of 52.81 ± 13.99% followed by G7C3CNC (55.05 ± 14.51%), G5C5CNC (61.66 ± 11.08%), G3C7CNC (67.37 ± 9.09%), and C10CNC (78.42 ± 9.49%). The results suggested that the porosities of the GCCNC scaffolds were proportional to the increasing concentration of chitosan content (Lan et al. 2015).
Water vapour transmission rate (WVTR)
The ability to transmit vapourised gas from the wound so that the wound exudate is neither overflooded nor dehydrated is known as WVTR (Fig. 2d). Given that only porous structure can transmit vapour, WVTR is important in skin tissue engineering. An ideal wound dressing should be able to absorb wound exudate to maintain a moist microenvironment for wound healing and allow vapour transmission to avoid excessive exudate accumulation, thus sequestering microbial away from the wound and promoting wound healing (Wu et al. 2004; Wang et al. 2018; Singh et al. 2022). G10CNC showed WVTR of 305.91 ± 50.75 g/m2/day followed by G7C3CNC (307.55 ± 37.25 g/m2/day), G5C5CNC (312.53 ± 27.88 g/m2/day), G3C7CNC (315.59 ± 25.27 g/m2/day), and the highest WVTR was demonstrated by C10CNC (357.69 ± 36.48 g/m2/day). The WVTR value in C10CNC showed to be significantly higher compared to G10CNC and G7C7CNC. Taken together, our results suggest that higher chitosan concentration gives higher WVTR in the GCCNC scaffolds. Normal skin has a WVTR of 204 g/m2/day, whereas injured skin records a WVTR of 279 g/m2/day up to 5138 g/m2/day in the granulating wound (Wu et al. 2004). In this current study, all scaffolds demonstrated WVTR of above 300 g/m2/day, which is an acceptable rate as a wound dressing and is slightly better than the pure gelatin sponge with a WVTR of 247 g/m2/day (Arif et al. 2020).
Water absorption capacity
As a porous skin substitute, the capability of absorbing wound exudate is crucial to create a moist wound microenvironment to promote wound healing and sequester microbial away from the wound (Singh et al. 2022). Therefore, the skin substitute has to be hydrophilic. The water absorption capacity represents the capability of a scaffold to absorb wound exudate (Fig. 2e). The swelling ratios in G10CNC, G7C3CNC, G5C5CNC, G3C7CNC, and C10CNC were 1096.72 ± 121.12%, 1105.79 ± 93.70%, 1123.85 ± 81.06%, 1071.11 ± 140.26% and 1290.09 ± 181.64%, respectively. The swelling ratio of C10CNC was significantly higher than G10CNC, G7C53CNC, and G3C7CNC. No significant difference was observed among GCCNC composite scaffolds and all of them achieved a swelling ratio of above 1000% despite being crosslinked (Salleh et al. 2022; Sallehuddin et al. 2022).
Contact angle
Surface wettability is another parameter to determine the hydrophilicity of a scaffold to absorb exudate from the wound surface (Law 2014). A contact angle of less than 90° indicates a hydrophilic scaffold and vice versa. The results showed that all scaffolds demonstrated contact angles lower than 90° (Fig. 2f). The contact angles of G10CNC, G7C3CNC, G5C5CNC, G3C7CNC, and C10CNC were 69.35 ± 4.80°, 71.46 ± 3.56°, 72.60 ± 3.89°, 70.21 ± 6.79° and 65.92 ± 4.20°, respectively. A significant difference in the contact angle (p < 0.05) was only observed between C10CNC and G5C5CNC scaffolds. These results suggest that all GCCNC scaffolds were hydrophilic with high surface wettability regardless of the concentration and the ratio of gelatin:chitosan.
In vitro biodegradation rate
As skin substitutes shall shield a wound and support cell attachment, proliferation, and differentiation until complete recovery of the wound is achieved, the degradation rate of a scaffold must not be too rapid in addition to an optimal mechanical property while absorbing and transmitting wound exudate. Wound exudates contain matrix metalloproteinase (MMP) regardless of being acute or chronic (Caley et al. 2015). For example, Collagenase type I is a type of MMP (MMP- I) that is present underlying a wound. Hence, the in vitro biodegradation rate of a scaffold can be evaluated experimentally with Collagenase type I to mimic the enzymatic biodegradation (Fig. 2g).
G10CNC degraded significantly with the fastest rate of 0.48 ± 0.07 mg/hr followed by G7C3CNC (0.10 ± 0.03 mg/hr), G5C5CNC (0.09 ± 0.04 mg/hr), G3C7CNC (0.06 ± 0.01 mg/hr), and C10CNC (0.05 ± 0.03 mg/hr). The degradation of the GCCNC scaffold was likely to be contributed by the gelatin degradation, which is consistent with a previous study (Han et al. 2014). Moreover, the incorporation of chitosan significantly reduced the biodegradation rate in the GCCNC scaffolds, which improves the resistance of the scaffold against enzymatic degradation. As compared to a study on collagen sponges with a biodegradation rate of 10 mg/h, which supported the complete wound closure in an in vivo model (Mh Busra et al. 2019), GCCNC scaffolds were suggested to be enzymatically stable as a skin substitute.
Mechanical tensile properties
The mechanical property of a scaffold is crucial during the handling and implantation of the wound. Young’s modulus is the elasticity modulus of a material in relation to tension and represents the linear slope of a stress–strain graph (Fig. 2g). In this study, G10CNC demonstrated the highest Young’s modulus of 310.70 ± 104.60 MPa and the lowest value was observed in C10CNC with 46.00 ± 5.29 MPa, whereas intermediate readings were seen in G7C3CNC (96.00 ± 70.49 MPa), G5C5CNC (62.67 ± 4.16 MPa), and G3C7CNC (64.67 ± 12.42 MPa). A skin scaffold is expected to mimic the strength of the human skin, which falls between 5 kPa to 140 MPa (Kalra and Lowe 2016) and 98.97 MPa (Gallagher et al. 2012). The GCCNC scaffolds in our study showed to have a suitable Young’s modulus that ranges between 62.67 MPa to 96 MPa with the gelatin-predominant scaffold being mechanically stronger than the chitosan-predominant scaffold.
Meanwhile, the tensile strain represents the ratio of elongation in the length of material before the fracture of the material (Fig. 2h). The highest tensile strain was seen in G5C5CNC (5.564 ± 0.601%) followed by G7C3CNC (5.105 ± 0.590%), G10CNC (4.239 ± 0.418%), C10CNC (3.348 ± 0.9357%), and G3C7CNC (3.012 ± 0.601%). The reducing trend in the mechanical strength of the scaffolds was observed when chitosan concentration was increased in our GCCNC scaffolds. This could be explained by the increasing porosity of the scaffolds, which may contribute to the weakened mechanical strength (Su et al. 2020).
Besides, this study also observed that a reduced mechanical strength was related to the increased tensile strain from G10CNC to G7C3CNC and G5C5CNC (Zhu et al. 2019). However, G3C7CNC and C10CNC did not follow the trend as their mechanical strength was further reduced. This could be due to the different porous structures in the G3C7CNC and C10CNC, which were columnar compared to the round-shaped pores in the other three scaffolds.
Energy dispersive X-ray spectrometry (EDX)
EDX mapping and spectrum (Fig. 3 and Table 1) demonstrated that carbon (C) was the main element among all of the scaffolds followed by oxygen (O) and nitrogen (N). Homogenous distribution over the three elements was seen in the mappings with no significant difference among the scaffolds.
Fourier transform InfraRed (FTIR)
The FTIR spectra were obtained from the gelatin, chitosan, CNCs, and the composite scaffolds (Fig. 4a). Gelatin showed characteristic absorption peaks at 3273 cm−1 due to amide A (NH stretching), 2936 cm−1 due to amide B (CH2 asymmetrical stretching), 1630 cm−1 due to amide I (NH bending), 1530 cm−1 due to amide II (CN stretching), and 1236 cm−1 due to amide III. Chitosan displayed characteristic peaks at 3299 cm−1 due to the overlapping of OH and NH2 stretching vibration, 2873 cm−1 due to CH and CH2 stretching, 1648 cm−1 due to amide I, 1566 cm−1 due to amide II, 1228 cm−1 due to amide III, 1054 cm−1 due to CO stretching vibration in a glycosidic linkage, and 895 cm−1 due to COC stretching in the glycosidic linkage. Meanwhile, CNCs are similar to chitosan with the only difference being the OH group in the C2 position instead of NH2, shared almost similar FTIR peaks with chitosan. FTIR peaks due to amino groups are absent in the CNCs, but peaks representing glycosidic linkage similar to chitosan will be present. Here, CNCs showed characteristic peaks at 3335 cm−1 due to OH stretching, 2900 cm−1 due to CH symmetrical, and CH2 stretching together with 1055 cm−1 and 897 cm−1 representing stretching of CO and COC in a glycosidic linkage, respectively.
The preservation of functional groups from the raw polymers was seen in all GCCNC scaffolds. This finding indicates the successful incorporation of the desired materials into the composite scaffold. G10CNC demonstrated two peaks at 1072 cm−1 and 897 cm−1 that were absent in gelatin. This hence supports that CO and COC glycosidic linkages in the pyranose ring of CNCs were successfully incorporated. All GCCNC scaffolds were shown to preserve the amides A, B, I, II, and III from the gelatin and chitosan together with the CO and COC glycosidic linkages in the pyranose ring of both polysaccharides named chitosan and CNCs.
Furthermore, all peak shifting observed in the GCCNC scaffolds when compared to their respective raw polymers indicated the presence of functional group interactions between different polymers during crosslinking polymers (Bhatnagar et al. 2021). The shifting of peaks to a higher wavelength number implied an increase in the bond energy. Evidently, the peak of COC glycosidic linkage of chitosan (895 cm−1) and CNCs (897 cm−1) was shifted to 899 cm−1 in both G5C5CNC and G3C7CNC. On the other hand, COC glycosidic linkage in the chitosan shifted from 895 cm−1 to a lower wavelength of 892 cm−1 in the C10CNC post-crosslinking with genipin. Hence, gelatin, CNCs, and chitosan were not superimposed onto each other but formed stable hydrogen bonds among them due to the genipin crosslinking (Lan et al. 2015).
X-ray diffraction study (XRD)
XRD is an analytical test used to determine the crystallographic structure of a material. The intensity peaks of the gelatin, chitosan, and CNCs are shown in Fig. 4b. Gelatin was the most amorphous material, followed by chitosan and rice husk-derived CNCs. CNCs displayed a major peak at 2θ = 22.4° and a minor peak at 2θ = 15.5°, whereas chitosan displayed a major intensity peak at 2θ = 20° and a minor peak at 2θ = 9.3°. Gelatin showed only a broad peak at 2θ = 21.1°.
All scaffolds displayed their major intensity peak at 2θ = 21.5° with a minor intensity peak at 2θ = 8.0°. G10CNC showed a higher intensity compared to gelatin due to the successful incorporation of highly-crystalline CNCs (Ooi et al. 2020). However, the intensity in semi-crystalline chitosan was lowered despite the addition of the same amount of the highly-crystalline CNCs in C10CNC. This interesting finding in C10CNC was due to the effect of genipin crosslinking, which alters the semi-crystalline structure of chitosan through the repositioning of the hydrogen bonds. Hence, C10CNC was found to be more amorphous than pure chitosan scaffold despite the successful incorporation of highly-crystalline CNCs (Zhu et al. 2019; Whitehead et al. 2020). Nevertheless, all GCCNC scaffolds were shown to be amorphous with G3C7CNC being the most amorphous scaffold Hence, the results herein demonstrated that all GCCNC scaffolds are suitable for skin tissue engineering.
Thermogravimetric analysis (TGA)
TGA is a study of polymeric materials including composites and it is widely used in the pharmaceutical field (Ng et al. 2018). TGA mainly shows the weight changes of a material in response to the increasing temperature in an inert (nitrogen gas) or oxidative (oxygen gas) environment, whereas DTG shows the rate of weight loss versus temperature that characterises transitions such as glass transition (Saadatkhah et al. 2020). All scaffolds showed a three-step weight loss during TGA (Fig. 4c). The first stage of TGA took place due to the loss of moisture absorbed by the scaffold via evaporation at a temperature below 100 °C, which was represented by the first peak in the DTG. This peak is a direct indicator of the hydrophilicity of the scaffolds and is consistent with the swelling ratio and contact angle that have been studied. All scaffolds displayed a similar trend in this stage.
The second stage of weight loss in TGA occurred from 100–500 °C, which is mainly due to the depolymerisation and decomposition of polymers in the scaffolds. All scaffolds demonstrated a second weight loss of around 300 °C except C10CNC. C10CNC showed two weight losses during the second stage in which 14.1% and 74.1% of the weight were lost at the Tmax of 159.1 °C and 303.6 °C, respectively (Fig. 4d and Table 2). The unique extra weight loss of C10CNC at Tmax of 159.1 °C was found to emerge around 100 °C before decomposition and depolymerisation of the scaffold (Tmax = 303.6 °C), which was attributed to the glass transition phenomenon. A study on chitosan film reported a glass transition temperature of 152 °C (Aesa and Walton 2018) while another study reported a 140–150 °C of glass transition temperature on chitosan film using four thermal analysis techniques (Dong et al. 2004), which both were similar to our finding. The glass transition phenomenon is a reversible event in which a solid semi-crystalline material such as chitosan is transformed into a liquid phase in the presence of increasing temperature. It is unfavourable in skin tissue engineering as the 3D porous structure of a scaffold might be damaged thus compromising the mechanical property and making the scaffold unable to support cellular activity (Qian et al. 2019). In our study, C10CNC reported a glass transition phenomenon but the incorporation of gelatin in the other GCCNC scaffolds stabilised the thermal property with the absence of the glass transition phenomenon. Although temperatures above 100 °C are unlikely to happen in the human body, however, there is a risk of this event occurring during the logistic and storage of the scaffold.
The last stage (500–800 °C) in the TGA was a slow reduction contributed by the decomposition of the residual mass. The residual mass for G10CNC, G7C3CNC, G5C5CNC, G3C7CNC, and C10CNC were 19.0%, 21.8%, 27.6%, 25.1% and 6.9%, respectively.
Cell attachment, cytotoxicity, and viability
The safety of a wound dressing is a prerequisite towards designing a successful wound dressing to ensure that cells underlying the skin can survive upon scaffold incorporation. The results herein demonstrated that all scaffolds were non-cytotoxic as both HEKs (Fig. 5) and HDFs (Fig. 6) remained viable in all of the scaffolds, which were represented by the green coloured-cells observed using the LIVE/DEAD® Cell Viability Assay. HDFs showed their typical spindle-shaped morphology only on G5C5CNC, G3C7CNC, and C10CNC but not on G10CNC and G7C3CNC as the HDFs appeared to be round in shape. Given the high porosity and pore size of the scaffold compared to HEKs and HDFs, the dead cells were probably detached and washed away during the washing step of the LIVE/DEAD® Cell Viability Assay.
Regarding HDF attachment in the scaffolds, G3C7CNC was demonstrated to possess the highest attachment (89.31 ± 5.58%) followed by G5C5CNC (88.35 ± 8.01%), C10CNC (88.24 ± 5.37%), G7C3CNC (87.43 ± 6.26%), and G10CNC (79.83 ± 4.16%). Moreover, G10CNC exhibited a significantly lowest attachment compared to the other scaffolds. Meanwhile, the attachment for HEKs on the scaffold was topped by C10CNC (96.34 ± 3.36%) followed by G3C7CNC (95.89 ± 2.86%), G5C5CNC (90.48 ± 1.62%), G7C3CNC (89.47 ± 4.18%), and G10CNC (83.83 ± 4.95%). There was no significant difference between C10CNC and G3C7CNC in the HEK attachment. Overall, all scaffolds showed good attachment for HDFs and HEKs. The incorporation of chitosan significantly increased the attachment of both HEKs and HDFs and was linked to the increased enzymatic and mechanical stability in the GCCNC compared to G10CNC, which is similar to previous studies (Arif et al. 2020; Loh et al. 2020). However, G3C7CNC and C10CNC showed a higher attachment compared to G7C3CNC and G5C5CNC (Fig. 5f), which might be attributed to the different surface morphology (Fig. 3). Surface roughness is well-known as a crucial factor for cellular attachment (Loh et al. 2018b). Nevertheless, this study showed that appropriate surface porous architecture could affect the attachment other than surface roughness. The spindle-shaped morphology of HDFs appeared in G5C5CNC, G3C7CNC, and C10CNC (Fig. 6) was an indicator of the extension of lamellipodium or filopodium in the migrating cells, which further supported compatibility of the scaffolds (Loh et al. 2018a).
Regarding the proliferation and viability of both HEKs and HDFs, MTT was performed to assess the metabolic activity of the seeded cells on the scaffolds. The MTT absorbance was shown to be increased from day 1 to day 7 for both HEKs (Fig. 7a) and HDFs (Fig. 7b), thus revealing that the GCCNC scaffolds were not cytotoxic and supported the growth of both HEKs and HDFs cells.
Immunofluorescence
Immunofluorescence staining of the HEKs-seeded scaffolds showed the presence of CK-14 expression in all GCCNC scaffolds regardless of the gelatin:chitosan ratio (Fig. 8). However, vinculin was not visible in all GCCNC scaffolds. On the other hand, the expression of the α-SMA was present in all GCCNC scaffolds, whereas COL-I was only observed in G7C3CNC and C10CNC (Fig. 9).
The expression of COL-I suggested that G3C7CNC and C10CNC were conducive for HDFs as COL-I represents the predominant protein in the ECM of the skin and it is needed in the fibroplasia for wound granulation (Chowdhury et al. 2018).
On the other hand, CK-14 (Fig. 8) and α-SMA (Fig. 9) were expressed in all scaffolds seeded with HEKs and HDFs, respectively. CK-14 is a marker of proliferative potential in basal keratinocytes (Purkis et al. 1990; Manira et al. 2014), whereas α-SMA is a hallmark of myofibroblasts in wound contracture and closure (Hinz 2016; Tai et al. 2021). All scaffolds showed similar expression of CK-14 and α-SMA, thus suggesting that these scaffolds can support the proliferative phase in wound healing.
The expression of vinculin, a tiny actin-binding protein that is involved in integrin-mediated cell–matrix focal adhesion (Wang et al. 2021), was, unfortunately, unable to be appreciated in any of the GCCNC scaffolds. Although studies have described vinculin as a marker of adherence between cell–cell and cell–matrix, a downregulation of vinculin implies higher cell motility and favour re-epithelisation (Kirfel and Herzog 2004; Ziegler et al. 2006; Koivisto et al. 2012). Nevertheless, incubation duration is another factor that influences the level of vinculin expression in a biomaterial scaffold. Our scaffolds were stained with vinculin after one day of culture, which was relatively short compared to other groups such as Veleirinho et al. and Jin et al. in which vinculin expression was only determined after 7 and 9 days of cell culture (Veleirinho et al. 2012; Jin et al. 2013). Therefore, we propose that focal adhesion might require a longer duration to occur as the HEKs could be still in the proliferating (presence of CK-14 expression) and migrating state after 1 day of culture, which could explain the absence of vinculin expression in all our scaffolds. Nevertheless, scaffold autofluorescence was noted in our study and it was also reported in other studies (Park et al. 2009; Arya et al. 2012; Nair et al. 2015; Ahadian et al. 2017; Akilbekova et al. 2018). Hence, the autofluorescence issue remains a significant challenge in tissue engineering.
All the parameters performed in this study were tabulated by providing a ranking for each of the scaffolds (Table 3). Besides, the overall mechanism of wound healing related to GCCNC scaffold is also illustrated in Fig. 10. In summary, C10CNC has shown to be the best candidate in terms of physicochemical, mechanical, and cellular biocompatibility with excellent expression of COL-I (Fig. 8). Unfortunately, the glass transition phenomenon in the TGA/DTG (Fig. 3c and 3d) hinders the use of C10CNC in tissue engineering. On the other hand, G3C7CNC showed optimal and stable characteristics in all studied parameters. Hence, we propose G3C7CNC as a potential candidate to serve as an acellular skin scaffold towards high exudative chronic wound management.
Proposed mechanism of GCCNC scaffold in maintaining the normal skin physiological function. The porous GCCNC scaffold absorbs wound exudate and maintains a moist wound microenvironment. The porous microstructure of the GCCNC scaffold promotes the migration, proliferation, and differentiation of human skin cells, which is evidenced by the expression of CK-14 in HEKs, as well as COL-I and α-SMA in HDFs. Once the wound is healed, the GCCNC scaffold will degrade into non-cytotoxic byproducts
Conclusion and future perspectives
GCCNC scaffolds were successfully fabricated with biopolymers into a potential acellular skin substitute. Compositing different biopolymers and their ratio affect the properties of the scaffolds and can overcome the shortcomings of the individual biopolymers. In this study, G3C7CNC was identified as the best model with its amorphous and interconnected porous structures. The good enzymatic and thermal stability and the absence of glass transition phenomenon with sufficient mechanical strength were observed. The excellent cell attachment, non-cytotoxicity, and biocompatibility toward human primary skin cells with the expression of CK-14, COL-I, and α-SMA further supported the potential of G3C7CNC in wound re-epithelisation and fibroplasia. Taken together, we suggest G3C7CNC be potentially used as an acellular skin substitute for chronic wound management. Nevertheless, in vivo animal studies are required in the future to further validate the antibacterial and wound healing properties of the GCCNC scaffold in a complex wound microenvironment of the fabricated scaffold.
References
Aesa AA, Walton CD (2018) 193 nm ArF laser ablation and patterning of chitosan thin films. Appl Phys A Mater Sci Process 124:1–10. https://doi.org/10.1007/s00339-018-1859-z
Afewerki S, Sheikhi A, Kannan S et al (2019) Gelatin-polysaccharide composite scaffolds for 3D cell culture and tissue engineering: towards natural therapeutics. Bioeng Trans Med. https://doi.org/10.1002/btm2.10124
Ahadian S, Davenport Huyer L, Estili M et al (2017) Moldable elastomeric polyester-carbon nanotube scaffolds for cardiac tissue engineering. Acta Biomater 52:81–91. https://doi.org/10.1016/j.actbio.2016.12.009
Akilbekova D, Shaimerdenova M, Adilov S, Berillo D (2018) Biocompatible scaffolds based on natural polymers for regenerative medicine. Int J Biol Macromol 114:324–333. https://doi.org/10.1016/j.ijbiomac.2018.03.116
Alizadeh M, Abbasi F, Khoshfetrat AB, Ghaleh H (2013) Microstructure and characteristic properties of gelatin/chitosan scaffold prepared by a combined freeze-drying/leaching method. Mater Sci Eng C 33:3958–3967. https://doi.org/10.1016/j.msec.2013.05.039
Arif MMA, Fauzi MB, Nordin A et al (2020) Fabrication of bio-based gelatin sponge for potential use as a functional acellular skin substitute. Polymers (basel) 12:1–19. https://doi.org/10.3390/polym12112678
Arya N, Sardana V, Saxena M et al (2012) Recapitulating tumour microenvironment in chitosan-gelatin three-dimensional scaffolds: an improved in vitro tumour model. J R Soc Interface 9:3288–3302. https://doi.org/10.1098/rsif.2012.0564
Ashrafi M, Alonso-Rasgado T, Baguneid M, Bayat A (2017) The efficacy of electrical stimulation in lower extremity cutaneous wound healing: a systematic review. Exp Dermatol 26:171–178. https://doi.org/10.1111/exd.13179
Bhatnagar P, Law JX, Ng S-F (2021) Chitosan reinforced with Kenaf nanocrystalline cellulose as an effective carrier for the delivery of platelet lysate in the acceleration of wound healing. Polymers (basel) 13:4392. https://doi.org/10.3390/polym13244392
Bin PS, Lih E, Park KS et al (2017) Biopolymer-based functional composites for medical applications. Prog Polym Sci 68:77–105. https://doi.org/10.1016/j.progpolymsci.2016.12.003
Caley MP, Martins VLC, O’Toole EA (2015) Metalloproteinases and wound healing. Adv Wound Care 4:225–234. https://doi.org/10.1089/wound.2014.0581
Cheah YJ, Buyong MR, Mohd Yunus MH (2021) Wound healing with electrical stimulation technologies: a review. Polymers (basel) 13:3790. https://doi.org/10.3390/polym13213790
Chowdhury SR, Mh Busra MF, Lokanathan Y et al (2018) Collagen type I: a versatile biomaterial. Nov Biomater Regen Med 1077:389–414. https://doi.org/10.1007/978-981-13-0947-2
Chu Y, Sun Y, Wu W, Xiao H (2020) Dispersion properties of nanocellulose: a review. Carbohydr Polym 250:116892. https://doi.org/10.1016/j.carbpol.2020.116892
Collins MN, Ren G, Young K et al (2021) Scaffold fabrication technologies and structure/function properties in bone tissue engineering. Adv Funct Mater 31:2010609. https://doi.org/10.1002/adfm.202010609
Daud JB, Lee K-Y (2017) Surface modification of nanocellulose. Handb Nanocell Cellul Nanocompos. https://doi.org/10.1002/9783527689972.ch3
Dong Y, Ruan Y, Wang H et al (2004) Studies on glass transition temperature of chitosan with four techniques. J Appl Polym Sci 93:1553–1558. https://doi.org/10.1002/app.20630
Gallagher AJ, Anniadh AN, Bruyere K et al (2012) Dynamic tensile properties of human skin. Dyn Tensile Properties Human Skin. 5:1–6
Grabska-Zielińska S, Sosik A, Małkowska A et al (2021) The characterization of scaffolds based on dialdehyde chitosan/hyaluronic acid. Materials (basel) 14:1–10. https://doi.org/10.3390/ma14174993
Graça MFP, Miguel SP, Cabral CSD, Correia IJ (2020) Hyaluronic acid—based wound dressings: a review. Carbohydr Polym 241:116364. https://doi.org/10.1016/j.carbpol.2020.116364
Guogien I, Kievi M, Varkalys K et al (2018) Split-thickness skin grafting using grafts of different thickness. Eur J Plast Surg 41:583–590. https://doi.org/10.1007/s00238-018-1424-1
Han F, Dong Y, Su Z et al (2014) Preparation, characteristics and assessment of a novel gelatin-chitosan sponge scaffold as skin tissue engineering material. Int J Pharm 476:124–133. https://doi.org/10.1016/j.ijpharm.2014.09.036
Hinz B (2016) The role of myofibroblasts in wound healing. Curr Res Transl Med 64:171–177. https://doi.org/10.1016/j.retram.2016.09.003
Ilyas RA, Sapuan SM, Ishak MR, Zainudin ES (2018) Development and characterization of sugar palm nanocrystalline cellulose reinforced sugar palm starch bionanocomposites. Carbohydr Polym 202:186–202. https://doi.org/10.1016/j.carbpol.2018.09.002
Jin G, Prabhakaran MP, Kai D et al (2013) Tissue engineered plant extracts as nanofibrous wound dressing. Biomaterials 34:724–734. https://doi.org/10.1016/j.biomaterials.2012.10.026
Johar N, Ahmad I, Dufresne A (2012) Extraction, preparation and characterization of cellulose fibres and nanocrystals from rice husk. Ind Crops Prod 37:93–99. https://doi.org/10.1016/j.indcrop.2011.12.016
Jones RE, Foster DS, Longaker MT (2018) Management of chronic wounds—2018. JAMA 320:1481–1482. https://doi.org/10.1001/jama.2018.12426
Kalra A, Lowe A (2016) Mechanical behaviour of skin: a review. J Mater Sci Eng 5:1000254. https://doi.org/10.4172/2169-0022.1000254
Kirfel G, Herzog V (2004) Migration of epidermal keratinocytes: Mechanisms, regulation, and biological significance. Protoplasma 223:67–78. https://doi.org/10.1007/s00709-003-0031-5
Koivisto L, Häkkinen L, Larjava H (2011) Re-epithelialization of wounds. Endodontic Topics 24(1):59–93
Lan G, Lu B, Wang T et al (2015) Chitosan/gelatin composite sponge is an absorbable surgical hemostatic agent. Colloids Surfaces B Biointerfaces 136:1026–1034. https://doi.org/10.1016/j.colsurfb.2015.10.039
Lanza R, Langer R, Vacanti J (2007) Principles of tissue engineering
Law KY (2014) Definitions for hydrophilicity, hydrophobicity, and superhydrophobicity: getting the basics right. J Phys Chem Lett 5:686–688
Leite ML, Soares DG, Anovazzi G et al (2021) Fibronectin-loaded Collagen/Gelatin hydrogel is a potent signaling biomaterial for dental pulp regeneration. J Endod 47:1110–1117. https://doi.org/10.1016/j.joen.2021.04.009
Loh QL, Choong C (2013) Three-dimensional scaffolds for tissue engineering applications: role of porosity and pore size. Tissue Eng Part B Rev 19:485–502. https://doi.org/10.1089/ten.teb.2012.0437
Loh EYX, Fauzi MB, Ng MH et al (2018a) Cellular and molecular interaction of human dermal fibroblasts with bacterial nanocellulose composite hydrogel for tissue regeneration. ACS Appl Mater Interfaces 10:39532–39543. https://doi.org/10.1021/acsami.8b16645
Loh EYX, Mohamad N, Fauzi MB et al (2018b) Development of a bacterial cellulose-based hydrogel cell carrier containing keratinocytes and fibroblasts for full-thickness wound healing. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-21174-7
Loh EYX, Fauzi MB, Ng MH et al (2020) Insight into delivery of dermal fibroblast by non-biodegradable bacterial nanocellulose composite hydrogel on wound healing. Int J Biol Macromol 159:497–509. https://doi.org/10.1016/j.ijbiomac.2020.05.011
Lu B, Wang T, Li Z et al (2016) Healing of skin wounds with a chitosan-gelatin sponge loaded with tannins and platelet-rich plasma. Int J Biol Macromol 82:884–891. https://doi.org/10.1016/j.ijbiomac.2015.11.009
Malette WG, Quigley HJ, Gaines RD et al (1983) Chitosan: a new hemostatic. Ann Thorac Surg 36:55–58. https://doi.org/10.1016/S0003-4975(10)60649-2
Manira M, Khairul Anuar K, Seet WT et al (2014) Comparison of the effects between animal-derived trypsin and recombinant trypsin on human skin cells proliferation, gene and protein expression. Cell Tissue Bank 15:41–49. https://doi.org/10.1007/s10561-013-9368-y
Mh Busra F, Rajab NF, Tabata Y et al (2019) Rapid treatment of full-thickness skin loss using ovine tendon collagen type I scaffold with skin cells. J Tissue Eng Regen Med 13:874–891. https://doi.org/10.1002/term.2842
Nair BP, Gangadharan D, Mohan N et al (2015) Hybrid scaffold bearing polymer-siloxane Schiff base linkage for bone tissue engineering. Mater Sci Eng C 52:333–342. https://doi.org/10.1016/j.msec.2015.03.040
Nandan D, Cates GA, Ball EH, Sanwal BD (1988) A collagen-binding protein involved in the differentiation of myoblasts recognizes the Arg-Gly-Asp sequence. Exp Cell Res 179:289–297. https://doi.org/10.1016/0014-4827(88)90368-0
Ng HM, Saidi NM, Omar FS et al (2018) Thermogravimetric Analysis of Polymers. Encycl Polym Sci Technol. https://doi.org/10.1002/0471440264.pst667
Okamoto Y, Kawakami K, Miyatake K et al (2002) Analgesic effects of chitin and chitosan. Carbohydr Polym 49:249–252. https://doi.org/10.1016/S0144-8617(01)00316-2
Ooi KS, Haszman S, Wong YN et al (2020) Physicochemical characterization of bilayer hybrid nanocellulose-collagen as a potential wound dressing. Materials (basel) 13:1–17. https://doi.org/10.3390/ma13194352
Park CJ, Clark SG, Lichtensteiger CA et al (2009) Accelerated wound closure of pressure ulcers in aged mice by chitosan scaffolds with and without bFGF. Acta Biomater 5:1926–1936. https://doi.org/10.1016/j.actbio.2009.03.002
Park S, Baker JO, Himmel ME et al (2010) Research Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuelsiofuels 3:1–10. https://doi.org/10.1186/1754-6834-3-10
Peng N, Huang D, Gong C et al (2020) Controlled arrangement of nanocellulose in polymeric matrix: from reinforcement to functionality. ACS Nano 14:16169–16179. https://doi.org/10.1021/acsnano.0c08906
Phang SJ, Arumugam B, Kuppusamy UR et al (2021) A review of diabetic wound models—Novel insights into diabetic foot ulcer. J Tissue Eng Regen Med 15:1051–1068. https://doi.org/10.1002/term.3246
Phang SJ, Basak S, Teh HX et al (2022) Advancements in extracellular matrix-based biomaterials and biofabrication of 3D organotypic skin models. ACS Biomater Sci Eng 8:3220–3241. https://doi.org/10.1021/acsbiomaterials.2c00342
Purkis PE, Steel JB, Mackenzie IC et al (1990) Antibody markers of basal cells in complex epithelia. J Cell Sci 97:39–50. https://doi.org/10.1242/jcs.97.1.39
Qian Z, Cao Z, Galuska L et al (2019) Glass transition phenomenon for conjugated polymers. Macromol Chem Phys 220:1–30. https://doi.org/10.1002/macp.201900062
Rasli SRAM, Ahmad I, Lazim AM, Hamzah A (2017) Extraction and characterization of cellulose from agricultural residue oil palm fronds. Malaysian J Anal Sci 21:1065–1073
Rýglová Š, Braun M, Suchý T (2017) Collagen and its modifications—crucial aspects with concern to its processing and analysis. Macromol Mater Eng. https://doi.org/10.1002/mame.201600460
Saadatkhah N, Carillo Garcia A, Ackermann S et al (2020) Experimental methods in chemical engineering: thermogravimetric analysis—TGA. Can J Chem Eng 98:34–43. https://doi.org/10.1002/cjce.23673
Salleh A, Mustafa N, Teow YH et al (2022) Dual-Layered approach of ovine collagen-gelatin/cellulose hybrid biomatrix containing graphene oxide-silver nanoparticles for cutaneous wound healing: fabrication, physicochemical, cytotoxicity and antibacterial characterisation. Biomedicines 10:1–17. https://doi.org/10.3390/biomedicines10040816
Sallehuddin N, Fadilah NI, Hwei NM et al (2022) Characterization and cytocompatibility of Collagen–Gelatin–Elastin (CollaGee) acellular skin substitute towards human dermal fibroblasts. In Vitro Assessment Biomed 10:1327. https://doi.org/10.3390/biomedicines10061327
Samimi Gharaie S, Habibi S, Nazockdast H (2018) Fabrication and characterization of chitosan/gelatin/thermoplastic polyurethane blend nanofibers. J Text Fibrous Mater 1:251522111876932. https://doi.org/10.1177/2515221118769324
Singh G, Byrne C, Thomason H, McBain AJ (2022) Investigating the microbial and metalloprotease sequestration properties of superabsorbent wound dressings. Sci Rep 12:1–17. https://doi.org/10.1038/s41598-022-08361-3
Snyder D, Sullivan N, Margolis BD, Schoelles K (2020) Technology assessment program-Skin substitutes for treating chronic wounds technical brief. Febr 2(2020):1–175
Su K, Wang C (2015) Recent advances in the use of gelatin in biomedical research. Biotechnol Lett 37:2139–2145. https://doi.org/10.1007/s10529-015-1907-0
Su SL, Rao QH, He YH, Xie W (2020) Effects of porosity on tensile mechanical properties of porous FeAl intermetallics. Trans Nonferrous Met Soc China 30:2757–2763. https://doi.org/10.1016/S1003-6326(20)65418-8
Tai Y, Woods EL, Dally J et al (2021) Myofibroblasts: function, formation, and scope of molecular therapies for skin fibrosis. Biomolecules 11:1–27. https://doi.org/10.3390/biom11081095
Tamara FR, Lin C, Mi FL, Ho YC (2018) Antibacterial effects of chitosan/cationic peptide nanoparticles. Nanomaterials 8:1–16. https://doi.org/10.3390/nano8020088
Thomas P, Duolikun T, Rumjit NP et al (2020) Comprehensive review on nanocellulose: recent developments, challenges and future prospects. J Mech Behav Biomed Mater 110:103884. https://doi.org/10.1016/j.jmbbm.2020.103884
Turissini JD, Elmarsafi T, Evans KK, Kim PJ (2019) Major risk factors contributing to split thickness skin graft failure. Georg Med Rev 3:7155. https://doi.org/10.52504/001c.7755
Veleirinho B, Coelho DS, Dias PF et al (2012) Nanofibrous poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/chitosan scaffolds for skin regeneration. Int J Biol Macromol 51:343–350. https://doi.org/10.1016/j.ijbiomac.2012.05.023
Wang HM, Chou YT, Wen ZH et al (2013) Novel biodegradable porous scaffold applied to skin regeneration. PLoS ONE 8:2–12. https://doi.org/10.1371/journal.pone.0056330
Wang L, Wang W, Liao J et al (2018) Novel bilayer wound dressing composed of SIS membrane with SIS cryogel enhanced wound healing process. Mater Sci Eng C 85:162–169. https://doi.org/10.1016/j.msec.2017.11.024
Wang Y, Yao M, Baker KB et al (2021) Force-dependent interactions between Talin and full-length vinculin. J Am Chem Soc 143:14726–14737. https://doi.org/10.1021/jacs.1c06223
Whitehead FA, Young SA, Kasapis S (2020) Swelling behaviour and glass transition in genipin-crosslinked chitosan systems. Int J Biol Macromol 164:3075–3083. https://doi.org/10.1016/j.ijbiomac.2020.08.178
Wu YB, Yu SH, Mi FL et al (2004) Preparation and characterization on mechanical and antibacterial properties of chitsoan/cellulose blends. Carbohydr Polym 57:435–440. https://doi.org/10.1016/j.carbpol.2004.05.013
Xue Y, Mou Z, Xiao H (2017) Nanocellulose as a sustainable biomass material: structure, properties, present status and future prospects in biomedical applications. Nanoscale 9:14758–14781. https://doi.org/10.1039/c7nr04994c
Zahari MJI, Jahi NM, Mohd NH et al (2018) Enhanced performance of cellulose from palm oil empty fruit bunch (EFB) via acetylation and Silylation. Preprints. https://doi.org/10.20944/preprints201807.0314.v1
Zhang J, Yang Z, Li C et al (2013) Cells behave distinctly within sponges and hydrogels due to differences of internal structure. Tissue Eng - Part A 19:2166–2175. https://doi.org/10.1089/ten.tea.2012.0393
Zhu T, Jiang J, Zhao J et al (2019) Regulating preparation of functional alginate-chitosan three-dimensional scaffold for skin tissue engineering. Int J Nanomedicine 14:8891. https://doi.org/10.2147/IJN.S210329
Ziegler WH, Liddington RC, Critchley DR (2006) The structure and regulation of vinculin. Trends Cell Biol 16:453–460. https://doi.org/10.1016/j.tcb.2006.07.004
Zinge C, Kandasubramanian B (2020) Nanocellulose based biodegradable polymers. Eur Polym J 133:109758. https://doi.org/10.1016/j.eurpolymj.2020.109758
Acknowledgments
The authors would like to acknowledge the research team in the Centre for Tissue Engineering and Regenerative Medicine, Faculty of Medicine, Universiti Kebangsaan Malaysia for the support in the laboratory works. This particular study has been performed in controlled facilities under the ISO9001:2015 management system. Additionally, authors are delighted to acknowledge Yosuke Hiraoka from Nitta-Gelatin Inc. Osaka, Japan for the gelatin supply, Mohd Reusmaazran Yusof from Malaysian Nuclear Agency for the mechanical tensile test service, and Nor Huwaida Binti Janil @ Jamil for guidance in OriginPro® 2022 software.
Funding
The research was funded by Ministry of Higher Education Fundamental Research Grant Scheme (FRGS/1/2020/SKK0/UKM/02/6).
Author information
Authors and Affiliations
Contributions
Conceptualization, YJC, MHMY, MBF, YT, MRB, and MDY; methodology; software, YJC and MHMY; validation, YJC, and MHMY; formal analysis, YJC; investigation; resources, YJC, MBF, YT, YH, SJP, MRC, and MHMY; data curation; writing—original draft preparation, YJC; writing—review and editing, MBF, MRB, SJP, and MHMY; visualization, YJC, SJP; supervision, MBF, and MHMY; project administration, MHMY. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors hereby declare that they have no conflict of interest.
Consent for publication
All authors are consented for publication.
Ethical approval and consent to participate
This research was approved by Universiti Kebangsaan Malaysia (UKM) Research Ethics Committee (JEP-2021-076).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cheah, Y.J., Yunus, M.H.M., Fauzi, M.B. et al. Gelatin–chitosan–cellulose nanocrystals as an acellular scaffold for wound healing application: fabrication, characterisation and cytocompatibility towards primary human skin cells. Cellulose 30, 5071–5092 (2023). https://doi.org/10.1007/s10570-023-05212-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-023-05212-w