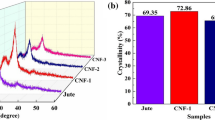
Abstract
TEMPO-oxidized cellulose nanofibril (TOCNF) hydrogel presents unique advantageous characteristics and has been envisioned as a building block to design novel biobased materials. However, the failure to maintain the gel shape and rapid decline of viscosity at elevated temperature (> 120 °C) limit the application of TOCNF hydrogel in high temperature environment of oil fields. In this study, dimethyl carbonate (DMC), a green methylation reagent, was employed to introduce methyl groups into TOCNFs to endow the DMC-TOCNF hydrogel with improved resistance at higher temperatures. The DMC-TOCNFs and original TOCNFs were characterized by fourier transform infrared (FTIR), X-ray photoelectron spectroscopy, atomic force microscopy, thermogravimetric analysis and rheological tests. The results show that the degree of substitution of DMC-TOCNFs was 0.078, 0.390 and 0.630, respectively. DMC-TOCNFs displayed the characteristics of gelation at high temperature, which increased with higher degree of substitution (DS). Of particular importance, the viscosity of DMC-TOCNFs dramatically raised from 226 to 2.9 × 104 mPa s when the temperature rose from 30 to 160 °C at a DS of 0.630. The methylated-TOCNF hydrogels displayed great potentials in expanding the application of TOCNFs at high temperatures.







Similar content being viewed by others
References
Abe K, Iwamoto S, Yano HJB (2007) Obtaining cellulose nanofibers with a uniform width of 15 nm from wood. Biomacromol 8:3276–3278. https://doi.org/10.1021/bm700624p
Albert et al (2004) Oxazoline-mediated interannular cyclopalladation of ferrocene: chiral palladium(II) catalysts for the enantioselective aza-claisen rearrangement. Angew Chem Int Ed 44(12):1865–1869. https://doi.org/10.1002/ange.200462434
Bajwa GS, Sammon C, Timmins P, Melia CD (2009) Molecular and mechanical properties of hydroxypropyl methylcellulose solutions during the sol:gel transition. Polymer 50:4571–4576. https://doi.org/10.1016/j.polymer.2009.06.075
Barcelo G, Grenouillat D, Senet JP, Sennyey GJT (1990) Pentaalkylguanidines as etherification and esterification catalysts. Tetrahedron 46:1839–1848. https://doi.org/10.1016/S0040-4020(01)89753-2
Candau F, Biggs S, Hill A, Selb J (1994) Synthesis, structure and properties of hydrophobically associating polymers. Prog Org Coat 24:11–19. https://doi.org/10.1016/0033-0655(94)85002-X
Chengmin H, Yufang C, Wenning C, Wei L (2011) Microwave-assisted methylation of cassava starch with dimethyl carbonate. Carbohyd Res 346(9):1178–1181. https://doi.org/10.1016/j.carres.2011.04.001
Duan P, Xu Q, Shen S, Zhang Y, Liu X (2019) One-pot modification on cotton fabric using an emulsion of Ag NPs protected by mercaptosuccinic acid to achieve durably antibacterial effect. Fibers and Polymers 20:1803–1811. https://doi.org/10.1007/s12221-019-9224-1
Habibi Y, Chanzy H, Vignon MR (2006) TEMPO-mediated surface oxidation of cellulose whiskers. Cellulose 13:679–687. https://doi.org/10.1007/s10570-006-9075-y
Hirrien M, Chevillard C, Desbrières J, Axelos M, Rinaudo MJP (1998) Thermogelation of methylcelluloses: new evidence for understanding the gelation mechanism. Polymer 39:6251–6259. https://doi.org/10.1016/S0032-3861(98)00142-6
Hirrien M, Ross-Murphy SB (2000) Thermogelation of methylcellulose: rheological considerations. Polymer 41:2451–2461. https://doi.org/10.1016/S0032-3861(00)00093-8
Hodges K, Kester W, Wiederrich D, Grover J (1979a) Determination of alkoxyl substitution in cellulose ethers by Zeisel gas chromatography. Anal Chem 51:2172–2176. https://doi.org/10.1021/ac50049a028
Hoeng F, Denneulin A, Reverdy-Bruas N, Krosnicki G, Bras J (2017) Rheology of cellulose nanofibrils/silver nanowires suspension for the production of transparent and conductive electrodes by screen printing. Appl Surf Sci 394:160–168. https://doi.org/10.1016/j.apsusc.2016.10.073
Hu Y, Tang L, Lu Q, Wang S, Chen X, Huang B (2014) Preparation of cellulose nanocrystals and carboxylated cellulose nanocrystals from borer powder of bamboo. Cellulose 21:1611–1618. https://doi.org/10.1007/s10570-014-0236-0
Khiari R, Rol F, Brochier-Salon MC, Bras J, Belgacem MN (2019) The efficiency of cellulose carbonates to produce cellulose nanofibers. ACS Sustain Chem Eng 7(9):8155–8167. https://doi.org/10.1021/acssuschemeng.8b06039
Khiari R, Salon M, Mhenni MF, Mauret E, Belgacem MN (2017) Synthesis and characterization of cellulose carbonate using greenchemistry: surface modification of Avicel. Carbohyd Polym 163:254–260. https://doi.org/10.1016/j.carbpol.2017.01.037
Kim SH, Lee CM, Kafle K (2013) Characterization of crystalline cellulose in biomass: basic principles, applications, and limitations of XRD, NMR, IR, Raman, and SFG. Korean J Chem Eng 30:2127–2141. https://doi.org/10.1007/s11814-013-0162-0
Labafzadeh SR, Helminen KJ, Kilpeläinen I, King A (2015) Synthesis of cellulose methylcarbonate in ionic liquids using dimethylcarbonate. Chemsuschem 8:77–81. https://doi.org/10.1002/cssc.201402794
Lee Y, Shimizu I (1998) Convenient O-methylation of phenols with dimethyl carbonate. Synlett 1998:1063–1064. https://doi.org/10.1055/s-1998-1893
Li L, Shan H, Yue CY, Lam YC, Tam KC, Hu X (2002) Thermally induced association and dissociation of methylcellulose in aqueous solutions. Langmuir 18:7291–7298. https://doi.org/10.1021/la020029b
Li MC, Mei C, Xu X, Lee S, Wu QJP (2016) Cationic surface modification of cellulose nanocrystals: toward tailoring dispersion and interface in carboxymethyl cellulose films. Polymer 107:200–210. https://doi.org/10.1016/j.polymer.2016.11.022
Liu M, Arshadi M, Javi F, Lawrence P, Davachi SM, Abbaspourrad A (2020) Green and facile preparation of hydrophobic bioplastics from tea waste. J Clean Prod 276(10):123353. https://doi.org/10.1016/j.jclepro.2020.123353
Liu X, Wang C, Wang A, Qu J, Wen Y, Wei B (2019) Application of cellulose and cellulose nanofibers in oil exploration. Paper Biomater 4(3):69–77
Lui MY et al (2016) Microwave-assisted methylation of dihydroxybenzene derivatives with dimethyl carbonate. RSC Adv 6(63):58443–58451. https://doi.org/10.1039/C6RA09841J
Ma S, Chen C, Sablong RJ, Koning CE, Benthem RATMV (2018) Non-isocyanate strategy for anionically stabilized water-borne polyurea dispersions and coatings. J Polym Sci Part A Polym Chem 56(10):1078–1090. https://doi.org/10.1002/pola.28986
Malladi R, Nagalakshmaiah M, Ro Be Rt M, Elkoun S (2018) Importance of agriculture and industrial waste in the field of nano cellulose and its recent industrial developments: a review. ACS Sustain Chem Eng 6:2807–2828. https://doi.org/10.1021/acssuschemeng.7b03437
Mei et al (2016) Cationic surface modification of cellulose nanocrystals: toward tailoring dispersion and interface in carboxymethyl cellulose films. Polymer 107:200–210. https://doi.org/10.1016/j.polymer.2016.11.022
Morozova SJPI (2020) Methylcellulose fibrils: a mini review. Polym Int 69:125–130. https://doi.org/10.1002/pi.5945
Nasatto PL, Pignon F, Silveira JLM, Duarte MER, Noseda MD, Rinaudo M (2015) Methylcellulose, a cellulose derivative with original physical properties and extended applications. Polymers 7(5):777–803. https://doi.org/10.3390/polym7050777
Niu et al (2018) Rosin modified cellulose nanofiber as a reinforcing and co-antimicrobial agents in polylactic acid/chitosan composite film for food packaging. Carbohyd Polym 183:102–109. https://doi.org/10.1016/j.carbpol.2017.11.079
Norinaga K, Kuniya M, Iino M (2002) Effect of associative interaction on the dynamic viscoelastic property of coal concentrated solution. Energy Fuels 16:62–68. https://doi.org/10.1021/ef010154f
Oliveira RL, Vieira JG, Barud HS, Assunção R, R Filho G, Ribeiro SJ, Messadeqq Y (2015) Synthesis and characterization of methylcellulose produced from bacterial cellulose under heterogeneous condition. J Braz Chem Soc 26(9):1861–1870. https://doi.org/10.5935/0103-5053.20150163
Saito T, Kimura S, Nishiyama Y, Isogai AJB (2007) Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromol 8:2485–2491. https://doi.org/10.1021/bm0703970
Sang CS, Shim HK, Cho TH (1985) Photoreaction of 5,7-dimethoxycoumarin with adenosine. Chem Lett 15:19–22. https://doi.org/10.1246/cl.1986.19
Selva M, Tundo P, Perosa A (2001) Reaction of primary aromatic amines with alkyl carbonates over NaY faujasite: a convenient and selective access to mono-N-alkyl anilines. J Org Chem 66:677–680. https://doi.org/10.1021/jo0006728
Shieh WC, Dell S, Repič O (2010) 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and microwave-accelerated green chemistry in methylation of phenols, indoles, and benzimidazoles with dimethyl carbonate. Org Lett 3:4279–4281. https://doi.org/10.1002/chin.200221056
Varghese HT, Panicker CY, Philip D, Mannekutla JR, Inamdar SR (2007) IR, Raman and SERS studies of methyl salicylate. Spectrochim Acta Part A Mol Biomol Spectrosc 66:959–963. https://doi.org/10.1016/j.saa.2006.04.034
Xia et al (2007) The effect of CaCl2 on the interaction between hydrolyzed polyacrylamide and sodium stearate: Rheological property study. Colloids Surf A 305:138–144. https://doi.org/10.1016/j.colsurfa.2007.04.057
Xiongli et al (2019) Improving salt tolerance and thermal stability of cellulose nanofibrils by grafting modification. Carbohyd Polym 211:257–265. https://doi.org/10.1016/j.carbpol.2019.02.009
Xla F et al (2019) Cellulose nanofibril-polymer hybrids for protecting drilling fluid at high salinity and high temperature. Carbohyd Polym 229:115465. https://doi.org/10.1016/j.carbpol.2019.115465
Yang J, Xu F, Han CR (2017) Metal ion mediated cellulose nanofibrils transient network in covalently cross-linked hydrogels: mechanistic insight into morphology and dynamics. Biomacromol 18:1019–1028. https://doi.org/10.1021/acs.biomac.6b01915
Yufang C, Jan L, Yaxi C, Wenning C, Chengmin H, Datian Z (2010) Methylation reaction of starch with dimethyl carbonate. FINE CHEMICALS 27(6):601–604
Zhou et al (2017) Study on salt thickening mechanism of the amphiphilic polymer with betaine zwitterionic group by β-cyclodextrin inclusion method. Colloid Polym Sci 295:1887–1895. https://doi.org/10.1007/s00396-017-4169-7
Zhu Z, Kang W, Sarsenbekuly B, Yang H, Dai C, Yang R, Fan H (2017) Preparation and solution performance for the amphiphilic polymers with different hydrophobic groups. J Appl Polym Sci 134(20):44744. https://doi.org/10.1002/app.44744
Zhuo X, Liu C, Pan R, Dong X, Li Y (2017) Nanocellulose Mechanically Isolated from Amorpha fruticosa Linn. ACS Sustain Chem Eng 5:4414–4420. https://doi.org/10.1021/acssuschemeng.7b00478
Acknowledgments
The authors are grateful for the financial support from the Natural Science Foundation of Tianjin, China (Grant No. 181CYBIC86500).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, L., Fang, Z., Zhang, W. et al. Preparation of methylated TEMPO-oxidized cellulose nanofibril hydrogel with high-temperature sensitivity. Cellulose 29, 8599–8609 (2022). https://doi.org/10.1007/s10570-022-04811-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04811-3