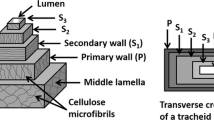
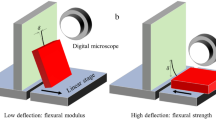
Abstract
Reported tensile mechanical properties of many natural plant fibres vary to a large extent due to very often inappropriate measurement of the fibre’s cross-sectional area by diameter estimation. Using natural ramie filament as a model testing elementary fibre, a more realistic stereological determination is presented, including microscopic imaging analysis of the fibres’ cross-sectional area. When applying the area data using this approach to calculate tensile strength, a far narrower variation in the fibres’ strength distribution according to Weibull analysis was found. The gauge length effects on the mechanical performance of the natural fibre were revealed and analysed. In addition, in situ SEM observations during microtensile measurements detected real time changes in the fibres’ structure during stress. It was found that fibre failure was mainly caused by macroscopic physical defects and associated microscopic slippage of the microfibrils. Furthermore, results of cyclic tensile tests indicated that the fibre underwent elastic deformations under progressive loading–unloading cycles, which is due to bonding restriction that the surrounding matrix presents against the slippage of the microfibrils and reorganisation of hydrogen bonds.
Graphical abstract











Similar content being viewed by others
References
Andersons J, Spārniņš E, Joffe R, Wallström L (2005) Strength distribution of elementary flax fibres. Compos Sci Technol 65:693–702
Andersons J, Poriķe E, Spārniņš E (2009) The effect of mechanical defects on the strength distribution of elementary flax fibres. Compos Sci Technol 69:2152–2157
Andersons J, Poriķe E, Spārniņš E (2011) Modeling strength scatter of elementary flax fibers: the effect of mechanical damage and geometrical characteristics. Compos Part A 42:543–549
Azizi Samir MAS, Alloin F, Dufresne A (2005) Review of recent research into cellulosic whiskers, their properties and their application in nanocomposite field. Biomacromol 6:612–626
Baley C (2002) Analysis of the flax fibres tensile behaviour and analysis of the tensile stiffness increase. Compos A 33:939–948
Banky EC, Slen SB (1956) Dimensional changes and related phenomena in wool fibers under stress. Text Res J 26:204–210
Bledzki AK, Gassan J (1999) Composites reinforced with cellulose based fibres. Prog Polym Sci 24:221–274
Bos H, Van Den Oever M, Peters O (2002) Tensile and compressive properties of flax fibres for natural fibre reinforced composites. J Mater Sci 37:1683–1692
Bourmaud A et al (2017) Exploring the mechanical performance and in-planta architecture of secondary hemp fibres. Ind Crops Prod 108:1–5
d’Almeida JRM, Mauricio MHP, Paciornik S (2012) Evaluation of the cross-section of lignocellulosic fibers using digital microscopy and image analysis. J Compos Mater 46:3057–3065
Del Masto A, Trivaudey F, Guicheret-Retel V, Placet V, Boubakar L (2017) Nonlinear tensile behaviour of elementary hemp fibres: a numerical investigation of the relationships between 3D geometry and tensile behaviour. J Mater Sci 52:6591–6610
Dijon G (2002) A study of the structure and the mechanical properties of flax as reinforcing fibre for composites. Imperial College of Science, Technology and Medicine, University of London, London
Edwards HGM, Farwell DW, Webster D (1997) FT Raman microscopy of untreated natural plant fibres. Spectrochim Acta, Part A 53:2383–2392
Eichhorn SJ, Young RJ (2003) Deformation micromechanics of natural cellulose fibre networks and composites. Compos Sci Technol 63:1225–1230
Eichhorn S et al (2010) Review: current international research into cellulose nanofibres and nanocomposites. J Mater Sci 45:1–33
Elices M, Llorca J (2002) Fiber fracture. Elsevier, Amsterdam
Faruk O, Bledzki AK, Fink H-P, Sain M (2012) Biocomposites reinforced with natural fibers: 2000–2010. Prog Polym Sci 37:1552–1596
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896
French AD, Kim HJ (2018) Cotton fiber structure. In: Fang D (ed) Cotton fiber: physics, chemistry and biology. Springer, Berlin, pp 13–39
Gassan J, Chate A, Bledzki AK (2001) Calculation of elastic properties of natural fibers. J Mater Sci 36:3715–3720
Goda K, Sreekala MS, Gomes A, Kaji T, Ohgi J (2006) Improvement of plant based natural fibers for toughening green composites—Effect of load application during mercerization of ramie fibers. Compos Part A 37:2213–2220
Hänninen T, Thygesen A, Mehmood S, Madsen B, Hughes M (2012) Mechanical processing of bast fibres: the occurrence of damage and its effect on fibre structure. Ind Crops Prod 39:7–11
Himmelsbach DS, Akin DE (1998) Near-infrared fourier-transform raman spectroscopy of flax (Linum usitatissimum L.) stems. J Agric Food Chem 46:991–998
Hughes M (2012) Defects in natural fibres: their origin, characteristics and implications for natural fibre-reinforced composites. J Mater Sci 47:599–609
Ilvessalo-Pfäffli M-S (1995) Fiber atlas: identification of papermaking fibers. Springer, Berlin
Jähn A, Schröder MW, Füting M, Schenzel K, Diepenbrock W (2002) Characterization of alkali treated flax fibres by means of FT Raman spectroscopy and environmental scanning electron microscopy. Spectrochim Acta, Part A 58:2271–2279
Kalia S, Kaith BS, Kaur I (2011) Cellulose fibers: bio- and nano-polymer composites. Springer, Berlin
Kelly SP, Sensenig A, Lorentz KA, Blackledge TA (2011) Damping capacity is evolutionarily conserved in the radial silk of orb-weaving spiders. Zoology 114:233–238
Le Cam JB, Huneau B, Verron E, Gornet L (2004) Mechanism of fatigue crack growth in carbon black filled natural rubber. Macromolecules 37:5011–5017
Lefeuvre A, Bourmaud A, Lebrun L, Morvan C, Baley C (2013) A study of the yearly reproducibility of flax fiber tensile properties. Ind Crops Prod 50:400–407
Lefeuvre A, Bourmaud A, Morvan C, Baley C (2014) Elementary flax fibre tensile properties: correlation between stress–strain behaviour and fibre composition. Ind Crops Prod 52:762–769
Lewin M (2006) Handbook of fiber chemistry. International fiber science and technology. CRC Press, Boca Raton
Lewin M (2007) Handbook of fiber chemistry. CRC, Boca Raton
Li Y, Mai Y-W, Ye L (2000) Sisal fibre and its composites: a review of recent developments. Compos Sci Technol 60:2037–2055
Liu M, Fernando D, Daniel G, Madsen B, Meyer AS, Ale MT, Thygesen A (2015) Effect of harvest time and field retting duration on the chemical composition, morphology and mechanical properties of hemp fibers. Ind Crops Prod 69:29–39
Mohanty AK, Misra M, Drzal LT (2005) Natural fibers, biopolymers, and biocomposites. Taylor & Francis Group, London
Pickering KL, Abdalla A, Ji C, McDonald AG, Franich RA (2003) The effect of silane coupling agents on radiata pine fibre for use in thermoplastic matrix composites. Compos Part A 34:915–926
Rong MZ, Zhang MQ, Liu Y, Yang GC, Zeng HM (2001) The effect of fiber treatment on the mechanical properties of unidirectional sisal-reinforced epoxy composites. Compos Sci Technol 61:1437–1447
Ščudla J, Raab M, Eichhorn KJ, Strachota A (2003) Formation and transformation of hierarchical structure of β-nucleated polypropylene characterized by X-ray diffraction, differential scanning calorimetry and scanning electron microscopy. Polymer 44:4655–4664
Shahid M, Mohammad F, Chen G, Tang R-C, Xing T (2016) Enzymatic processing of natural fibres: white biotechnology for sustainable development. Green Chem 18:2256–2281
Spārniņš E, Andersons J (2009) Diameter variability and strength scatter of elementary flax fibers. J Mater Sci 44:5697–5699
Thamae T, Baillie C (2007) Influence of fibre extraction method, alkali and silane treatment on the interface of Agave americana waste HDPE composites as possible roof ceilings in Lesotho. Compos Interfaces 14:821–836
Thomason JL, Carruthers J (2012) Natural fibre cross sectional area, its variability and effects on the determination of fibre properties. J Biobased Mater Bioenergy 6:424–430
Truss RW (2011) Natural fibers for biocomposites. MRS Bull 36:711–715
Vincent JFV (2000) A unified nomenclature for plant fibres for industrial use. Appl Compos Mater 7:269–271
Vuure AV (2008) Natural fibre composites: recent developments. KU Leuven, Leuven
Wambua P, Ivens J, Verpoest I (2003) Natural fibres: can they replace glass in fibre reinforced plastics? Compos Sci Technol 63:1259–1264
Weibull W (1951) A statistical distribution function of wide applicability. J Appl Mech 18:293–305
Wu Y et al (2017) Bioinspired supramolecular fibers drawn from a multiphase self-assembled hydrogel. Proc Natl Acad Sci USA 114:8163–8168
Young RA (2000) Fibers, vegetable. In: Suslick KS (ed) Kirk-othmer encyclopedia of chemical technology. Wiley, Hoboken
Zhang Y, Wang X, Pan N, Postle R (2002) Weibull analysis of the tensile behavior of fibers with geometrical irregularities. J Mater Sci 37:1401–1406
Zhang H et al (2012) Nanocavitation in carbon black filled styrene–butadiene rubber under tension detected by real time small angle X-ray scattering. Macromolecules 45:1529–1543
Acknowledgments
The authors are thankful for financial support from the National Natural Science Foundation of China (21706039, 21676058) and the Natural Science Foundation of Guangdong Province (2017A030310300, 2016A030313694). PSS also gratefully acknowledges the Spanish Ministry Economy and Competitivity (MINECO) for a Ramón y Cajal fellowship (RYC-2014-16759) and a Proyecto de Investigación en el Programa Estatal de Fomento de la Investigación Científica y Técnica de Excelencia (MAT2017-88382-P). We are grateful to Dr. Álvaro Ridruejo and Editor Alfred D. French, Ph.D. for their kind suggestion and discussion.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yue, H., Rubalcaba, J.C., Cui, Y. et al. Determination of cross-sectional area of natural plant fibres and fibre failure analysis by in situ SEM observation during microtensile tests. Cellulose 26, 4693–4706 (2019). https://doi.org/10.1007/s10570-019-02428-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-019-02428-7