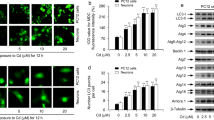
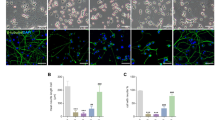
Abstract
Mitochondrial-associated endoplasmic reticulum (ER) membranes (MAMs) play a key role in several physiological functions, including calcium ion (Ca2+) transfer and autophagy; however, the molecular mechanism controlling this interaction in cadmium (Cd)-induced neurotoxicity is unknown. This study shows that Cd induces alterations in MAMs and mitochondrial Ca2+ levels in PC12 cells and primary neurons. Ablation or silencing of mitofusin 2 (Mfn2) in PC12 cells or primary neurons blocks the colocalization of ER and mitochondria while reducing the efficiency of mitochondrial Ca2+ uptake. Moreover, Mfn2 defects reduce interactions or colocalization between GRP75 and VDAC1. Interestingly, the enhancement of autophagic protein levels, colocalization of LC3 and Lamp2, and GFP-LC3 puncta induced by Cd decreased in Mfn2−/− or Grp75−/− PC12 cells and Mfn2- or Grp75-silenced primary neurons. Notably, the specific Ca2+ uniporter inhibitor RuR blocked both mitochondrial Ca2+ uptake and autophagy induced by Cd. Finally, this study proves that the mechanism by which IP3R-Grp75-VDAC1 tethers in MAMs is associated with the regulation of autophagy by Mfn2 and involves their role in mediating mitochondrial Ca2+ uptake from ER stores. These results give new evidence into the organelle metabolic process by demonstrating that Ca2+ transport between ER-mitochondria is important in autophagosome formation in Cd-induced neurodegeneration.
Graphical abstract









Similar content being viewed by others
Data availability
All relevant data and materials are available from the authors upon reasonable request.
Code availability
Not applicable.
References
Axe EL, Walker SA, Manifava M, Chandra P, Roderick HL, Habermann A, et al. Autophagosome formation from membrane compartments enriched in phosphatidylinositol 3-phosphate and dynamically connected to the endoplasmic reticulum. J Cell Biol. 2008;182(4):685–701. https://doi.org/10.1083/jcb.200803137.
Baughman JM, Perocchi F, Girgis HS, Plovanich M, Belcher-Timme CA, Sancak Y, et al. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature. 2011;476(7360):341–5. https://doi.org/10.1038/nature10234.
Böckler S, Westermann B. Mitochondrial ER contacts are crucial for mitophagy in yeast. Dev Cell. 2014;28(4):450–8. https://doi.org/10.1016/j.devcel.2014.01.012.
Cárdenas C, Miller RA, Smith I, Bui T, Molgó J, Müller M, et al. Essential regulation of cell bioenergetics by constitutive InsP3 receptor Ca2+ transfer to mitochondria. Cell. 2010;142(2):270–83. https://doi.org/10.1016/j.cell.2010.06.007.
Cárdenas C, Müller M, McNeal A, Lovy A, Jaňa F, Bustos G, et al. Selective vulnerability of cancer cells by inhibition of Ca(2+) transfer from endoplasmic reticulum to mitochondria. Cell Rep. 2016;14(10):2313–24. https://doi.org/10.1016/j.celrep.2016.02.030.
Chin-Chan M, Navarro-Yepes J, Quintanilla-Vega B. Environmental pollutants as risk factors for neurodegenerative disorders: Alzheimer and Parkinson diseases. Front Cell Neurosci. 2015;9:124. https://doi.org/10.3389/fncel.2015.00124.
Csordás G, Renken C, Várnai P, Walter L, Weaver D, Buttle KF, et al. Structural and functional features and significance of the physical linkage between ER and mitochondria. J Cell Biol. 2006;174(7):915–21. https://doi.org/10.1083/jcb.200604016.
de Brito OM, Scorrano L. Mitofusin 2 tethers endoplasmic reticulum to mitochondria. Nature. 2008a;456(7222):605–10. https://doi.org/10.1038/nature07534.
De Stefani D, Raffaello A, Teardo E, Szabò I, Rizzuto R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature. 2011;476(7360):336–40. https://doi.org/10.1038/nature10230.
Filadi R, Greotti E, Turacchio G, Luini A, Pozzan T, Pizzo P. Presenilin 2 modulates endoplasmic reticulum-mitochondria coupling by tuning the antagonistic effect of mitofusin 2. Cell Rep. 2016;15(10):2226–38. https://doi.org/10.1016/j.celrep.2016.05.013.
Garofalo T, Matarrese P, Manganelli V, Marconi M, Tinari A, Gambardella L, et al. Evidence for the involvement of lipid rafts localized at the ER-mitochondria associated membranes in autophagosome formation. Autophagy. 2016;12(6):917–35. https://doi.org/10.1080/15548627.2016.1160971.
Gomez-Suaga P, Paillusson S, Stoica R, Noble W, Hanger DP, Miller CCJ. The ER-mitochondria tethering complex VAPB-PTPIP51 regulates autophagy. Curr Biol. 2017;27(3):371–85. https://doi.org/10.1016/j.cub.2016.12.038.
Hailey D, Rambold A, Satpute-Krishnan P, Mitra K, Sougrat R, Kim P, et al. Mitochondria supply membranes for autophagosome biogenesis during starvation. Cell. 2010;141(4):656–67. https://doi.org/10.1016/j.cell.2010.04.009.
Hamasaki M, Furuta N, Matsuda A, Nezu A, Yamamoto A, Fujita N, et al. Autophagosomes form at ER-mitochondria contact sites. Nature. 2013;495(7441):389–93. https://doi.org/10.1038/nature11910.
Hayashi T, Rizzuto R, Hajnoczky G, Su TP. MAM: more than just a housekeeper. Trends Cell Biol. 2009;19(2):81–8. https://doi.org/10.1016/j.tcb.2008.12.002.
Honrath B, Culmsee C, Dolga AM. One protein, different cell fate: the differential outcome of depleting GRP75 during oxidative stress in neurons. Cell Death Dis. 2018;9(2):32. https://doi.org/10.1038/s41419-017-0148-7.
Itakura E, Kishi C, Inoue K, Mizushima N. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell. 2008;19(12):5360–72. https://doi.org/10.1091/mbc.e08-01-0080.
Itoh T, Fujita N, Kanno E, Yamamoto A, Yoshimori T, Fukuda M. Golgi-resident small GTPase Rab33B interacts with Atg16L and modulates autophagosome formation. Mol Biol Cell. 2008;19(7):2916–25. https://doi.org/10.1091/mbc.e07-12-1231.
Järup L, Berglund M, Elinder CG, Nordberg G, Vahter M. Health effects of cadmium exposure—a review of the literature and a risk estimate. Scand J Work Environ Health. 1998;24(suppl 1):1–52.
Khan MT, Joseph SK. Role of inositol trisphosphate receptors in autophagy in DT40 cells. J Biol Chem. 2010;285(22):16912–20. https://doi.org/10.1074/jbc.M110.114207.
Leal NS, Schreiner B, Pinho CM, Filadi R, Wiehager B, Karlström H, et al. Mitofusin-2 knockdown increases ER-mitochondria contact and decreases amyloid β-peptide production. J Cell Mol Med. 2016;20(9):1686–95. https://doi.org/10.1111/jcmm.12863.
Liu Y, Li Y, Liu K, Shen J, Boone DL. Exposing to cadmium stress cause profound toxic effect on microbiota of the mice intestinal tract. PLoS ONE. 2014;9(2):e85323.
Mallilankaraman K, Cárdenas C, Doonan PJ, Chandramoorthy HC, Irrinki KM, Golenár T, et al. MCUR1 is an essential component of mitochondrial Ca2+ uptake that regulates cellular metabolism. Nat Cell Biol. 2012;14(12):1336–43. https://doi.org/10.1038/ncb2622.
Matsunaga K, Morita E, Saitoh T, Akira S, Ktistakis NT, Izumi T, et al. Autophagy requires endoplasmic reticulum targeting of the PI3-kinase complex via Atg14L. J Cell Biol. 2010;190(4):511–21. https://doi.org/10.1083/jcb.200911141.
Mizushima N, Yoshimori T, Ohsumi Y. The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol. 2011;27:107–32. https://doi.org/10.1146/annurev-cellbio-092910-154005.
Pi H, Xu S, Zhang L, Guo P, Li Y, Xie J, et al. Dynamin 1-like-dependent mitochondrial fission initiates overactive mitophagy in the hepatotoxicity of cadmium. Autophagy. 2013;9(11):1780–800. https://doi.org/10.4161/auto.25665.
Rapizzi E, Pinton P, Szabadkai G, Wieckowski MR, Vandecasteele G, Baird G, et al. Recombinant expression of the voltage-dependent anion channel enhances the transfer of Ca2+ microdomains to mitochondria. J Cell Biol. 2002;159(4):613–24. https://doi.org/10.1083/jcb.200205091.
Rowland AA, Voeltz GK. Endoplasmic reticulum-mitochondria contacts: function of the junction. Nat Rev Mol Cell Biol. 2012;13(10):607–25. https://doi.org/10.1038/nrm3440.
Shukla A, Shukla GS, Srimal RC. Cadmium-induced alterations in blood-brain barrier permeability and its possible correlation with decreased microvessel antioxidant potential in rat. Hum Exp Toxicol. 1996;15(5):400–5. https://doi.org/10.1177/096032719601500507.
Steffen J, Koehler CM. ER-mitochondria contacts: actin dynamics at the ER control mitochondrial fission via calcium release. J Cell Biol. 2018;217(1):15–7. https://doi.org/10.1083/jcb.201711075.
Szabadkai G, Bianchi K, Várnai P, De Stefani D, Wieckowski MR, Cavagna D, et al. Chaperone-mediated coupling of endoplasmic reticulum and mitochondrial Ca2+ channels. J Cell Biol. 2006;175(6):901–11. https://doi.org/10.1083/jcb.200608073.
Vicencio JM, Ortiz C, Criollo A, Jones AW, Kepp O, Galluzzi L, et al. The inositol 1,4,5-trisphosphate receptor regulates autophagy through its interaction with Beclin 1. Cell Death Differ. 2009;16(7):1006–17. https://doi.org/10.1038/cdd.2009.34.
Wang QW, Zhu JQ, Zhang KB, Jiang CY, Wang Y, Yuan Y, et al. Induction of cytoprotective autophagy in PC-12 cells by cadmium. Biochem Biophys Res Commun. 2013;438(1):186–92. https://doi.org/10.1016/j.bbrc.2013.07.050.
Wang T, Wang Q, Song R, Zhang Y, Yang J, Wang Y, et al. Cadmium induced inhibition of autophagy is associated with microtubule disruption and mitochondrial dysfunction in primary rat cerebral cortical neurons. Neurotoxicol Teratol. 2016a;53:11–8. https://doi.org/10.1016/j.ntt.2015.11.007.
Wang T, Wang Q, Song R, Zhang Y, Zhang K, Yuan Y, et al. Autophagy plays a cytoprotective role during cadmium-induced oxidative damage in primary neuronal cultures. Biol Trace Elem Res. 2015;168(2):481–9. https://doi.org/10.1007/s12011-015-0390-8.
Wang T, Yuan Y, Zou H, Yang J, Zhao S, Ma Y, et al. The ER stress regulator Bip mediates cadmium-induced autophagy and neuronal senescence. Sci Rep. 2016b;6:38091. https://doi.org/10.1038/srep38091.
Wang T, Zhu Q, Cao B, Yuan Y, Wen S, Liu Z. Cadmium induces mitophagy via AMP-activated protein kinases activation in a PINK1/Parkin-dependent manner in PC12 cells. Cell Prolif. 2020;53(6): e12817. https://doi.org/10.1111/cpr.12817.
Wong A, Grubb DR, Cooley N, Luo J, Woodcock EA. Regulation of autophagy in cardiomyocytes by Ins(1,4,5)P(3) and IP(3)-receptors. J Mol Cell Cardiol. 2013;54:19–24. https://doi.org/10.1016/j.yjmcc.2012.10.014.
Wu W, Lin C, Wu K, Jiang L, Wang X, Li W, et al. FUNDC1 regulates mitochondrial dynamics at the ER-mitochondrial contact site under hypoxic conditions. Embo J. 2016;35(13):1368–84. https://doi.org/10.15252/embj.201593102.
Yan Y, Bian JC, Zhong LX, Zhang Y, Sun Y, Liu ZP. Oxidative stress and apoptotic changes of rat cerebral cortical neurons exposed to cadmium in vitro. Biomed Environ Sci. 2012;25(2):172–81. https://doi.org/10.3967/0895-3988.2012.02.008.
Yokouchi M, Hiramatsu N, Hayakawa K, Kasai A, Takano Y, Yao J, et al. Atypical, bidirectional regulation of cadmium-induced apoptosis via distinct signaling of unfolded protein response. Cell Death Differ. 2007;14(8):1467–74. https://doi.org/10.1038/sj.cdd.4402154.
Yuan Y, Jiang CY, Xu H, Sun Y, Hu FF, Bian JC, et al. Cadmium-induced apoptosis in primary rat cerebral cortical neurons culture is mediated by a calcium signaling pathway. PLoS ONE. 2013;8(5): e64330. https://doi.org/10.1371/journal.pone.0064330.
Zhang L, Xia Q, Zhou Y, Li J. Endoplasmic reticulum stress and autophagy contribute to cadmium-induced cytotoxicity in retinal pigment epithelial cells. Toxicol Lett. 2019;311:105–13. https://doi.org/10.1016/j.toxlet.2019.05.001.
Zhou H, Wang S, Hu S, Chen Y, Ren J. ER-mitochondria microdomains in cardiac ischemia-reperfusion injury: a fresh perspective. Front Physiol. 2018;9:755. https://doi.org/10.3389/fphys.2018.00755.
Acknowledgements
We thank Dr. Liqi Zhu for giving us the plasmid as gift, Dr. Mahmoud for providing language help, and the Testing Center of Yangzhou University for providing technical support for the use of laser confocal microscopy. We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Funding
This study was supported by grants from the National Natural Science Foundation of China (Nos. 31802260, 31772808, 31872533, and 31702305), Natural Science Research of Higher Education in Jiangsu Province (18KJB230009), Jiangsu Provincial Natural Science Foundation of China (BK20180917), and the National Key Research and Development Program of China (No. 2016YFD0501208), a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Contributions
TW and YY conceived and designed the study. TW, QPZ, and BBC performed the experiments. SQW, JCB, HZ, XZL, and JHG contributed materials and methods. YC and RLS analyzed the data. TW wrote the manuscript. ZPL performed the final approval of the version to be submitted.
Corresponding authors
Ethics declarations
Ethics approval
The ethics committee of Yangzhou University (Yangzhou, China) approved all animal experiments.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Cd triggers alterations of MAMs and mitochondrial Ca2+ levels in neuronal cells.
• Ablation or silencing of Mfn2 blocks the colocalization of ER and mitochondria.
• Mfn2 defect reduces Grp75-VDAC1 interaction and mitochondrial Ca2+ uptake.
• Mutation of Mfn2 or Grp75 suppressed Cd-induced autophagy.
• Autophagy induced by Cd is blocked by the inhibition of mitochondrial Ca2+ uptake.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, T., Zhu, Q., Cao, B. et al. Ca2+ transfer via the ER-mitochondria tethering complex in neuronal cells contribute to cadmium-induced autophagy. Cell Biol Toxicol 38, 469–485 (2022). https://doi.org/10.1007/s10565-021-09623-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09623-y