Abstract
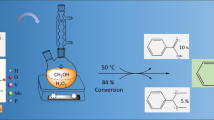
In this work, vanadium replaced molybdenum atoms generating catalysts with general formulae H3+nPMo12-nVnO40 (n = 0,1, 2 and 3), which were used in the acetalization of alkyl alcohols with terpene aldehyde (β-citronellal and methyl alcohol were model substrates). The initial focus was evaluating how the vanadium load impact the catalytic activity of phosphomolybdic acids and trying to link this effect with their structural properties. A comparison of performance achieved by the catalysts revealed that among phosphomolybdic acids (i.e., with V1, V2, and V3 atoms/per anion), the vanadium monosubstituted phosphomolybdic acid was the most active and selective toward the formation of β-citronellyl acetal. The effects of main reaction variables such as time, temperature, catalyst load, type of alcohol, and vanadium load on conversion and selectivity of the reactions were investigated. Remarkably, while in methyl alcohol, only acetal was formed, in the presence of other alkyl alcohols terpene ethers (geranyl and β-citronellyl) were also obtained. Their highest activity of H4PMo11VO40 was attributed to the greatest Brønsted acidity strength, as demonstrated by the acidity measurements and infrared spectroscopy analysis. This catalyst has advantages over traditional liquid mineral acid catalysts and provides an alternative route to synthesize acetal and terpene ethers.
Graphical Abstract









Similar content being viewed by others
References
Plößer J, Lucas M, Claus P (2014) J Catal 320:189–197. https://doi.org/10.1016/j.jcat.2014.10.007
Bhatia SP, Ginty DMC, Letizia CS, Api AM (2008) Food Chem Toxicol 46:S209–S214. https://doi.org/10.1016/j.fct.2008.06.059
Tsolakis N, Bam W, Srai JS, Kumar M (2019) J Clean Prod 222:802–822. https://doi.org/10.1016/j.jclepro.2019.02.108
Gallezot P (2012) Chem Soc Rev 41:1538–1558. https://doi.org/10.1039/C1CS15147A
Lenardão EJ, Botteselle GV, de Azambuja F, Perin G, Jacob RG (2007) Tetrahedron 63:6671–6712. https://doi.org/10.1016/j.tet.2007.03.159
Wu L, Moteki T, Gokhale AA, Flaherty DW, Toste FD (2016) Chem 1:32–58. https://doi.org/10.1016/j.chempr.2016.05.002
Sanchez LM, Thomas HJ, Climent MJ, Romanelli GP, Iborra S (2016) Catal Rev 58(2016):497–586. https://doi.org/10.1080/01614940.2016.1248721
da Silva MJ, Liberto NA, Leles LCA, Pereira UA (2016) J Mol Catal A 422:69–83
Roelofs JCAA, van Dillen AJ, de Jong KP (2001) Catal Lett 74:91–94. https://doi.org/10.1023/A:1016626521403
Chaves DM, Ferreira SO, da Silva RC, Natalino R, da Silva MJ (2019) Energ Fuel 33:7705–7716. https://doi.org/10.1021/acs.energyfuels.9b01583
da Silva MJ, Chaves DM, Ferreira SO, da Silva RC, Gabriel Filho JB, Bruziquesi CGO, Al-Rabiah AA (2022) Chem Eng Sci 247:116913. https://doi.org/10.1016/j.ces.2021.116913
Dong J-L, Yu L-S, Yu H, Xie J-W (2018) ACS Omega 3:4974–4985. https://doi.org/10.1021/acsomega.8b00159
Hamada N, Kazahaya K, Shimizu H, Sato T (2004). Synlett. https://doi.org/10.1055/s-2004-820038
Corma A, García H (2003) Chem Rev 103:4307–4366. https://doi.org/10.1021/cr030680z
Wegenhart BL, Liu S, Thom M, Stanley D, Abu-Omar MM (2012) ACS Catal 2:2524–2530. https://doi.org/10.1021/cs300562e
Umbarkar SB, Kotbagi TV, Biradar AV, Pasricha R, Chanale J, Dongare MK, Mamede A-S, Lancelot C, Payen E (2009) J Mol Catal A 310:150–158. https://doi.org/10.1016/j.molcata.2009.06.010
Hartati PBD, Firda HB, Bakar MB (2021) Flavour Fragr J 36:509–525. https://doi.org/10.1002/ffj.3671
Serafim H, Fonseca IM, Ramos AM, Vital J, Castanheiro JE (2011) Chem Eng J 178:291–296. https://doi.org/10.1016/j.cej.2011.10.004
Anaç O, Talinli N (2010) Bull Des Soc Chim Belg 102:79–87. https://doi.org/10.1002/bscb.19931020203
Rubio-Caballero JM, Saravanamurugan S, Maireles-Torres P, Riisager A (2014) Catal Today 234:233–236. https://doi.org/10.1016/j.cattod.2014.03.004
Kopa ID, Barakov RY, Sotnik SO, Shcherban ND (2022) Mater Today Proc 62:7686–7690. https://doi.org/10.1016/j.matpr.2022.03.146
da Silva MJ, Teixeira MG, Natalino R (2019) New J Chem 43:8606–8612. https://doi.org/10.1039/C9NJ01284B
Teixeira MG, Natalino R, da Silva MJ (2020) Catal Today 344:143–149. https://doi.org/10.1016/j.cattod.2018.11.071
Silva MJ, Andrade da Silva PH, Ferreira SO, Silva RC, Brusiquezi CGO (2022). ChemiSelect. https://doi.org/10.1002/slct.202104174
Wang S-S, Yang G-Y (2015) Chem Rev 115:4893–4962. https://doi.org/10.1021/cr500390v
López X, Carbó JJ, Bo C, Poblet JM (2012) Chem Soc Rev 41:7537. https://doi.org/10.1039/c2cs35168d
Vilanculo CB, da Silva MJ (2021) RSC Adv 11:34979–34987. https://doi.org/10.1039/D1RA06718D
Coronel NC, da Silva MJ (2018) J Clust Sci 29:195–205. https://doi.org/10.1007/s10876-018-1343-0
da Silva MJ, Liberto NA (2016) Curr Org Chem 20:1263–1283. https://doi.org/10.2174/1385272819666150907193100
da Silva MJ, Lopes NPG, Ferreira SO, da Silva RC, Natalino R, Chaves DM, Texeira MG (2021) Chem Papers 75:153–168. https://doi.org/10.1007/s11696-020-01288-x
Batalha DC, Ferreira SO, da Silva RC, da Silva MJ (2020) ChemSelect 5:1976–1986
Vilanculo CB, da Silva MJ (2020) New J Chem 2020:2813–2820
da Silva MJ, da Silva Andrade PH, Sampaio VFC (2021) Catal Lett 151:2094–2106. https://doi.org/10.1007/s10562-020-03449-9
da Silva MJ, de Oliveira CM (2018) Curr Catal 7:26–34. https://doi.org/10.2174/2211544707666171219161414
Mizuno N, Kamata K (2011) Coord Chem Rev 255:2358–2370. https://doi.org/10.1016/j.ccr.2011.01.041
Shatalov AA (2019) Carbohydr Polym 206:80–85. https://doi.org/10.1016/j.carbpol.2018.10.106
Villabrille P, Romanelli G, Vázquez P, Cáceres C (2004) Appl Catal A 270:101–111. https://doi.org/10.1016/j.apcata.2004.04.028
Barteau KP, Lyons JE, Song IK, Barteau MA (2006) Top Catal 41:55–62. https://doi.org/10.1007/s11244-006-0094-6
Lee JK, Melsheimer J, Berndt JS, Mestl G, Schlögl R, Köhler K (2001) Appl Catal A 214:125–148. https://doi.org/10.1016/S0926-860X(01)00485-9
Vilanculo CB, da Silva MJ, Rodrigues AA, Ferreira SO, da Silva RC (2021) RSC Adv 11:24072–24085. https://doi.org/10.1039/D1RA04191F
da Silva MJ, Ribeiro CJA, Vilanculo CB (2022). Catal Lett. https://doi.org/10.1007/s10562-022-04132-x
Vilanculo CB, da Silva MJ (2021) Mol Catal 512:111780. https://doi.org/10.1016/j.mcat.2021.111780
Jing F, Katryniok B, Dumeignil F, Bordes-Richard E, Paul S (2014) J Catal 309(2014):121–135. https://doi.org/10.1016/j.jcat.2013.09.014
Tsigdinos GA, Hallada CJ (1968) Inorg Chem 7:437–441. https://doi.org/10.1021/ic50061a009
Chen CY, Li HX, Davis ME (1993) Microporous Mat 2:17–26
Pizzio LR, Blanco MN (2007) Microporous Mesoporous Mat 103:40–47. https://doi.org/10.1016/j.micromeso.2007.01.036
Serwicka EM, Bruckman K, Haber J, Paukshtis EA, Yurchenko EN (1991) Appl Catal 73:153–163. https://doi.org/10.1016/0166-9834(91)85133-G
Acknowledgements
The authors are grateful for the financial support from CNPq and FAPEMIG (Brasil). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Silva, M.J., Ribeiro, C.J.A. & Rodrigues, A.A. H4PMo11VO40-Catalyzed β-Citronellal Condensation Reactions. Catal Lett 153, 3829–3836 (2023). https://doi.org/10.1007/s10562-023-04274-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-023-04274-6