Abstract
Periodic and molecular density functional theory calculations have been applied to elucidate the associative mechanism for hydrazine and ammonia synthesis in the gas phase and hydrazine formation on Co3Mo3N. We find that there are two activation barriers for the associative gas phase mechanism with barriers of 730 and 658 kJ/mol, corresponding to a hydrogenation step from N2 to NNH2 and H2NNH2 to H3NNH3, respectively. The second step of the mechanism is barrierless and an important intermediate, NNH2, can also readily form on Co3Mo3N surfaces via the Eley–Rideal chemisorption of H2 on a pre-adsorbed N2 at nitrogen vacancies. Based on this intermediate a new heterogeneous mechanism for hydrazine synthesis is studied. The highest relative barrier for this heterogeneous catalysed process is 213 kJ/mol for Co3Mo3N containing nitrogen vacancies, clearly pointing towards a low-energy process for the synthesis of hydrazine via a heterogeneous catalysis route.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The development of alternative routes to convert N2 to ammonia (NH3) and hydrazine (H2NNH2) could have significant economic and environmental impact, since more that 50% of ammonia for soil fertilisers is produced industrially and the global annual production of hydrazine is more than 80 thousand tons [1]. Ammonia is industrially produced mostly via the classical Haber–Bosch (H–B) process [2,3,4], by a Fe–K2O–Al2O3 catalyst under high temperatures (>400 °C) and pressures (150–200 atm). Some production has shifted towards the Kellogg advanced ammonia process, which uses a graphite-supported alkali/alkaline-earth promoted Ru catalyst and operates at milder conditions [5].
Hydrazine is produced by the Bayern ketazine process, which is a variation of the Olin Raschig process [6]. The two main reactions of this process are the formation and hydrolysis of 1,2-di(propan-2-ylidene)hydrazine (i.e. acetone azine),
In brief, in this process ammonia is oxidised by sodium hypochlorite in acetone to form acetone azine (ketazine) which in the presence of water decomposes to hydrazine and acetone. A by product of the reaction is sodium chloride. The details of this process are: sodium hypochlorite (NaOCl) solution, ammonia (NH3) and acetone (Me2CO) are mixed at 35 °C. A solution results consisting of about 7% by weight acetone azine together with sodium chloride and excess ammonia. Ammonia is distilled off and returned to the reaction. The acetone azine-water-azeotrope (b.p. 95 °C) is distilled off leaving the sodium chloride solution. The acetone azine is hydrolyzed with water at temperatures up to 180 °C and pressures of 8–12 bar into an acetone and an aqueous hydrazine solution. The hydrazine solution is then concentrated by distillation.
Co3Mo3N is one of the most active catalysts for ammonia synthesis at mild conditions, especially when doped with caesium [7,8,9,10,11]. In two recent density functional theory (DFT) studies we have identified possible sites for the adsorption and activation of the reactants of the ammonia synthesis reaction on a model Co3Mo3N surface, with heterogeneity due to surface nitrogen vacancies [12]. Such vacancies are present in large concentrations even at ambient temperatures (i.e. ~1013 cm−2) and can efficiently activate N2 [13]. In particular, we found that N2 can be activated at a surface cavity where N2 is bound side-on a Co8 cluster at the 16e Wyckoff site or it can be activated at surface 3f-nitrogen vacancies where N2 is activated in an end-on configuration [12]. There are other DFT studies that have shown that N-vacancies can participate in the mechanism for ammonia synthesis, such as in the electrochemical reduction of ammonia on Zr, Nb, Cr, V mononitrides [14,15,16] and in the two-phase solar-energy driven ammonia synthesis on metal-nitrides [17, 18]. End-on activated N2 also exists in Fe(0) complexes (i.e. Fe(Et2PCH2CH2PEt2)2(N2)) found to be selective for the formation of hydrazine from N2 with NH3 forming at a ratio of 25:1 [19]. These complexes were initially designed to resemble the binding of N2 to FeMo-cofactor [20], the enzyme responsible for the biological fixation of nitrogen in nitrogenases, for which further atomistic details on the mechanism have recently been reported [21].
To assist our understanding of the hydrazine synthesis reaction, we have first modelled the reaction mechanism for associative synthesis of hydrazine and ammonia in the gas phase, via hybrid DFT calculations and also modelled how some of the intermediates can be formed on the surface of Co3Mo3N. The results of this paper are presented in the following order. First we present the various hydrogenated intermediates of the hydrazine and ammonia synthesis reaction in the gas phase. Then we present a gas-phase, free energy diagram that explores the kinetic pathway for hydrazine and ammonia synthesis. Lastly we present a new mechanism that shows how hydrazine could be synthesized over a heterogeneous catalyst (i.e. Co3Mo3N).
2 Methods and Models
Molecular open and closed-shell DFT computations have been perfomed with Gaussian 09 [22] with the use of Becke’s three-parameter hybrid exchange functional [23] (XC) combined with the Lee–Yang–Parr non-local correlation functional [24], abbreviated as B3LYP. For the basis functions we have used the spherical version (i.e. 5d, 7f), the correlation consistent augmented valence triple zeta basis set [25,26,27,28,29], abbreviated as aug-cc-pVTZ.
The potential free energy diagrams were obtained by plotting the formation Gibbs free energy change \({({\Delta} G}_{f,PES})\) of the various intermediates and transitions states with respect to gas phase reactants, given by the following expression,
In the computed free energy changes we have included the entropy changes for gas phase molecules the vibrational energy of the adsorbates bound to a single metal atom, therefore entropy changes for the solid phase have been omitted. These were calculated at P = 1 atm and T = 298.15 K. The equation from which the vibrational energies of the adsorbates was calculated is given in the supporting information file. Intermediates and transition states (TS) have been confirmed using vibrational analysis, by the absence and presence of one imaginary vibrational frequency, respectively. Transition state structures were either located using the Synchronous Transit-Guided Quasi-Newton (STQN) method of Schlegel and co-workers [30, 31] or by scanning a particular bond length at a 0.05 Å resolution and relaxing the remaining atoms. The imaginary frequency of the located TS was animated to ensure that it corresponds to the desired reaction coordinate. Every structure was calculated at a spin multiplicity (s.m.) of 1, 3 and 5, but only the lowest energy structure is reported. Calculations were checked for spin contamination, which was found to be negligible. In most cases, the structure with s.m. = 1 had the lowest energy. The molecular structure, point group (p.g.) symmetry, vibrational streching frequency (νN−N), bond length (rN−N), spin multiplicity and formation energy (ΔGf) of the various NxHy intermediates are shown in Table 1. The table is partioned to show the various stable hydrogenation intermediates NxHy that were found. These can assist infrared spectroscopic efforts in identifying the intermediates in the gas phase reaction mechanism. The intermediate of interest, F, could be identified through IR spectroscopy as its νN−N differs by more than 400 cm−1 from that of isomeric G, H and I. Furthermore, Table 1 lists the computed free energies of formation of the various intermediates at P = 1 atm and T = 298.15 K. These formation energies show the relative trends of the stability of the various intermediates of the same molecular weight and are therefore useful in the experimental isolation of such intermediates during the synthesis of ammonia and hydrazine. We find good agreement between the computed formation free energy of ammonia −23 kJ/mol with the one listed in thermeodynamic tables (i.e. CRC Handbook of Chemistry and Physics) −27 kJ/mol, indicative that a relatively accurate computational methodology has been followed.
Periodic calculations were performed with the VASP code with a 650 eV cutoff for the planewave expansion and the revPBE XC functional. A 4 × 4 × 1 Γ-point centered MP grid for the 2 × 2 surface supercell with one 3f-hollow nitrogen vacancy and a vacuum gap of 20 Å was used. All calculations were spin-polarised.
3 Results and Discussion
3.1 Hydrazine and Ammonia Synthesis in Gas Phase
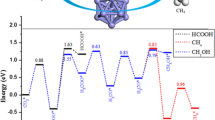
We have studied the associative mechanism for ammonia and hydrazine synthesis, where N2 does not dissociate but rather reacts directly with H2, which necessitates very high pressure rather than thermal activation of the reactants. This mechanism of ammonia synthesis in the gas phase can be directly compared to the associative mechanism occurring on the (111) surface of Co3Mo3N. The resulting free energy diagram for the associative mechanism of ammonia synthesis in the gas phase is shown in Fig. 1, where we only present the kinetic pathway (the pathway which has the lowest barrier for its rate-determining step (RDS)). We note that there is also another mechanism which proceeds via monotamic hydrogen atoms and provides an energetically viable alternative as a result of the high activation barrier of the first hydrogenation step (730 kJ/mol) for the associative mechanism which based on the bond-dissociation enthalpy (BDE) of H2 would cause the H–H bond to break before the formation of intermediate N2H2 (F). However, we only consider the associative mechanism due to its relevance to the reaction occurring on the Co3Mo3N surface. We find that for the gas phase reaction, the various mechanistic pathways generally consist of three activated steps, where each step corresponds to a hydrogenation reaction of H2 with N2 forming N2(H2)n (n = 0, 1, 2) intermediates. For the kinetic pathway only the first and the last hydrogenation steps have considerably high barriers, whereas in the second the barrier was essentially absent. The kinetic pathway was determined on the basis that its RDS had the lowest barrier among the four possible associative pathways studied. We find that the RDS step for ammonia synthesis in the gas phase (without a catalyst) has a barrier of 730 kJ/mol, which is followed by a barrier of 658 kJ/mol which corresponds to the third hydrogenation step. From the relative barrier heights of the 1st and 3rd hydrogenation steps in Fig. 1 it is suggested that based on an associative mechanism, there would be some hydrazine formation during ammonia synthesis. This has been previously observed in glow discharge reactions of N2 and H2 over various solid surfaces (e.g. SiO2, TiO2, MoO3, Al2O3, MgO, Mg(OH)2) having a zeroth order dependence on the reactants total pressure, producing 4–28% hydrazine and 72–96% ammonia [37]. The 658 kJ/mol barrier could be overcome if the O isomer of hydrazine radiatively undergoes isomerization through intermediate N (see Table 1), which brings the hydrazine intermediate N to the right stereochemical structure to undergo subsequent hydrogenation to form H3NNH3 (S). We have calculated that this requires far-infrared radiation of a wavelength of 2842 cm−1. S thermally decomposes to form two ammonia molecules in a barrierless process. The intriguing aspect of this mechanism is that the 2nd hydrogenation step is barrierless, which corresponds to the adsorption of molecular hydrogen on the hydrogen pre-activated intermediate N2H2 (F). Interestingly we also found that this intermediate can form readily on (111)-surfaces of Co3Mo3N, at nitrogen vacancy sites. This intermediate in the gas phase reaction mechanism requires 730 kJ/mol to form but only 107 kJ/mol at the nitrogen vacancies on Co3Mo3N. We seek this intermediate and the mechanism for hydrazine synthesis in the associative mechanism for ammonia synthesis in the following section.
Gibbs free energy diagram for associative hydrazine and ammonia synthesis mechanism in gas phase calculated at B3LYP/aug-cc-pVTZ (5d, 7f). Free energies of formation of the various intermediates and transition states have been calculated using Eq. 1 and are tabulated in Table 1 at P = 1 atm and T = 298.15 K
3.2 Hydrazine Synthesis on Co3Mo3N
In this study we have found that intermediate F on Co3Mo3N can form at nitrogen vacancy sites, similar to its formation on Mo = NNH2 [32] and Fe-NNH2 [33] complexes and in a DFT study of the Schrock cycle molybdenum complex [Mo(hiptN3N)] [34]. It is noted that this activation site of Co3Mo3N shows some similarities with the FeMo-cofactor with the main difference the bridging-N rather than bridging-S sites. A detailed study of the various adsorption sites on Co3Mo3N surfaces is given in Ref. [13]. The D3-corrected adsorption energy of N2 is moderately exothermic, evidence that the adsorption of N2 at the nitrogen vacancies can occur at ambient temperatures. N2 in this position is activated by 11%, based the adsorption induced N–N bond lengthening. Once N2 is adsorbed to the surface in an end-on configuration it reacts with H2 coming from the gas phase by an activated step of only 107 kJ/mol in order to form intermediate X. The bonding of intermediate X resembles intermediate L in the gas phase mechanism. The optimal molar ratio of N2:H2 in the gas feed-stream is found to be 1:1, in order manifest N2 adsorption at nitrogen vacancies and H2 co-adsorption at Co8 clusters [13]. This prohibits poisoning of the nitrogen activation sites by chemisorbed hydrogen although we have previously shown via DFT that even at ambient T, there are more than 1013 nitrogen vacancies per cm2 which can act as active sites for nitrogen activation [12]. The energy required to generate nitrogen vacancies, the vacancy formation energy (VFE) was found to be 162 kJ/mol for three-fold (3f) bound nitrogen. This clearly suggests that if the reaction is run with a net input of energy of 213 kJ/mol (see barrier for desorption of intermediate X from the nitrogen vacancy in Fig. 2) then nitrogen activation sites will constantly be present and replenished even if some hydrogen adsorption occurs at these sites, whose adsorption is generally more exothermic than nitrogen chemisorption. Once intermediate X is formed on the catalyst surface, from the free energy diagram shown in Fig. 2, we see that the energy required to achieve its desorption is relatively high but still much lower than the barrier of the gas phase mechanism (730 kJ/mol). Therefore some decomposition of intermediate X back to the reactants N2 and H2, should be expected as this is observed also over MoNx/γ–Al2O3 [35] and activated carbon supported tungsten carbide [36]. Once NNH2 is in the gas phase (i.e. intermediate F, a small desorption barrier has been ignored) it reacts in a barrierless process with H2(g) as shown in Fig. 2. It is noted that NNH2 can also undergo surface hydrogenation reactions which lead to the formation of ammonia rather then hydrazine. Through this newly suggested mechanism the synthesis of hydrazine is possible on Co3Mo3N with a relative barrier of the RDS of only 213 kJ/mol, which is 517 kJ/mol lower than the gas phase associative mechanism for hydrazine synthesis. It is therefore suggested that this Eley–Rideal synthesis of hydrazine should be tested under the conditions described with the Co3Mo3N catalyst.
Gibbs free energy diagram of heterogeneous mechanism occurring on Co3Mo3N in the presence of nitrogen vacancies (black square). XC: revPBE (650 eV) for the surface mechanism and B3LYP/aug-cc-pVTZ for the gas phase steps. The choice of these XC functionals is justified based on the mean-average-percent-error that various GGA and hybrid-GGA functionals have in evaluating bond-dissociation enthalpies in the supporting information of Ref. [12]. P = 1 atm and T = 298.15 K. Structures of the surface reactions on Co3Mo3N are shown in Fig. 3
To summarise, our DFT study shows that hydrazine can be formed with relatively small energy input (213 kJ/mol) on Co3Mo3N-(111) surfaces, compared to the gas phase mechanism which requires 730 kJ/mol for the associative mechanism. The free energy diagrams presented are critical for the design of new experiments to show the possibility of hydrazine synthesis from N2 and H2. The infrared absorption frequencies presented of the various ammonia synthesis intermediates are necessary for spectroscopic identification of the reaction intermediates. We therefore anticipate that this study will initiate experimental efforts to establish a heterogeneous catalytic route for the synthesis hydrazine.
References
Schirmann J-P, Bourdauducq P (2002) Hydrazine. In: Ullmann’s encyclopedia of industrial chemistry. Wiley, Weinheim
Haber F (1922) Naturwissenschaften 10:1041–1049
Haber F (1923) Naturwissenschaften 11:339–340
Smil V (2004) Enriching the earth: Fritz Haber, Carl Bosch, and the transformation of world food production. MIT Press, Cambridge
Saadatjou N, Jafari A, Sahebdelfar S (2015) Chem Eng Commun 202(4):420–448
Ritz J, Fuchs H, Perryman HG (2000) Hydroxylamine. Ullmann’s Encyclop Ind Chem
Boisen A, Dahl S, Jacobsen CJH (2002) J Catal 208(1):180–186
Jacobsen CJH (2000) Chem Commun 12:1057–1058
Jacobsen CJH, Dahl S, Clausen BS, Bahn S, Logadottir A, Nørskov J (2001) J Am Chem Soc 123:8404–8405
Kojima R, Aika K-I (2000) Chem Lett 29(5):514–515
Kojima R, Aika K-I (2001) Appl Catal A 215:149–160
Zeinalipour-Yazdi CD, Hargreaves. JSJ, Catlow CRA (2015) J Phys Chem C 119:28368–28376
Zeinalipour-Yazdi CD, Hargreaves JSJ, Catlow CRA (2016) J Phys Chem C 120:21390–21398
Abghoui Y, Garden AL, Hlynsson VF, Bjorgvinsdottir S, Olafsdottir H, Skulason E (2015) Phys Chem Chem Phys 17:4909–4918
Abghoui Y, Garden AL, Howat JG, Vegge T, Skulason E (2016) ACS Catal 6:635–646
Abghoui Y, Skúlason E (2016) Catal Today 6(2):635–646
Michalsky R, Pfromm PH, Steinfeld A (2015) Interface Focus 5:20140084
Michalsky R, Avram AM, Peterson BA, Pfromm PH, Peterson AA (2015) Chem Sci 6:3965–3974
Hill PJ, Doyle LR, Crawford AD, Myers WK, Ashley AE (2016) J Am Chem Soc 138(41):13521–13524
Hoffman BM, Dean DR, Seefeldt LC (2009) Acc Chem Res 42:609–619
Siegbahn PEM (2016) J Am Chem Soc 138(33):10485–10495
Gaussian 09 RC, Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA et al (2009) Gaussian Inc., Wallingford CT
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Woon DE, Dunning TH Jr (1993) J Chem Phys 98:1358–1371
Wilson A, van Mourik T, Dunning TH Jr (1997) J Mol Struct 388:339–349
Peterson KA, Woon DE, Dunning TH Jr (1994) J Chem Phys 100:7410–7415
Kendall RA, Dunning TH Jr, Harrison RJ (1992) J Chem Phys 96:6796–6806
Dunning TH Jr (1989) J Chem Phys 90:1007–1023
Peng C, Schlegel HB (1993) Isr J Chem 33:449–454
Peng C, Ayala PY, Schlegel HB, Frisch MJ (1996) J Comput Chem 17:49–56
Rittle J, Peters JC (2016) J Am Chem Soc 138(12):4243–4248
Del Castillo TJ, Thompson NB, Peters JC (2016) J Am Chem Soc 138(16):5341–5350
Studt F, Tuczek F (2005) Angew Chem Int Ed 44:5639–5642
Chen X, Zhang T, Xia L, Li T, Zheng M, Wu Z, Wang X, Wei Z, Xin Q, Li C (2002) Catal Lett 79:21–25
Sun J, Zheng M, Wang X, Wang A, Cheng R, Li T, Zhang T (2008) Catal Lett 123:150–155
Miyahara K (1984) J Res Inst Catal 32:1–18
Acknowledgements
We are grateful for useful discussions with Dr. Justin Hargreaves.
Funding
No competing financial interests have been declared. The authors acknowledge EPSRC funding (EP/L02537X/1, EP/L000202/1).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zeinalipour-Yazdi, C.D., Catlow, C.R.A. A Computational Study of the Heterogeneous Synthesis of Hydrazine on Co3Mo3N. Catal Lett 147, 1820–1826 (2017). https://doi.org/10.1007/s10562-017-2080-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-017-2080-y