Abstract
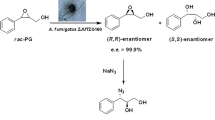
l-2-Aminobutyric acid (l-ABA), an unnatural amino acid, is a key intermediate of several important drugs. Although some methods have been developed to prepare pure chiral l-ABA, there are still many drawbacks, including low catalytic efficiency, cumbersome steps and high cost due to the addition of some expensive catalysts or coenzymes. Herein, with chemical and biological approaches together, we discovered a newly isolated Aspergillus tamarii ZJUT ZQ013 strain containing a microbial lipase which could be employed to resolve racemic methyl N-Boc-2-aminobutyrate to produce l-ABA with high enantioselectivity (e.e.s > 99.9%, E = 257). Moreover, the subsequent gram scale experiment confrimed that A. tamarii ZJUT ZQ013 could be an attractive biocatalyst for the efficient preparation of optically pure acid.
Graphical Abstract








Similar content being viewed by others
References
Taylor PP, Pantaleone DP, Senkpeil RF, Fotheringham IG (1998) Novel biosynthetic approaches to the production of unnatural amino acids unnatural amino acids using transaminases. Trends Biotechnol 16:412–418
Zhu L, Tao RS, Wang Y et al (2011) Removal of L-alanine from the production of l-2-aminobutyric acid by introduction of alanine racemase and d-amino acid oxidase. Appl Microbiol Biotechnol 90(3):903–910
Shin JS, Kim BG (2009) Transaminase-catalyzed asymmetric synthesis of L-2-aminobutyric acid from achiral reactants. Biotechnol Lett 31(10):1595–1599
Futagawa T, Canvat JP, Cavoy E, et al. Process for the preparation of levetiracetam. US Patent 6,107,492, 22 Aug 2000
Ragonese R, Macka M, Hughes J et al (2002) The use of the Box-Behnken experimental design in the optimisation and robustness testing of a capillary electrophoresis method for the analysis of ethambutol hydrochloride in a pharmaceutical formulation. J Pharm Biomed Anal 27(6):995–1007
Jefery E A, Meisters A (1978) Electrochemical synthesis of amino acids by reductive amination of keto acids. I. Reduction at mercury electrodes. Aust J Chem 31(1):79–84
Babievskii KK, Belikov VM, Tikhonova NA (1965) Synthesis of D, L—threonine and D, L-2-aminobutyrie acid based on condensations of nitroacetie ester. Russ Chem Bull, 14(1):76–81
Chiyuki F. Process for production of (+)-2-amino-1-butanol. US Patent 3,979,457, 07 Sept 1976
Tao RS, Jiang Y, Zhu FY, Yang S (2014) A one-pot system for production of L-2-aminobutyric acid from L-threonine by L-threonine deaminase and a NADHregeneration system based on L-leucine dehydrogenase and formate dehydrogenase. Biotechnol Lett 36:835–841
Seo YM, Mathew S, Bea HS, Khang YH, Lee SH, Kim BG, Yun H (2012) Deracemization of unnatural amino acid: homoalanine using D-amino acid oxidase and omega-transaminase. Org Biomol Chem 10(12):2482–2485
Fotheringham IG, Grinter N, Pantaleone DP, Senkpeil RF, Taylor PP (1999) Engineering of a novel biochemical pathway for the biosynthesis of L-2-aminobutyric acid in Escherichia coli K12. Bioorg Med Chem 7:2209–2213
Alvaro G, Decor A, Fontana S, Hamprecht D, Large C, Marasco A. Imidazolidinedione derivatives. WO 2011069951 A1, 16 July 2011
Brenner M, Vecchia LL, Leutert T, Seebach D (2003) (4 S)-4- (1-methylethyl)-5,5- diphenyl- 2-oxazolidinone[(2-oxazolidinone,4-(1-methylethyl)-5,5- diphenyl-, (4 S)-)]. Organ Synth 80:57–65.
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Xu F, Deng G, Cheng S et al (2012) Molecular cloning, characterization and expression of the phenylalanine ammonia-lyase gene from Juglans Regia. Molecules 17(7):7810–7823
Tamura K, Peterson D, Peterson N et al (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Chen CH, Sih CJ (1989) General aspects and optimization of enantioselective biocatalysis in organic solvents: the use of lipases. Angew Chem Int Ed 28(6):695–707
Baldwin BG (1992) Phylogenetic utility of the internal transcribed spacers of nuclear ribosomal DNA in plants: an example from the compositae. Mol Phylogenet Evol 1(1):3–16
Dosanjh NS, Kaur J (2002) Immobilization, stability and esterification studies of a lipase from a Bacillus sp. Appl Biochem 36(1):7–12
Dong HP, Wang YJ, Zheng YG (2010) Enantioselective hydrolysis of diethyl 3-hydroxyglutarate to ethyl (S)-3-hydroxyglutarate by immobilized Candida antarctica lipase B. J Mol Catal B Enzym 66(1):90–94
Long WS, Kow PC, Kamaruddin AH (2005) Comparison of kinetic resolution between two racemic ibuprofen esters in an enzymic membrane reactor. Process Biochem 40(7):2417–2425
Guo JL, Mu XQ, Xu Y (2010) Integration of newly isolated biocatalyst and resin-based in situ product removal technique for the asymmetric synthesis of (R)-methyl mandelate. Bioproc Biosyst Eng 33(7):797–804
Zhang Z, Sheng Y, Jiang K (2010) Bio-resolution of glycidyl (o,m,p)-methylphenyl ethers by Bacillus megaterium. Biotechnol Lett 32:513–516
Chang CS, Ho SC (2011) Enantioselective esterification of (R, S)-2-methylalkanoic acid with Carica papaya lipase in organic solvents. Biotechnol Lett 33(11):2247–2253
Acknowledgements
This work was supported by National Science Foundation of China (Nos. 21272212, 21472172) and Zhejiang Natural Science Fund (Nos. LY17B060009, LY12B02018), Project of Science Technology Department of Zhejiang Province (2014C33141), and Project of Department of Science Technology Jinhua City (2013-3-003).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
An, Z., Gu, X., Liu, Y. et al. Bioproduction of l-2-Aminobutyric Acid by a Newly-Isolated Strain of Aspergillus tamarii ZJUT ZQ013. Catal Lett 147, 837–844 (2017). https://doi.org/10.1007/s10562-017-1999-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-017-1999-3