Abstract
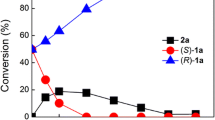
The enantioselective reduction of methyl benzoylformate to (R)-methyl mandelate, an important pharmaceutical intermediate and a versatile resolving agent, was investigated in this study. After minimizing the reaction-specific constraints (constraints dependent on the nature of the substrate and product) by preliminary selection of the reaction parameters, an effective whole cell biocatalyst (Saccharomyces cerevisiae AS2.1392) was obtained by simple screening procedures. Under further optimized conditions, a product concentration of 103 mmol L−1 could be attained within 5 h with a yield of 85.8% and an enantiometric excess of 95.4%, indicating S. cerevisiae AS2.1392 an efficient biocatalyst for the asymmetric synthesis of (R)-methyl mandelate. Furthermore, resin-based in situ product removal (ISPR) technique was applied to alleviate the substrate and product inhibition or toxicity to the whole cells. The integration of newly isolated biocatalyst and proper ISPR technique provides a practical route for the preparation of optically active pharmaceutical intermediates.






Similar content being viewed by others
References
Breuer M, Ditrich K, Habicher T, Hauer B, Kesseler M, Sturmer R, Zelinski T (2004) Industrial methods for the production of optically active intermediates. Angew Chem Int Ed 43:788–824
Coppola GM, Schuster HF (2003) Mandelic acid. In: Walter G (ed) Alpha-hydroxy acids in enantioselective syntheses, 2nd edn. Wiley, New York, pp 137–165
Furlenmeier A, Quitt P, Vogler K, Lanz P (1976) 6-Acyl derivatives of aminopenicillanic acid. US Patent 3,957,758
Bichel H, Kocsis K, Peter H (1978) Cephalosporins having an alpha-acyloxyacetic acid side chain. US Patent 4,067,979
Surivet JP, Vatele JM (1999) Total synthesis of antitumor Goniothalamus styryllactones. Tetrahedron 55:13011–13028
Mills J, Schmiegel KK, Shaw WN (1983) Phenethanolamines, compositions containing the same, and method for effecting weight control. US Patent 4,391,826
Kinbara K, Sakai K, Hashimoto Y, Nohira H, Saigo K (1996) Design of resolving reagents: p-substituted mandelic acids as resolving reagents for 1-arylalkylamines. Tetrahedron Asymmetry 7:1539–1542
Kesslin G, Kelly KW (1982) Resolution of racemic mandelic acid. US Patent 4,322,548
Yang JW, List B (2006) Catalytic asymmetric transfer hydrogenation of alpha-ketoesters with Hantzsch esters. Org Lett 8:5653–5655
Schoemaker HE, Mink D, Wubbolts MG (2003) Dispelling the myths—biocatalysis in industrial synthesis. Science 299:1694–1697
Pollard DJ, Woodley JM (2007) Biocatalysis for pharmaceutical intermediates: the future is now. Trends Biotechnol 25:66–73
Strauss UT, Faber K (1999) Deracemization of (±)-mandelic acid using a lipase-mandelate racemase two-enzyme system. Tetrahedron Asymmetry 10:4079–4081
Groger H (2001) Enzymatic routes to enantiomerically pure aromatic alpha-hydroxy carboxylic acids: A further example for the diversity of biocatalysis. Adv Synth Catal 343:547–558
He YC, Xu JH, Pan J, Ouyang LM, Xu Y (2008) Preparation of (R)-(−)-mandelic acid and its derivatives from racemates by enantioselective degradation with a newly isolated bacterial strain Alcaligenes sp. ECU0401. Bioprocess Biosyst Eng 31:445–451
Nakamura K, Yamanaka R, Matsuda T, Harada T (2003) Recent developments in asymmetric reduction of ketones with biocatalysts. Tetrahedron Asymmetry 14:2659–2681
Schmid A, Dordick JS, Hauer B, Kiener A, Wubbolts M, Witholt B (2001) Industrial biocatalysis today and tomorrow. Nature 409:258–268
Kayser MM, Mihovilovic MD, Kearns J, Feicht A, Stewart JD (1999) Baker’s yeast-mediated reductions of alpha-keto esters and an alpha-keto-beta-lactam. Two routes to the paclitaxel side chain. J Org Chem 64:6603–6608
Milagre HMS, Milagre CDF, Moran PJS, Santana MHA, Rodrigues JAR (2005) Reduction of ethyl benzoylformate mediated by Saccharomyces cerevisiae entrapped in alginate fibers with double gel layers in a continuously operated reactor. Enzyme Microb Technol 37:121–125
Kratzer R, Egger S, Nidetzky B (2008) Integration of enzyme, strain and reaction engineering to overcome limitations of baker’s yeast in the asymmetric reduction of alpha-keto esters. Biotechnol Bioeng 101:1094–1101
Chen YZ, Xu JG, Xu XY, Xia Y, Lin H, Xia SW, Wang LX (2007) Enantiocomplementary preparation of (S)- and (R)-mandelic acid derivatives via alpha-hydroxylation of 2-arylacetic acid derivatives and reduction of alpha-ketoester using microbial whole cells. Tetrahedron Asymmetry 18:2537–2540
He CM, Chang DL, Zhang J (2008) Asymmetric reduction of substituted alpha- and beta-ketoesters by Bacillus pumilus Phe-C3. Tetrahedron Asymmetry 19:1347–1351
Ema T, Ide S, Okita N, Sakai T (2008) Highly efficient chemoenzymatic synthesis of methyl (R)-o-chloromandelate, a key intermediate for clopidogrel, via asymmetric reduction with recombinant Escherichia coli. Adv Synth Catal 350:2039–2044
Kratzer R, Pukl M, Egger S, Nidetzky B (2008) Whole-cell bioreduction of aromatic alpha-keto esters using Candida tenuis xylose reductase and Candida boidinii formate dehydrogenase co-expressed in Escherichia coli. Microb Cell Fact 7:12
Hilker I, Gutierrez MC, Furstoss R, Ward J, Wohlgemuth R, Alphand V (2008) Preparative scale Baeyer–Villiger biooxidation at high concentration using recombinant Escherichia coli and in situ substrate feeding and product removal process. Nat Protoc 3:546–554
Xu MW, Mu XQ, Xu Y (2009) Enhancement of substrate concentration in microbial stereoinversion through one-pot oxidation and reduction by aqueous two-phase system. Bioprocess Biosyst Eng. doi:10.1007/s00449-009-0334-9
Kansal H, Banerjee UC (2009) Enhancing the biocatalytic potential of carbonyl reductase of Candida viswanathii using aqueous-organic solvent system. Bioresour Technol 100:1041–1047
Burton SG, Cowan DA, Woodley JM (2002) The search for the ideal biocatalyst. Nat Biotechnol 20:37–45
Pscheidt B, Glieder A (2008) Yeast cell factories for fine chemical and API production. Microb Cell Fact 7:1–49
Phillips RS (1996) Temperature modulation of the stereochemistry of enzymatic catalysis: Prospects for exploitation. Trends Biotechnol 14:13–16
Sakai T (2004) ‘Low-temperature method’ for a dramatic improvement in enantioselectivity in lipase-catalyzed reactions. Tetrahedron Asymmetry 15:2749–2756
Leon R, Fernandes P, Pinheiro HM, Cabral JMS (1998) Whole-cell biocatalysis in organic media. Enzyme Microb Technol 23:483–500
Chen Y, Lin H, Xu X, Xia S, Wang L (2008) Preparation the key intermediate of angiotensin-converting enzyme (ACE) inhibitors: High enantioselective production of ethyl (R)-2-hydroxy-4-phenylbutyrate with Candida boidinii CIOC21. Adv Synth Catal 350:426–430
Goldberg K, Schroer K, Lutz S, Liese A (2007) Biocatalytic ketone reduction—a powerful tool for the production of chiral alcohols—part II: whole-cell reductions. Appl Microbiol Biotechnol 76:249–255
Acknowledgments
The authors gratefully acknowledged the financial support of this work by the National Key Basic Research and Development Program of China (973 program) (2009CB724706), the Hi-Tech Research and Development Program of China (863 Program No. 2007AA02Z226) and the Program for Changjiang Scholars and Innovative Research Team in University (IRT0532).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, JL., Mu, XQ. & Xu, Y. Integration of newly isolated biocatalyst and resin-based in situ product removal technique for the asymmetric synthesis of (R)-methyl mandelate. Bioprocess Biosyst Eng 33, 797–804 (2010). https://doi.org/10.1007/s00449-009-0401-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-009-0401-2