Abstract
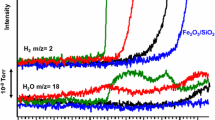
The dehydrogenation of propane over a silica-supported vanadium oxide catalyst was investigated at 823 K under atmospheric pressure in the presence/absence of CO2. The yield of propene and the selectivity to propene were higher in the dehydrogenation in the presence of CO2 than those in the dehydrogenation in the absence of CO2. On the other hand, the yield of aromatics and the selectivity to aromatics were much higher in the dehydrogenation in the absence of CO2 than those in the dehydrogenation in the presence of CO2. TPR measurements, NH3 desorption studies and in-situ UV–vis studies on the catalyst were also performed to elucidate the effects of CO2 on the behavior of the vanadium oxide in the catalyst during the dehydrogenation of propane.
Similar content being viewed by others
References
M. Saito S. Watanabe I. Takahara K. Murata M. Inaba (2003) Catal. Lett. 89 213 Occurrence Handle10.1023/A:1025754413131
I. Takahara M. Saito M. Inaba K. Murata (2004) ibid 96 29
X. Gao M.A. Banares I.E. Wachs (1999) J. Catal. 188 325 Occurrence Handle10.1006/jcat.1999.2647
X. Gao J.-M. Jehng I.E. Wachs (2002) ibid 209 43
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takahara, I., Saito, M., Inaba, M. et al. Dehydrogenation of propane over a silica-supported vanadium oxide catalyst. Catal Lett 102, 201–205 (2005). https://doi.org/10.1007/s10562-005-5856-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10562-005-5856-4