Abstract
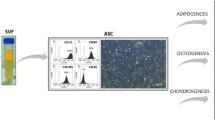
Adipose tissue-derived stem cells (ADSCs) are an available source of mesenchymal stem cells with the appropriate capacity to in vitro survive, propagate, and differentiate into cells from three lineages of ectoderm, mesoderm, and endoderm. The biological features of ADSCs depend on the donor physiology and health status, isolation procedure, culture conditions, and differentiation protocols used. Adipose tissue samples are provided by surgery and lipoaspiration-based methods and subjected to various mechanical and chemical digestion techniques to finally generate a heterogeneous mixture named stromal vascular fraction (SVF). ADSCs are purified through varied cell populations that exist within SVF and cultured under standard conditions to give rise to a highly rich resource of stem cells directly applied in the clinic or differentiated into a wide range of cells. The development and optimization of conventional isolation, expansion, and differentiation methods seem noteworthy to preserve the desirable biological functions of ADSCs in pre-clinical and clinical investigations.


Similar content being viewed by others
Change history
13 May 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10561-021-09928-6
Abbreviations
- ADSCs:
-
Adipose-derived stem cells
- aFGF:
-
Acidic FGF
- ALP:
-
Alkaline phosphatase
- AT:
-
Adipose tissue
- bFGF:
-
Basic fibroblast growth factor
- BDNF:
-
Brain-derived neurotrophic factor
- BMP:
-
Bone morphogenetic protein
- CaSR:
-
Calcium-sensing receptor
- CTGF:
-
Connective tissue growth factor
- Dex:
-
Dexamethasone
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- DMEM/F-12:
-
Dulbecco’s modified Eagle’s medium/Ham’s F12
- DSG3:
-
Desmoglein 3
- ECs:
-
Endothelial cells
- EGF:
-
Epidermal growth factor
- EGM-2:
-
Endothelial cell growth medium-2
- FBS:
-
Fetal bovine serum
- FCS:
-
Fetal calf serum
- FGF:
-
Fibroblast growth factor
- GalC:
-
Galactosylceramidase
- GCM2:
-
Glial cells missing homolog 2
- GFAP:
-
Glial fibrillary acidic protein
- GLP-1:
-
Glucagon-like peptide1
- GMP:
-
Good manufacturing practice
- HEM:
-
Human epidermal melanocyte
- HGF:
-
Hepatocyte growth factor
- IBMX:
-
1-Methyl-3-isobutyl xanthine
- IL-3:
-
Interleukin 3
- IL-6:
-
Interleukin 6
- ITS:
-
Insulin, transferrin, selenious acid
- KGF:
-
Keratinocyte growth factor
- LAL:
-
Laser-assisted liposuction
- MAP2:
-
Microtubule-assisted protein 2
- MEM:
-
Minimal essential medium
- MHC:
-
Heavy chain myosin
- MLANA:
-
Protein melan-A
- MSH:
-
Melatonin stimulating hormone
- MSCs:
-
Mesenchymal stem cells
- NeuN:
-
Neuronal nuclei
- NRP1:
-
Neuropilin 1
- PAL:
-
Power-assisted liposuction
- PDGF:
-
Platelet-derived growth factor
- PMEL17:
-
Premelanosome protein 17
- PPARγ:
-
Peroxisome proliferator-activated receptor-gamma
- PRP:
-
Platelet-rich plasma
- PTH:
-
Parathyroid hormone
- RUNX2:
-
Runt-related transcription factor 2
- SHH:
-
Sonic hedgehog
- SMA:
-
Smooth muscle actin
- SPC:
-
Sphingosylphosphorylcholine
- SR:
-
Surgical resection
- STAT3:
-
Signal transducer and activator of transcription 3
- SVF:
-
Stromal vascular fraction
- TGFβ:
-
Transforming growth factor β
- TRP1/2:
-
Tryptophan biosynthesis protein 1/2
- TxA2:
-
Thromboxane A2
- TYR:
-
Tyrosinase
- VEGF:
-
Vascular endothelial growth factor
References
Aguena M, Fanganiello RD, Tissiani LA, Ishiy FA, Atique R, Alonso N, Passos-Bueno MR (2012) Optimization of parameters for a more efficient use of adipose-derived stem cells in regenerative medicine therapies. Stem Cells Int 2012:303610. https://doi.org/10.1155/2012/303610
Al-Salleeh F, Beatty MW, Reinhardt RA, Petro TM, Crouch L (2008) Human osteogenic protein-1 induces osteogenic differentiation of adipose-derived stem cells harvested from mice. Arch Oral Biol 53:928–936. https://doi.org/10.1016/j.archoralbio.2008.05.014
Algire C, Medrikova D, Herzig S (2013) White and brown adipose stem cells: from signaling to clinical implications. Biochim Biophys Acta 1831:896–904. https://doi.org/10.1016/j.bbalip.2012.10.001
Amer MG, Embaby AS, Karam RA, Amer MG (2018) Role of adipose tissue derived stem cells differentiated into insulin producing cells in the treatment of type I diabetes mellitus. Gene 654:87–94. https://doi.org/10.1016/j.gene.2018.02.008
Anghileri E et al (2008) Neuronal differentiation potential of human adipose-derived mesenchymal stem cells. Stem Cells Dev 17:909–916. https://doi.org/10.1089/scd.2007.0197
Aurich H et al (2009) Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut 58:570–581. https://doi.org/10.1136/gut.2008.154880
Awad HA, Halvorsen Y-DC, Gimble JM, Guilak F (2003) Effects of transforming growth factor β 1 and dexamethasone on the growth and chondrogenic differentiation of adipose-derived stromal cells. Tissue Eng 9:1301–1312
Bacakova L et al (2018) Stem cells: their source, potency and use in regenerative therapies with focus on adipose-derived stem cells—a review. Biotechnol Adv 36:1111–1126. https://doi.org/10.1016/j.biotechadv.2018.03.011
Bajek A et al (2017) Does the harvesting technique affect the properties of adipose-derived stem cells?—The comparative biological characterization. J Cell Biochem 118:1097–1107. https://doi.org/10.1002/jcb.25724
Barbatelli G et al (2010) The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol Endocrinol Metab 298:E1244–E1253. https://doi.org/10.1152/ajpendo.00600.2009
Bayati V, Abbaspour MR, Dehbashi FN, Neisi N, Hashemitabar M (2017) A dermal equivalent developed from adipose-derived stem cells and electrospun polycaprolactone matrix: an in vitro and in vivo study. Anat Sci Int 92:509–520. https://doi.org/10.1007/s12565-016-0352-z
Bonora-Centelles A, Jover R, Mirabet V, Lahoz A, Carbonell F, Castell JV, Gómez-Lechón MJ (2009) Sequential hepatogenic transdifferentiation of adipose tissue-derived stem cells: relevance of different extracellular signaling molecules, transcription factors involved, and expression of new key marker genes. Cell Transplant 18:1319–1340. https://doi.org/10.3727/096368909x12483162197321
Bourin P et al (2013) Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 15:641–648. https://doi.org/10.1016/j.jcyt.2013.02.006
Câmara DAD, Shibli JA, Müller EA, De-Sá-Junior PL, Porcacchia AS, Blay A, Lizier NF (2020) Adipose tissue-derived stem cells: the biologic basis and future directions for tissue engineering. Materials (Basel Switzerland) 13:3210. https://doi.org/10.3390/ma13143210
Cannella V et al (2018) Differentiation and characterization of rat adipose tissue mesenchymal stem cells into endothelial-like cells. Anat Histol Embryol 47:11–20. https://doi.org/10.1111/ahe.12318
Cao Y, Sun Z, Liao L, Meng Y, Han Q, Zhao RC (2005) Human adipose tissue-derived stem cells differentiate into endothelial cells in vitro and improve postnatal neovascularization in vivo. Biochem Biophys Res Commun 332:370–379. https://doi.org/10.1016/j.bbrc.2005.04.135
Carvalho PP, Leonor IB, Smith BJ, Dias IR, Reis RL, Gimble JM, Gomes ME (2014) Undifferentiated human adipose-derived stromal/stem cells loaded onto wet-spun starch-polycaprolactone scaffolds enhance bone regeneration: nude mice calvarial defect in vivo study. J Biomed Mater Res A 102:3102–3111. https://doi.org/10.1002/jbm.a.34983
Ceccarelli S et al (2018) Neuropilin 1 mediates keratinocyte growth factor signaling in adipose-derived stem cells: potential involvement in adipogenesis. Stem Cells Int 2018:1075156. https://doi.org/10.1155/2018/1075156
Chavez-Munoz C, Nguyen KT, Xu W, Hong SJ, Mustoe TA, Galiano RD (2013) Transdifferentiation of adipose-derived stem cells into keratinocyte-like cells: engineering a stratified epidermis. PLoS ONE 8:e80587. https://doi.org/10.1371/journal.pone.0080587
Chen DC et al (2014) Purification of human adipose-derived stem cells from fat tissues using PLGA/silk screen hybrid membranes. Biomaterials 35:4278–4287. https://doi.org/10.1016/j.biomaterials.2014.02.004
Ciuffi S, Zonefrati R, Brandi ML (2017) Adipose stem cells for bone tissue repair. Clin Cases Miner Bone Metab 14:217–226. https://doi.org/10.11138/ccmbm/2017.14.1.217
Colazzo F, Chester AH, Taylor PM, Yacoub MH (2010) Induction of mesenchymal to endothelial transformation of adipose-derived stem cells. J Heart Valve Dis 19:736–744
Cuevas-Diaz Duran R, González-Garza MT, Cardenas-Lopez A, Chavez-Castilla L, Cruz-Vega DE, Moreno-Cuevas JE (2013) Age-related yield of adipose-derived stem cells bearing the low-affinity nerve growth factor receptor. Stem Cells Int 2013:372164. https://doi.org/10.1155/2013/372164
Czapla J et al (2019) The effect of culture media on large-scale expansion and characteristic of adipose tissue-derived mesenchymal stromal cells. Stem Cell Res Ther 10:235. https://doi.org/10.1186/s13287-019-1331-9
Devitt SM, Carter CM, Dierov R, Weiss S, Gersch RP, Percec I (2015) Successful isolation of viable adipose-derived stem cells from human adipose tissue subject to long-term cryopreservation: positive implications for adult stem cell-based therapeutics in patients of advanced age. Stem Cells Int 2015:146421. https://doi.org/10.1155/2015/146421
Diekman BO, Estes BT, Guilak F (2010) The effects of BMP6 overexpression on adipose stem cell chondrogenesis: interactions with dexamethasone and exogenous growth factors. J Biomed Mater Res A 93A:994–1003. https://doi.org/10.1002/jbm.a.32589
Edwards NJ, Stone R, Christy R, Zhang CK, Pollok B, Cheng X (2018) Differentiation of adipose derived stem cells to keratinocyte-like cells on an advanced collagen wound matrix. Tissue Cell 53:68–75. https://doi.org/10.1016/j.tice.2018.06.001
Fideles SOM et al (2019) Effect of cell source and osteoblast differentiation on gene expression profiles of mesenchymal stem cells derived from bone marrow or adipose tissue. J Cell Biochem. https://doi.org/10.1002/jcb.28463
Frese L, Dijkman PE, Hoerstrup SP (2016) Adipose tissue-derived stem cells in regenerative medicine. Transfus Med Hemother 43:268–274
Gaustad KG, Boquest AC, Anderson BE, Gerdes AM, Collas P (2004) Differentiation of human adipose tissue stem cells using extracts of rat cardiomyocytes. Biochem Biophys Res Commun 314:420–427
Graziano ACE, Avola R, Perciavalle V, Nicoletti F, Cicala G, Coco M, Cardile V (2018) Physiologically based microenvironment for in vitro neural differentiation of adipose-derived stem cells. World J Stem Cells 10:23–33. https://doi.org/10.4252/wjsc.v10.i3.23
Guo DL, Wang ZG, Xiong LK, Pan LY, Zhu Q, Yuan YF, Liu ZS (2017) Hepatogenic differentiation from human adipose-derived stem cells and application for mouse acute liver injury. Artif Cells Nanomed Bbiotechnol 45:224–232. https://doi.org/10.3109/21691401.2016.1138495
Gwak S-J, Bhang SH, Yang HS, Kim S-S, Lee D-H, Lee S-H, Kim B-S (2009) In vitro cardiomyogenic differentiation of adipose-derived stromal cells using transforming growth factor-β1. Cell Biochem Funct 27:148–154. https://doi.org/10.1002/cbf.1547
Hasani S, Javeri A, Asadi A, Fakhr Taha M (2020) Cardiac differentiation of adipose tissue-derived stem cells is driven by BMP4 and bFGF but counteracted by 5-azacytidine and valproic acid. Cell J 22:273–282. https://doi.org/10.22074/cellj.2020.6582
Hasegawa T, Sakamoto A, Wada A, Fukai T, Iida H, Ikeda S (2015) Keratinocyte progenitor cells reside in human subcutaneous adipose tissue. PLoS ONE 10:e0118402. https://doi.org/10.1371/journal.pone.0118402
He J et al (2017) P019 the impact of HLA haplotype and allele mismatches of donor-recipient pairs on outcomes of haploidentical stem cell transplantation with a third part cord blood unit. Hum Immunol 78:67
Hendijani F (2017) Explant culture: an advantageous method for isolation of mesenchymal stem cells from human tissues. Cell Prolif 50:e12334. https://doi.org/10.1111/cpr.12334
Hennig T, Lorenz H, Thiel A, Goetzke K, Dickhut A, Geiger F, Richter W (2007) Reduced chondrogenic potential of adipose tissue derived stromal cells correlates with an altered TGFβ receptor and BMP profile and is overcome by BMP-6. J Cell Physiol 211:682–691. https://doi.org/10.1002/jcp.20977
Higuchi A et al (2015) A hybrid-membrane migration method to isolate high-purity adipose-derived stem cells from fat tissues. Sci Rep 5:10217. https://doi.org/10.1038/srep10217
Hu F, Wang X, Liang G, Lv L, Zhu Y, Sun B, Xiao Z (2013) Effects of epidermal growth factor and basic fibroblast growth factor on the proliferation and osteogenic and neural differentiation of adipose-derived stem cells. Cell Reprogram 15:224–232. https://doi.org/10.1089/cell.2012.0077
Hu R, Ling W, Xu W, Han D (2014) Fibroblast-like cells differentiated from adipose-derived mesenchymal stem cells for vocal fold wound healing. PLoS ONE 9:e92676. https://doi.org/10.1371/journal.pone.0092676
Hur W et al (2017) Regeneration of full-thickness skin defects by differentiated adipose-derived stem cells into fibroblast-like cells by fibroblast-conditioned medium. Stem Cell Res Ther 8:92. https://doi.org/10.1186/s13287-017-0520-7
Iyyanki T, Hubenak J, Liu J, Chang EI, Beahm EK, Zhang Q (2015) Harvesting technique affects adipose-derived stem cell yield. Aesthet Surg J 35:467–476. https://doi.org/10.1093/asj/sju055
Jang S, Cho HH, Cho YB, Park JS, Jeong HS (2010) Functional neural differentiation of human adipose tissue-derived stem cells using bFGF and forskolin. BMC Cell Biol 11:25. https://doi.org/10.1186/1471-2121-11-25
Jankowski M et al (2020) In vitro cultures of adipose-derived stem cells: An overview of methods, molecular analyses, and clinical applications. Cells 9:1783. https://doi.org/10.3390/cells9081783
Keshavarz G, Jalili C, Pazhouhi M, Khazaei M (2020) Resveratrol effect on adipose-derived stem cells differentiation to chondrocyte in three-dimensional culture. Adv Pharm Bull 10:88–96. https://doi.org/10.15171/apb.2020.011
Kim YM, Jeon ES, Kim MR, Jho SK, Ryu SW, Kim JH (2008) Angiotensin II-induced differentiation of adipose tissue-derived mesenchymal stem cells to smooth muscle-like cells. Int J Biochem Cell Biol 40:2482–2491. https://doi.org/10.1016/j.biocel.2008.04.016
Kim H-J, Im G-I (2009) Chondrogenic differentiation of adipose tissue-derived mesenchymal stem cells: greater doses of growth factor are necessary. J Orthop Res 27:612–619. https://doi.org/10.1002/jor.20766
Kim SC, Han DJ, Lee JY (2010) Adipose tissue derived stem cells for regeneration and differentiation into insulin-producing cells. Curr Stem Cell Res Ther 5:190–194. https://doi.org/10.2174/157488810791268717
Knippenberg M, Helder MN, Zandieh Doulabi B, Wuisman PIJM, Klein-Nulend J (2006) Osteogenesis versus chondrogenesis by BMP-2 and BMP-7 in adipose stem cells. Biochem Biophys Res Commun 342:902–908. https://doi.org/10.1016/j.bbrc.2006.02.052
Kolodziej M, Strauss S, Lazaridis A, Bucan V, Kuhbier JW, Vogt PM, Könneker S (2019) Influence of glucose and insulin in human adipogenic differentiation models with adipose-derived stem cells. Adipocyte 8:254–264. https://doi.org/10.1080/21623945.2019.1636626
Krampera M et al (2007) Induction of neural-like differentiation in human mesenchymal stem cells derived from bone marrow, fat. spleen thymus Bone 40:382–390. https://doi.org/10.1016/j.bone.2006.09.006
Lee W-CC, Sepulveda JL, Rubin JP, Marra KG (2009) Cardiomyogenic differentiation potential of human adipose precursor cells. Int J Cardiol 133:399–401. https://doi.org/10.1016/j.ijcard.2007.11.068
Lee M-S et al (2017) Enhanced cell growth of adipocyte-derived mesenchymal stem cells using chemically-defined serum-free media. Int J Mol Sci 18:1779
Li H, Zimmerlin L, Marra KG, Donnenberg VS, Donnenberg AD, Rubin JP (2011) Adipogenic potential of adipose stem cell subpopulations. Plast Reconstr Surg 128:663–672. https://doi.org/10.1097/PRS.0b013e318221db33
Li H, Han Z, Liu D, Zhao P, Liang S, Xu K (2013a) Autologous platelet-rich plasma promotes neurogenic differentiation of human adipose-derived stem cells in vitro. Int J Neurosci 123:184–190. https://doi.org/10.3109/00207454.2012.742077
Li W, Li K, Wei W, Ding S (2013b) Chemical approaches to stem cell biology and therapeutics. Cell Stem Cell 13:270–283. https://doi.org/10.1016/j.stem.2013.08.002
Li CJ, Madhu V, Balian G, Dighe AS, Cui Q (2015) Cross-talk between VEGF and BMP-6 pathways accelerates osteogenic differentiation of human adipose-derived stem cells. J Cell Physiol 230:2671–2682. https://doi.org/10.1002/jcp.24983
Liu L, Rando TA (2011) Manifestations and mechanisms of stem cell aging. J Cell Biol 193:257–266. https://doi.org/10.1083/jcb.201010131
Liu W, Liu H, Wang Y, Zhang L, Wang C, Li H (2019) Ascorbic acid induces cardiac differentiation of white adipose tissue-derived stem cells. Mol Cell Biochem 450:65–73. https://doi.org/10.1007/s11010-018-3373-1
Lund P, Pilgaard L, Duroux M, Fink T, Zachar V (2009) Effect of growth media and serum replacements on the proliferation and differentiation of adipose-derived stem cells. Cytotherapy 11:189–197
Markarian CF et al (2014) Isolation of adipose-derived stem cells: a comparison among different methods. Biotechnol Lett 36:693–702. https://doi.org/10.1007/s10529-013-1425-x
Meligy FY, Shigemura K, Behnsawy HM, Fujisawa M, Kawabata M, Shirakawa T (2012) The efficiency of in vitro isolation and myogenic differentiation of MSCs derived from adipose connective tissue, bone marrow, and skeletal muscle tissue. In Vitro Cell Dev Biol Anim 48:203–215. https://doi.org/10.1007/s11626-012-9488-x
Merfeld-Clauss S, Lupov IP, Lu H, Feng D, Compton-Craig P, March KL, Traktuev DO (2014) Adipose stromal cells differentiate along a smooth muscle lineage pathway upon endothelial cell contact via induction of activin A. Circ Res 115:800–809. https://doi.org/10.1161/circresaha.115.304026
Mihaila SM, Frias AM, Pirraco RP, Rada T, Reis RL, Gomes ME, Marques AP (2013) Human adipose tissue-derived SSEA-4 subpopulation multi-differentiation potential towards the endothelial and osteogenic lineages. Tissue Eng A 19:235–246. https://doi.org/10.1089/ten.TEA.2012.0092
Mohamad Buang ML, Seng HK, Chung LH, Saim AB, Idrus RB (2012) In vitro generation of functional insulin-producing cells from lipoaspirated human adipose tissue-derived stem cells. Arch Med Res 43:83–88. https://doi.org/10.1016/j.arcmed.2012.01.012
Morandi E, Verstappen R, Zwierzina M, Geley S, Pierer G, Ploner C (2016) ITGAV and ITGA5 diversely regulate proliferation and adipogenic differentiation of human adipose derived stem cells. Sci Rep 6:28889
Mushahary D, Spittler A, Kasper C, Weber V, Charwat V (2018) Isolation, cultivation, and characterization of human mesenchymal stem cells. Cytom Part A J Int Soc Anal Cytol 93:19–31. https://doi.org/10.1002/cyto.a.23242
Najar M et al (2014) Proliferative and phenotypical characteristics of human adipose tissue-derived stem cells: comparison of Ficoll gradient centrifugation and red blood cell lysis buffer treatment purification methods. Cytotherapy 16:1220–1228. https://doi.org/10.1016/j.jcyt.2014.05.021
Nincheri P et al (2009) Sphingosine 1-phosphate induces differentiation of adipose tissue-derived mesenchymal stem cells towards smooth muscle cells. Cell Mol Life Sci 66:1741–1754. https://doi.org/10.1007/s00018-009-9181-8
Oberbauer E, Steffenhagen C, Wurzer C, Gabriel C, Redl H, Wolbank S (2015) Enzymatic and non-enzymatic isolation systems for adipose tissue-derived cells: current state of the art. Cell Regen (London England) 4:7. https://doi.org/10.1186/s13619-015-0020-0
Orbay H, Devi K, Williams PA, Dehghani T, Silva EA, Sahar DE (2016) Comparison of endothelial differentiation capacities of human and rat adipose-derived stem cells. Plast Reconstr Surg 138:1231–1241. https://doi.org/10.1097/prs.0000000000002791
Palumbo P, Lombardi F, Siragusa G, Cifone MG, Cinque B, Giuliani M (2018) Methods of isolation, characterization and expansion of human adipose-derived stem cells (ASCs): an overview. Int J Mol Sci 19:1897. https://doi.org/10.3390/ijms19071897
Park WS et al (2013) Functional expression of smooth muscle-specific ion channels in TGF-β(1)-treated human adipose-derived mesenchymal stem cells. Am J Physiol Cell Physiol 305:C377–C391. https://doi.org/10.1152/ajpcell.00404.2012
Patrikoski M, Juntunen M, Boucher S, Campbell A, Vemuri MC, Mannerström B, Miettinen S (2013) Development of fully defined xeno-free culture system for the preparation and propagation of cell therapy-compliant human adipose stem cells. Stem Cell Res Ther 4:27. https://doi.org/10.1186/scrt175
Patrikoski M, Mannerström B, Miettinen S (2019) Perspectives for clinical translation of adipose stromal/stem cells. Stem Cells Int 2019:5858247. https://doi.org/10.1155/2019/5858247
Petry L et al (2018) Directing adipose-derived stem cells into keratinocyte-like cells: impact of medium composition and culture condition. J Eur Acad Dermatol Venereol 32:2010–2019. https://doi.org/10.1111/jdv.15010
Phetfong J, Tawonsawatruk T, Seenprachawong K, Srisarin A, Isarankura-Na-Ayudhya C, Supokawej A (2017) Re-using blood products as an alternative supplement in the optimisation of clinical-grade adipose-derived mesenchymal stem cell culture. Bone Jt Res 6:414–422
Planat-Benard V et al (2004a) Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation 109:656–663. https://doi.org/10.1161/01.cir.0000114522.38265.61
Planat-Bénard V et al (2004b) Spontaneous cardiomyocyte differentiation from adipose tissue stroma cells. Circ Res 94:223–229. https://doi.org/10.1161/01.res.0000109792.43271.47
Raposio E, Bertozzi N (2017) Isolation of ready-to-use adipose-derived stem cell (ASC) pellet for clinical applications and a comparative overview of alternate methods for ASC isolation. Curr Protoc Stem Cell Biol 41:1F – 17
Raposio E, Caruana G, Petrella M, Bonomini S, Grieco MP (2016) A standardized method of isolating adipose-derived stem cells for clinical applications. Ann Plast Surg 76:124–126. https://doi.org/10.1097/sap.0000000000000609
Raposio E, Simonacci F, Perrotta RE (2017) Adipose-derived stem cells: comparison between two methods of isolation for clinical applications. Ann Med Surg 20:87–91. https://doi.org/10.1016/j.amsu.2017.07.018
Ratushnyy A, Ezdakova M, Buravkova L (2020) Secretome of senescent adipose-derived mesenchymal stem cells negatively regulates angiogenesis. Int J Mol Sci 21:1802. https://doi.org/10.3390/ijms21051802
Razavi S, Razavi MR, Ahmadi N, Kazemi M (2015) Estrogen treatment enhances neurogenic differentiation of human adipose derived stem cells in vitro Iran. J Basic Med Sci 18:799–804
Reumann MK et al (2018) Donor site location is critical for proliferation, stem cell capacity, and osteogenic differentiation of adipose mesenchymal stem/stromal cells: implications for bone tissue engineering. Int J Mol Sci 19:1868. https://doi.org/10.3390/ijms19071868
Rodbell M (1966) Metabolism of isolated fat cells. II. The similar effects of phospholipase C (Clostridium perfringens alpha toxin) and of insulin on glucose and amino acid metabolism. J Biol Chem 241:130–139
Rodriguez A-M et al (2004) Adipocyte differentiation of multipotent cells established from human adipose tissue. Biochem Biophys Res Commun 315:255–263
Schubert T, Lafont S, Beaurin G, Grisay G, Behets C, Gianello P, Dufrane D (2013) Critical size bone defect reconstruction by an autologous 3D osteogenic-like tissue derived from differentiated adipose MSCs. Biomaterials 34:4428–4438. https://doi.org/10.1016/j.biomaterials.2013.02.053
Seo BF, Kim KJ, Kim MK, Rhie JW (2016) The effects of human keratinocyte coculture on human adipose-derived stem cells. Int wound J 13:630–635. https://doi.org/10.1111/iwj.12335
Seo E, Kang H, Lim OK, Jun HS (2018) Supplementation with IL-6 and muscle cell culture conditioned media enhances myogenic differentiation of adipose tissue-derived stem cells through STAT3 activation. Int J Mol Sci 19:1557. https://doi.org/10.3390/ijms19061557
Seo Y, Shin TH, Kim HS (2019) Current strategies to enhance adipose stem cell function: an update. Int J Mol Sci 20:3827. https://doi.org/10.3390/ijms20153827
Shah FS et al (2015) The relative functionality of freshly isolated and cryopreserved human adipose-derived stromal/stem cells. Cells Tissues Organs 201:436–444. https://doi.org/10.1159/000446494
Taha MF, Javeri A, Karimipour M, Yamaghani MS (2019) Priming with oxytocin and relaxin improves cardiac differentiation of adipose tissue-derived stem cells. J Cell Biochem 120:5825–5834. https://doi.org/10.1002/jcb.27868
Taléns-Visconti R, Bonora A, Jover R, Mirabet V, Carbonell F, Castell JV, Gómez-Lechón MJ (2006) Hepatogenic differentiation of human mesenchymal stem cells from adipose tissue in comparison with bone marrow mesenchymal stem cells. World J Gastroenterol 12:5834–5845. https://doi.org/10.3748/wjg.v12.i36.5834
Tarte K et al (2010) Clinical-grade production of human mesenchymal stromal cells: occurrence of aneuploidy without transformation. Blood 115:1549–1553
Tevlin R et al (2016) A novel method of human adipose-derived stem cell isolation with resultant increased cell yield. Plast Reconstr Surg 138:983e–996e. https://doi.org/10.1097/prs.0000000000002790
Timper K et al (2006) Human adipose tissue-derived mesenchymal stem cells differentiate into insulin, somatostatin, and glucagon expressing cells. Biochem Biophys Res Commun 341:1135–1140. https://doi.org/10.1016/j.bbrc.2006.01.072
Van Pham P, Vu NB (2016) In vitro expansion of mesenchymal stem cells for clinical use. Prog Stem Cell 3:87–96
Van Pham P et al (2013) Activated platelet-rich plasma improves adipose-derived stem cell transplantation efficiency in injured articular cartilage. Stem Cell Res Ther 4:91
Wang C, Yin S, Cen L, Liu Q, Liu W, Cao Y, Cui L (2010) Differentiation of adipose-derived stem cells into contractile smooth muscle cells induced by transforming growth factor-beta1 and bone morphogenetic protein-4. Tissue Eng A 16:1201–1213. https://doi.org/10.1089/ten.TEA.2009.0303
Wang F, Zachar V, Pennisi CP, Fink T, Maeda Y, Emmersen J (2018) Hypoxia enhances differentiation of adipose tissue-derived stem cells toward the smooth muscle phenotype. Int J Mol Sci 19:517. https://doi.org/10.3390/ijms19020517
Wu CH et al (2012) The isolation and differentiation of human adipose-derived stem cells using membrane filtration. Biomaterials 33:8228–8239. https://doi.org/10.1016/j.biomaterials.2012.08.027
Xu LJ et al (2017) Adipose-derived stromal cells resemble bone marrow stromal cells in hepatocyte differentiation potential in vitro and in vivo. World J Gastroenterol 23:6973–6982. https://doi.org/10.3748/wjg.v23.i38.6973
Yang XF et al (2011) High efficient isolation and systematic identification of human adipose-derived mesenchymal stem cells. J Biomed Sci 18:59. https://doi.org/10.1186/1423-0127-18-59
Yun DH, Song HY, Lee MJ, Kim MR, Kim MY, Lee JS, Kim JH (2009) Thromboxane A(2) modulates migration, proliferation, and differentiation of adipose tissue-derived mesenchymal stem cells. Exp Mol Med 41:17–24. https://doi.org/10.3858/emm.2009.41.1.003
Zavala G, Sandoval C, Meza D, Contreras R, Gubelin W, Khoury M (2019) Differentiation of adipose-derived stem cells to functional CD105neg CD73low melanocyte precursors guided by defined culture condition. Stem Cell Res Ther 10:249. https://doi.org/10.1186/s13287-019-1364-0
Zhang R, Jack GS, Rao N, Zuk P, Ignarro LJ, Wu B, Rodríguez LV (2012) Nuclear fusion-independent smooth muscle differentiation of human adipose-derived stem cells induced by a smooth muscle environment. Stem Cells 30:481–490. https://doi.org/10.1002/stem.1023
Zhang P, Zhang H, Dong W, Wang Z, Qin Y, Wu C, Dong Q (2020) Differentiation of rat adipose-derived stem cells into parathyroid-like cells. Int J Endocrinol 2020:1860842. https://doi.org/10.1155/2020/1860842
Zhou ZQ et al (2019) Adipose extracellular matrix promotes skin wound healing by inducing the differentiation of adipose–derived stem cells into fibroblasts. Int J Mol Med 43:890–900. https://doi.org/10.3892/ijmm.2018.4006
Zhu M, Heydarkhan-Hagvall S, Hedrick M, Benhaim P, Zuk P (2013) Manual isolation of adipose-derived stem cells from human lipoaspirates. J Vis Exp JoVE. https://doi.org/10.3791/50585
Zollino I, Sibilla MG, Gianesini S, Menegatti E, Tessari M, Malagoni AM (2017) Technique for intraoperatory harvesting of adipose derived stem cells: towards cell treatment of recalcitrant ulcers. Veins Lymphatics 6:6814
Zuk PA et al (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295. https://doi.org/10.1091/mbc.e02-02-0105
Acknowledgements
The authors would like to show their gratitude to the Fertility and Infertility Research Center, Kermanshah University of Medical Sciences for supporting the ideas of the present manuscript.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khazaei, S., Keshavarz, G., Bozorgi, A. et al. Adipose tissue-derived stem cells: a comparative review on isolation, culture, and differentiation methods. Cell Tissue Bank 23, 1–16 (2022). https://doi.org/10.1007/s10561-021-09905-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-021-09905-z