Abstract
Patients with hypertrophic cardiomyopathy may exhibit impaired functional capacity, associated with increased morbidity and mortality. Systolic function is one of the determinants of functional capacity. Early identification of systolic disfunction may identify patients at risk for adverse outcomes. Myocardial deformation parameters, derived from three-dimensional (3D) speckle-tracking echocardiography (3DSTE) are useful tools to assess left ventricular systolic function, and are often abnormal before a decline in ejection fraction is seen. The aim of this study was to evaluate the correlation between myocardial deformation parameters obtained by 3DSTE and functional capacity in patients with hypertrophic cardiomyopathy. Seventy-four hypertrophic cardiomyopathy adult patients were prospectively evaluated. All patients underwent a dedicated 2D and 3D echocardiographic examination and cardiopulmonary exercise testing (CPET). Values of 3D global radial (GRS), longitudinal (3DGLS) and circumferential strain (GCS) were overall reduced in our population: 99% (n = 73) of the patients had reduced GLS, 82% (n = 61) had reduced GRS and all patients had reduced GCS obtain by 3DSTE. Average peak VO2 was 21.01 (6.08) ml/Kg/min; 58% (n = 39) of the patients showed reduced exercise tolerance (predicted peak VO2 < 80%). The average VE/VCO2 slope was 29.0 (5.3) and 16% (n = 11) of the patients had impaired ventilatory efficiency (VE/VCO2 > 34). In multivariable analysis, 3D GLS (β1 = 0.10, 95%CI: 0.03;0.23, p = 0.014), age (β1 = -0.15, 95%CI: -0.23; -0.05, p = 0.002) and female gender (β1 = -5.10, 95%CI: -7.7; -2.6, p < 0.01) were independently associated with peak VO2. No association was found between left ventricle ejection fraction obtain and peak VO2 (r = 0.161, p = 0.5). Impaired myocardial deformation parameters evaluated by 3DSTE were associated with worse functional capacity assessed by peak VO2.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypertrophic cardiomyopathy (HCM) is the most frequent genetic heart disease, with an approximate prevalence of 0.2% [1]. It is characterized by heterogenous left ventricular (LV) hypertrophy and histologically by cardiomyocyte hypertrophy, myocardial fibre disarray and interstitial fibrosis [2].
Patients with hypertrophic cardiomyopathy may exhibit reduced functional capacity, which is associated with increased morbidity and mortality. Progression to heart failure (HF) is a major concern in these patients and can be in part predicted by impaired functional capacity [3].
The pathophysiology of reduced functional capacity in patients with HCM has been mainly linked to left ventricular diastolic and systolic disfunction, chronotropic incompetence and impaired stroke volume response [4].
Cardiopulmonary exercise testing (CPET) is indicated by international guidelines as a safe and reliable test to characterize severity and mechanisms of functional limitation in patients with HCM. Reduced values of peak oxygen, ventilatory efficiency and anaerobic threshold are associated with progression to HF [5].
Systolic function evaluation based on ejection fraction (EF) is not a sensitive method, since the former is typically normal or even increased in this group of patients. Myocardial deformation parameters derived from two-dimension (2D) and three-dimension (3D) speckle tracking echocardiography, on the other hand, are often abnormal before a decline in EF is seen [6].
The heterogeneous LV hypertrophy and interstitial fibrosis that characterize HCM leads to variability in regional and global systolic and diastolic deformation parameters. Characterization and quantification of myocardial segmental and rotational mechanics by 2D and 3D speckle tracking echocardiography (3DSTE) is an evolving and promising technique [2].
3DSTE provides a better quantification of cardiac chambers size and function, minimizing errors provoked by foreshortened views that can occur in standard 2D echocardiography [7].
The aim of this study was to evaluate the association between myocardial deformation parameters obtained by 3DSTE and functional capacity in patients with hypertrophic cardiomyopathy.
Materials and methods
Study population
This prospective study included 74 HCM adult patients evaluated at the outpatient cardiomyopathy clinic at Hospital de Santa Marta, Centro Hospitalar Universitário de Lisboa and Hospital Garcia de Orta, Almada between December 2017 and August 2020.
Diagnosis of HCM was made based on the European Society of Cardiology guidelines on Hypertrophic Cardiomyopathy [6]. Briefly a maximal wall thickness (MWT) ≥ 15 mm in probands or ≥ 13 mm in relatives or likely pathogenic variant carriers was considered diagnostic. HCM was considered obstructive if a systolic gradient ≥ 30 mmHg in the left ventricle outflow tract obstruction (LVOT) at rest or after provocative manoeuvre was obtained.
Patients with LV ejection fraction < 50% and LV wall thinning were excluded. Epicardial coronary artery disease and previous septal reduction therapy were also exclusion criteria.
Epicardial coronary artery disease was excluded by invasive coronary angiography or cardiac computerized tomography in symptomatic patients or asymptomatic patients older than 40 years.
The investigation followed the principles outlined in the Declaration of Helsinki. All patients gave written consent.
Echocardiography
All patients underwent a dedicated 2D and 3D echocardiographic examination (Vivid E95; General Electric). LV morphology and function was assessed according to the recommendations of the American Society of Echocardiography [7]. Evaluation of diastolic function included assessment of transmitral flow pattern with pulse Doppler and tissue Doppler at the mitral annulus, left atrial volume index and maximal velocity of tricuspid regurgitation.
2D global longitudinal strain (GLS) was used to evaluate LV myocardial deformation. Regarding 3D speckle-tracking echocardiography GLS, global circumferential strain (GCS) and global radial strain (GRS) were obtained.
To perform 3D speckle tracking echocardiography (3DSTE) using the acquired full-volume 3D dataset of the left ventricle (LV), the 3D strain analysis software automatically detected the LV’s endocardial and epicardial borders, generating a precise 3D mesh model of the myocardium. Manual adjustments were made by experienced cardiologists. The software utilized “block matching” to compare 3D patterns of acoustic markers within regions of interest (ROIs), identifying and removing outliers. Spatial averaging was then performed for accurate strain analysis, calculating longitudinal, circumferential, and radial strain values for each LV segment throughout the cardiac cycle [8].
2D GLS of -22.5 ± 2.7% was considered normal [9]. Normal 3D values considered were: GLS − 21.0 ± 2.6%, GCS − 30.3 ± 4.0%, GRS 43.2 ± 4.5 [10] [11].
Cardiopulmonary exercise testing
CPET was performed using a treadmill with the application of the modified Bruce protocol. The duration of the exercise was limited only by patients’ symptoms when reaching maximal effort.
Ventilatory expired gas analysis was performed using Ergostik, Geratherm®, Cardio Solutions systems. The equipment was calibrated in a standard fashion before each test.
A standard 12-lead electrocardiogram (ECG) monitoring, diastolic blood pressure (DBP) and systolic blood pressure (SBP), saturation probe and measure of expiratory gases were recorded.
Oxygen consumption (VO2), CO2 production (VCO2), minute ventilation (VE) and other CPET variables were acquired breath by breath.
Peak VO2 (pVO2) was measured in each patient and reported as mL kg − 1 min − 1. The expected peak VO2 was calculated based on age, gender, and body index (adjusted for body mass/fat-free mass in obese patients) [12].
VE/VCO2 at peak exercise was used to obtain the ventilatory class. Anaerobic threshold was measured by V-slope method and by the analysis of ventilatory equivalents.
Statistical analysis
Data are expressed as mean and standard deviation (SD). The Spearman rank-order correlation coefficient was used to assessed univariable correlation between variables. Variables with significant correlations (p < 0.05) where analyzed using multivariable linear regression. Comparisons of groups were analyzed using 1-way analysis of variance (ANOVA) for parametric data or Kruskal-Wallis for non-parametric data. Analysis within groups was assessed by post-hoc Bonferroni test. A probability value of p < 0.05 was considered significant.
Results
Clinical and echocardiographic findings
Of 74 patients with HCM, mean age 56 (15) years, 43 (58%) males, 40 patients (54%) were in New York Heart Association (NYHA) functional class I, 29 (39%) in class II and 5 (7%) in class III. Fifty-five patients (74%) had non obstructive HCM. Maximum LV wall thickness (MWT) was 20 (7) mm. Study population characteristics are shown in Table 1.
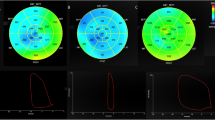
Mean values of global longitudinal, radial, and circumferential strain are reported in Table 2. All patients had reduced 2D GLS. Values of 3D GRS, GLS and GCS were overall reduced in our population – 73 (99%) of the patients had reduced GLS, 61 (82%) had reduced GRS and all patients had reduced GCS obtained by 3DSTE. An example of a echocardiographic study is represented in Fig. 1.
Regarding the basal characteristics of our population, an association between NYHA class and 3D GLS was found (F (2.71) = 4.07; p = 0.02). Post hoc analysis with the Bonferroni test revealed that 3D GLS values were significantly lower in patients in NYHA III compared with patients in NYHA I (-4.75 (95% CI, 0.66–8.84%, p = 0.01), but not between patients in NYHA III compared to patients NYHA II (-4.08, CI, -8.26-0.09, p = 0.057) (Fig. 2). The remaining 3D strain measures had no correlation with NYHA class: 3D GRS (F (2,71) = 1.22; p = 0.30) and 3D GCS (F (2,71) = 1.38, p = 0.26).
A relationship between 2D GLS and NYHA class was also found (F (2) = 7.7; p = 0.001).
MWT was not related with 3DSTE strain measures (3D GLS: β1 = -0.14, 95%IC -0.35;0.06, p = 0.12; 3D GRS: β1 = -0.28, 95%IC -0.94;0.37, p = 0.39; 3D GCS: β1 = -0.10, 95%IC -0.45;0.25, p = 0.57), neither was left ventricular outflow tract obstruction (β1 = -0.002, 95%IC -0.02;0.02, p = 0.80).
Cardiopulmonary exercise test findings
Regarding CPET findings respiratory exchange ratio was 1.03 (0.09) revealing adequate exercise effort. Mean time of exercise was 12.4 (4.3) minutes. Average pVO2 was 21.01 (6.08) ml/Kg/min; 39 (58%) showed reduced exercise tolerance (predicted pVO2 < 80%) and 31 (42%) had a pVO2 < 20 ml/Kg/min. The average VE/VCO2 slope was 29 (5.3) and 11 (16%) of the patients had impaired ventilatory efficiency (VE/VCO2 > 34). CPET findings are shown in Table 2.
There was an association between pVO2 and gender (t (72) = 5.52, p < 0.01), pVO2 was higher in males – mean 24 (5.8) vs. 17 (5.3) ml/Kg/min than in females.
Regarding the echocardiographic findings, E/e’ showed a inverse relation with pVO2 (β1 = -0.05, 95%IC -0.9;0.3, p < 0.01) and so did RV-RA peak velocity of tricuspid regurgitant jet (β1 = -0.06, 95%IC -0.09;0.01, p = 0.01), however no association was found after adjusting for baseline characteristics in multivariable analysis (p = 0.34; p = 0.25) (Table 3). No association was found between left atrium index volume and pVO2 (β1 = -0.09, 95%IC -0.2;0.004, p = 0.06).
No association was found between pVO2 and MWT (β1 = 0.024, 95%IC -0.35;0.39, p = 0.10) or between the presence of obstruction and pVO2 (t (-65) = − 1.11, p = 0.83).
Univariable analysis revealed a correlation between 3D GRS (r = 0.34, p = 0.003), 3D GLS (r = 0.40, p < 0.001) and 3D GCS (r = 0.35, p = 0.002) with pVO2. On the other hand, no correlation was found between LVEF (r = 0.161, p = 0.5) or GLS (r = 0.169, p = 0.5) obtained by 2D method and pVO2 (Fig. 3). No correlation was found between 3D LVEF and pVO2 (r=-0.1, p = 0.40).
In multivariable analysis, 3D GLS (β1 = 0.1, 95%CI: 0.03;0.23, p = 0.014), age (β1 = -0.15, 95%CI: -0.23; -0.05, p = 0.002) and female gender (β1=-5.1, 95%CI: -7.7; -2.6, p < 0.01) were independently associated with pVO2. The remaining 3D strain parameters (GRS and GCS) were not considered significantly related to pVO2 in multivariable analysis.
Discussion
In this study we found that that 3DGLS was independently associated with worse functional capacity determined by peak VO2, 3DGLS was also significantly decreased in patients with higher NYHA classes.
The high variability of left ventricular hypertrophy and contractile capacity between different segments of the myocardium of a single HCM patient, makes 3DSTE a more sensitive method than 2D LVEF to characterize systolic function, considering its higher accuracy at assessing regional cardiac mechanisms. In our study we found a correlation between 3D evaluation of myocardial deformation and functional capacity, which could not be achieved with 2D LVEF.
Overall strain parameters were reduced in our population, as expected for a cohort of HCM patients [13]. Regarding GCS, it was diminished in all the patients of our cohort. Previous studies described an initial compensatory increase in GCS compared do GLS, which is later lost due to disease progression, culminating in an overall fall of strain parameters, which we verified in our cohort [14]. Furthermore, GCS reflects changes in the middle layer of the myocardium where fibres are oriented in a circumferential pattern, as opposed to the inner longitudinal and outer oblique myocardium. Hypertrophy and fibrosis mainly take place in this middle layer, which might explain the overall reduction of this measure verified in our population [15].
As hypothesized, impaired myocardial deformation parameters evaluated by 3DSTE, namely 3D GLS, were associated with worse functional capacity objectively assessed by CPET parameters. On the other hand, there was not a significant correlation between LVEF or GLS obtain by 2D method and pVO2.
Absolute values of 3D GLS showed the strongest correlation with pVO2 in univariate analysis and were independently associated with pVO2 in multivariable analysis.
3D GLS was also significantly decreased from patients in NYHA class I to class III, which further confirms the association of this parameter with functional capacity. On the other hand, significant GLS variations between patients in NYHA class II and III could not be found. However, 2DGLS was also associated with NYHA class changes.
3D speckle tracking echocardiography has been recognized has a useful tool in the global evaluation of HCM: some studies have reported its utility in predicting atrial fibrillation [13], family screening for HCM [14] and arrhythmic risk stratification [15]. Our findings suggest that 3DSTE might have additional applications in predicting functional capacity.
Incorporating 3DSTE in daily clinical practice can have positive prognostic implications in these patients, allowing for an early identification of patients with worse outcomes, facilitating more effective management strategies and timely interventions. Furthermore, prediction of functional capacity through 3DSTE can represent an alternative for patients who cannot undergo CPET.
Diastolic disfunction and functional capacity
Our population showed reduced early diastolic mitral annular velocities measured using Doppler tissue imaging. Myocardial hypertrophy, ischemia secondary to coronary microvascular dysfunction and interstitial fibrosis causing chamber stiffness are responsible for impaired ventricular myocardial relaxation, largely described in patients with HCM [6], with consequent increase in intracavitary pressure.
In our cohort average E/e’ ratio correlated with functional capacity in univariate analysis. Unlike conventional Doppler indices alone, the transmitral E to e’ ratio has been particularly correlated with NYHA functional class in patients with HCM [16].
Exercise intolerance in patients with HCM can be, at least in part, attributed to raised left atrial pressures. However, the relation between average E/e’ and left atrium index volume and pVO2 were not significant when applied a multivariate analysis, which may indicate that other mechanisms such as reduced stroke volume response, ventilation/perfusion mismatch and abnormal peripheral oxygen utilisation may also influence exercise capacity [17].
Controversy remains regarding left ventricular diastolic pressure and functional capacity at rest as major determinants of exercise capacity in patients with HCM, and more investigation is needed to clear the true mechanisms.
Study limitations
The present work has some limitations. The sample size is relatively small, which may limit the generalisation of these findings and contributed for the absence of strong correlations in our analysis. Echocardiographic parameters were obtained at rest and not simultaneously with the physical effort.
Conclusions
Impaired myocardial deformation parameter 3DGLS was independently associated with worse functional capacity assessed by CPET.
Our study supports the emerging data about 3DSTE applications in HCM and its advantages over standard echocardiographic methods, namely in the evaluation of systolic function.
Abbreviations
- CMR:
-
Cardiovascular magnetic resonance
- CPET:
-
Cardiopulmonary exercise testing
- GCS:
-
Global circumferential strain
- GLS:
-
Global longitudinal strain
- GRS:
-
Global radial strain
- HCM:
-
Hypertrophic cardiomyopathy
- LV:
-
Left ventricular
- LVOT:
-
Left ventricular outflow tract
- MWT:
-
Maximum wall thickness
- NYHA:
-
New York Heart Association
- NSVT:
-
Non-sustained ventricular tachycardia
- TAPSE:
-
Tricuspid annular plane systolic excursion
- VCO2:
-
CO2 production
- VE:
-
Ventilation
- VO2 :
-
Oxygen uptake
- 2D:
-
Two-dimensional
- 3D:
-
Three-dimensional
References
Maron BJ, McKenna WJ, Danielson GK et al (2003) American College of Cardiology/European Society of Cardiology Clinical Expert Consensus Document on hypertrophic cardiomyopathy: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus documents and the European Society of Cardiology Committee for Practice Guidelines. Journal of the American College of Cardiology. Elsevier Inc., pp 1687–1713
McLeod CJ, Bos JM, Theis JL et al (2009) Histologic characterization of hypertrophic cardiomyopathy with and without myofilament mutations. Am Heart J 158:799–805. https://doi.org/10.1016/j.ahj.2009.09.006
Magrì D, Re F, Limongelli G et al (2016) Heart failure progression in hypertrophic cardiomyopathy - possible insights from cardiopulmonary exercise testing. Circ J 80:2204–2211. https://doi.org/10.1253/circj.CJ-16-0432
Smith JR, Medina-Inojosa JR, Layrisse V et al Clinical Medicine Predictors of Exercise Capacity in patients with hypertrophic obstructive cardiomyopathy. https://doi.org/10.3390/jcm7110447
Ommen SR, Mital S, Burke MA et al (2020) 2020 AHA/ACC Guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 142:e558–e631. https://doi.org/10.1161/CIR.0000000000000937
Cecchi F, Charron P, Alain Hagege A, et al ESC et al GUIDELINES 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy the Task Force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). https://doi.org/10.1093/eurheartj/ehu284
Mitchell C, Rahko PS, Blauwet LA et al (2019) Guidelines for performing a comprehensive transthoracic echocardiographic examination in adults: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr 32:1–64. https://doi.org/10.1016/j.echo.2018.06.004
Muraru D, Niero A, Rodriguez-Zanella H et al (2018) Three-dimensional speckle-tracking echocardiography: benefits and limitations of integrating myocardial mechanics with three-dimensional imaging. Cardiovasc Diagn Ther 8:101–117
Sugimoto T, Dulgheru R, Bernard A et al (2017) Echocardiographic reference ranges for normal left ventricular 2D strain: results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging 18:833–840. https://doi.org/10.1093/EHJCI/JEX140
Bernard A, Addetia K, Dulgheru R et al (2017) 3D echocardiographic reference ranges for normal left ventricular volumes and strain: results fromthe EACVI NORRE study. Eur Heart J Cardiovasc Imaging 18:475–483. https://doi.org/10.1093/ehjci/jew284
Saraiva RM, Scolin EMB, Pacheco NP et al 3-Dimensional Echocardiography and 2-D strain analysis of Left Ventricular, Left Atrial and right ventricular function in healthy brazilian volunteers. https://doi.org/10.5935/abc.20190155
Masri A, Pierson LM, Smedira NG et al (2015) Predictors of long-term outcomes in patients with hypertrophic cardiomyopathy undergoing cardiopulmonary stress testing and echocardiography. Am Heart J 169:684–692e1. https://doi.org/10.1016/j.ahj.2015.02.006
Urbano-Moral JA, Rowin EJ, Maron MS et al (2014) Investigation of global and regional myocardial mechanics with 3-dimensional speckle tracking echocardiography and relations to hypertrophy and fibrosis in hypertrophic cardiomyopathy. Circ Cardiovasc Imaging 7:11–19. https://doi.org/10.1161/CIRCIMAGING.113.000842
Rakesh K, Rajesh GN, Vellani H (2021) 3D speckle tracking echocardiographic strain pattern in hypertrophic cardiomyopathy and its relation with Sudden Cardiac Death risk markers. Indian Heart J 73:451–457. https://doi.org/10.1016/j.ihj.2020.11.144
Pu C, Fei J, Lv S et al (2021) Global circumferential strain by Cardiac magnetic resonance tissue tracking Associated with ventricular arrhythmias in hypertrophic cardiomyopathy patients. Front Cardiovasc Med 8. https://doi.org/10.3389/fcvm.2021.670361
Inciardi RM, Galderisi M, Nistri S et al (2018) Echocardiographic advances in hypertrophic cardiomyopathy: three-dimensional and strain imaging echocardiography. Echocardiography 35:716–726
Aly MFA, Brouwer WP, Kleijn SA et al (2014) Three-dimensional speckle tracking echocardiography for the preclinical diagnosis of hypertrophic cardiomyopathy. Int J Cardiovasc Imaging 30:523–533. https://doi.org/10.1007/s10554-014-0364-5
Funding
Open access funding provided by FCT|FCCN (b-on).
Author information
Authors and Affiliations
Contributions
I.C, J.V. and S.A.R. wrote the main manuscript. L.L, R.F., L.M.B and A.G. revised the manuscript. The remaining authors contributed to data collection.
Corresponding author
Ethics declarations
Conflict of interest
Nothing to Disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cardoso, I., Viegas, J.M., Rosa, S.A. et al. Three-dimensional echocardiography for the evaluation of hypertrophic cardiomyopathy patients: relation to symptoms and exercise capacity. Int J Cardiovasc Imaging 39, 2475–2481 (2023). https://doi.org/10.1007/s10554-023-02952-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-023-02952-5