Abstract
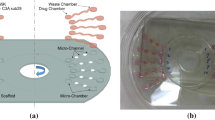
Mammalian cells are sensitive to extracellular microenvironments. In order to faithfully explore the physiological responses of cells to extracellular conditions, a steady, homogenous, and three-dimensional (3-D) culture environment is required because it can provide a more quantifiable and biologically-relevant culture condition. To achieve this, this study reports a perfusion micro cell culture platform encompassing 22 microbioreactor units for high throughput 3-D cell culture. The cell culture platform structurally consisting of a plug and a microbioreactor chamber module was simply fabricated by replica molding of polydimethylsiloxane (PDMS) polymer. The platform features in the proposed plug module with multiple molds incorporated, facilitating the preparation of cell encapsulated 3-D hydrogel constructs in a precise and efficient manner. This trait is found particularly useful for high-precision and high-throughput micro 3-D cell culture-based assay. In this study, the real value of the proposed platform to maintain a stable and homogenous culture condition was discussed. Besides, the application of the presented platform for precisely investigating the effect of serum concentration on the metabolic activities and biosynthetic abilities of articular chondrocytes was also demonstrated. As a whole, the proposed device has paved an alternative route to carry out high throughput micro-scale 3-D perfusion cell culture in a simple, cost-effective and precise manner. The promising applications include 3-D cell culture-based high throughput drug or toxicity testing/screening, or other investigations on the cell biology, where the precise quantification of the links between the cellular responses and extracellular conditions is required.




Similar content being viewed by others
References
A. Abbott, Cell culture: biology’s new dimensions. Nature 424, 870–872 (2003)
F. Abbasi, H. Mirzadeh, A.-A. Katbab, Modification of polysiloxane polymers for biomedical applications: a review. Polym. Int. 50, 1279–1287 (2001)
J.R. Anderson, D.T. Chiu, J.C. McDonald, R.J. Jackman, O. Cherniavskaya, H. Wu, S. Whitesides, G.M. Whitesides, Fabrication of topologically complex three-dimensional microfluidic systems in PDMS by rapid prototyping. Anal. Chem. 72, 3158–3164 (2000)
K. Boxshall, M.H. Wu, Z. Cui, Z.F. Cui, J.F. Watts, M.A. Baker, Simple surface treatments to modify protein adsorption and cell attachment properties within a poly(dimethylsiloxane) microbioreactor. Surf. Interface Anal. 38, 198–201 (2006)
L. Bian, E.G. Lima, S.L. Angione, K.W. Ng, D.Y. Williams, D. Xu, A.M. Stoker, J.L. Cook, G.A. Ateshian, C.T. Hung, Mechanical and biochemical characterization of cartilage explants in serum-free culture. J. Biomech. 41, 1153–1159 (2008)
T. Braschler, R. Johann, M. Heule, L. Metref, P. Renaud, Gentle cell trapping and release on a microfluidic chip by in situ alginate hydrogel formation. Lab Chip 5, 553–559 (2005)
E. Cukierman, R. Pankov, D.R. Stevens, K.M. Yamada, Taking cell-matrix adhesions to the third dimension. Science 294, 1708–1712 (2001)
E. Cukierman, R. Pankov, K.M. Yamada, Cell interactions with three dimensional matrices. Curr. Opin. Cell Biol. 14, 633–639 (2002)
Y. Choi, J. Vukasinovic, A. Glezer, M.G. Allen, MEMS-based fabrication and microfluidic analysis of three-dimensional perfusion systems. Biomed. Microdevices 10, 437–446 (2008)
Y.S. Choi, M.L. Sang, W.L. Chang, K. Dong-IL, Development of serum-free media for primary culture of human articular chondrocytes. J. Microbiol. Biotechnol. 15, 1299–1303 (2005)
Z.F. Cui, X. Xu, N. Trainor, J.T. Triffitt, J.P. Urban, U.K. Tirlapur, Application of multiple parallel perfused microbioreactors and three-dimensional stem cell culture for toxicity testing. Toxicol. In Vitro 21, 1318–1324 (2007)
J. El-Ali, P.K. Sorger, K.F. Jensen, Cells on chips. Nature 442, 403–411 (2006)
A.M. Freyria, D. Cortial, M.C. Ronziere, S. Guerret, D. Herbage, Influence of medium composition, static and stirred conditions on the proliferation and matrix protein expression of bovine articular chondrocytes cultured in a 3-D collagen scaffold. Biomaterials 25, 687–697 (2004)
R.W. Farndale, D.J. Buttle, A.J. Barrett, Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim. Biophys. Acta 883, 173–177 (1986)
G. Gstraunthaler, Alternative to the use of fetal bovine serum: serum-free cell culture. ALTEX 20, 275–281 (2003)
J. Glowacki, K.E. Yates, R. MacLean, S. Mizuno, In vitro engineering of cartilage: effect of serum substitutes TGF-β and IL-1α. Orthod. Craniofac. Res. 8, 200–2008 (2005)
M.J. Grimshaw, R.M. Mason, Modulation of bovine articular chondrocyte gene expression in vitro by oxygen tension. Osteoarthritis Cartilage 9, 357–364 (2001)
C.D. Hoemann, J. Sun, V. Chrzanowski, M.D. Buschmann, A multivalent assay to detect glycosaminoglycan, protein, collagen, RNA, DNA content in milligram samples of cartilage or hydrogel-based repair cartilage. Anal. Biochem. 300, 1–10 (2002)
B.S. Kim, S.P. Yoo, H.W. Park, Tissue engineering of cartilage with chondrocytes cultured in a chemically-defined, serum-free medium. Biotechnol. Lett. 26, 709–712 (2004)
M.S. Kim, J.H. Yeon, J.K. Park, A microfluidic platform for 3-dimensional cell culture and cell-based assays. Biomed. Microdevices 9, 25–34 (2007)
S.H. Kamil, K. Kojima, M.P. Vacanti, V. Zaporojan, C.A. Vacanti, R.D. Eavey, Tissue engineered cartilage: utilization of autologous serum and serum-free media for chondrocyte culture. Int. J. Pediatr. Otorhinolaryngol. 71, 71–75 (2007)
J.N. Lee, X. Jiang, D. Ryan, G.M. Whitesides, Compatibility of mammalian cells on surfaces of polydimethylsiloxane. Langmuir 20, 11684–11691 (2004)
R.B. Lee, J.P.G. Urban, Evidence of a negative Pasteur effect in articular cartilage. Biochem. J. 321, 95–102 (1997)
J.L. Lin, M.H. Wu, C.Y. Kuo, K.D. Lee, Y.L. Shen, Application of indium tin oxide (ITO)-based microheater chip with uniform thermal distribution for perfusion cell culture outside a cell incubator. Biomed. Microdevices 12, 389–398 (2010)
E.W. Mandl, S.W. Van Der Veen, J.A.N. Verhaar, G.J.V.M. Van Osch, Serum-free medium supplemented with high-concentration FGF2 for cell expansion culture of human ear chondrocytes promotes re-differentiation capacity. Tissue Eng. 8, 573–580 (2002)
T.I. Morales, The role and content of endogenous insulin-like growth factor-binding protein in bovine articular cartilage. Arch. Biochem. Biophys. 343, 164–172 (1997)
S. Ostrovidov, J. Jiang, Y. Sakai, T. Fujii, Membrane-based PDMS microbioreactor for perfused 3D primary rat hepatocyte cultures. Biomed. Microdevices 6, 279–287 (2004)
T.D. Pollard, W.C. Earnshaw, Cell biology, (Elsevier Science, 2002), pp. 536–537
S. Razaq, R.J. Wilkins, J.P.G. Urban, The effect of extracellular pH on matrix turnover by cells of the bovine nucleus pulposus. Eur. Spine J. 12, 341–349 (2003)
H. Stegemann, K.H. Stalder, Determination of hydroxyproline. Clin. Chim. Acta 18, 267–273 (1967)
M. Sittinger, O. Schultz, G. Keyszer, W.W. Minuth, G.R. Burmester, Artificial tissues in perfusion culture. Int. J. Artif. Organs 20, 57–62 (1997)
Y.C. Toh, J. Zhang, Y.M. Khong, S. Chang, V.D. Samper, D. van Noort, D.W. Hutmacher, H. Yu, A novel 3D mammalian cell perfusion culture system in microfluidic channels. Lab Chip 7, 302–309 (2007)
J.P. Urban, J.F. McMullin, Swelling pressure of the intervertebral disc: Influence of proteoglycan and collagen content. Biorheology 22, 145–157 (1985)
J.P. Urban, M.T. Bayliss, Regulation of proteoglycan synthesis rate in cartilage in Vitro: Influence of extracellular ionic composition. Biochim. Biophys. Acta 992, 59–65 (1989)
M.H. Wu, J.P.G. Urban, Z.F. Cui, Z. Cui, X. Xu, Effect of extracellular pH on matrix synthesis by chondrocytes in 3D agarose gel. Biotechnol. Prog. 23, 430–434 (2007)
M.H. Wu, J.P.G. Urban, Z. Cui, Z.F. Cui, Development of PDMS microbioreactor with well-defined and homogenous culture environment for chondrocyte 3-D culture. Biomed. Microdevices 8, 331–340 (2006)
M.H. Wu, S.B. Huang, Z.F. Cui, Z. Cui, G.B. Lee, A high throughput perfusion-based microbioreactor platform integrated with pneumatic micropumps for three-dimensional cell culture. Biomed. Microdevices 10, 309–319 (2008)
M.H. Wu, Simple poly(dimethylsiloxane) surface modification to control cell adhesion. Surf. Interface Anal. 41, 11–16 (2009)
K.E. Yates, F. Allemann, J. Glowacki, Phenotypic analysis of bovine chondrocytes cultured in 3-D collagen sponges: effect of serum substitutes. Cell Tissue Bank. 6, 45–54 (2005)
Acknowledgements
The authors would like to thank finical support from the National Science Council (NSC) (NSC 97-2218-E-182-002-MY2) and Chang Gung Memorial Hospital in Taiwan (CMRPD170281 and CMRPD170282).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, MH., Kuo, CY. Application of high throughput perfusion micro 3-D cell culture platform for the precise study of cellular responses to extracellular conditions -effect of serum concentrations on the physiology of articular chondrocytes. Biomed Microdevices 13, 131–141 (2011). https://doi.org/10.1007/s10544-010-9478-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-010-9478-2