Abstract
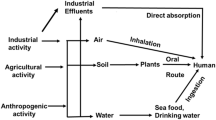
The viability, cellular uptake and subcellular distribution of heavy metal Hg, were determined in human mammary cell lines (MCF-7, MDA-MB-231 and MCF-10A). It was observed that Hg had the capacity of being excluded from the cells with a different type of possible transporters. MCF-7 cells showed the lowest viability, while the other two cell lines were much more resistant to Hg treatments. The intracellular concentration of Hg was higher at lower exposure times in MCF-10A cells and MCF-7 cells; but as the time was increased only MDA-MB-231 showed the capacity to continue introducing the metal. In MCF-7 and MCF-10A cells the subcellular distribution of Hg was higher in cytosolic fraction than nucleus and membrane, but MDA-MB-231 showed membrane and nucleus fraction as the enriched one. The analysis of RNA-seq about the genes or family of genes that encode proteins which are related to cytotoxicity of Hg evidenced that MCF-10A cells and MCF-7 cells could have an active transport to efflux the metal. On the contrary, in MDA-MB-231 no genes that could encode active transporters have been found.






Similar content being viewed by others
References
Afgan E, Baker D, van den Beek M et al (2016) The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2016 update. Nucleic Acids Res 44(W1):W3–W10. https://doi.org/10.1093/nar/gkw343
Aitio A, Alessio L, Axelson O et al (1993) IARC monographs on the evaluation of carcinogenic risks to humans: beryllium, cadmium, mercury, and exposures in the glass manufacturing industry. IARC monographs on the evaluation of carcinogenic risks to humans, vol 58. IARC, Lyon, pp 1–415
Anders S, Pyl PT, Huber W (2015) HTSeq-A python framework to work with high-throughput sequencing data. Bioinformatics 31(2):166–169. https://doi.org/10.1093/bioinformatics/btu638
Andrews S (2010) FastQC: a quality control tool for high throughput sequence data (Galaxy version 0.65). Babraham Institute, Cambridge
Baghirova S, Hughes BG, Hendzel MJ, Schulz R (2015) Sequential fractionation and isolation of subcellular proteins from tissue or cultured cells. MethodsX 2:440–445. https://doi.org/10.1016/j.mex.2015.11.001
Board PG, Menon D (2016) Structure, function and disease relevance of Omega-class glutathione transferases. Arch Toxicol 90(5):1049–1067. https://doi.org/10.1007/s00204-016-1691-1
Bose-O’Reilly S, Lettmeier B, Roider G, Siebert U, Drasch G (2008) Mercury in breast milk—a health hazard for infants in gold mining areas? Int J Hyg Environ Health 211(5):615–623. https://doi.org/10.1016/j.ijheh.2007.09.015
Bridges CC, Zalups RK (2005) Molecular and ionic mimicry and the transport of toxic metals. Toxicol Appl Pharmacol 204(3):274–308. https://doi.org/10.1016/j.taap.2004.09.007
Bridges CC, Zalups RK (2010) Transport of inorganic mercury and methylmercury in target tissues and organs. J Toxicol Environ Health B 13(5):385–410. https://doi.org/10.1080/10937401003673750
Bucio L, García C, Souza V et al (1999) Uptake, cellular distribution and DNA damage produced by mercuric chloride in a human fetal hepatic cell line. Mut Res 423(1–2):65–72. https://doi.org/10.1016/s0027-5107(98)00226-7
Byrne C, Divekar SD, Storchan GB, Parodi DA, Martin MB (2013) Metals and breast cancer. J Mammary Gland Biol Neoplasia 18(1):63–73. https://doi.org/10.1007/s10911-013-9273-9
Costa V, Aprile M, Esposito R, Ciccodicola A (2013) RNA-Seq and human complex diseases: recent accomplishments and future perspectives. Eur J Hum Genet 21(2):134–142. https://doi.org/10.1038/ejhg.2012.129
Ding J, Wang J, Xiang Z et al (2017) The organic anion transporting polypeptide 1a5 is a pivotal transporter for the uptake of microcystin-LR by gonadotropin-releasing hormone neurons. Aquat Toxicol 182:1–10. https://doi.org/10.1016/j.aquatox.2016.11.005
Dos Santos FA, Cavecci B, Vieira JCS et al (2015) A metalloproteomics study on the association of mercury with breast milk in samples from lactating women in the Amazon Region of Brazil. Arch Environ Contam Toxicol 69(2):223–229. https://doi.org/10.1007/s00244-015-0161-8
Fujiyama J, Hirayama K, Yasutake A (1994) Mechanism of methylmercury efflux from cultured astrocytes. Biochem Pharmacol 47(9):1525–1530. https://doi.org/10.1016/0006-2952(94)90527-4
Heggland I, Kaur P, Syversen T (2009) Uptake and efflux of methylmercury in vitro: comparison of transport mechanisms in C6, B35 and RBE4 cells. Toxicol In Vitro 23(6):1020–1027. https://doi.org/10.1016/j.tiv.2009.06.018
Ho RH, Kim RB (2010) Uptake transporters comprehensive toxicology, vol 4, 2nd edn. Vanderbilt University School of Medicine, Nashville, pp 519–556
Hollman AL, Tchounwou PB, Huang HC (2016) The association between gene-environment interactions and diseases involving the human GST superfamily with SNP variants. Int J Environ Res Public Health 13(4):379. https://doi.org/10.3390/ijerph13040379
Irvine GW, Pinter TBJ, Stillman MJ (2016) Defining the metal binding pathways of human metallothionein 1a: balancing zinc availability and cadmium seclusion. Metallomics 8(1):71–81. https://doi.org/10.1039/c5mt00225g
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360. https://doi.org/10.1038/nmeth.3317
Kindla J, Rau TT, Jung R et al (2011) Expression and localization of the uptake transporters OATP2B1, OATP3A1 and OATP5A1 in non-malignant and malignant breast tissue. Cancer Biol Ther 11(6):584–591. https://doi.org/10.4161/cbt.11.6.14533
Kiss K, Kucsma N, Brozik A et al (2015) Role of the N-terminal transmembrane domain in the endo-lysosomal targeting and function of the human ABCB6 protein. Biochem J 467(1):127–139. https://doi.org/10.1042/BJ20141085
Klijn C, Durinck S, Stawiski EW et al (2015) A comprehensive transcriptional portrait of human cancer cell lines. Nat Biotechnol 33(3):306–312. https://doi.org/10.1038/nbt.3080
Legge SE, Hamshere ML, Ripke S et al (2016) Genome-wide common and rare variant analysis provides novel insights into clozapine-associated neutropenia. Mol Psychiatry. https://doi.org/10.1038/mp.2016.137
Llop S, Ballester F, Broberg K (2015) Effect of gene-mercury interactions on mercury toxicokinetics and neurotoxicity. Curr Environ Health Rep 2(2):179–194. https://doi.org/10.1007/s40572-015-0047-y
Lohren H, Bornhorst J, Galla HJ, Schwerdtle T (2015) The blood-cerebrospinal fluid barrier—first evidence for an active transport of organic mercury compounds out of the brain. Metallomics 7(10):1420–1430. https://doi.org/10.1039/c5mt00171d
Mohammadi M, Riyahi Bakhtiari A, Khodabandeh S (2014) Concentration of Cd, Pb, Hg, and Se in different parts of human breast cancer tissues. J Toxicol. https://doi.org/10.1155/2014/413870
Nabi S (2014) Toxic effects of mercury. Springer, New Delhi
Palacios Ò, Capdevila M (2013) Toxicology (Pb, Hg, Cd, As, Al, Cr, and Others) comprehensive inorganic chemistry II: from elements to applications, vol 3, 2nd edn. Pergamon Press, Oxford, pp 51–63
Parks JM, Smith JC (2016) Modeling mercury in proteins. Methods Enzymol 578:103–122. https://doi.org/10.1016/bs.mie.2016.05.041
Patnaik R, Padhy RN (2015) Cellular and nuclear toxicity of HgCl < inf > 2</inf > to in vitro grown lymphocytes from human umbilical cord blood. Proc Natl Acad Sci India Sect B 85(3):821–830. https://doi.org/10.1007/s40011-015-0503-3
Sebastian K, Detro-Dassen S, Rinis N et al (2013) Characterization of SLCO5A1/OATP5A1, a solute carrier transport protein with non-classical function. PLoS ONE. https://doi.org/10.1371/journal.pone.0083257
Stacchiotti A, Morandini F, Bettoni F et al (2009) Stress proteins and oxidative damage in a renal derived cell line exposed to inorganic mercury and lead. Toxicology 264(3):215–224. https://doi.org/10.1016/j.tox.2009.08.014
Straka E, Ellinger I, Balthasar C et al (2016) Mercury toxicokinetics of the healthy human term placenta involve amino acid transporters and ABC transporters. Toxicology 340:34–42. https://doi.org/10.1016/j.tox.2015.12.005
Vázquez M, Vélez D, Devesa V, Puig S (2015) Participation of divalent cation transporter DMT1 in the uptake of inorganic mercury. Toxicology 331:119–124. https://doi.org/10.1016/j.tox.2015.03.005
Waalkes MP, Fox DA, States JC, Patierno SR, McCabe MJ Jr (2000) Metals and disorders of cell accumulation: modulation of apoptosis and cell proliferation. Toxicol Sci 56(2):255–261
Zalups RK, Lash LH (1997) Binding of mercury in renal brush-border and basolateral membrane-vesicles: implication of a cysteine conjugate of mercury involved in the luminal uptake of inorganic mercury in the kidney. Biochem Pharmacol 53(12):1889–1900. https://doi.org/10.1016/S0006-2952(97)00138-X
Zhang X, Wang Y, Zhao Y, Chen X (2008) Experimental study on the estrogen-like effect of mercuric chloride. Biometals 21(2):143–150. https://doi.org/10.1007/s10534-007-9102-y
Zhou H, Brock J, Liu D, Board PG, Oakley AJ (2012) Structural insights into the dehydroascorbate reductase activity of human omega-class glutathione transferases. J Mol Biol 420(3):190–203. https://doi.org/10.1016/j.jmb.2012.04.014
Acknowledgements
This work was supported by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Agencia Nacional de Promoción Científica y Tecnológica (FONCYT) (PICT-BID), Universidad Nacional de Cuyo (Argentina) and Instituto Nacional del Cáncer (INC). We gratefully acknowledge access to RNAseq data base from Genentech.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maniero, M.Á., Guerrero-Gimenez, M.E., Fanelli, M.A. et al. Inorganic mercury in mammary cells: viability, metal uptake but efflux?. Biometals 31, 69–80 (2018). https://doi.org/10.1007/s10534-017-0068-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-017-0068-0