Abstract
Transport of terrestrial carbon through riverine systems to coastal water has a negative impact on oxygen concentration in coastal areas. However, information on seasonal variation and the impact of catchment composition on the bioavailability of allochthonous carbon is lacking. In this project we address this knowledge gap by investigating the reactivity of dissolved organic carbon (DOC) at river mouths which originate from agricultural or forested dominated catchments over a year. Using a high-capacity oxygen sensing system biological oxygen demand (BOD) was measured and converted to carbon utilization rate. This allowed a spatial and temporal resolution necessary to understand how concentrations of total nitrogen, total phosphorus, DOC, as well as carbon composition influence carbon bioavailability. Seasonality and the differing catchment compositions yielded variable results about which factors were significantly contributing to reactivity. In addition, we found that carbon utilization rate was highest during April, June, and October for most rivers. The bioavailable fraction (BFc) was significantly higher in rivers with agricultural compared to forest dominated catchments during January, April, June, and October. However, rivers with agricultural dominated catchment had a significantly higher carbon utilization rate in August. This indicate that rivers dominated by forest transport larger and more refractory pools of carbon, while rivers with agricultural dominated catchments have a higher percentage of BFc. Based on these results we suggest that management efforts, to reduce the transport of bioavailable carbon, would be most efficient during spring and autumn with equal importance on rivers with catchment areas dominated by agriculture and forest.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Baltic Sea is suffering from severe anthropogenic nutrient input causing anoxic conditions (Kotta et al. 2020), with permanent anoxia in deep bottom waters and periodic hypoxia in shallower coastal regions (Conley et al. 2011). The focus of research over the past couple of decades has been to identify and to remediate the anoxic conditions caused by excess nitrogen and phosphorus run off (Kotta et al. 2020). However, hypoxia in the Baltic Sea persists, even if management efforts have successfully reduced the nutrient loading (Gustafsson et al. 2012; HELCOM 2021). This has led current research into investigating the effects of increased transport of terrestrial organic matter on coastal marine environments (Soares et al. 2019). The utilization of bioavailable carbon by bacteria, a process which requires oxygen, may contribute to a lower oxygen concentration in coastal areas (Dagg et al. 2008). Rivers flowing into the Baltic Sea supply organic matter and as a consequence 86% of the DOM in the Baltic Sea is thought to be terrestrially derived (Deutsch et al. 2012). The transported organic matter is made up of 45% organic carbon, with around 90% of the carbon existing as DOC and a small fraction as particulate organic matter (POC) (Maciejewska and Pempkowiak 2014).
The molecular composition of DOC depends on the source of carbon (Autio et al. 2016), which affects the reactivity and utilization by microbes (Reyes and Crisosto 2016). Autochthonous carbon which is produced by autotrophs within the system (Kritzberg et al. 2004) has a low molecular weight and aromaticity and is bioavailable to bacteria (Autio et al. 2016), but has previously been believed to be completely taken up in the riverine system before reaching coastal waters (Catalán et al. 2016). Allochthonous carbon which is carbon transported from the catchment area to the system (Kritzberg et al. 2004), was earlier believed to be refractory to microbial degradation, but several recent studies have shown microbial utilization of terrestrial derived organic carbon (Figueroa et al. 2016; Kritzberg et al. 2004; Kuliński et al. 2016). The quantity and composition of allochthonous carbon in rivers is affected by the characteristics and land-use of the catchment area (Agren et al. 2007; Reader et al. 2014), but also on precipitation, anthropogenic alterations such as clear cutting, and soil type (Sepp et al. 2019). In addition, composition is related to water residence time (WRT) with longer WRT allowing more time for photodegradation and microbial utilization, resulting in less bioavailable carbon (Autio et al. 2016). The source of allochthonous carbon determine the bioreactivity, for example peat land and boreal forest have been found to export carbon with high molecular weight and low bioavailability (Autio et al. 2016). Whereas agricultural land has shown to export carbon with a low molecular weight and high bioavailability (Autio et al. 2016).
Previous studies that focus on the transport of bioavailable carbon into the Baltic Sea have either investigated only a few sampling sites (Reader et al. 2014) or have sampled only during seasons with highest productivity (spring and autumn) (Asmala et al. 2013; Soares et al. 2019) and therefore lack an understanding of seasonal changes and how that affects the reactivity and loading (Autio et al. 2016). In this research we investigated how biological oxygen demand (BOD), which was converted to carbon utilization rate (R = ΔC/Δt (mg C L−1 d−1), and the bioavailable fraction (BFc) of riverine carbon (% of DOC utilized by bacteria within 24 h) is influenced by catchment composition and how it varies during different seasons. We hypothesize that carbon utilization rate and the BFc will be related to carbon composition and concentrations of nitrogen and phosphorus. Further we hypothesized that carbon utilization rate and the BFc peak in spring/early summer during highest terrestrial productivity. Lastly, we believe that rivers with agricultural dominated catchments contain a higher bioavailable fraction compared to forest dominated catchments. The results from this study provide insight into the seasonality of allochthonous carbon inputs and BOD relative to hypoxia in the Baltic Sea.
Materials and methods
Sampling and set-up
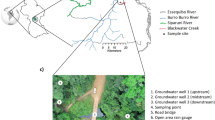
Sampling of nine rivers draining into the Baltic Sea was performed five times during 2020 (January, April, June, August, and October). At each time point 10 L of water were collected close to the river mouths of Emån, Alsterån, Ljungbyån, Lyckebyån, Mörrumsån, Helgeå, Skivarpsån, Kävlingeån, and Råån (Fig. 1). Rivers were selected to encompass the varying landscapes and land-use of catchment areas across Sweden (Table 1). Rivers 1–6 have catchment areas dominated by forest, while catchments of rivers 7–9 are dominated by agriculture land (Table 1; Fig. 1). The water from the rivers were filtered through type A/E glass fiber filters (142 mm, PALL Corporation) to remove particles and collected in clean 10 L bottles.
Nutrients and DOC
Nutrient and DOC samples were subsampled from the 10-liter bottles and collected in acid-washed polyethylene bottles (50 mL and 100 mL respectively) and then frozen. For samples collected in January, nitrogen concentrations were processed at Denmark Technical University using standard protocols (Hansen 1999) on a SmartChem 2000 discrete analyzer (AMS Alliance). Nitrogen concentrations in April, June, August, and October, as well as all DOC samples were analyzed by the department of Biology, Lund University, using a TOC V-CPH/CPN (Shimadzu) with the method Non-Purgeable Organic Carbon (NPOC). Total phosphorus was analyzed manually using MRP-method (Menzel & Corwin 1965).
Carbon utilization rate and bioavailable fraction
Biological oxygen demand (BOD) was measured and converted to carbon utilization rate. Water from each river was used to start triplicate incubations in 2 L glass bottles. The bottles were filled carefully to avoid bubble formation and to remove all the headspace and then sealed with gas tight stoppers. In addition to the nine rivers, three controls with milliQ water were also included, resulting in a set-up with 30 incubation bottles. The incubation bottles were incubated in darkness for 5 days in a temperature controlled room at 20 °C (Wilhelm 2009). Oxygen (mg/L) was measured every 30 min using optical oxygen sensors (optodes) and three 10-channel fiber oxygen transmitters (oxy-10, PreSens®). It took a few hours for the system to reach 20 °C. The first 15 h were therefore removed in the data analysis. However, in January stabilization took longer and the first 24 h were removed. The slope for oxygen consumption was calculated using excel (R = ΔO2/Δt (mg O2 L−1 d−1), Fig. S1). The oxygen consumption was converted to carbon units using a molar respiratory quotient of 1.0 (Soares et al. 2019) which is close to estimates for rivers (Berggren et al. 2012). BFc was calculated by dividing the carbon utilized in 24 h by the DOC concentration.
Absorbance
A subsample of the filtered water from each river was used for absorbance measurements and to calculate absorbance ratios for carbon size, watercolor, and aromaticity as well as specific ultraviolet absorbance (SUVA) (De Haan HaDB 1987; Joo et al. 2008; Li et al. 2016; Reyes and Crisosto 2016; Uyguner and Bekbolet 2005). Absorbance was measured every 1 nm from 200 to 800 nm on a UV-2600, UV–VIS spectrophotometer (Shimadzu) using a 1 cm quartz cuvette and with milli-Q water as a reference and a blank. The ratio a250/a365 provides an index of the size of the carbon molecules, where a low value indicates larger molecules, and a high value indicates smaller molecules (Reyes and Crisosto 2016). Watercolor was calculated based on a420 (Pennanen 1986). Specific UV-absorbance (SUVA (m2 g−1 C) was calculated as the ratio between a254 nm (m−1) and total organic carbon (mg L−1) (Goodman et al. 2011). SUVA is used as an indicator of the aromatic content of DOC and provides information about the source and reactivity of the carbon (Goodman et al. 2011). The high SUVA value is related to high aromatic content (Goodman et al. 2011). Aromaticity of carbon molecules is also described by the ratio a254/a204, with a high value indicating high aromaticity (Reyes and Crisosto 2016). This absorbance ratio provides understanding of the composition of carbon but in contrast to SUVA it does not account for total organic carbon concentration.
Statistics
All statistical analysis were performed in Prism graphpad 9.2.0. One-way anova was used to compare carbon utilization rate and bioavailable fraction between sampling months for each river. Rivers were grouped into forest dominated and agricultural dominated catchment areas based on land use data from SMHI (Table 1). Unpaired t tests were used to compare carbon utilization rate and bioavailable fraction between rivers with catchment areas dominated by agriculture or forest. Multiple linear regressions were used to investigate which variables (DOC, carbon composition, concentration of total nitrogen (TN) and concentration of total phosphorus (TP)) explain the variation in carbon utilization rate and the BFc. However, the results in the manuscript are based on single regressions because the multiple linear regressions did not yield any significant results.
Results
Seasonal and spatial variations in nutrient concentrations and carbon composition
Concentration of DOC, total N and total P
Concentrations of DOC, TN and TP varied both seasonally and between rivers with catchment areas dominated by agriculture and forest, respectively. The highest DOC concentrations were found in April, June, or October in rivers with catchment areas dominated by forest, whereas rivers with catchment area dominated by agricultural land had highest concentrations of DOC in August or October. Rivers with forested catchments generally had lowest DOC concentrations in August. The only exception was Alsterån which had the lowest concentration in January (Table 2). In January and April DOC was generally higher in rivers with forest dominated catchments (14.9–27.7 mg/L) than in rivers with agricultural dominated catchment areas (5.5–9.5 mg/L) (Table 2, t test, p = 0.0013 (January), p = 0.0018 (April)). The TN concentration was significantly higher in April compared to other months in all rivers (ANOVA, p < 0.0001). On the other hand, TP was highest in June or August (Table 2). In all months except January and April, TP concentrations were higher in rivers with agricultural dominated catchments than in rivers with forest dominated catchments (ANOVA, p < 0.001, Table 2).
Carbon composition
Carbon composition also varied both seasonally and between rivers with catchment areas dominated by agriculture or forest. In rivers with catchment areas dominated by forest, the values of the ratio a250/a365 (molecular size) was generally lowest in January and highest in August (Table 2). However, for rivers with catchment area dominated by agriculture no clear pattern was found (Table 2). Values of the ratio a254/a204 (aromatic content) was generally lower in rivers with agriculture dominated catchment areas than in rivers with forest dominated catchment areas (Table 2). Watercolor (a420) was significantly lower in rivers with agriculturally dominated catchment areas over the whole year compared to rivers with forest dominated catchment areas (Table 2, Agriculture, 20–92.5 mg Pt/L, Forested, 30–285 mg Pt/L, t test, p < 0.001). In addition, the seasonal variations in watercolor were lower in rivers with agricultural dominated catchment areas and the lowest values are detected in April. In contrast, watercolor in forest dominated rivers was lowest in August or October Highest values were detected in January or April for all rivers with forest dominated catchment areas except Alsterån where watercolor was highest in June (Table 2). SUVA fluctuated over the year with highest and lowest values found in different months for the different rivers (Table 2).
Comparison between rivers with forest and agricultural dominated catchments
To investigate the effect of catchment area on carbon utilization rate and BFc, the rivers were divided into two groups based on dominating land use activity in the catchment area (Table 1). There was a significant difference in carbon utilization rate between rivers with forest dominated catchment areas and agricultural dominated catchment areas in August (t test, p = 0.04, Fig. 2), and at this timepoint the carbon utilization rate was highest in rivers with catchment areas dominated by agricultural land. However, the BFc was significantly higher in agricultural dominated catchments than forested dominated catchments in January (t test, p = 0.016), April (t test, p = 0.001), June (t test, p = 0.007) and August (t test, p = 0.009, Fig. 2).
Seasonal variation in carbon utilization rate and bioavailable fraction
Carbon utilization rate and the BFc varied seasonally. In rivers with forest dominated catchment areas carbon utilization rate and the BFc was highest during spring or early summer (April and June) as well as in October (Fig. 3). Ljungbyån showed the largest seasonal variations for carbon utilization rate and the BFc, with values comparable to the other rivers for January - August (2.23–6.85 µmol C L−1 Day −1, 0.13–0.45% BFc) but significantly higher in October (31.97 µmol C L−1 Day −1, 2.45% BFc)(October carbon utilization rate vs. January, April, June, and August carbon utilization rate, ANOVA, p < 0.01) (October BFc vs. January, April, June, and August BFc, ANOVA, p < 0.01, Fig. 3). All forest dominated rivers had lowest values in January and August, except Mörrumsån where the BFc was lowest in January and June (Fig. 3).
Skivarpsån showed the same pattern as the rivers with forest dominated catchment area, where carbon utilization rate and the BFc increased from January to June, decreased in August, then increased again in October (Fig. 3). No significant differences were found for the carbon utilization rate, but the BFc yielded significant differences between January and June respectively June and August (ANOVA, p = 0.044 and 0.022, Fig. 3). Carbon utilization rate and BFc was stable throughout the year in Kävlingeån whereas in Råån carbon utilization rate and the BFc increased from January through August and then declined in October (Fig. 3). In Råån, carbon utilization rate was significantly higher in August compared to January, April, June, and October (ANOVA, p < 0.05, Fig. 3).
Seasonal variations in factors explaining total utilization and bioavailable fraction
Linear regressions were used to investigate which factors explain the variation in carbon utilization rate and the BFc. Based on data from all rivers and all months, there were no significant factors explaining the carbon utilization rate or BFc (Table S1).
Examining each month separately, different factors significantly explained differences in carbon utilization rate and the BFc during different months. Watercolor and SUVA were positively related to the carbon utilization rate in April whereas in August phosphorus concentration was positively related to carbon utilization rate (Table 3). Focusing on the BFc, there were positive relations to nitrogen concentration and a250/a365, and negative relations to DOC concentration, watercolor, a254/a204, and SUVA in April (Table 3). In June phosphorus was positively related, while DOC and a254/a204 were negatively related (Table 3). In August nitrogen concentration is positively related to BFc (Table 3).
Discussion
Carbon transport from terrestrial environments to aquatic systems has been a major player in coastal hypoxia over the years (Conley et al. 2011; Reader et al. 2014). This topic has been examined in many studies, but these primarily focused on bacterial utilization rate during spring and autumn (Ågren et al. 2008; Autio et al. 2016). Few studies have explored the complex nature of carbon utilization and the BFc over the year, which would further expand our understanding of how seasonality effects transport. In addition, there is a gap in understanding how catchment composition influences carbon utilization rate and the BFc.
Comparison between rivers with forest and agricultural dominated catchment areas
Catchment composition plays an important role when it comes to the quality of carbon that is leached into riverine systems (Sepp et al. 2019). Boreal forest, when compared to agricultural dominated catchments, leads to loadings of DOM that are higher in humic content and are a major driver for watercolor (Reader et al. 2014; Sepp et al. 2019). In our study, the 3 rivers with agricultural dominated catchments had significantly lower watercolor throughout the year compared to rivers with forest dominated catchments (t test, p < 0.0001, Table 2). Similarly, in these rivers a254/a204 values were consistently lower (Table 2). Data on watercolor and a254/a204 therefore suggest that carbon exported from agricultural catchments is more bioavailable (Reader et al. 2014). This agrees with a previous study showing that agricultural catchments in Europe export greater amounts of reactive carbon compared to boreal forested catchments (Autio et al. 2016). In addition, studies have shown that high TP and TN concentrations close to river mouths lead to a higher bioreactivity due to primary production being mostly P limited (Soares et al. 2019). This phosphorus allows for increased primary production which leads to a release of labile carbon (Soares et al. 2019). Our study found elevated nutrient concentrations for the three agricultural dominated rivers compared to the rivers with forested catchments, most likely caused by lowland agriculture and urban environments leaching nutrients (Soares et al. 2019). Based on carbon composition and nutrient concentrations we therefore expected higher carbon utilization rates in rivers with catchment area dominated by agricultural activity compared to forest.
However, we only found a significant difference in carbon utilization rate in August, and at this time point carbon utilization rate was highest in rivers with agricultural catchments (Fig. 2). In contrast, the BFc was significantly higher in rivers with catchment areas dominated by agriculture at all timepoints except October. One reason for the lack of difference in carbon utilization rate at most time points is probably related to rivers with forested catchments having higher DOC concentrations but a lower BFc compared to rivers with an agricultural dominated catchment. This is in agreement with a previous study showing that bioavailability and carbon utilization rate depend on both the quality and quantity of carbon (Berggren et al. 2018).
Seasonal variation in utilization rate and bioavailable fraction
This study sheds light on the seasonal variation in carbon transport and shows that carbon utilization rate and the BFc increased from winter to spring followed by a decrease in August (Fig. 1). This study is in general in agreement with a previous study which found that carbon utilization rate and the BFc are lowest during the summer and are higher during spring and autumn (Reader et al. 2014). The low carbon utilization rate and BFc during winter is most likely due to low water temperatures, hindering bacterial processes and primary production (Scavia and Laird 1987; Wetzel 2001). In agreement it has previously been described that low winter temperatures preserve organic matter in the catchment due to low metabolic rates and a low flow of water through soils (Reader et al. 2014). During spring when the soils thaw, carbon utilization rate and the BFc increases which is related to increased carbon mobility as well as bacterial respiration.
Forested catchments showed a significant decrease in carbon utilization rate and BFc between June and August, while agricultural catchments had a more stable carbon utilization rate and BFc (Fig. 2). The decrease in carbon utilization has previously been explained by that many of these rivers experience a relatively dry summer which decreases loading to the aquatic systems (Reader et al. 2014). However, in our study this is probably related to low flow rates during the summer months (Table S2). Agricultural catchments are not as effected by less loading possibly due to a majority of the carbon being produced within the system by primary producers (Soares et al. 2019). In conclusion we found that forested catchments transport more DOC with a lower BFc, while agriculture catchments transport less DOC with a higher BFc.
Rivers with forested catchments showed large variability for both carbon utilization rate and BFc in October. Ljungbyån, Lyckebyån, and Mörrumsån had their highest carbon utilization rate and BFc during October, and for Lyckebyån and Mörrumsån the carbon utilization rate was twice as high compared to other months (Fig. 2). Possible explanations could be the increase in DOC concentration and a decrease in SUVA and/or a254/a204 between August and October (Table 2), suggesting loading of more reactive carbon. Another possible explanation could be increased loading from precipitation. In Ljungbyån there was 62.4 mm of rainfall in October (Table S3) which is 26 mm higher than the historical average (SMHI 2021). Mörrumsån experienced 87.3 mm (Table S3) of rainfall which is 14 mm higher than the historical averages (SMHI 2021). Similarly, it has previously been shown that forested catchments export bacterial productive carbon during flooding (Ågren et al. 2008) resulting in a delivery of reactive material increasing the BOD (Reader et al. 2014). Our results show different seasonal loading patterns for rivers with forested catchments and agricultural catchments, understanding these seasonal fluctuations as well as the driving factors are important for future management efforts.
Seasonal variations in explanatory factors for carbon utilization rate and the bioavailable fraction
Carbon utilization rate has previously been shown to be correlated with both quantity and quality of carbon (Kritzberg et al. 2004) as well as nutrient concentrations (Soares et al. 2019). In our study none of the included parameters (DOC concentration, carbon composition or nutrient concentration) could explain the carbon utilization rate or the BFc in all rivers over the whole year (Table S1). We therefore analyzed the data for carbon utilization rate and BFc for each sampling time separately and found that different factors were important for carbon utilization rate and BFc during different seasons (Table 3). In April carbon utilization rate was positively correlated with both watercolor and SUVA. Generally, more aromatic carbon does not increase bacterial utilization since more complex carbon has been shown to be difficult for bacteria to degrade (Kritzberg et al. 2004). However, bacteria can produce ectoenzymes to break down recalcitrant DOC into sources of energy and nutrients that are more easily utilized (Chróst 1990; Kritzberg et al. 2004). If only a small fraction of the carbon is labile this might be an important strategy to obtain nutrients and carbon. Although DOC concentration did not significantly correlate with carbon utilization rate (Table 3), we found that DOC is positively correlated with watercolor and SUVA (p < 0.0001, p 0.0491). This indicates that the DOC concentration and not the composition is important for the carbon utilization rate. In August, nitrogen concentrations have a significant positive effect on carbon utilization rate (Table 3). A potential explanation for the correlation with nitrogen could be the rivers being nitrogen limited, although other studies have shown that increased phosphorus concentrations resulted in higher rates of microbial growth (Soares et al. 2019). However, the fraction of nitrogen and phosphorus that is bioavailable to bacterioplankton varies over space and between seasons (Muscarella et al. 2014; Soares et al. 2017; Stepanauskas et al. 1999). It has been shown that for dissolved organic nitrogen (DON) anywhere from 2 to 75% (Pellerin et al. 2006) of the nitrogen pool was biologically available whereas for phosphorus the estimates vary from less than 5 to 50% of the TP pool.
Most of the rivers contained their lowest BFc in January whereas the highest BFc was found in June, August, or October and the variation was significantly explained by several factors. Carbon composition has previously been shown to affect carbon utilization rate and the BFc (Berggren et al. 2018). In April, the BFc was negatively correlated to DOC concentration, watercolor (a420), a254/a204 (aromatic content), and SUVA and positively related to a250/a365 (molecular size), whereas in June BFc was negatively correlated to a254/a204 (aromatic content). In general, these correlations show that BFc is lower when the carbon has a higher molecular complexity (negative correlations to watercolor, aromatic content, and SUVA) which is in agreement with that carbon becomes more bioavailable as colored DOM (CDOM) is broken down by bacteria and photodegradation (Goodman et al. 2011).
The BFc was significantly related to nutrient concentration. In April and August there was a positive correlation to TN and in June to TP. During April and June this seems to be related to the loading of nutrients; nitrogen concentrations increased between January and April followed by an increase in phosphorus concentration between April and June (Table 2). As mentioned above the variation in which nutrient is correlated to the BFc might be related to seasonal variations in the bioavailability of the nitrogen and phosphorus pools (Pellerin et al. 2006).
To summarize, the results from the linear regressions show that carbon quantity and nutrient concentrations seem most important for carbon utilization rate whereas carbon quality and nutrient concentrations are more important for BFc.
Conclusion and implications
Our data provides insight to the complex nature of carbon transport and how carbon composition and nutrient concentrations are driving carbon utilization rate and BFc. We have found that while rivers with agricultural catchments have a lower DOC concentration, they have a higher BFc. Contrary, rivers with forested catchments have a higher DOC concentration containing a lower BFc. However, the discharge of bioavailable carbon will be higher from rivers with forested catchments since the flow rate is generally higher than for rivers with agricultural dominated catchments. Knowing which form is more detrimental when it reaches coastal zones is difficult and more studies are needed to investigate the fate of carbon. Climate change, with heavier and more frequent precipitation events, will undoubtedly increase carbon transport and thereby further lower the status of coastal water. A recommendation for management would be to focus efforts during spring and autumn when carbon utilization rate and BFc is highest. A potential way of doing this could be by increasing water retention time with the implementation of ponds and wetlands along the river continuum.
Data availability
The datasets generated during and/or analyzed during the current study is publicly available on Mendeley Data https://doi.org/10.17632/8xc8fn7wjm.1 and from the authors on reasonable request.
References
Agren A, Buffam I, Jansson M, Laudon H (2007) Importance of seasonality and small streams for the landscape regulation of dissolved organic carbon export. J Geophys Res-Biogeosci 112(G3):11
Ågren A, Berggren M, Laudon H, Jansson M (2008) Terrestrial export of highly bioavailable carbon from small boreal catchments in spring floods. Freshw Biol 53(5):964–972
Asmala E, Autio R, Kaartokallio H, Pitkänen L, Stedmon CA, Thomas DN (2013) Bioavailability of riverine dissolved organic matter in three Baltic Sea estuaries and the effect of catchment land use. Biogeosciences 10(11):6969–6986
Autio I, Soinne H, Helin J, Asmala E, Hoikkala L (2016) Effect of catchment land use and soil type on the concentration, quality, and bacterial degradation of riverine dissolved organic matter. Ambio 45(3):331–349
Berggren M, Lapierre JF, del Giorgio PA (2012) Magnitude and regulation of bacterioplankton respiratory quotient across freshwater environmental gradients. Isme J 6(5):984–993
Berggren M, Klaus M, Selvam BP, Ström L, Laudon H, Jansson M, Karlsson J (2018) Quality transformation of dissolved organic carbon during water transit through lakes: contrasting controls by photochemical and biological processes. Biogeosciences 15(2):457–470
Catalán N, Marcé R, Kothawala DN, Tranvik LJ (2016) Organic carbon decomposition rates controlled by water retention time across inland waters. Nat Geosci 9(7):501–504
Chróst RJ (1990) Microbial ectoenzymes in aquatic environments. Springer, New York
Conley DJ, Carstensen J, Aigars J, Axe P, Bonsdorff E, Eremina T, Haahti BM, Humborg C, Jonsson P, Kotta J, Lannegren C, Larsson U, Maximov A, Medina MR, Lysiak-Pastuszak E, Remeikaite-Nikiene N, Walve J, Wilhelms S, Zillen L (2011) Hypoxia is increasing in the Coastal Zone of the Baltic Sea. Environ Sci Technol 45(16):6777–6783
Dagg M, Sato R, Liu HB, Bianchi TS, Green R, Powell R (2008) Microbial food web contributions to bottom water hypoxia in the northern Gulf of Mexico. Cont Shelf Res 28(9):1127–1137
De Haan HaDB T (1987) Applicability of light absorbance and fluorescence as measures of concentration and molecular size of dissolved organic carbon in humic Lake Tjeukemeer. Water Res 21:731–734
Deutsch B, Alling V, Humborg C, Korth F, Mörth CM (2012) Tracing inputs of terrestrial high molecular weight dissolved organic matter within the Baltic Sea ecosystem. Biogeosciences 9(11):4465–4475
Figueroa D, Rowe OF, Paczkowska J, Legrand C, Andersson A (2016) Allochthonous Carbon—a major driver of Bacterioplankton Production in the Subarctic Northern Baltic Sea. Microb Ecol 71(4):789–801
Goodman KJ, Baker MA, Wurtsbaugh WA (2011) Lakes as buffers of stream dissolved organic matter (DOM) variability: temporal patterns of DOM characteristics in mountain stream-lake systems. J Phys Res. https://doi.org/10.1029/2011JG001709
Gustafsson BG, Schenk F, Blenckner T, Eilola K, Meier HEM, Muller-Karulis B, Neumann T, Ruoho-Airola T, Savchuk OP, Zorita E (2012) Reconstructing the development of Baltic Sea Eutrophication 1850–2006. Ambio 41(6):534–548
Hansen HP, Koroleff F (1999) Determination of nutrients. In: Kremling K, Ehrhardt M (eds) Methods of seawater analysis. Wiley, Hoboken
HELCOM (2021) HELCOM Baltic Sea Action Plan – 2021 update
Joo JC, Shackelford CD, Reardon KF (2008) Association of humic acid with metal (hydr)oxide-coated sands at solid-water interfaces. J Colloid Interface Sci 317(2):424–433
Kotta J, Futter M, Kaasik A, Liversage K, Ratsep M, Barboza FR, Bergstrom L, Bergstrom P, Bobsien I, Diaz E, Herkul K, Jonsson PR, Korpinen S, Kraufvelin P, Krost P, Lindahl O, Lindegarth M, Lyngsgaard MM, Muhl M, Sandman AN, Orav-Kotta H, Orlova M, Skov H, Rissanen J, Siaulys A, Vidakovic A, Virtanen E (2020) Cleaning up seas using blue growth initiatives: Mussel farming for eutrophication control in the Baltic Sea. Sci Total Environ 709:12
Kritzberg ES, Cole JJ, Pace ML, Graneli W, Bade DL (2004) Autochthonous versus allochthonous carbon sources of bacteria: results from whole-lake C-13 addition experiments. Limnol Oceanogr 49(2):588–596
Kuliński K, Hammer K, Schneider B, Schulz-Bull D (2016) Remineralization of terrestrial dissolved organic carbon in the Baltic Sea. Mar Chem 181:10–17
Li SJ, Zhang JQ, Mu GY, Ju HY, Wang R, Li DJ, Shabbir AH (2016) Spatiotemporal characterization of Chromophoric dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for highly polluted Rivers. Water 8(9):21
Maciejewska A, Pempkowiak J (2014) DOC and POC in the water column of the southern Baltic. Part I. evaluation of factors influencing sources, distribution and concentration dynamics of organic matter**This study was supported by the Baltic-C/BONUS Plus EUFP6 Project, statutory activities of Institute of Oceanology PAN, Sopot and the Polish Ministry of Science and Higher Education, grant no. N N306 404338. Oceanologia 56(3):523–548
Menzel DW, Corwin N (1965) The measurement of total phosphorus in seawater based on the liberation of organically bound fractions by persulfate oxidation1. Limnol Oceanogr 10(2):280–282
Muscarella M, Bird K, Larsen M, Placella S, Lennon J (2014) Phosphorus resource heterogeneity in microbial food webs. Aquat Microb Ecol 73(3):259–272
Pellerin BA, Kaushal SS, McDowell WH (2006) Does Anthropogenic Nitrogen Enrichment increase Organic Nitrogen concentrations in runoff from forested and human-dominated Watersheds? Ecosystems 9(5):852–864
Pennanen V, Kortelainen P, Mannio J (1986) Comparative study on the estimation of humic matter in natural waters. Publications of the Water Research Institute, National Board of Waters, Finland, No. 65
Reader HE, Stedmon CA, Kritzberg ES (2014) Seasonal contribution of terrestrial organic matter and biological oxygen demand to the Baltic Sea from three contrasting river catchments. Biogeosciences 11(12):3409–3419
Reyes TG, Crisosto JM (2016) Characterization of dissolved organic matter in river water by conventional methods and direct sample analysis-time of flight-mass spectrometry. J Chem. https://doi.org/10.1155/2016/1537370
Scavia D, Laird GA (1987) Bacterioplankton in Lake Michigan: Dynamics, controls, and significance to carbon flux1. Limnol Oceanogr 32(5):1017–1033
Sepp M, Koiv T, Noges P, Noges T (2019) The role of catchment soils and land cover on dissolved organic matter (DOM) properties in temperate lakes. J Hydrol 570:281–291
SMHI (2021) Swedish Meteorological and Hydrological Institute. https://vattenwebb.smhi.se/modelarea/. Accessed 25 Oct 2021
Soares ARA, Bergström A-K, Sponseller RA, Moberg JM, Giesler R, Kritzberg ES, Jansson M, Berggren M (2017) New insights on resource stoichiometry: assessing availability of carbon, nitrogen, and phosphorus to bacterioplankton. Biogeosciences 14(6):1527–1539
Soares ARA, Lapierre J-F, Selvam BP, Lindström G, Berggren M (2019) Controls on dissolved organic carbon bioreactivity in river systems. Sci Rep. https://doi.org/10.1038/s41598-019-50552-y
Stepanauskas R, Leonardson L, Tranvik LJ (1999) Bioavailability of wetland-derived DON to freshwater and marine bacterioplankton. Limnol Oceanogr 44(6):1477–1485
Uyguner CS, Bekbolet M (2005) Evaluation of humic acid photocatalytic degradation by UV–Vis and fluorescence spectroscopy. Catal Today 101(3–4):267–274
Wetzel R (2001) Limnology: Lakes and River Ecosystems. Academic Press, San Diego
Wilhelm FM (2009) Pollution of aquatic ecosystems I. In: Likens GE (ed) Encyclopedia of inland waters. Academic Press, Oxford, pp 110–119
Funding
Open access funding provided by Lund University. This research was supported by the Swedish Research Council FORMAS (2020-00730) and Carl Tryggers foundation (CTS 19:337) to Johanna Sjöstedt.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by KJ and JS. The first draft of the manuscript was written by KJ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Handling Editor: J.M. Melack.
Publisher’s Note
Springer nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jones, K., Berggren, M. & Sjöstedt, J. Seasonal variation and importance of catchment area composition for transport of bioavailable carbon to the Baltic Sea. Biogeochemistry 165, 265–276 (2023). https://doi.org/10.1007/s10533-023-01079-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-023-01079-y