Abstract
Climate change is causing major changes in terrestrial ecosystems and biomes around the world. This is particularly concerning in oceanic islands, considered reservoirs of biodiversity, even more in those with a significant altitudinal gradient and high complexity in the vegetation they potentially harbour. Here, in Tenerife (Canary Islands), we have evaluated the changes in potential vegetation belts during the last 20 years by comparing them with a previous study. Considering the intimate linkage between vegetation and climate, we used a methodology based on phytosociological knowledge, ordination techniques and geostatistics, using multivariate spatial interpolations of bioclimatic data. This has allowed us to spatially detect the variations experienced by eight vegetation units during the last 20 years and incorporating a set of vulnerability metrics. New bioclimatic and vegetation cartography are provided according to the current scenario studied (1990–2019). Our results indicate that summit vegetation, humid laurel forest and thermo-sclerophyllous woodland are the habitats that have experienced a very high area loss and mismatch index, strong changes, if we consider that we are only comparing a period of 20 years. Simultaneously, the more xeric vegetation belts, the dry laurel forest and the pine forest would have benefited from this new warmer and drier climate, by gaining area and experiencing strong upward movements. These changes have not been spatially uniform, indicating that the elevational gradient studied not explain completely our results, showing the influence of the complex island topography. Effective landscape management should consider current remnants, transition capacity and movement limitations to better understand current and future vegetation responses in a global change context.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High reciprocity exists between climate variables and distribution and expression of plant communities (Woodward 1987; Woodward and Williams 1987). This relationship is part of a dynamic process that has undergone notable changes in recent decades due to global warming. Consequently, the need to update vegetation maps can be considered an opportunity to understand the mismatch that communities are experiencing. The construction of models that best explain this link represents one of the most significant contributions to understand the distribution of communities in accordance with phytosociology and landscape ecology- This process facilitates a better diagnosis and potential delimitation of ecosystems and plant communities. Moreover, vegetation mapping is also helpful for analysing spatial and temporal dynamics, contributing to more refined reconstruction of vegetation series and potential vegetation (Rivas-Martínez 1983; Pedrotti 2013; Álvarez-Santacoloma et al. 2022).
Rapid human-induced climate change is disturbing species populations and ecosystems (Lenoir and Svenning 2015). Species range shifts are among the main expected effects (Walther et al. 2010), posing a significant challenge in mountainous and near-peak regions, where suitable habitats are diminishing, and species migration is impeded (Esperon-Rodriguez et al. 2019). In these regions, the replacement of alpine ecosystems by coniferous forest (upward shift of the tree line) is expected if suitable soil conditions and effective propagule arrival occur (Harsch et al. 2009). Climate change is also causing variations in vegetation patterns, leading to transitions from cool, wet forests to more temperate or dry mixed formations. Additionally, lowland and desert shrublands may shift upward in elevation. Climate change can result in community realignments and compositional shifts, often favouring the development of new, warmer, and drier vegetation (Holsinger et al. 2019). In this context, many authors suggest the inability of the current protected area network to cope with anticipated responses and paths, based on factors such as high climate-analog distance and sensitivity to human land use, among others (e.g., Parks et al. 2023).
When mapping vegetation or bioclimatic units, it is imperative to carefully select an adequate spatial scale and climatic/bioclimatic classification method (Canu et al. 2015). This selection is particularly relevant in small and highly heterogeneous territories that harbour high biodiversity, such as oceanic islands. These areas may require more detailed consideration than mainland regions because representing their vegetation units involves accounting for an abundance of locally varying factors. Therefore, island size and scale (resolution and extent) are fundamental features to be considered (Otto et al. 2020), along with factors such as aspect and different exposure to prevailing winds, topographic complexity, non-uniformity in the spatialization of climate phenomena (anisotropy), elevation as an environmental gradient driver, etc. (Irl et al. 2015). Therefore, using coarse resolutions could not be adequate to summarise the full complexity of the target area working on islands (Vondrakova et al. 2013), and it is necessary to approach this question on an island scale to identify and quantify spatial reconfiguration of the vegetation units along the island elevational gradient. In order to avoid those issues, the Worldwide Bioclimatic Classification System (WBCS) promoted by Rivas-Martínez, and co-authors (2011) can be applied at different scales and macrobioclimates, being considered a valid and widely used method in Botany and Geobotany (Pesaresi et al. 2017; Minh et al. 2023). It constitutes a continuous and hierarchical numerical framework capable of accommodating the most relevant climatic variables when defining plant communities in the Mediterranean climate (Torregrosa et al. 2013). Some authors emphasize the complementary use of clustering techniques to identify homogeneous spatial or temporal patterns or the combination of both (Gavilan et al. 1998; Cutini et al. 2021).
Interpolation techniques and vegetation classification methods have also been revised or changed in recent decades. In these types of studies, the use of spatial interpolations and phytosociological supervision to obtain continuous rasters of thermotypes (spaces of the territory characterized by thermal values) and ombrotypes (territorial areas characterized by rainfall values nuanced with temperature) is emphasized, which facilitates their integration into a GIS environment. This fact implies a required update according to the latest WBCS mentioned earlier (Rivas-Martínez et al. 2011), representing a significant improvement compared to previous bioclimatic studies based on Rivas-Martínez (1997), where arithmetic and gradient-based methods were used to delimit thermotypes and ombrotypes. These new approaches consider the upper and lower horizons of each thermotype and ombrotype to get a more detailed and updated approximation of the correspondence between bioclimatic combinations and their translation into climatophilous vegetation series. Moreover, regression interpolation techniques have shown good accuracy on islands, even in low-density networks of stations (Garzón-Machado et al. 2013). These methods provide territorial adscription based on quantitative methods as additional information to the phytosociological characterization of vegetation and are considered replicable and scalable at both insular and continental scales. In the Macaronesian context, there are different studies related to vegetation mapping and plant communities classification, either based on bioclimatology and interpolations (Mesquita et al. 2004) or on supervised remote sensing techniques (Masseti et al. 2016) for Madeira Island. In the case of the Azores archipelago, studies are mainly based on the effects of climate change on a subset of native species using a species distribution modeling approach (Ferreira et al. 2016) or confronting invasive species with native species (Silva et al. 2019).
The Canary Islands are a good testing bench for the application of bioclimatic criteria and spatial characterization of climatophilous vegetation series. These islands harbour high diversity featuring five main vegetation belts from coastal to high mountain ecosystems (Del Arco et al. 2006 a). Moreover, steep inter-island and intra-island climatic gradients exist. In this context, Tenerife, the largest and most diverse island of the archipelago, is the most complete choice to test these changes in the communities. In this island, global warming is already producing a significant rise in temperatures (0.09 ± 0.04 ºC/decade), particularly at the summit (0.14 ± 0.07 ºC/decade) (Martín-Esquivel et al. 2012), accompanied by a differential distribution of decreasing rainfall across the island. This process would lead to readjustments in the community’s composition and species distribution (Ibarrola et al. 2019; Martín-Esquivel et al. 2020), resulting in predictions of elevational changes in vegetation (Del Arco 2008; Bello-Rodríguez et al. 2021). Over the last two decades, climatic conditions have changed significantly due to global warming, leading us to anticipate substantial changes, even over this short period time.
Considering all this, the general goal of this study is to synthesize the bioclimatic and vegetational complexity of Tenerife Island, updating climate data and methods to know the bioclimatic factors changing the potential distribution of climatophilous vegetation belts. Specifically, we want to (a) analyse how the effects of climate change have modified in only two decades the distribution and spatial coverage of vegetation belts in an oceanic island with a broad elevational gradient (b) how different approaches to addressing the spatial mismatch between present and past vegetation maps can explain the accounted changes. We hypothesize that global warming is causing non-homogeneous shifts in the circumscription of the bioclimatic belts and their corresponding translation into climatophilous vegetation series previously defined by Del Arco et al. (2006b). This contribution aims to develop a methodological approach applicable to other complex environments and their potential vegetation mapping and diagnosis. Since the ongoing climate change accelerates the need for ecosystems active management (Dollinger et al. 2023), our methodology could serve as a base for current landscape restoration actions, establishing a framework for other future spatial vegetation models that can help identifying potential areas for different types of vegetation.
Methods
Study area
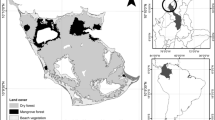
Tenerife is situated geographically at 28°35’15’’- 27°59’59’’ N and 16°5’27’’ − 16°55’4’’ W, within the Canary Islands archipelago (Fig. 1). As the largest (2035 km2) and highest island of the region, with an elevation reaching 3715 m a.s.l at Mt. Teide. We selected Tenerife because of its climatic heterogeneity, often likened to as a mini-continent (Irl et al. 2015; Dorta et al. 2021). Also, Tenerife harbours a representation of all the vegetation belts defined to the Canary Islands, which is a distinct orographic-climatic relationship in this is island featuring a clear-to-see temperature elevational gradient, a higher humidity, and rainfall in the northern slopes, especially on those areas that directly receive the fog water from the trade winds and a high radiation in areas above the treeline. (Fernández-Palacios and de Nicolás 1995; Sperling et al. 2004).
Tenerife possesses a high degree of Canary Island endemicity in its vascular flora elements (about 26%). According to Del Arco and Rodríguez (2018), in this island, we can distinguish the following elevational vegetation belts:
-
Euphorbia scrubs, xerophytic vegetation framed within the African Rand flora, currently distributed in the infra- and thermo-Mediterranean thermotypes and in the hyperarid, arid, semi-arid and lower-dry ombrotypes.
-
Thermo-sclerophyllous woodland, related to the Mediterranean flora, are bioclimatically established in the upper-inframediterranean, thermo-Mediterranean. They cover an ombrotypical range from lower-semiarid to dry, especially in areas lacking trade winds clouds.
-
Laurel forests, remnants of the Tertiary-Tethyan stage of the Pliocene, are currently confined to areas affected by the trade winds clouds. They settle in regions with varying ombrotypes, ranging from dry to subhumid, and thermotypes spanning from infra- to mesomediterranean.
-
Canary pine forest, primarily affiliated with the Afro-European-Mediterranean and Paleo-Mediterranean of the Tertiary-Tethyan. Their enormous plasticity and resilience allow them to occupy a broad spectrum of bioclimatic combinations, from the thermomediterranean to the upper-mesomediterranean thermotypes, the latter, a bioclimatic space that can coexist with the summit broom and tolerate ombrotypes ranging from semi-arid to upper-subhumid.
-
Summit vegetation, dominated by species related to Mediterranean and North African-Mediterranean flora. It develops on the high mountain and summit bioclimatic belts, represented by the upper-mesomediterranean, supramediterranean, oromediterranean and cryoromediterranean thermotypes, and the arid, semi-arid, and dry ombrotypes.
The potential climatophilous plant communities of these belts show a different degree of conservation: the sweet spurge scrub occupies 10,548 ha (32.6% of its possible area); the cardón scrub 8,372 ha (34.2%); the scarce remnants of thermo-sclerophyllous woodland occupy 437 ha (1.47%); the reminiscences of dry laurel forest 671 ha (5.3%); the humid laurel forest 860 ha (5.1%), and cold laurel forest 85 ha (4.6%); the Canary pine forest and its variants considered as climatophilous maintain 22.606 ha (68.91%); and the summit vegetation 13,679 ha (92.6%) (Del Arco et al. 2010).
Methods
Climatic data
Data from 121 meteorological stations (68 pluviometric, 14 thermometric, and 39 thermo-pluviometric) operated by the Spanish Meteorological Agency (AEMET) were used for this study (Fig. 1). The temporal resolution of the climate data covers a period of 30 years (1990–2019). On the other hand, our reference climate period is 1960–1999 (which covers data from Del Arco et al. 2006 b), which is the baseline of the potential vegetation map of Tenerife generated prior to this work (Del Arco et al. 2006 a).
Firstly, we verified the exact location of each meteorological station and performed an exploratory analysis of the data. Climatic, bioclimatic, and topographic variabilities are shown in Table 1.
Worldwide bioclimatic classification
According to the WBCS of Rivas-Martínez et al. (2011), the Canary Islands fit within the Mediterranean macro-bioclimate, which is primarily characterized by having at least two consecutive months after the summer solstice in which P < 2T (this lack of rainfall can persist throughout the year in the desertic and hyperdesertic Mediterranean bioclimates). This classification allows defining bioclimatic belts from the unique and exclusive combination of thermotypes and ombrotypes without ignoring the importance of trade-wind cloud effects in the context of the westernmost islands (Del Arco and Rodríguez-Delgado 2018). The same combination of thermotype and ombrotype under the influence or absence of clouds generates bioclimatic territories of different climatophilous vegetation series (Del Arco and Rodríguez 2018).
Thermotypes encompass ranges based on the compensated thermicity index (hereafter Itc) or positive temperature (hereafter Tp) gradient, while ombrotypes involve ranges of the ombrothermal index (hereafter Io) (Table 2). Factors related to thermotypic and ambrotypes, in conjunction with the presence or absence of clouds, establish the bioclimatic belts along an altitudinal gradient for the Canary Islands (Del Arco et al. 2006b; Garzón et al. 2013). Each bioclimatic combination is associated with specific climatophilous plant communities that succeed each other in a staggered manner along an elevational cliseries (Rivas-Martínez et al. 2011) and constitute an expression of the vegetation zonation phenomenon (Rivas-Martínez et al. 2017). This successional process is framed within a discipline known as symphytosociology, whose fundamental unit of study is the vegetation series or sigmeta (Loidi 2021). For the case study, we will address the finalstage of succession or the cuspidal climax community within each vegetation series (Loidi 2017). A few bioclimatic indices are used to establish thermotypes and ombrotypes. The Itc is defined as follows:
\(Itc = It \pm\) C
Given that It (thermicity index) = (T + M + m) ×10, where T is the mean annual temperature, M the mean maximum temperature of the coldest month of the year, and m is the mean minimum temperature of the coldest month of the year. C is the compensation value used when the continentality index (Ic = difference between the mean temperatures of the warmest and coldest month) is lower than 8 (oceanicity) or higher than 18 (continentality). In the Canary Islands, this compensation value is always used subtractively and is obtained as follows: C = (8.0-Ic) ×10.
The Io is used to establish ombrotypes and is defined as an annual mean value of a quotient between precipitation values and temperature, attempting to consider the weight of temperature in the availability of water to plants:
Positive precipitation (hereafter Pp) is the annual rainfall in mm, only considering months whose average temperature is higher than 0 ºC. Tp (positive temperature) is the value in tenths of degree resulting from the sum of the average temperature of all the months with an average temperature higher than 0 ºC. This index is well adjusted to the elevational limits of the vegetation (Del Arco et al. 2006b) and considers the overall water availability of the plant, indirectly assessing evapotranspiration by comparing precipitation and temperature (Mesquita et al. 2004).
In addition to the threshold values for thermotypes and ombrotypes according to WBCS (Rivas-Martínez et al. 2011) (see supplementary material), to obtain a better adjustment for some climatophilous series, the upper horizon of the mesomediterranean thermotype has been further divided into two intervals, values greater than 230 and values between 230 and 220. Similarly, the upper horizon of the arid ombrotype was adjusted to values between 0.7 and 0.9 and 0.9-1.
Topographic and vegetation dataset
Elevation was obtained from a digital elevation model (DEM) at 25 m of resolution (GRAFCAN 2021). A trade-wind effect raster and current patches vegetation were generated from the Vegetation Map of the Canary Islands (Del Arco et al. 2006a) based on the most indicative degradation communities of the climatophilous vegetation series of the cloud forest and georeferenced remnants of potential vegetation, respectively.
For our analysis purposes, we selected only the cuspidal communities of each climatophilous series. Each series was rasterized to the identical resolution of bioclimatic and vegetation layers.
Statistical and geostatistical analysis
To generate raster maps depicting thermotype and ombrotype, we conducted spatial interpolation by leveraging a specific set of bioclimatic parameters (Itc, Tp, and Io). Employing regression kriging (RK) for Itc and Tp. This interpolation method involves a regression analysis of the dependent variable against auxiliary factors (such as elevation, latitude, and longitude in our case) and ordinary kriging (OK) of the regression residuals (Hengl et al. 2007). Further details can be found in Ninyerola et al. (2007), Garzón et al. (2013), and Canu et al. (2015). To train and validate the regression models, we used caret package (Kuhn 2022). Variogram fitting and geostatistic interpolation of the residuals using OK were performed with the package gstat (Gräler et al. 2016) and regression metrics were obtained using Metrica package (Correndo et al. 2022).
For Io interpolation, we opted for thin plate splines (TPS) utilizing elevation data (derived from a DEM) as a covariate along with the default smooth parameter (lambda), internally determined through generalized cross-validation (Li 2022). The ‘Tps’ function from the fields R Package (Nychka et al. 2021) facilitated this process, applied to both our 1990–2019 (30 years) climatic dataset and the reference period (1960–1999) included in Del Arco et al. 2006 b. Detailed interpolation procedure, cross-validation metrics and interpolated surfaces can be found in supplementary material I. This process involves the use of the function ‘tpscv’ included in spm2 R Package (Li 2023).
New bioclimatic indices surfaces were reclassified according to lower and upper thresholds using map algebra language implemented in the raster calculator tool (Pesaresi et al. 2017). Thermotypes, ombrotypes, and trade-wind cloud rasters were crossed using a raster calculator and raster attribute table tool (RAT) and then assigned to each combination its corresponding vegetation series under phytosociological criteria. The use of spatial interpolations and the support of the expert phytosociologist are fundamental for the correct ascription and interpretation of bioclimatic combinations and their corresponding climatophilous vegetation series (Capelo et al. 2007), being a necessary task if detailed classification is required. Considering a combination of two bioclimatic parameters related to temperature (Tp and Itc) was fundamental in the more detailed obtention of thermotypes, especially in those located at the summit of the island.
To explain the bioclimatic complexity of Tenerife and the vegetation belts, we applied non-metric multidimensional scaling (NMDS) based on bioclimatic variables (Io, Itc, Tp, Pp, Cloud effect) and elevation with the Bray-Curtis distance and number of axes fixed to two. We assessed the goodness-of-fit of this analysis with the stress value. Finally, we carried out a permutational multivariate analysis of variance (PERMANOVA) to test the significance of NMDS with 999 permutations. We used the corrplot package (Wei and Sinko 2021) to show Pearson’s pairwise correlations between the obtained dimensions and bioclimatic variables in a correlation matrix.
We employed three different functions from the vegan package (Oksanen et al. 2022): ‘metaMDS’ and ‘vegdist’ functions to perform NMDS ordination and generate distance-matrix, and the ‘adonis2’ function to carry out permutational multivariate analysis of variance. All statistical analyses were performed using R software version 4.2.2 (R Development Core Team 2022).
Assessment of changes in the vegetation belts
Kappa index
To detect spatial changes in the vegetation belts, we compared our vegetation series map with that obtained from Del Arco et al. 2006 b. For this purpose, we selected Cohen’s Kappa (1960), a metric deeper than simple accuracy (correct classification ratio) widely used when analyzing spatial classifications or comparing between categorical rasters. (Zhao et al. 2011).
P0 is the proportion of overlapping pixels (or observed similarity), while Pe is the expected agreement by chance, which can reduce the noise of random effects (Delmeyer et al. 2018). A Kappa value ranges from − 1 (absolute disagreement) to 1 (absoluteagreement). We interpreted values of 0.8 or above as indicating the highest similarity between maps, 0.6–0.8 as very high, 0.4–0.6 as high, 0.2–0.4 as medium, and < 0.2 as low (with 0 indicating no better similarity than expected by chance). For this purpose, we use the r.kappa algorithm implemented in the GRASS GIS Module (2023). We used Kappa as an indicator of agreement and spatial change.
Vegetation belts distribution changes
The maintenance, gain, or loss of bioclimatic suitability was calculated for each vegetation belt under the past and present scenarios considered. The percentage of predicted future range change (which refers to the change from past to present, hereafter C) by cell was also estimated following Del Rio et al. (2021) as follows: C = 100* (RG-RL)/PR, where RG (range gain) is the number of grid cells projected as being unsuitable under past conditions (1960–1999) but suitable in current conditions; RL (range loss) is the number of grid cells projected as suitable under past conditions but unsuitable in current conditions; and PR (past range) is the number of grid cells projected as appropriate under past reference conditions. A positive C value means an increase in overall range size, and a negative value indicates a loss. We used that metric as a range expansion or contraction of each vegetation belt.
Vulnerability assessment
The vulnerability of the vegetation belts of Tenerife under observed climate change was analyzed applying the Mismatch Index (González-Mancebo et al. 2023) (hereafter MI), also proposed as the Vulnerability Index in Felicísimo et al. (2012). This index in defined as:
VI = 1-((CPA ∩ COA) *(CPA ∩ EPA)) where: CPA ∩ COA is the intersection of the present potential area (in surface units) and the current occupied area (patches) (extracted from Del Arco et al. 2006 a) in the whole study area, and EPA ∩ CPA is the intersection of the present potential area (in surface units) and the earlier potential area (past).
The MI moves from 0 to 1, with 0 and 1 indicating complete and no overlap, respectively, between ther bioclimatic surface at present and both potential area and occupied patches at past reference time. We use this indicator as a metric of vulnerability and persistence.
Maps and spatial analysis were produced in QGIS 3.26.1 Buenos Aires (2022). The system reference is UTM (WGS84) zone 28 N (if not explicit). Area sizes and coverages were calculated in hectares (Ha).
Results
Thermotypes and ombrotypes
The spatial interpolations and the subsequent reclassification of the continuous rasters have allowed the identification of 6 thermotypes (inframediterranean, thermomediterranean, mesomediterranean, supramediterranean, oromediterranean and cryoromediterranean) and 5 ombrotypes (hyperarid, arid, semiarid, dry, subhumid) on the island, all with their respective lower and upper horizons except the cryoromediterranean. These are shown in Figs. 2 and 3, respectively. Equations of the regression models are reported in Table 2. The cross-validation metrics of the interpolations carried out to generate these rasters returned satisfactory results in the indicators evaluated (more details in supplementary material I). The most considerable thermotypes in terms of coverage are the lower and upper inframediterranean (Fig. 2), with their area decreasing according to the elevational gradient and the pyramidal topographic configuration of the island. Concerning the ombrotypes, the semiarid has the highest area, generally covering the midlands of the south of the island. The dry and subhumid ombrotypes, more prominently characterized by their lower horizons, extend along the middle zones of the northern slope of the island, coinciding with the areas with the highest annual rainfall. On the contrary, those ombrotypes linked to greater aridity mainly appear on the southern slope, where water stress persists throughout the year.
Bioclimatic belts
Because of the combination of thermotypes, ombrotypes, and the influence of the trade-winds clouds, a total of 90 bioclimatic combinations have been obtained (see supplementary material II).
Climatophilous vegetation map
A map of climatophilous vegetation series has been produced from the interpolated rasters and phytosociological interpretation (Fig. 4), considering the most recent global bioclimatic classification of Rivas-Martínez et al. (2011) and the high correlation between bioclimatic combinations and climatophilous vegetation series. These series have been extensively described and characterized in the phytosociological literature (e.g., Del Arco et al. 2006a; Del Arco and Rodríguez 2018). Sweet spurge scrub, cardón scrub, and the pine forest sensu lato are the series that occupy the most extensive areas. The thermo-sclerophyllous woodland traces a ring under the pine forest in southern areas and below the laurel forest (in general) on the northern slope of the island, the latter under the influence of the frequent trade winds clouds. Within the laurel forest, the dry laurel forest has the highest coverage, followed by the humid variety. The cold or summer-dry type is restricted to those areas free of clouds during summer and represents the shorter vegetation zone in coverage, followed by the summit vegetation.
The two axes of the NMDS (Fig. 5) comprised enough variation in the main bioclimatic gradients of Tenerife (stress = 0.04). In the first axis of the ordination, there is a tendency to group the pixels based on the distribution of vegetation along the island’s elevational gradient. This grouping contrasts the bioclimatic parameters associated with thermicity (Tp and Itc) and ombrothermic gradient versus elevation. This fact is consistent with the correlations obtained between the first dimension and elevation (r > 0.8, p < 0.001), Tp, and Itc (r < − 0.8, p < 0.001) (Fig. 6). The second underlying trend is related to humidity. Annual precipitation and the fog effect contribute to discerning the pixels affected by the presence of clouds and characterizing the laurel forest, the humid faciation of the thermos-sclerophyllous forest, and the humid pine forest. Mist effect, Pp, and Io are the variables best correlated with this second dimension. The PERMANOVA based on Bray-Curtis dissimilarities identified that at least two of the climatophilous vegetation belts obtained differ significantly in terms of their characteristics along the bioclimatic gradients of the island, which is consistent with the ordination pattern yielded by the NMDS.
Non-metric multidimensional scaling (NMDS) analysis of bioclimatic and elevational variables. The bioclimatic characteristics of vegetation differ significantly between NMDS dimensions obtained (Permutational multivariate analysis of variance: (PERMANOVA): R2 = 0.78, p < 0.0001 in the case of NMDS1; R2 = 0.04, p < 0.0001 in the case of NMDS2)
Considering the whole island, we obtained a global Kappa value of 0.80 and 82.6% of pixels correctly classified, although with significant differences between series (Table 3). Kappa values offer the best consistent results (> 0.8) for sweet spurge scrub, cold laurel forest, dry laurel forest, and Canary pine forest. However, fair results were obtained for other ecosystems, reaching the worst correspondence in the thermo-sclerophyllous woodland with its disposition at the beginning of the 21st century (Fig. 7a). According to C, dry laurel forest, sweet spurge scrub, cardón scrub and Canary pine forest tent to increase their bioclimatic surfaces and this translates into spatial expansion at the expense of adjacent vegetation (Fig. 7a, b, c). On the contrary, the humid laurel forest, the summit vegetation, and the thermo-sclerophyllous woodland suffered a loss of area and a subsequent range contraction (-28.4 and − 19.8% respectively) (Fig. 7c), especially noteworthy in the humid laurel forest (-31.1%) (Table 3). These results have strong correspondence with the values of the Mismatch Index for each vegetation belt (Fig. 8). In this index, the component linked to the persistence of current patches showed lower values than the persistence of potential area (more details in supplementary material III). The lowest values of MI correspond to the vegetation belts that expand or maintain their bioclimatic suitable area and current distribution; conversely, the highest values are associated with the vegetation belts that reduce their potential area.
Changes in potential vegetation distribution in Tenerife. (a) Maps of vegetation under past and present conditions. (b) Proportion of vegetation series in each scenario considered (c) Projected transitions among vegetation series from past to present climatic conditions, where the sector size in the outer circle indicates the distribution of change over vegetation series and the thickness of connecting flows depicts the number of transitions from one vegetation series to another (series without changes were excluded)
Spatial change assessment between past and present scenarios. (a) Loss, gain, or maintenance (not change) of suitable bioclimatic area for thermo-sclerophyllous woodland under past and current climatic conditions. (b) Relationships between the Kappa index, percentage of predicted future range change (c), and mismatch index (MI)
Discussion
The bioclimatic analyses performed reveal a significant spatial variation in the vegetation belts in the context of climate change, representing only a 20-year interval since the last study. The high orographic complexity of Tenerife Island appears to contribute to the varied response of ecosystems to the new drier and warmer conditions, as hypothesized. While some communities tend to increase their surface strongly (sweet spurge scrub and dry laurel forest), others diminish it, such as the thermo-sclerophyllous woodland, the humid laurel forest, and the summit vegetation. The pine forest and the cardón scrub experience a slight increase in terms of surface. According to spatial change metrics, the lowest C values (indicated by negative values) occur in climatophilous communities with less capacity for spatial expansion. This limitation is mainly attributed to the constriction caused by the ascend of the lower vegetation belts and the lack of potential areas to migrate, further exacerbated by the low persistence of the current patches. Contrary, the vegetation belts that exhibit the highest level of gained area are those less constrained or limited by cloud effect and present less thermic and hydric limitations. This shift towards more xeric communities is reported in vegetation dynamics, ultimately resulting in new drier and warmer environments (Batllori et al. 2020).
The increase in the sweet spurge scrub is explained by the absence of losses at lower altitudes and a strong upward shift. Occurring despite it being precisely in coastal areas where communities’ constrictions are more commonly reported for vascular plants (Ferreira et al. 2016) due to the temperature increase and lower rainfall patterns. The advance in elevation is especially relevant in the southern slopes of the island, which is consistent with the strong position of the sweet spurge scrub in the elevational-thermic gradient of the NMDS. Therefore, more thermic and drier scenarios could conduct more profound upward shifts in this lowland communities (Kelly and Goulden 2008), at least for those species with sufficient adaptive capacity to respond to global warming. The increase in the dry laurel forest is narrowly related to the island orography since this type of forest is expanding within the current area of humid laurel forest located at a higher elevation; meanwhile, the lower limits of the past are still suitable zones. This gain is limited by the effect of trade-wind clouds since laurel forest communities have been restricted to these areas in our framework, as they are entirely dependent on their intensity and frequency (Marzol et al. 2010). In contrast, thermo-sclerophyllous woodland is limited at both boundaries. In its lower extreme, there is a contraction caused by the advance of the cardón scrub on both slopes of the island, which could be facilitated by the current strong disturbance that presents these formations (Otto et al. 2012). The gain opportunities of this community are in the inframediterranean and windward situations under the influence of the trade winds (Fig. 7c), leading to the formation of mixed communities with elements of dry laurel forest (Fig. 7c). This faciation is characterized by humid juniper forests and other arboreal elements, enriched with characteristic species of dry laurel forest such as Visnea mocanera, Ilex canariensis, Apollonias barbujana or Morella faya, forming more heterogeneous communities (Rodríguez-Delgado et al. 1990; Fernández-Palacios et al. 2008; Otto et al. 2012). Even then, the gained area does not neutralize the large amount of lost area according to our results (Fig. 7c). Likewise, the high value of the mistmatch index and the few remnants currently persisting in Tenerife aggravate the situation of these communities. On the other hand, on the leeward slope, the thermo-sclerophyllous woodland is being transgressed by Pinus canariensis, with some elements of the understory of the thermo-sclerophyllous forest present in the lower levels of the pine forest (Otto et al. 2012).
Humid laurel forest has a very narrow range in terms of temperature, humidity and precipitation (Fig. 9). As with many other cloud-dependent forests, they are highly vulnerable to changes in the cloud formation regime and to spatial and temporal changes in rainfall patterns (Sperling et al. 2004; Van Beusekom et al. 2017). In the Canary Islands, there is uncertainty about these changes due to the difficulty of predicting shifts in frequency, height, and strength of stratocumulus clouds (Carrillo et al. 2023). Therefore, there is still considerable uncertainty about the real migration possibilities and future climatic niche availability for laurel forest species beyond their adaptive capacity to global warming. According to our data, the most significant imbalances in humid laurel forest occur in Anaga and Teno, which represent islands within islands in the northeastern and northwestern sectors of the island. The observed phenomenon can be attributed to the increase in the lower thermomediterranean and inframediterranean thermotypes (see supplementary material 1), which facilitates the advance of dry laurel forest. This fact is especially relevant since both areas contains the best-preserved remnants of this relict formation (Santos-Guerra 1990; Ohsawa et al. 1999). The central-northern sector of the island seems to be the area in which the humid laurel forest has more opportunities to persist. Nevertheless, currently, this represents highly impoverished areas due to past uses. Regarding the cold laurel forest, the coincidence between maps is almost complete (Fig. 7a), which agrees with its appearance only in areas without clouds during the summer. Given the Mediterranean character of the Canary Islands’ macroclimate, the contribution of fog precipitation during the summer drought is essential in maintaining the island’s biodiversity in windward slopes (Marzol 2008).
Characteristically, high mountain bioclimatic belts are undergoing a constraint process due to global warming (Bonannella et al. 2023), a phenomenon also observed in the summit vegetation of Tenerife (Martín-Esquivel et al. 2021). Cubas et al. 2022 reported a drastic reduction of the broom area in the upper-mesomediterranean belt concerning its past distribution. This observation is consistent with the expected decrease in keystone species, a result that becoming more prevalent because of climate change (Holsinger et al. 2019). The reduction of the highest vegetation belt on the island is combined with an advance of the surrounding pine forest (Bello-Rodríguez et al. 2019), with a subsequent change in the communities (Martín-Esquivel et al. 2020, 2021). This pattern is ordinary in many sub-alpine forests and depends on soil availability, seed dispersal, production, and herbivory pressure (Annadol-Rossell et al. 2019; Bello-Rodríguez et al. 2019). Some authors underline the potential existence in the past of a widely open formation of Canary cedar (Juniperus cedrus) just in the timberline area of the pine forest Sideritido solutae-Pinetum canariensis subass. cistetosum osbaeckiaefolii (= Junipero cedri-Pinetum canariensis) (Rivas-Martínez et al. 1993; Garzón-Machado et al. 2011; Rumeu and Nogales 2021). Besides, Adenocarpo viscosi-Juniperetum cedri was described as a possibly past open woodland of Juniperus cedrus on the summits of Tenerife (Martín-Osorio et al. 2007). However, we don’t know the possibilities of the recent effort for the recovery of this type of non-evaluated vegetation. We know the existence of testimonial specimens in inaccessible places in Tenerife or La Palma (Del Arco et al. 1999; Sangüesa-Barreda et al. 2021), but there is only a fragmentary information about this theoretical formation.
Pine forest and cardón scrub are the only vegetation belts that have not experienced significant changes during the analyzed period. The Canary pine forest is the most bioclimatically heterogeneous vegetation series and appears extremely well clarified in both gradients of the NMDS (Fig. 9). Spatial change metrics indicate an increase in area and a low level of contraction which demonstrates that climate change wouldn’t affect this formation too much. However, forest fires and invasive herbivores prevent considering this community as a global warming play winner as its understory is currently highly impoverished (Garzón-Machado et al. 2012; Cubas et al. 2021). Its main gain occurs in the upper mesomediterranean belt to the detriment of the summit vegetation. However, the arrival of species from this ecosystem could not compensate for the loss of summit species, an area of greater diversity and richness (Bello-Rodríguez et al. 2023). At the lower limit, in the transition zone with the thermo-sclerophyllous woodland, the pine forest experiences only minimal losses (Fig. 7). This fact is especially relevant because this contact mainly occurs in the southern slopes, where the most natural stands persist (Del Arco et al. 1992). However, those losses are not entirely occurring, as only some patches of thermo-sclerophyllous woodland remain in these areas (Otto et al. 2012). In fact, the abundance of salic substrata on this part of the island already allows the introgression of the pine stands or sparce individuals to lower levels in the current area of thermo-sclerophyllous woodland. These introgressions also occur in areas of potential laurel forest in the northern slope, where there are salic outcrops, which highlights the importance of considering lithology in the modification of natural climatophilous vegetation on oceanic islands (Salas-Pascual et al. 1998; Del Arco et al. 2013). Thus, there are marked differences in the lower levels of the pine forest on the windward and leeward slopes of the island. Regarding the cardón scrub, this community increases its climatophilous surface gently, mainly due to an increase in the semiarid inframediterranean bioclimatic belt on both sides of the island. The advance of the sweet spurge scrub d is noticeable at its lower limit, causing significant interweaving and contraction. This increase is not homogeneous since the cardón scrub can carry out ascending and descending soil movements on lava flows and recent salic outcrops or ‘malpaíses’ (Rivas-Martínez et al. 1993; Del Arco et al. 2006b).
Our study underscores how rapid spatial changes in plant communities and bioclimatic belts can be detected due to global warming. This work highlights the necessity, especially in the prevailing climate change scenario, to periodically update vegetation maps using bioclimatic indices. This type of assessment facilitates decision-making on territorial management and land use planning (Küchler and Zonneveld 1988; Pedrotti 2013) and contributes to planning and even questioning the current limits that protected areas should have (Parks et al. 2023) by detecting the zones and vegetation types that are best persisting over time. In addition, these changes include considerations in the evaluating ecosystem services (Garzón-Machado et al. 2011; Mesquita et al. 2017). Landscape planning and management requires a vision of the past, present, and future, so any conservation effort must also consider what will happen in the future, as more profound changes in the distribution and composition of these plant formations are expected due to climate change (Keane et al. 2020). In addition, the various stochastic processes affecting oceanic islands, as well as the occurrence of invasive species and ongoing habitat degradation, must be taken into account for forecasting purposes, as they are considered major threats for island conservation.
Data availability
Bioclimate data is provided within the manuscript supplementary information files (thermic stations and ombrothermic stations).
References
Álvarez-Santacoloma A, Ferreiro-Lera GB, González-Pérez A, Penas A, Del Río S (2022) Bioclimatic characterizacion of Northwest Spain (Asturias, Galicia Y León). Int J Geobotany Res 11:63–80. https://doi.org/10.5616/ijgr
Anadon-Rossell A, Talavera M, Ninot JM, Carillo E, Batllori E (2019) Seed production and dispersal limit treeline advance in the Pyrenees. J Veg Sci 00:1–14. https://doi.org/10.1111/jvs.12849
Batllori E, Lloret F, Aakala T et al (2020) Forest and woodland replacement patterns following drought-related mortality. PNAS 117(47):29720–29729. https://doi.org/10.1073/pnas.2002314117
Bello-Rodríguez V, Cubas J, Del Arco MJ, Martín-Esquivel JL, González-Mancebo JM (2019) Elevational and structural shifts in the threeline of an oceanic island (Tenerife, Canary Island) in the context of global warming. Int J Appl Earth Obs Geoinf 82:101918. https://doi.org/10.1016/j.jag.2019.101918
Bello-Rodríguez V, Hamman A, Martín-Esquivel JL, Cubas J, Del Arco MJ, González-Mancebo JM (2023) Habitat loss and biotic velocity response to climate change for alpine species in Atlantic Oceanic Islands. Diversity 15(7):864. https://doi.org/10.3390/d15070864
Bonannella C, Hengl T, Parente L, de Bruin S (2023) Biomes of the world under climate change scenarios: increasing aridity and higher temperatures lead to significant shifts in natural vegetation. PeerJ 11:e15593. https://doi.org/10.7717/peerj.15593
Canu S, Rosati L, Fiori M, Motroni A, Rosella F, Farris E (2015) Bioclimate Map of Sardinia (Italy). J Maps 11(5):711–718. https://doi.org/10.1080/17445647.2014.988187
Capelo J, Mesquita S, Costa JC, Ribeiro S, Arsenio P, Neto C, Monteiro-Henriques T, Aguiar C, Honrado J, Espirito-Santo D, Lousa M (2007) A methodological approach to potential vegetation modeling using GIS techniques and phytosociological expert-knowledge: application to mainland Portugal. Phytocoenologia 37:399–415. 10.1 127/0340-269X/2007/0037–0399
Carrillo J, Hernández-Barrera S, Expósito FJ, Díaz JP, González A, Pérez JC (2023) The uneven impact of climate change on drought with elevation in the Canary Islands. Clim Atmospheric Sci 20239:6–31. https://doi.org/10.17632/94gmt9c7fj.1
Cohen J (1960) A coefficient of agreement for nominal scales. Educ Psychol Meas 20:37–46
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Correndo A, Moro Rosso L, Schwalbert R, Hernandez C, Bastos L, Nieto L, Holzworth D, Ciampitti I (2022) metrica: Prediction Performance Metrics. R package version 2.0.0
Cubas J, Kluge J, Bello-Rodríguez V, Del Arco MJ, Cooke B, González-Mancebo JM (2021) Can habitat type predict the abundance of European rabbits on oceanic islands? Community Ecol 22:93–105. https://doi.org/10.1007/s42974-021-00039-6
Cubas J, Martín-Esquivel JL, Marrero-Gómez M, Docoito JR, Rodríguez F, González-Mancebo JM (2022) Climate change causes rapid collapse of a keystone shrub from insular alpine ecosystems. J Nat Conserv 69:126263. https://doi.org/10.1016/j.jnc.2022.126263
Cutini M, Marzialetti F, Barbato G, Riana G, Theurillat JP (2021) Bioclimatic pattern in a Mediterranean mountain area: assesment from a classification approach on a regional scale. Int J Biometeorol 65:1085–1097. https://doi.org/10.1007/s00484-021-02089-x
Dallmeyer A, Clausen M, Brovkin V (2018) Harmonising plant functional types distributions for evaluating Earth system models. Clim Past 15:335–366. https://doi.org/10.5194/cp-15-335-2019
Del Arco MJ (2008) La flora Y La vegetación canaria ante El Cambio climático actual. Naturaleza Amenazada Por Los cambios en El Clima Actas III Semana Científica Telesforo Bravo. Instituto de Estudios Hispánicos de Canarias, Tenerife, pp 105–140
Del Arco MJ, Rodríguez-Delgado O (2018) Vegetation of the Canary Islands. Plant and Vegetation, 16. Editorial Springer. 437 pp. https://doi.org/10.1007/978-3-319-77255-4_6
Del Arco MJ, De Pérez PL, Rodríguez-Delgado O, Salas M, Wildpret W (1992) Atlas Cartográfico de los Pinares Canarios. vol. 2, Tenerife. Viceconsejería de Medio Ambiente. Gobierno de Canarias, Santa Cruz de Tenerife. 228 pp + 41 maps
Del Arco MJ, Acebes J, De Pérez PL, Marrero MC (1999) Bioclimatology and climatophilous vegetation of Hierro (part II) and La Palma (Canary Islands). Phytocoenologia 29:253–290
Del Arco MJ, Wildpret W, Pérez PL, Rodríguez-Delgado O, Acebes JR, García A, Martín-Osorio VE, Reyes-Betancort A, Salas-Pascual M, Díaz MA, Bermejo JA, González R, Cabrera MV, García S (2006a) Mapa De Vegetación De Canarias. GRAFCAN SL, p 552
Del Arco MJ, De Pérez PL, Acebes JR, González-Mancebo JM, Reyes-Betancort JA, Bermejo JA, De-Armas S, González R (2006b) Bioclimatology and climatophilous vegetation of Tenerife (Canary Islands). Ann Botanici Fennici 43:167–192
Del Arco MJ, González R, Garzón V, Pizarro B (2010) Actual and potencial natural vegetation on the Canary Islands and its conservation status. Biodivers Conserv 19:3089–3140. https://doi.org/10.1007/s10531-010-9881-2
Del Arco MJ, Rodríguez-Delgado O, Pérez-de Paz PL (2013) The salic pine forest of Tenerife: Morello fayae-pinetum canariensis ass. Nova. Int J Geobotanical Res 3:37–45. https://doi.org/10.5616/ijgr
Del Río S, Canas R, Cano E, Cano-Ortiz A, Musarella C, Pinto-Gomes C, Penas A (2021) Modelling the impacts of climate change on habitat suitability and vulnerability in deciduous forests in Spain. Ecol Ind 131:108202. https://doi.org/10.1016/j.ecolind.2021.108202
Dollinger C, Rammer W, Seidl R (2023) Climate change accelerates ecosystem restoration in the mountain forests of Central Europe. J Appl Ecol 0:1–11. https://doi.org/10.1111/1365-2644.14520
Dorta P, López A, Díaz J (2021) Los Rasgos Climáticos De Tenerife. https://doi.org/10.25145/c.27.Asociaci?n
Esperon-Rodríguez M, Beaumont LJ, Lenoir J, Baumgartner JB, McGowan J, Correa-Metrio A, Camac JS (2019) Climate change threatens the most diversity regions of Mexico. Biol Conserv 240:108215. https://doi.org/10.1016/j.biocon.2019.108215
Felicísimo AM, Muñoz J, Mateo R, Villalba C (2012) Vulnerabilidad De la flora Y La vegetación españolas ante El Cambio climático. Ecosistemas 21(3):1–6. https://doi.org/10.7818/ECOS.2012.21-3.01
Fernández-Palacios JM, de Nicolás JP (1995) Altitudinal pattern of vegetation variation on Tenerife. J Veg Sci 6:183–190
Fernández-Palacios JM, Otto R, Delgado JD, Naranjo A, González F, Morici C, Barone R (2008) Los bosques termófilos de canarias. Proyecto LIFE04/NAT/ES/000064. Cabildo Insular De Tenerife. Santa Cruz de Tenerife, p 192
Ferreira MT, Cardoso P, Borges PAV, Gabriel R, Brito de Azevedo E, Reis F, Araujo MB, Bento R (2016) Effects of climate change on the distribution of indigenous species in oceanic islands (Azores). Clim Change 138:603–615. https://doi.org/10.1007/s10584-016-1754-6
Garzón-Machado V, Del Arco MJ, Pérez de Paz PL (2011) A tool set for description and mapping vegetation on protected natural areas: an example from the Canary Islands. Biodivers Conserv 20:3605–3625. https://doi.org/10.1007/s10531-011-0153-6
Garzón-Machado V, Del Arco MJ, Váldes F, Pérez-de-Paz PL (2012) Fire as a threatening factor for endemic plants of the Canary Islands. Biodivers Conserv 21:2621–2632. https://doi.org/10.1007/s10531-012-0321-3
Garzón-Machado V, Otto R, Del Arco MJ (2013) Bioclimatic and vegetation mapping of topographically complex oceanic island applying different interpolation techniques. Int J Biometeorol 58:887–899. https://doi.org/10.1007/s00484-013-0670-y
Gavilán RG, Fernández-González F, Blasi C (1998) Climatic classification and ordination of the Spanish Sistema Central: relationships with potential vegetation. Plant Ecol 139:1–11
González-Mancebo JM, Bello-Rodríguez V, Cubas J, Parada-Díaz J, Bañares-Baudet A, Palomares A, Martín-Esquivel JL, Del Arco MJ (2023) Assessing global warming vulnerability of restricted and common plant species in alpine habitats of two Oceanic islands. Biodivers Conserv. https://doi.org/10.1007/s10531-023-02731-7
GRAFCAN (2021) MDT 25x25 metros de la isla de Tenerife. https://opendata.sitcan.es/dataset/modelo-digital-de-terreno-mdt-de-25x25-metros
Gräler B, Pebesma E, Heuvelink G (2016) Spatio-temporal interpolation using gstat. R J 8:204–218. https://doi.org/10.32614/RJ-2016-014
GRASS Development Team (2023) GRASS GIS 8.3.1dev Reference Manual
Harsch MA, Hulme PA, McGlone MS, Duncan RP (2009) Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol Lett 12:1040–1049. https://doi.org/10.1111/j.1461-0248.200901355.x
Hengl T, Heuvelink GBM, Rossiter DG (2007) About regression kriging: From equations to case studies. Computer & Geosciences 33: 1301–1315. 2007 https://doi.org/10.1016/j.cageo.2007.05.001
Holsinger LM, Parks SA, Parisien MA, Miller C, Batllori E, Moritz MA (2019) Climate change likely reshape vegetation in American’s largest protected areas. Conserv Sci Pract e50. 10.111/csp2.50
Ibarrola-Ulzurrun E, Marcello J, Gonzalo-Martín C, Martín JL (2019) Temporal dynamic analysis of a mountain ecosystem based on multi-source and multi-scale remote sensing data. Ecosphere 10(6). https://doi.org/10.1002/ecs2.2708
Irl SDH, Harter DEV, Steinbauer MJ, Gallego D, Fernández-Palacios JM, Jentsch A, Beierkuhnlein C (2015) Climate vs topography – spatial patterns of plant species diversity and endemism on a high-elevation island. J Ecol. https://doi.org/10.1111/1365-2745.12463
Keane RA, Holsinger LM, Loechman R (2020) Bioclimatic modelling of potential vegetation types as an alternative to species distribution models for projecting plant species shifts under changing climates. For Ecol Manag 447:118498. https://doi.org/10.1016/j.foreco.2020.118498
Kelly AE, Goulden ML (2008) Rapid shifts in plant distribution with recent climate change. PNAS 105(33):11823–11826. https://doi.org/10.1073/pnas.0802891105
Küchler AW, Zonneveld IS (1988) Vegetation mapping. Handbook of Vegetation Science, vol 10. p. Kluwer Academic, Dordrecht, p 632
Kuhn M (2022) caret: Classification and Regression Training. R package version 6.0–92
Lenoir J, Svenning JC (2015) Climated-related range shifts – a global multidimensional sysnthesis and new research directions. Ecography 38:15–28. https://doi.org/10.1111/ecog.00967
Li J (2022) Spatial Predictive Modeling with R. Taylor & Francis. pp. 333. https://doi.org/10.1201/9781003091776
Li J (2023) spm2: Spatial Predictive Modelling. R Package Version 1.1.3
Loidi J (2017) Dynamism in Vegetation. Vegetation Change on a Short Time Scale. In: Loidi J, editor. The Vegetation of the Iberian Peninsula. Springer International Publishing, 1 (2). pp. 83–99. https://doi.org/10.1007/978-3-319-54784-8
Loidi J (2021) Dynamic-Catenal Vegetation Mapping as a Tool for Ecological Restoration and Conservation Policy. In: Pedrotti F, Owen E (editors). Tools for Landscape-Scale Geobotany and Conservation. pp. 37–61
Martín-Esquivel JL, Bethencourt J, Cuevas-Agulló E (2012) Assessment of global warming on the island of Tenerife, Canary Islands (Spain). Trends in minimum, maximum and mean temperatures since 1944. Clim Change 114(2):343–355. https://doi.org/10.1007/s10584-012-0407-7
Martín-Esquivel JL, Marrero MV, Cubas J, González-Mancebo JM, Olano JM, Del Arco MJ (2020) Climate warming and introduced herbivores disrupt alpine plant community of an oceanic island (Tenerife, Canary Islands). Plant Ecol 221:1117–1131. https://doi.org/10.1007/s11258-020-01066-5
Martín-Esquivel JL, Marrero-Gómez MV, González Mancebo JM (2021) Efectos Del cambio climático en la vegetación de la alta montaña de Tenerife. Ecosistemas 30(1):2189–2189. https://doi.org/10.7818/ECOS.2189
Martín-Osorio VE, Wildpret W, del Arco M, Pérez de Paz PL, Hernández Bolaños B, Rodríguez O, Acebes JR, García Gallo A (2007) Estudio Bioclimático Y Fitocenótico comparativo de la alta cumbre canaria: Tenerife-La Palma. Islas Canarias Phytocoenologia 37:663–697. https://doi.org/10.1127/0340-269X/2007/0037-0663
Marzol MV (2008) Temporal characteristics and fog water collection during summer in Tenerife (Canary Islands, Spain). Atmos Res 87:352–361. https://doi.org/10.1016/j.atmosres.2007.11.019
Marzol MV, Sanchez-Mejía J, García G (2010) Effects of fog on climatic conditions at a sub-tropical montane cloud forest site in northern Tenerife (Canary Islands, Spain). Science for conservation and management: 359–364
Massetti A, Menezes-Sequeira M, Pupo A, Figueiredo A, Guiomar N, Gil A (2016) Assessing the effectiveness of RapidEye multispectral imagery for vegetation mapping in Madeira Island (Portugal). Eur J Remote Sens 49:643–672. https://doi.org/10.5721/EuJRS20164934
Mesquita S, Capelo J, Gama I, Marta-Pedroso C, Reis M, Domingos T (2017) Using Geobotanical Tools to Operationalize Mapping and Assesment of Ecosystem Services (MAES) in Southern Portugal in Pedrotii F, Owen E (editors). Tools for Landscape-Scale Geobotany and Conservation. pp. 127–159. https://doi.org/10.1007/978-3-030-74950-7
Mihn T, Cao H, Khanh V, Hoang H, Thi N, Thi G, Thi H, Anh T (2023) Application of the Worldwide Bioclimatic classification system to determine bioclimatic features and potential natural vegetation distribution in Van Chan district, Vietnam. Trop Ecol 64:765–780. https://doi.org/10.1007/s42965-023-00300-1
Ninyerola M, Pons X, Roure JM (2007) Monthly precipitation mapping of the Iberian Peninsula using spatial interpolation tools implemented in a Geographic Information System. Theor Appl Climatol 89:195–209. https://doi.org/10.1007/s00704-006-0264-2
Nyckha D, Furrer R, Paige J, Sain S (2021) Fields: tools for spatial data. R Package Version 15.2.
Ohsawa M, Nakamura Y, Eguchi T, Takada M, Ohtsuka T, Wildpret W, Del Arco MJ, Shumiya T, Reyes-Betencort JA (1999) Comparative ecological study on the evergreen broad-leaved forest in the Canaries and Japan. In: Suzuki K (ed.). Researches related to the UNESCO’s Man and Biosphere Programme in Japan 1997–1998. Japanese Coordinating Committee for MAB: 9–16. Japón. L
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O’Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6-4
Otto R, Barone R, Delgado JD et al (2012) Diversity and distribution of the last remnants of endemic juniper woodlands on Tenerife, Canary Islands. Biodivers Conserv 21:1811–1834. https://doi.org/10.1007/s10531-012-0278-2
Otto R, Fernández-Lugo S, Blandino C, Manganelli G, Chiarucci A, Fernández-Palacios JM (2020) Biotic homogenization of oceanic islands depends on taxon, spatial scale and the quantification approach. Ecography 43:1–12. https://doi.org/10.1111/ecog.04454
Parks SA, Holsinger LM, Abatzoglou JT, Littlefield CE, Zeller KA (2023) Protected areas not likely to serve as steppingstones for species undergoing climate-induce range shifts. Glob Change Biol 29:2681–2696. https://doi.org/10.1111/gcb.16629
Pedrotti P (2013) Plant and vegetation mapping. Springer International Publishing, p 294
Pesaresi S, Biondi E, Casaveccchia S (2017) Bioclimates of Italy. J Maps 13(2):955–960. https://doi.org/10.1080/17445647.2017.1413017
QGIS Development Team (2022) QGIS Geographic Information System. Open Source Geospatial Foundation Project. https://qgis.org
Rivas-Martínez S (1983) Pisos bioclimáticos De España. Lazaroa 5:33–43
Rivas-Martínez S (1997) Syntaxonomical synopsis of the north American natural potential vegetation communities 1. Itinera Geobotánica 10:5–148
Rivas-Martínez S, Wildpret W, Del Arco M, Rodríguez O, de Paz Pérez, García PL, Acebes A, Díaz JR, T.E., and, Fernández F (1993) Las comunidades vegetales de la Isla De Tenerife (Islas Canarias). Itinera Geobotanica 7:169–374
Rivas-Martínez S, Rivas S, Penas A (2011) Worldwide bioclimatic classification system. Global Geobotany 1:1–634 + 4 maps
Rodríguez-Delgado O, Wildpret W, Del Arco MJ, Pérez-de Paz P PL (1990) Contribución Al Estudio fitosociológico De Los restos de sabinares y otras comunidades termófilas de la Isla De Tenerife (Canarias). Rev Acad Canar Cienc 2(1990):121–142
Rumeu B, Nogales M (2021) Los bosques de cedros en El Parque Nacional Del Teide in Durbán. In: Martín M, JL (ed) La Ciencia en El Parque Nacional Del Teide Durante El período 2009–2019. Editorial Turquesa and Cabildo Insular de Tenerife, pp 217–230
Salas-Pascual M, Del Arco MJ, Pérez-de-Paz PL (1998) Contribución Al Estudio fitosociológico Del pinar grancanario (Islas Canarias). Lazaroa 19:99–117
Sangüesa-Barreda G, García-Cervigón AI, García-Hidalgo M, Rozas V, Martín-Esquivel JL, Martín-Carbajal J, Martínez R, Olano JM (2021) Vertical cliffs harbor millenia-old junipers in the Canary Islads. Ecology 103:e3633. https://doi.org/10.1002/ecy.3633
Santos-Guerra A (1990) Bosques de Laurisilva en la región macaronésica. Consejo de Europa. Colección Naturaleza y Medio Ambiente 49. Estrasburgo. 79 pp
Silva LD, Brito de Azevedo E, Vieira F, Bento R, Silva L (2019) Limitations of species distribution models based on available climate Change Data: a Case Study in the Azorean Forest. Forests 10:575. https://doi.org/10.3390/f10070575
Sperling FN, Washington R, Whittaker RJ (2004) Future climate change of the subtropical North Atlantic: implications of the cloud forests of Tenerife. Clim Change 65:103–123. https://doi.org/10.1023/B:CLIM.0000037488.33377.bf
Torregrosa A, Taylor MD, Flint LE, Flint AL (2013) Present, Future and Novel bioclimates of San Francisco, California Region. PLoS ONE 8:1–14. https://doi.org/10.1371/journal.pone
Van Beusekom AE, GonzálezG., Scholl MA (2017) Analyzing cloud base at local and regional scales to understand tropical montane cloud forest vulnerability to climate change. Atmos Chem Phys 17(11):7245–7259. https://doi.org/10.5194/acp-17-7245-2017
Vondrakova A, Vávra A, Vozelinek V (2013) Climatic regions of Czech Republic. J Maps 9:425–430. https://doi.org/10.1080/17445647.2013.800827
Walther GR (2010) Community and ecosystem responses to recent climate change. Philos Trans R Soc B 365(1549):2019–2024
Wei T, Simko W (2021) R package ‘corrplot’: Visualization of a Correlation Matrix (Version 0.92). https://github.com/taiyun/corrplot
Woodward FI (1987) Climate and plant distribution. Cambridge Univ. Press, Cambridge, p 174
Woodward FI, Williams BG (1987) Climate and plant distribution at global and local scales. Vegetatio 69:189–197
Zhao D, Wu S, Yin Y, Y ZY (2011) Vegetation distribution on tibetan plateau under climate change scenario. Reg Envriron Chang 11:905–915. https://doi.org/10.1007/s10113-011-0228-7
Acknowledgements
We thank the financial support from the Canary Islands Government. The grant was awarded to the first author and included in a Fellowship Scheme for a Doctoral Training Program in Biodiversity and Conservation: Universidad de La Laguna. We also thank Sandra Guerrero for the English revision of original manuscript.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
JJGA, VBR, JMGM & MJD wrote the main manuscript. JJGA & VBR prepared figures. JJGA & JMGM conceptualization & statistical analysis. MJD phytosociological and vegetal communities supervision. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Daniel Sanchez Mata.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
García-Alvarado, J.J., Bello-Rodríguez, V., González-Mancebo, J.M. et al. Updating knowledge of vegetation belts on a complex oceanic island after 20 years under the effect of climate change. Biodivers Conserv 33, 2441–2463 (2024). https://doi.org/10.1007/s10531-024-02864-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-024-02864-3