Abstract
Invasive non-native plants challenge ecosystems restoration, and understanding the factors that determine the establishment of invasive plants is crucial to improve restoration outcomes. However, the drivers of invasibility of plant communities are not sufficiently clear, and combined effects are not understood. Therefore, we investigated the contribution of the main drivers of invasion success during early phases of restoration, i.e., biotic resistance, invasive propagule pressure, and environmental fluctuations. We compared the contribution of these drivers in a series of mesocosms experiments using designed grasslands as a model system, and Solidago gigantea as invasive model species. Two grassland communities were designed according to competitive trait hierarchies with different sowing patterns, reflecting variation in biotic resistance. We then manipulated invader propagule pressure and applied different scenarios of environmental fluctuation, i.e., flood, heat, and N fertilization. Invasive biomass was considered as proxy for invasion success, while native biomass represented restoration success. There were consistent effects of biotic resistance to S. gigantea invasion via competitive trait hierarchies in the three experiments. Communities dominated by species with high-competition traits were more resistant regardless of environmental fluctuation. Clumped seeding of the native community reduced invasibility, whereas high non-native propagule density increased invasion. The effects of environmental fluctuation were less consistent and context-dependent, thus playing a secondary role when compared to biotic drivers of invasion. Restoration initiatives on grasslands impacted by invasive plants should consider biotic resistance of the restored community as a key driver and the importance of controlling further arrivals of invasive species during community assembly.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Given the negative impact of invasive non-native plants on degraded ecosystems, restoration activities that constrain the spread of such species and promote the colonization of native ones are increasingly needed (Bakker and Wilson 2004). However, some activities associated with restoration, e.g., clearcutting, site preparation, and topsoil removal, create windows of opportunity (sensu Johnstone 1986) for further invasions (Torres et al. 2018). Thus, an emerging field in restoration ecology is the design of resident communities that are resistant to invasions during community assembly. In the case of grasslands, where the design and reintroduction of native seed mixtures is common restoration practice (Kiehl et al. 2010), most evidence shows that revegetation can decrease the performance of invasive plants, although the magnitude of such effect is variable (Schuster et al. 2018). Since biotic resistance is only one aspect explaining to what extent invasion can succeed, other invasion drivers need to be considered as well, i.e., invasive propagule pressure and time of arrival of the invasive species, and the influence of environmental fluctuations (Byun et al. 2015).
The biotic resistance of a resident community implies a reduction of invasibility mainly through competition (Levine et al. 2004). Although there is little evidence that species interactions can repel invaders completely, some resident communities reduce the establishment, abundance, and fitness of invaders to a certain extent (Levine et al. 2004). The understanding of biotic resistance as driven by interspecific competitive interactions has been addressed by various hypotheses, based on diversity effects and the saturation of niche space (Elton 1958; Hooper et al. 2005), limiting trait similarity (Funk et al. 2008), phylogenetic relatedness (Yannelli et al. 2017a), or trait hierarchies, with the focus on the identity and value of fitness-related traits that define which species are most likely to invade successfully (Kunstler et al. 2012).
An additional mechanism to increase biotic resistance and diminish invasion success in early community assembly is the management of community density (Yannelli et al. 2018). Threshold seed densities and sowing patterns can play an important role in invader suppression in newly restored ecosystems (Reinhardt Adams and Galatowitsch 2007) by increasing competition for light (Lindig-Cisneros and Zedler 2002) and, thus, reducing the establishment of invaders even when arriving at higher densities (Byun et al. 2015). In turn, the propagule pressure of invasive plants is a key determinant of invasion success (Von Holle and Simberloff 2005; Simberloff 2009). It comprises the number of seeds in a propagule (‘propagule density’), and the rate at which propagules arrive per unit of time (‘propagule arrival rate’; Lockwood et al. 2005). Previous studies acknowledge propagule pressure as a key factor explaining invasion success, thus the minimization of propagule pressure, along with rapid revegetation, should reduce the impact of invasive plants in restored areas (Colautti et al. 2006; Hufbauer et al. 2013; Cassey et al. 2018).
Abiotic constraints and environmental variability significantly interact with biological invasions. Harsh abiotic conditions can affect the performance of invasive and native species (Melbourne et al. 2007; Parepa et al. 2013), and thus influence invasion outcomes (Going et al. 2009). Since invasive species can capitalize on greater resource availability after a disturbance or reduced competition, climate-change effects are thought to increase biological invasions (Sorte et al. 2013; Funk et al. 2020). Abiotic fluctuations such as floods (Gerhardt and Collinge 2003; Collinge et al. 2011), eutrophication (Goldstein and Suding 2014), or extreme temperatures (Collinge et al. 2011; Goldstein and Suding 2014) play a decisive role in invasibility, and therefore need more experimental studies, especially when the likelihood and frequency of floods, heatwaves, and droughts increase with climate change (Kam et al. 2018; Knutson et al. 2018; Schiermeier 2018).
While the importance of trait similarity and phylogenetic relatedness on community invasibility is ambiguous (Byun et al. 2015; Funk and Wolf 2016; Yannelli et al. 2017a; Byun et al. 2020), the effects of environmental stress (Conti et al. 2018), trait hierarchies (Funk and Wolf 2016; Carmona et al. 2019), and seed density (Yannelli et al. 2017b; Yannelli et al. 2018; Byun et al. 2020) are increasingly supported as suitable proxies of competition outcomes, and thus of the biotic resistance of a plant community. However, interactions of trait hierarchies and environmental fluctuations with propagule pressure and priority effects (i.e., time of arrival of the species) need further research to elucidate their relative effect, and hence to improve ecological restoration under climate change.
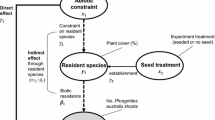
In this study, we investigate specific and combined effects of the following drivers of plant invasions during early phases of community assembly in experimental grasslands: (1) biotic resistance via competitive trait hierarchies and (2) via seed clumping of the resident community, (3) propagule pressure of the invasive species, and (4) abiotic constraints. We applied the concept of trait hierarchies by designing two native communities a priori, in which species abundances were manipulated to obtain one grassland dominated by highly competitive species, and another one dominated by low-competitive species. On these communities, the effects of seed clumping, propagule pressure, and abiotic constraints were experimentally tested to answer the following questions: (1) Is reduced invasibility attributable to the biotic resistance attained via trait hierarchies within the native resident community? (2) What is the role of seed clumping of the native species on the establishment of the invasive species? (3) To what extent do propagule density or propagule arrival rate affect invasion success? (4) Do environmental fluctuations increase grassland invasibility? (5) Are there cascading effects between biotic resistance, propagule pressure, and the abiotic constraints, that favor grassland invasion?
Materials and methods
Study system
We assessed invasion success in three mesocosm experiments: one in a greenhouse and two in climate chambers. Semi-natural mesic grasslands of Central Europe were the model system in which we investigated the relative importance of different drivers on the invasion success of Solidago gigantea Aiton (Asteraceae), a common invasive forb from North America, that was introduced as an ornamental to Europe in the eighteenth century (Weber and Jakobs 2005).
The experiments simulated early phases of active restoration of grasslands, in which particular mixtures of native species are sown on bare soil (Kiehl et al. 2010), while seeds of S. gigantea concomitantly arrived. Trait hierarchies, sowing patterns, the two aspects of propagule pressure of the invasive species, and environmental fluctuations were manipulated to assess the ability of the resident communities to hinder invasion. Environmental fluctuation scenarios consisted of manipulations of N fertilization in the greenhouse experiment, and heat and flood experiments in climate chambers. For the experiments, the seeds of S. gigantea were collected along the River Isar in Freising, southern Germany (48°23′57″N, 11°45′16″E). The seeds of the native grasses were supplied by a regional seed producer based on local provenances (Johan Krimmer, Pulling, Germany).
Design of native communities
Two community types were established for testing biotic resistance as a function of fitness-related trait hierarchies of the study species. For this, five native grass species were selected and allocated in the trait hierarchy based on their ecological indicator values for nutrients (EIV-N) (Ellenberg et al. 1991). EIV-N values positively correlated to fitness-determining traits (e.g., seed mass, relative growth rate, specific leaf area), and thus they are good proxies for ecophysiological characterization of the native species, especially during establishment (Bartelheimer and Poschlod 2016). The resulting hierarchy was Arrhenatherum elatius (L.) P.Beauv. ex J.Presl & C.Presl. > Holcus lanatus L. > Dactylis glomerata L. > Festuca rubra L. > Festuca ovina L. Experimental grassland communities composed of native grasses were chosen because this functional group generates high biotic resistance to invasions, compared to other growth forms (Sheley and James 2010; Török et al. 2010), and rapidly colonizes open areas (Young et al. 2009). Traditionally used mixtures to establish highly productive grasslands were not intended, but instead, a synthetic community portraying a gradient of trait values.
The two grassland types were designed based on the relative abundances of the native grasses, and this way manipulating the dominance of the fitness-related traits of interest. In community ARR50, the species with the highest competitive ability was more abundant (A. elatius dominating), whereas, in community ARR5, species with a lower competitive ability were more abundant (F. ovina dominating; Table 1). For all experiments, 3 g m−2 was set as target sowing density, mimicking recommendations in grassland revegetation (Kiehl et al. 2010). The effective density of the seeds needed for ARR5 and ARR50 was based on seed mass, adjusted considering the a priori germination rates tested for each species (Table S1).
Trait-hierarchy, sowing-pattern and N-fertilization experiment
To test for the effect of competitive trait hierarchy (‘community type’) and seed clumping as the expression of biotic resistance in combination with abiotic constraints, a greenhouse experiment was conducted for 18 weeks. Forty plastic trays (48 × 33 × 6 cm3, i.e., 0.16 m2 area and 0.0098 m3 volume each) were filled with two parts of potting soil and one part of vermiculite and distributed in five blocks. Each tray was divided into two halves by polystyrene pieces allowing for a split-plot design. Each tray contained a combination of community type, seeding pattern, and N fertilization, resulting in four possible treatment combinations and five replicates per treatment combination (Fig. S1.1).
Half of the trays contained the community ARR5, while the other half ARR50. In the ‘random’ treatment, the native mixture was spread over the entire tray; in the ‘clumped’ treatment, the same amount of seed mixture was sown in 59% of the area by applying seeds in a concentric pattern to simulate clumped establishment of the plant communities, while 31% of the area was left open (Fig. S1.2). Solidago gigantea was sown simultaneously with the native mixture with 1 g m−2 in one half of each tray, while the second half remained uninvaded.
To test the influence of N-fertilization on the competitive outcome of native and invasive species, we applied fertilizer in two different levels at the early stages of community establishment. Half of the trays received only a basic amount of NPK (Peters Excel Grower 15/5/15, 100 g/100 l), while the second half received additional nitrogen (Novatec 18 Fluid, 85 g/100 l). Fertilization was done once a week by supplying 1 l of dissolved nutrients; in total, 0.6 g N together with P and K were added to provide a basic nutrition supply. An extra supply of 0.6 g N simulated fluctuating fertilization. The N fertilization levels were equivalent to 37.5 or 75.0 kg N ha−1, representing low- and high-intensity grasslands, respectively (Vitousek et al. 1997; Butterbach-Bahl et al. 2011). No extra water was added during the enrichment period, to keep soil moisture levels similar. Afterward, tap water was added 1–3 times per week, depending on the growth stage of the mesocosms and temperatures inside the greenhouse.
After 16 weeks, the aboveground biomass of the invasive species and the native community was harvested 1 cm above the soil level, and the biomass was oven-dried at 65 °C for 48 h and weighed.
Trait-hierarchy and heat-flood pulse experiment
To test for the effect of community type in combination with heat and flood pulses, an experiment was conducted in two walk-in climate chambers (2.4 × 3.2 × 2.2 m3; area 7.7 m2; Fig. S1.3) at TUMmesa (TUM Model EcoSystem Analyser) at Technical University of Munich (Roy et al. 2021). During the experiment, the chambers were maintained at 12 h light and 12 h dark, air temperature was set to 25/15 °C (day/night range), and 60–65% relative humidity. Air O3 concentration was constant at 0.58 ppb, while CO2 varied between 380 and 450 ppm. Commercial sterilized potting soil (Floradur® Pot substrate) was placed in 48 trays of the above-mentioned size. Each tray was again split into two halves, allowing for a split-plot design. The trays were placed on standard greenhouse floodable tables with six trays per table and four tables per chamber (Fig. S1.4 and S1.5).
In each of the floodable tables, three trays per community type (ARR5 and ARR50) were randomly allocated, and the position within each table was biweekly re-randomized. S. gigantea was simultaneously sown with the native mixture in a density of 1 g m−2 in one-half of each tray, the second half was not sown with the invasive species. Water was supplied to the plants every second day, by pouring 10 l of tap water directly in the table, enabling water uptake by the roots. To assess the effect of heat and flood pulses, the experimental communities were subjected to interspersed simulated temperature increases and floods after the third week since start of the experiment. For this, one chamber was randomly selected and its temperature increased from 25/15 to 35/20 °C for 48 h every second week. To simulate the flood events, two tables per chamber were randomly selected and watered with 50 l of tap water and maintained flooded for 48 h every two weeks.
To measure the competitive outcome of the communities under such environmental fluctuations, the aboveground biomass of the invasive species and the native community was harvested 1 cm above the soil level after 14 weeks of community development; it was oven-dried at 65 °C for 48 h and weighed.
Trait-hierarchy, invasive propagule-pressure, and heat-flood experiment
To assess the role of invasive propagule density and arrival rate in combination with heat and flood pulses on the biotic resistance of the communities, a similar experimental approach was tested in four climate chambers of TUMmesa. The two community types were subjected to competition with the invasive species given three propagule pressure treatments: (1) One single introduction, simultaneously with the sowing of the native seeds, with a density of 1 g m−2; (2) three biweekly introductions, each of them with a density of 1 g m−2; and (3) one single introduction, with a density of 3 g m−2, simultaneously with the sowing of the native mixture. The chosen propagule densities fall within recorded seed rain values for Solidago altissima L., another aggressive invader in Europe, in unmowed old fields under succession (Meyer and Schmid 1999) and then extrapolated to S. gigantea. The experiment was carried out with three fully crossed factors: (1) community type, (2) invasive propagule pressure (i.e., propagule density or arrival rate), and (3) environmental fluctuation (i.e., extreme heat and floods).
In each chamber, two tables contained trays with the community ARR50 and two tables ARR5. Each of the 96 trays was split into two halves. By using the split-plot approach of the trays, each treatment combination had a corresponding control at the side, i.e., with no addition of S. gigantea seeds. The three propagule pressure levels were randomly applied in each half of the 96 trays, so in each table, each propagule level was applied in two half-trays (Fig. S1.6). Whenever not submitted to flooding, water was supplied to the plants every second day, by pouring 10 l of tap water into the table.
To assess the effect of environmental fluctuations, two of the chambers were randomly selected to simulate six interspersed heat pulses and floods after the third week of community development under the conditions described in the previous experiment. To simulate extreme heat pulses, the temperature was increased from 25 to 35 °C for 72 h every second week. To simulate six flood events, two tables per chamber corresponding to two different treatment combinations were randomly selected and supplied with 50 l of tap water and maintained flooded for 72 h every two weeks.
After 17 weeks, the aboveground biomass was harvested by clipping the shoots 1 cm above the soil level; the material was dried at 65 °C for 96 h and weighed. Aboveground biomass of the plants was considered appropriated to relate to the biotic resistance of the native species, and the invasion success in the treatments applied.
Data analyses
To test the effect of each driver, i.e., biotic resistance (two levels of trait hierarchy), sowing pattern of resident communities, propagule pressure of the invasive species (three levels), and environmental fluctuations (N-fertilization, heat waves, and flood pulses, with two levels each), linear mixed-effects models (LMMs) with random intercepts were fitted using the function lme from the R package nlme (version 3.1.137) (Pinheiro et al. 2021). Experimental blocks or climate chambers and experimental tables were defined as random effects, accordingly, whereas biomass of the native community, competitive hierarchy, sowing pattern, propagule pressure, heat waves, and flood pulses, were included as fixed effects explaining biomass of S. gigantea in the respective experiments. Assumptions of normality of residuals and homoscedasticity were checked using model plots. The response variable ‘aboveground biomass’ was log-transformed to meet the assumption of a normal distribution of residuals whenever needed. To account for cases of non-homogeneity of variances, we used a variance structure (VarIdent) for the propagule level (Experiment 3).
Since the trait hierarchies, sowing pattern, propagule pressure, and abiotic conditions were expected to have complex direct and indirect effects on the resulting biomass of native and invasive plants, confirmatory path analyses (i.e., piecewise Structural Equation Models, SEM) were applied to assess the causal relationships depicting them. Piecewise SEM combines information from multiple separated models into a single causal network (Shipley 2009). Moreover, because the individual models used for piecewise SEM included random effects and did not always have a normal distribution, piecewise SEM was advantageous to conduct SEM analyses (Lefcheck and Duffy 2015). For each experiment, a single causal network relating to the paths of interest was constructed. Since the a priori models were saturated, i.e., all possible paths were considered, no overall goodness-of-fit based on the test of directed separation (Shipley 2000) is provided (here Fischer’s C = 0, with p-value = 1). Marginal model fit (R2) of individual path models ranged from 19 to 91% of explained variance (Fig. 4). We assessed the relative importance of the variables included in the final SEMs using standardized path coefficients scaled by mean and variance. LMMs using the package NLME (Pinheiro et al. 2021) were the base for fitting the SEM in the package piecewiseSEM version 2.0.2 (Lefcheck 2016).
Results
Effects of trait-hierarchy, sowing-pattern and N-fertilization
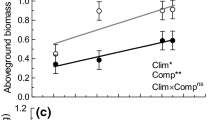
Grass communities dominated by high-competition species had a more negative effect on the biomass production of the invasive S. gigantea (mean ± SE: ARR50: 0.45 ± 0.08 g vs. ARR5: 1.00 ± 0.17 g; Fig. 1, Table S3). Also, sowing patterns had a significant effect on the biomass production of the invasive species. Clumping led to a direct increase per unit area of native biomass production (clumped: 50.0 ± 1.3 g vs. random: 43.5 ± 1.3 g). An increased input of nitrogen did not stimulate the biomass of the invasive species. Instead, it favored the biomass of the native community (N-enriched: 54.9 ± 1.3 g vs. Non-enriched: 38.6 ± 1.3 g), which might result in negative effects on invasive biomass (Fig. 1). The total community biomass, i.e., the biomass of native and invasive species added up, was overall positively affected by N-enrichment (p < 0.001) and a clumped sowing (p < 0.001; Fig. S2.1, Table S2).
Effects of the community type, sowing pattern, and N fertilization on aboveground biomass of native grass mixtures (upper panel) and the invasive Solidago gigantea (lower panel), 16 weeks after sowing. The two communities (ARR5 and ARR50) had low vs. high competitive traits and were subjected to pulses of N fertilization, which had a positive effect on native biomass (p < 0.001), albeit without differences among communities. Clumped sowing resulted in more native biomass (p < 0.001), and S. gigantea was significantly less productive in the high-competition community ARR50. Note that different scales were used for the upper (‘Native community biomass’) and lower panel (‘Invasive species biomass’)
Effects of trait-hierarchy and heat-flood pulses
Similar to previous results, there were negative effects of the high-competition community on the biomass of the invasive species (ARR50: 5.42 ± 0.67 g vs. ARR5: 9.45 ± 0.70 g; mean ± SE) (Fig. 2, Table S3). Moreover, environmental fluctuation (i.e., interspersed flood pulses and heat) had a positive effect on the biomass of the native community (Fig. 2). The total community biomass was significantly higher in invaded communities (p < 0.001), and positively affected by flood and heat pulses (p < 0.001), with ARR50 being less productive (p < 0.005; Fig. S2.2, Table S3).
Effects of the community type, flood, and heat pulses on aboveground biomass of the two native communities (upper panel) and the invasive Solidago gigantea (lower panel), 14 weeks after sowing. The communities ARR5 and ARR50 were established based on trait hierarchies, and subjected to weekly interspersed pulses of heat and flood, that had a positive effect on the biomass of the two native communities (p < 0.001). Community ARR50 produced more native biomass (p < 0.05), and S. gigantea was significantly less abundant here, while more biomass was produced when the invaded communities experienced flood pulses (p < 0.001). Note that different scales were used for the upper (‘Native community biomass’) and lower panel (‘Invasive species biomass’)
Effects of trait-hierarchy, propagule-pressure, and heat-flood pulses
We found strong evidence of biotic resistance conferred by trait hierarchies also when the propagule pressure of the invasive species was manipulated, in addition to environmental fluctuations. The community dominated by high-competition species had a negative effect on the biomass of the invasive species (ARR50: 1.00 ± 0.17 g vs. ARR5: 1.53 ± 0.17 g; Fig. 3, Table S3). Flooding did not affect the biomass of either native or invasive plants (Fig. 3a). In contrast to the previous experiment, heat had a positive effect on the invasive biomass (heat: 1.45 ± 0.17 g vs. non-heat: 1.08 ± 0.17 g; Fig. 3b). Added early in the assembly process, a propagule in high density of S. gigantea had a negative effect on native biomass and increased biomass of the invasive species (Fig. 3). The propagule in the largest density of the invasive species led to highest invasive biomass (3: 2.79 ± 0.42 g vs. 1: 0.52 ± 0.10 g). Contrastingly, three introduction events of propagules were not different from a single seeding event (1: 0.52 ± 0.10 g 1 + 1 + 1: 0.48 ± 0.08 g; Fig. 3). The total community biomass was only significantly higher in invaded communities (p = 0.005; Fig. S2.3, Table S2).
Effects of the community type and invasive propagule pressure on aboveground biomass of native grass communities (upper panel) and the invasive Solidago gigantea (lower panel), 17 weeks after sowing and subjected to flood (a) and heat (b) pulses. The two communities ARR5 and ARR50 were established based on trait hierarchies and sown with three different levels of invasive propagule pressure, i.e., 1 g m−2 simultaneously with native sowing, 1 + 1 + 1 g m−2 (three weekly introductions of 1 g m−2 each), and 3 g m−2 simultaneously with native sowing. A large density propagule added early in the assembly process had a negative effect on the biomass of the two native communities (p < 0.1). Community ARR50 affected negatively the biomass of S. gigantea (p < 0.01), while more invasive biomass was produced with an invasive propagule density of 3 g m−2 (p < 0.001). Note that different scales were used for the upper (‘Native community biomass’) and lower panel (‘Invasive species biomass’)
Direct and indirect drivers of plant invasion
The structural equation models indicated that the high-competition communities were more resistant against the invasive species irrespective of environmental fluctuations (Fig. 4a–c). While clumped sowing and high competition traits of the grass communities were positive for native biomass, environmental drivers positively affected native and invasive biomass in the first and second experiment, respectively (Fig. 4a, b). In contrast, no effects of environmental drivers were observed in the third experiment (Fig. 4c).
Structural Equation Model fitted with standardized coefficients to test direct and indirect effects of a competitive traits, sowing pattern, and N enrichment, b competitive traits, and heat and flood pulses, and c competitive traits, propagule pressure, and heat and flood pulses, on aboveground biomass of the invasive Solidago gigantea and the native grass communities. Solid and dashed arrows indicate that the drivers had a significant positive or negative effect on biomass, respectively, while half-transparent arrows indicate no detectable influence of the driver (p > 0.05). Standardized coefficients are presented for each significant path; conditional R2 values for the individual models are shown for each response variable
In the first experiment, native biomass was positively affected by clumped sowing (estimate = 0.32, p ≤ 0.001) and N enrichment (estimate = 0.80, p ≤ 0.001), with indirect negative effects on invasive biomass (Fig. 4a). Thus, native biomass exerted a negative control over invasive biomass (estimate = − 0.59, p ≤ 0.05). Together with the effects of native biomass, trait hierarchy also negatively affected invasive biomass (estimate = − 0.44, p ≤ 0.01, Fig. 4a). In the second experiment, native biomass was positively affected by environmental fluctuations (estimate = 0.55, p ≤ 0.001 for heat; estimate = 0.51, p ≤ 0.001 for flood). However, no influence of native biomass on invasion was observed (Fig. 4b). This time, communities dominated by highly competitive plants were positively affecting native biomass (estimate = 0.27, p ≤ 0.05), while again suppressing invasion (estimate = − 0.46, p ≤ 0.001). Finally, flood pulses positively affected invasive biomass (estimate = 0.58, p ≤ 0.001), indicating that, depending on the scenario, environmental drivers can also facilitate invasion (Fig. 4b). In the third experiment, invasive propagule pressure positively controlled invasive biomass (estimate = 7.58, p ≤ 0.001), while native biomass was negatively affected (estimate = − 0.20, p ≤ 0.05). Once more, the more competitive community had a lower invasive biomass (estimate = − 0.12, p ≤ 0.05). However, due to a stronger effect of invasive propagule pressure on native and invasive biomasses, this effect was almost four times smaller than what was previously observed (Fig. 4c).
Discussion
Biotic resistance in early stages of plant communities
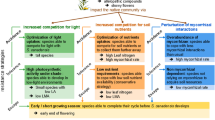
In all three experiments, the effect of the biotic resistance derived from trait hierarchies led to a reduction in biomass on the invasive species during the establishment of our synthetic grassland communities, irrespective of environmental fluctuations in nutrients, water, and temperature, and for every level of invasive propagule pressure. This result supports the hypothesis of biotic resistance attained by competitive hierarchies based on the identity and value of fitness-related functional traits (Laughlin 2014; Funk and Wolf 2016; Conti et al. 2018; Carmona et al. 2019; Ferenc and Sheppard 2020). Moreover, it agrees with the mass ratio theory (Grime 1998), posing that the effects of a certain community on competition are driven by the traits of the dominant species. This explains why the high-competition community ARR50 was more efficient in suppressing S. gigantea in the three experiments.
These findings should be tested under field conditions due to: (1) the ‘artificial’ species composition including only grasses in our experiments; (2) the use of ‘soil-limited’ mesocosms to test our experimental approach; and (3) the impossibility of disentangling effects of the manipulated relative abundances (evenness) from those expected to result from trait values (competitive trait hierarchy). Still, we argue that our approach can be, to a certain degree, associated with practical aspects of grassland management via reseeding. Because our synthetic communities are exclusively composed by grasses, we avoided potential confounding effects related to the differences in the ecological niches occupied by species belonging to other functional groups (i.e. forbs or legumes) and the values yielded for the fitness-related traits considered here. Because seed mixtures applied in the reseeding of similar grasslands are, mostly, grass-dominated (Gentili et al. 2015), assessing the role of commonly occurring grasses is very important to managing such systems when dealing with invasive species. This is particularly important if we consider that grass-dominated grasslands can produce higher native cover and stem density (Meissen et al. 2020); aspects which are most desirable to outcompete invasive plants.
Sowing patterns and invasibility
The pattern of seed sowing showed that clumping, and thus locally higher densities both of the invasive species and the native grass community, reduced the biomass of S. gigantea. By sowing in circular clumps within the experimental trays, the plasticity of plant growth may have allowed the roots of the native grasses to rapidly forage the surrounding un-vegetated areas of high (soil) resource levels. Similar mechanisms were suggested by Fargione et al. (2003), Török et al. (2010), and Lu et al. (2020). The resulting growth rate advantage of the native grasses led to stronger asymmetric (aboveground) competition (Freckleton and Watkinson 2001; Weiner et al. 2001), and thus enhanced suppression of S. gigantea.
Additionally, the clumped sowing pattern in our experiment generated a local-scale increase in density of the native community. The negative effects on invasive biomass due to this higher local density implies that the competition for light benefitted the native plants (Weiner et al. 2001; Lindig-Cisneros and Zedler 2002; Nemec et al. 2013; Byun et al. 2015, 2020; Yannelli et al. 2018), especially in the community ARR50, whose dominant species were able to grow rapidly, to fill the available space, and to create a dense canopy (Colbach et al. 2014). This result underpins the importance of rapid and uniform occupancy of space combined with higher density and highly productive species to suppress unwanted species (Hess et al. 2020; Lu et al. 2020). However, even though clumped seeding reduced S. gigantea invasion, in a real-world setup of bare ground revegetation, gaps between clumps must be reduced to minimize invasibility of the community (Lu et al. 2020).
Propagule pressure and invasiveness
Our study agrees with published evidence on the importance of propagule pressure for invasion success (Simberloff 2009; Byun et al. 2015, 2020). A large propagule density (i.e., high seed density) in the early stages (i.e., priority effect) of colonization of bare soil resulted in the highest aboveground biomass of S. gigantea, and thus most successful establishment of the invasive species (Hess et al. 2019). Priority effects were identified as a key factor explaining the assembly of communities, together with trait-based assembly rules (Fukami et al. 2005). Since resources (e.g., space, nutrients, light) are fully available at very early phases of colonization, most of the propagule of S. gigantea rapidly pre-empted or modified the available niche (cf. Fukami 2015), with negative effects on the native species (Dickson et al. 2012). Furthermore, our results also suggest propagule density, and not propagule arrival rate as a stronger determinant of the invasion success in S. gigantea.
This finding further supports the importance of priority effects for invasion resistance (Gillhaussen et al. 2014; Viana et al. 2016; Hess et al. 2019; Yannelli et al. 2020; Weidlich et al. 2021). The strikingly lower biomass production of S. gigantea when introduced in three pulses of 1 g m−2 each compared to a single event of 3 g m−2 suggest a marked priority effect of the resident community (Hess et al. 2020), capturing resources and thus, limiting the establishment of S. gigantea. As stated by Simberloff (2009), propagule arrival rate becomes more important if it interplays with demographic or environmental stochasticity, and has a notable effect when a large number of introduction events triggers the establishment of the invasive species. Notwithstanding, regardless of the propagule pressure, the community ARR50 was negatively affecting invasion success, since competitive hierarchies offset priority effects to a high degree. Thus, arrival order and propagule density are less important when highly competitive native species are present in the invaded community (Fukami 2015).
The role of abiotic fluctuations
In our experiments, we applied environmental fluctuations as pulse events following Jentsch and White (2019), i.e., some system resources or abiotic factors were abruptly modified to test their effects on grassland invasibility. Native communities capitalized on pulses of nitrogen supply, especially ARR50, and prevented the invasive species from taking advantage of extra nitrogen availability, given the dominance of highly competitive native species (Walker et al. 2015; Conradi et al. 2017; Liu et al. 2018). Despite S. gigantea growing best in nitrogen-rich habitats (Weber and Jakobs 2005), and invasive species, in general, responding faster to environmental fluctuations (Davis et al. 2000; Liu and Kleunen 2017; Liu et al. 2019) and enhancing nutrient uptake (Dassonville et al. 2008; Liu et al. 2019), our experiment did not show advantages for invasion of S. gigantea with a pulsed supply of nitrogen in our model systems, that represent early stages of grassland establishment. This might be different in later stages where S. gigantea with rapid resource exploitation, high growth rates, a tall and dense canopy, and clonal propagation could dominate disturbed grasslands (Weber and Jakobs 2005).
Thus, with the duration of the environmental fluctuations simulated in our experiments, we detected no clear benefits for the invader, suggesting that environmental fluctuations need broader testing, e.g., accounting also for different magnitudes and temporal patterns of resource or environmental pulses of flood and heatwaves (Parepa et al. 2013; Koerner et al. 2015; Matsubara and Sakai 2016). This way, the fluctuating conditions depicting the superior ability of the invasive plants to capitalize on periods of high resource availability (rapid response or higher maximum growth rate) can be better identified.
Cascading effects on plant invasions
Our study suggests that designing resident grassland communities based on trait hierarchies has a consistent direct effect on the biomass of the invasive species during early stages of community assembly, even when other invasibility drivers are at stake. Moreover, clumped sowing patterns increased the asymmetry of competition in favor of the native communities, improved their productivity, and thereby indirectly affected the productivity of the invasive species. Conversely, we found that the large propagule density of S. gigantea in the third experiment influenced the establishment of that species in two ways: On the one hand, larger propagule density of the invasive established first, and quickly produced a canopy to capitalize on the available resources, thus having a direct negative effect on the biomass production of the native species. On the other hand, an invasive propagule arriving in higher density promoted a greater biomass production of the invasive species (Byun et al. 2015), while the biomass of the native community was not consistently mediating negative effects on the invasive biomass across the experiments. In the invaded communities, the biomass production of communities was not always different, yet the community ARR50 was always more effective in decreasing the biomass of S. gigantea.
The suppression mechanism on the invasive species may be therefore related not only to higher resource capture but to the expression of other competition traits (Yannelli et al. 2020). Only when there was a pulsed, extra supply of nitrogen and a clumped sowing pattern, the performance of ARR50 disproportionally increased its biomass production, with a negative effect on the biomass of S. gigantea. In contrast to common expectations (Hood and Naiman 2000; Kercher and Zedler 2004; Collinge et al. 2011; Goldstein and Suding 2014), the fluctuation in physical parameters (heat and flood pulses) did not have a consistent effect on the performance of the native community nor the productivity of the invasive species in the two experiments that tested for it, suggesting that even small differences in magnitude or frequency of pulsed environmental fluctuations can have significantly different outcomes in competition between native communities and invasive species.
Study limitations
As the three experiments were conducted in shallow mesocosms, belowground processes involved in competition and establishment of the native grasses and the invasive species might have been overlooked to some degree. Invasion experiments suggest that competition for belowground resources plays a significant role in the survival and productivity of invasive species introduced in designed grassland communities (Brown and Rice 2010; Scherber et al. 2010; Broadbent et al. 2018), without implying that other resources (e.g. light and water) involved in competitive relations among plants are not limiting. Effects of belowground competition can be even more pronounced when considering grass-dominated communities, because of their extensive root system (Fargione et al. 2003, Yannelli et al. 2017b). Therefore, further efforts to disentangle invasion drivers should account for the belowground component at near-natural soil depth conditions.
Still, our study reflects only early phases of grassland establishment, ranging from 14 to 18 weeks after sowing. Thus, seasonal development of the communities, natural disturbances occurring over years, or management measures (mowing or grazing) could lead to different outcomes in a field, long-term developed grassland. Despite this, much can be learned from the patterns we observed, and we argue that the results of this study provide helpful insights into early phases of grassland restoration in areas with invasion pressure.
Conclusions
In this study, we aimed at understanding specific and combined effects of known drivers of invasive plant invasions. We showed that biotic resistance based on hierarchies of traits related to competition has a consistent effect on invasion outcomes in early grassland establishment. Plant communities dominated by strong competitors can reduce invasibility by limiting the productivity of invasive plants. The propagule pressure of the invasive species plays a substantial role when greater propagule densities are introduced early in the assembly process stressing the importance of priority effects. The environmental fluctuations, in contrast, were an inconsistent predictor of invasion outcomes, suggesting context-dependent effects. Previous studies have confirmed how different drivers such as seed provenance, prior arrival of invasive species, and light fluctuations can also contribute to invasion outcomes. This calls for the need of conducting follow-up studies combining also these drivers in more complex setups. However, our results suggest that revegetation or restoration practice needs to account for biotic resistance of the restored community and also the importance of controlling the further arrival of invasive species to limit their effect in early phases, e.g. increasing seed clumping rates.
Data availability
All data included in this manuscript will be made available via MediaTUM.
References
Bakker JD, Wilson SD (2004) Using ecological restoration to constrain biological invasion. J Appl Ecol 41:1058–1064. https://doi.org/10.1111/j.0021-8901.2004.00962.x
Bartelheimer M, Poschlod P (2016) Functional characterizations of Ellenberg indicator values—A review on ecophysiological determinants. Funct Ecol 30:506–516. https://doi.org/10.1111/1365-2435.12531
Broadbent A, Stevens CJ, Peltzer DA, Ostle NJ, Orwin KH (2018) Belowground competition drives invasive plant impact on native species regardless of nitrogen availability. Oecologia 186:577–587. https://doi.org/10.1007/s00442-017-4039-5
Brown CS, Rice KJ (2010) Effects of belowground resource use comlementarity on invasion of constructed grassland plant communities. Biol Invasions 12:1319–1334. https://doi.org/10.1007/s10530-009-9549-6
Butterbach-Bahl K, Gundersen P, Ambus P, Augustin J, Beier C, Boeckx P, Dannenmann M, Gimeno BS, Ibrom A, Kiese R, Kitzler B, Rees RM, Smith KA, Stevens C, Vesala T, Zechmeister-Boltenstern S (2011) Nitrogen processes in terrestrial ecosystems. In: Sutton MA, Howard CM, Erisman JW, Billen G, Bleeker A, Grennfelt P, van Grinsven H, Grizzetti B (eds) The European nitrogen assessment. Cambridge University Press, Cambridge, pp 99–125
Byun C, de Blois S, Brisson J (2015) Interactions between abiotic constraint, propagule pressure, and biotic resistance regulate plant invasion. Oecologia 178:285–296. https://doi.org/10.1007/s00442-014-3188-z
Byun C, Oh M, Lee EJ, Kang H (2020) Seed density is as important as limiting similarity, diversity effect, and propagule pressure in plant restoration to control invasion. Ecol Eng 144:105712. https://doi.org/10.1016/j.ecoleng.2019.105712
Carmona CP, Bello F, Azcárate FM, Mason NWH, Peco B (2019) Trait hierarchies and intraspecific variability drive competitive interactions in Mediterranean annual plants. J Ecol 107:2078–2089. https://doi.org/10.1111/1365-2745.13248
Cassey P, Delean S, Lockwood JL, Sadowski JS, Blackburn TM (2018) Dissecting the null model for biological invasions: a meta-analysis of the propagule pressure effect. PLoS Biol 16:e2005987.https://doi.org/10.1371/journal.pbio.2005987
Colautti RI, Grigorovich IA, MacIsaac HJ (2006) Propagule pressure: a null model for biological invasions. Biol Invasions 8:1023–1037. https://doi.org/10.1007/s10530-005-3735-y
Colbach N, Collard A, Guyot SHM, Mézière D, Munier-Jolain N (2014) Assessing innovative sowing patterns for integrated weed management with a 3D crop:weed competition model. Eur J Agron 53:74–89. https://doi.org/10.1016/j.eja.2013.09.019
Collinge SK, Ray C, Gerhardt F (2011) Long-term dynamics of biotic and abiotic resistance to exotic species invasion in restored vernal pool plant communities. Ecol Appl A Pub Ecol Soc Am 21:2105–2118. https://doi.org/10.1890/10-1094.1
Conradi T, Temperton VM, Kollmann J (2017) Resource availability determines the importance of niche-based versus stochastic community assembly in grasslands. Oikos 126:1134–1141. https://doi.org/10.1111/oik.03969
Conti L, Block S, Parepa M, Münkemüller T, Thuiller W, Acosta ATR, van Kleunen M, Dullinger S, Essl F, Dullinger I, Moser D, Klonner G, Bossdorf O, Carboni M (2018) Functional trait differences and trait plasticity mediate biotic resistance to potential plant invaders. J Ecol 106:1607–1620. https://doi.org/10.1111/1365-2745.12928
Dassonville N, Vanderhoeven S, Vanparys V, Hayez M, Gruber W, Meerts P (2008) Impacts of alien invasive plants on soil nutrients are correlated with initial site conditions in NW Europe. Oecologia 157:131–140. https://doi.org/10.1007/s00442-008-1054-6
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534. https://doi.org/10.1046/j.1365-2745.2000.00473.x
Dickson TL, Hopwood JL, Wilsey BJ (2012) Do priority effects benefit invasive plants more than native plants? An experiment with six grassland species. Biol Invasions 14:2617–2624. https://doi.org/10.1007/s10530-012-0257-2
Ellenberg H, Weber HE, Düll R, Wirth V, Werner W, Paulissen D (1991) Zeigerwerte von Pflanzen in Mitteleuropa. Scripta Geobot 18:1–248
Elton CS (1958) The Ecology of invasions by animals and plants. Methuen and Co Ltd, London, UK
Fargione J, Brown CS, Tilman D (2003) Community assembly and invasion: an experimental test of neutral versus niche processes. Proc Natl Acad Sci USA 100:8916–8920. https://doi.org/10.1073/pnas.1033107100
Ferenc V, Sheppard CS (2020) The stronger, the better – Trait hierarchy is driving alien species interaction. Oikos 129:1455–1467. https://doi.org/10.1111/oik.07338
Freckleton RP, Watkinson AR (2001) Asymmetric competition between plant species. Funct Ecol 15:615–623
Fukami T (2015) Historical contingency in community assembly: integrating niches, species pools, and priority effects. Annu Rev Ecol Evol Syst 46:1–23. https://doi.org/10.1146/annurev-ecolsys-110411-160340
Fukami T, Martijn Bezemer T, Mortimer SR, Putten WH (2005) Species divergence and trait convergence in experimental plant community assembly. Ecol Lett 8:1283–1290. https://doi.org/10.1111/j.1461-0248.2005.00829.x
Funk JL, Cleland EE, Suding KN, Zavaleta ES (2008) Restoration through reassembly: plant traits and invasion resistance. Trends Ecol Evol 23:695–703. https://doi.org/10.1016/j.tree.2008.07.013
Funk JL, Parker IM, Matzek V, Flory SL, Aschehoug ET, D’Antonio CM, Dawson W, Thomson DM, Valliere J (2020) Keys to enhancing the value of invasion ecology research for management. Biol Invasions 22:2431–2445. https://doi.org/10.1007/s10530-020-02267-9
Funk JL, Wolf AA (2016) Testing the trait-based community framework: Do functional traits predict competitive outcomes? Ecology 97:2206–2211. https://doi.org/10.1002/ecy.1484
Gentili R, Gilardelli F, Ciappetta S, Ghiani A, Citterio S (2015) Inducing competition: intensive grassland seeding to control Ambrosia artemisiifolia. Weed Res 55:278–288. https://doi.org/10.1111/wre.12143
Gerhardt F, Collinge SK (2003) Exotic plant invasions of vernal pools in the Central Valley of California, USA. J Biogeogr 30:1043–1052. https://doi.org/10.1046/j.1365-2699.2003.00911.x
Going BM, Hillerislambers J, Levine JM (2009) Abiotic and biotic resistance to grass invasion in serpentine annual plant communities. Oecologia 159:839–847. https://doi.org/10.1007/s00442-008-1264-y
Goldstein LJ, Suding KN (2014) Applying competition theory to invasion: resource impacts indicate invasion mechanisms in California shrublands. Biol Invasions 16:191–203
Grime JP (1998) Benefits of plant diversity to ecosystems: immediate, filter and founder effects. J Ecol 86:902–910
Hess MCM, Buisson E, Fontes H, Bacon L, Sabatier F, Mesléard F (2020) Giving recipient communities a greater head start and including productive species boosts early resistance to invasion. Appl Veg Sci 23:340–352. https://doi.org/10.1111/avsc.12502
Hess MCM, Mesléard F, Buisson E (2019) Priority effects: emerging principles for invasive plant species management. Ecol Eng 127:48–57. https://doi.org/10.1016/j.ecoleng.2018.11.011
Hood WG, Naiman RJ (2000) Vulnerability of riparian zones to invasion by exotic vascular plants. Plant Ecol 148:105–114. https://doi.org/10.1023/A:1009800327334
Hooper DU, Chapin FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawton JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setälä H, Symstad AJ, Vandermeer J, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75:3–35. https://doi.org/10.1890/04-0922
Hufbauer RA, Rutschmann A, Serrate B, Vermeil de Conchard H, Facon B (2013) Role of propagule pressure in colonization success: disentangling the relative importance of demographic, genetic and habitat effects. J Evol Biol 26:1691–1699. https://doi.org/10.1111/jeb.12167
Jentsch A, White P (2019) A theory of pulse dynamics and disturbance in ecology. Ecology 100:e02734. https://doi.org/10.1002/ecy.2734
Johnstone IM (1986) Plant invasion windows: a time-based classification of invasion potential. Biol Rev 61:369–394. https://doi.org/10.1111/j.1469-185X.1986.tb00659.x
Kam J, Knutson TR, Zeng F, Wittenberg AT (2018) CMIP5 model-based assessment of anthropogenic influence on highly anomalous arctic warmth during November–December 2016. Bull Am Meteor Soc 99:S34–S38. https://doi.org/10.1175/BAMS-D-17-0115.1
Kercher SM, Zedler JB (2004) Multiple disturbances accelerate invasion of reed canary grass (Phalaris arundinacea L.) in a mesocosm study. Oecologia 138:455–464. https://doi.org/10.1007/s00442-003-1453-7
Kiehl K, Kirmer A, Donath TW, Rasran L, Hölzel N (2010) Species introduction in restoration projects—Evaluation of different techniques for the establishment of semi-natural grasslands in Central and Northwestern Europe. Basic Appl Ecol 11:285–299
Knutson TR, Kam J, Zeng F, Wittenberg AT (2018) CMIP5 model-based assessment of anthropogenic influence on record global warmth during 2016. Bull Am Meteor Soc 99:S11–S15. https://doi.org/10.1175/BAMS-D-17-0104.1
Koerner SE, Avolio ML, Chang CC, Gray J, Hoover DL, Smith MD (2015) Invasibility of a mesic grassland depends on the time-scale of fluctuating resources. J Ecol 103:1538–1546. https://doi.org/10.1111/1365-2745.12479
Kunstler G, Lavergne S, Courbaud B, Thuiller W, Vieilledent G, Zimmermann NE, Kattge J, Coomes DA (2012) Competitive interactions between forest trees are driven by species’ trait hierarchy, not phylogenetic or functional similarity: implications for forest community assembly. Ecol Lett 15:831–840. https://doi.org/10.1111/j.1461-0248.2012.01803.x
Laughlin DC (2014) Applying trait-based models to achieve functional targets for theory-driven ecological restoration. Ecol Lett 17:771–784. https://doi.org/10.1111/ele.12288
Lefcheck JS (2016) Piecewisesem: piecewise structural equation modelling in r for ecology, evolution, and systematics. Methods Ecol Evol 7:573–579. https://doi.org/10.1111/2041-210X.12512
Lefcheck JS, Duffy JE (2015) Multitrophic functional diversity predicts ecosystem functioning in experimental assemblages of estuarine consumers. Ecology 96:2973–2983. https://doi.org/10.1890/14-1977.1
Levine JM, Adler PB, Yelenik SG (2004) A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7:975–989. https://doi.org/10.1111/j.1461-0248.2004.00657.x
Lindig-Cisneros R, Zedler JB (2002) Phalaris arundinacea seedling establishment: effects of canopy complexity in fen, mesocosm, and restoration experiments. Can J Bot 80:617–624. https://doi.org/10.1139/b02-042
Liu Y, Kleunen M (2017) Responses of common and rare aliens and natives to nutrient availability and fluctuations. J Ecol 105:1111–1122. https://doi.org/10.1111/1365-2745.12733
Liu Y, Zhang X, Kleunen M (2018) Increases and fluctuations in nutrient availability do not promote dominance of alien plants in synthetic communities of common natives. Funct Ecol 32:2594–2604. https://doi.org/10.1111/1365-2435.13199
Liu Y-Y, Sun Y, Müller-Schärer H, Yan R, Zhou Z-X, Wang Y-J, Yu F-H (2019) Do invasive alien plants differ from non-invasives in dominance and nitrogen uptake in response to variation of abiotic and biotic environments under global anthropogenic change? Sci Total Environ 672:634–642. https://doi.org/10.1016/j.scitotenv.2019.04.024
Lockwood JL, Cassey P, Blackburn T (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228. https://doi.org/10.1016/j.tree.2005.02.004
Lu P, Jiang B, Weiner J (2020) Crop spatial uniformity, yield and weed suppression. In: Elsevier (ed) Advances in agronomy, Elsevier, pp. 117–178
Matsubara Y, Sakai S (2016) The role of flood regime on invasive success of exotic species growing in riparian environments. Biol Invasions 18:793–808. https://doi.org/10.1007/s10530-015-1049-2
Meissen JC, Glidden AJ, Sherrard ME, Elgersma KJ, Jackson LL (2020) Seed mix design and first year management influence multifunctionality and cost-effectiveness in prairie reconstruction. Restor Ecol 28:807–816. https://doi.org/10.1111/rec.13013
Melbourne BA, Cornell HV, Davies KF, Dugaw CJ, Elmendorf S, Freestone AL, Hall RJ, Harrison S, Hastings A, Holland M, Holyoak M, Lambrinos J, Moore K, Yokomizo H (2007) Invasion in a heterogeneous world: Resistance, coexistence or hostile takeover? Ecol Lett 10:77–94. https://doi.org/10.1111/j.1461-0248.2006.00987.x
Meyer AH, Schmid B (1999) Seed dynamics and seedling establishment in the invading perennial Solidago altissima under different experimental treatments. J Ecol 87:28–41. https://doi.org/10.1046/j.1365-2745.1999.00316.x
Nemec KT, Allen CR, Helzer CJ, Wedin DA (2013) Influence of richness and seeding density on invasion resistance in experimental tallgrass prairie restorations. Ecol Restor 31:168–185. https://doi.org/10.3368/er.31.2.168
Parepa M, Fischer M, Bossdorf O (2013) Environmental variability promotes plant invasion. Nat Commun 4:1604. https://doi.org/10.1038/ncomms2632
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2021) nlme: linear and nonlinear mixed effects models https://CRAN.R-project.org/package=nlme
Reinhardt Adams C, Galatowitsch SM (2007) The transition from invasive species control to native species promotion and its dependence on seed density thresholds. Appl Veg Sci 11:131–138. https://doi.org/10.3170/2007-7-18335
Roy J, Rineau F, de Boeck HJ, Nijs I, Pütz T, Abiven S, Arnone JA, Barton CVM, Beenaerts N, Brüggemann N, Dainese M, Domisch T, Eisenhauer N, Garré S, Gebler A, Ghirardo A, Jasoni RL, Kowalchuk G, Landais D, Larsen SH, Leemans V, Le Galliard J-F, Longdoz B, Massol F, Mikkelsen TN, Niedrist G, Piel C, Ravel O, Sauze J, Schmidt A, Schnitzler J-P, Teixeira LH, Tjoelker MG, Weisser WW, Winkler B, Milcu A (2021) Ecotrons: powerful and versatile ecosystem analysers for ecology, agronomy and environmental science. Glob Change Biol 27:1387–1407. https://doi.org/10.1111/gcb.15471
Scherber C, Mwangi PN, Schmitz M, Scherer-Lorenzen M, Bessler H, Engels C, Eisenhauer N, Migunova VD, Scheu S, Weisser WW, Schulze E-D, Schmid B (2010) Biodiversity and belowground interactions mediate community invasion resistance against a tall herb invader. J Plant Ecol 3:99–108. https://doi.org/10.1093/jpe/rtq003
Schiermeier Q (2018) Droughts, heatwaves and floods: how to tell when climate change is to blame. Nature 560:20–22. https://doi.org/10.1038/d41586-018-05849-9
Schuster MJ, Wragg PD, Reich PB (2018) Using revegetation to suppress invasive plants in grasslands and forests. J Appl Ecol 55:2362–2373. https://doi.org/10.1111/1365-2664.13195
Sheley RL, James J (2010) Resistance of native plant functional groups to invasion by Medusahead (Taeniatherum caput-medusae). Invasive Plant Sci Manag 3:294–300. https://doi.org/10.1614/IPSM-D-09-00056.1
Shipley B (2000) A new inferential test for path models based on directed acyclic graphs. Struct Equ Modeling 7:206–218. https://doi.org/10.1207/S15328007SEM0702_4
Shipley B (2009) Confirmatory path analysis in a generalized multilevel context. Ecology 90:363–368
Simberloff D (2009) The role of propagule pressure in biological invasions. Annu Rev Ecol Evol Syst 40:81–102. https://doi.org/10.1146/annurev.ecolsys.110308.120304
Sorte CJB, Ibáñez I, Blumenthal DM, Molinari NA, Miller LP, Grosholz ED, Diez JM, D’Antonio CM, Olden JD, Jones SJ, Dukes JS (2013) Poised to prosper? A cross-system comparison of climate change effects on native and non-native species performance. Ecol Lett 16:261–270. https://doi.org/10.1111/ele.12017
Török P, Deák B, Vida E, Valkó O, Lengyel S, Tóthmérész B (2010) Restoring grassland biodiversity: sowing low-diversity seed mixtures can lead to rapid favourable changes. Biol Cons 143:806–812. https://doi.org/10.1016/j.biocon.2009.12.024
Torres A, Alarcón P, Rodríguez-Cabal M, Nuñez M (2018) Secondary invasions hinder the recovery of native communities after the removal of nonnative pines along a precipitation gradient in Patagonia. Forests 9:394. https://doi.org/10.3390/f9070394
Viana DS, Cid B, Figuerola J, Santamaría L (2016) Disentangling the roles of diversity resistance and priority effects in community assembly. Oecologia 182:865–875. https://doi.org/10.1007/s00442-016-3715-1
Vitousek PM, Aber JD, Howarth RW, Likens GE, Matson PA, Schindler DW, Schlesinger WH, Tilman DG (1997) Human alteration of the global nitrogen cycle: sources and consequences. Ecol Appl 7:737–750
von Gillhaussen P, Rascher U, Jablonowski ND, Plückers C, Beierkuhnlein C, Temperton VM (2014) Priority effects of time of arrival of plant functional groups override sowing interval or density effects: a grassland experiment. PLoS ONE 9:e86906. https://doi.org/10.1371/journal.pone.0086906
Von Holle B, Simberloff D (2005) Ecological resistance to biological invasion overwhelmed by propagule pressure. Ecology 86:3212–3218. https://doi.org/10.1890/05-0427
Walker EA, Conradi T, Meimberg H, Kollmann J (2015) Seed selection for grassland restoration: competitive effect of a dominant grass is mediated by seed source and nutrient availability. Restor Ecol 23:261–267. https://doi.org/10.1111/rec.12171
Weber E, Jakobs G (2005) Biological flora of central Europe: Solidago gigantea Aiton. Flora—Morphol, Distrib Func Ecol Plants 200:109–118. https://doi.org/10.1016/j.flora.2004.09.001
Weidlich EWA, Nelson CR, Maron JL, Callaway RM, Delory BM, Temperton VM (2021) Priority effects and ecological restoration. Restor Ecol. https://doi.org/10.1111/rec.13317
Weiner J, Griepentrog H-W, Kristensen L (2001) Suppression of weeds by spring wheat Triticum aestivum increases with crop density and spatial uniformity. J Appl Ecol 38:784–790. https://doi.org/10.1046/j.1365-2664.2001.00634.x
Yannelli FA, Koch C, Jeschke JM, Kollmann J (2017a) Limiting similarity and Darwin’s naturalization hypothesis: understanding the drivers of biotic resistance against invasive plant species. Oecologia 183:775–784. https://doi.org/10.1007/s00442-016-3798-8
Yannelli FA, Hughes P, Kollmann J (2017b) Preventing plant invasions at early stages of revegetation: the role of limiting similarity in seed size and seed density. Ecol Eng 100:286–290. https://doi.org/10.1016/j.ecoleng.2016.12.001
Yannelli FA, Karrer G, Hall R, Kollmann J, Heger T (2018) Seed density is more effective than multi-trait limiting similarity in controlling grassland resistance against plant invasions in mesocosms. Appl Veg Sci 21:411–418. https://doi.org/10.1111/avsc.12373
Yannelli FA, MacLaren C, Kollmann J (2020) Moving away from limiting similarity during restoration: timing of arrival and native biomass are better proxies of invasion suppression in grassland communities. Front Ecol Evol 8:128. https://doi.org/10.3389/fevo.2020.00238
Young SL, Barney JN, Kyser GB, Jones TS, DiTomaso JM (2009) Functionally similar species confer greater resistance to invasion: Implications for grassland restoration. Restor Ecol 17:884–892. https://doi.org/10.1111/j.1526-100X.2008.00448.x
Acknowledgements
We appreciate the contributions of Holger Paetsch, Jie Si Ma, Robin Fahle, Andrea Frank, and Johannes Prifling during the experimental development of the study and the pre-processing of data. The staff at Greenhouse Laboratory Center Dürnast provided continuous technical support during the experiments. The technical and scientific staff at the TUMmesa facility contributed to the implementation of the experiments. Funding for conducting the research was provided by DFG (INST 95/1184- FUGG) the LandKlif Project by StMWK (F.7-F5121.14.2.3/14/9). We would like to thank all reviewers and the Associate Editor for their valuable comments and important contributions that improved this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. SR-B conducted her research in the frame of the joint project LandKlif, funded by the Bavarian Ministry of Science and the Arts via the Bavarian Climate Research Network (bayklif; F.7-F5121.14.2.3/14/9) and DFG (INST 95/1184- FUGG) establishing TUMmesa.
Author information
Authors and Affiliations
Contributions
LHT and JK conceived and designed the experiments; SR-B and LHT conducted the experiments; SR-B and LHT collected, analyzed, and interpreted the data; SR-B and LHT led the writing and editing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rojas-Botero, S., Kollmann, J. & Teixeira, L.H. Competitive trait hierarchies of native communities and invasive propagule pressure consistently predict invasion success during grassland establishment. Biol Invasions 24, 107–122 (2022). https://doi.org/10.1007/s10530-021-02630-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-021-02630-4