Abstract
Alien species in urban areas have a large effect on overall species diversity. A suitable metric of flora’s response to environmental change is functional diversity (FD) that refers to the multivariate space of species’ trait compositions, reflecting their ecological niches. We studied how FD changed over 320 years of urbanization in the city of Halle (Saale), Germany. Selected functional traits (related to stress-tolerance, reproduction, competitiveness and phenology) were examined for the difference in FD between native and alien plant species, the latter specifically for archaeophytes, neophytes and invasive species. Functional diversity for each trait was calculated using Rao’s Q index followed by a linear model to test for changes in Rao’s Q over time between the groups. Over the 320 years, overall FD remained constant despite species turnover, but FD significantly increased for neophytes and invasive species compared to native species. Plant height was the only trait showing increase in FD as main effect, while for the other traits examined FD decreased over time. Considering invasive species separately, the majority of traits exhibit a significant increase in FD except for seed mass where it decreased. Finally, FD of multiple functional traits combined decreased over time. This can be due to homogenization of functional trait between native and alien species, as a consequence of habitats becoming more similar and subsequent habitat filtering. Our results demonstrate that during the last three centuries, urbanization influenced plant FD in various ways and may contribute to future uniformity of urban floras and greater invasiveness.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
“There was no doubt about it: the City was the culmination of man’s mastery over the environment”, Isaac Asimov wrote in his 1953 novel The Caves of Steel. The major factors behind the changes in species composition are human activities, particularly as their settlements expand and many habitats change or disappear. Processes associated with urbanization are mainly related to human population growth and increased area of infrastructure built for living, industry, and traffic (Sukopp 2008; Hua et al. 2017). These, further lead to habitat loss and fragmentation (Kowarik 1995a; Syphard et al. 2011), pollution, changes in climate (temperature, moisture), hydrological systems (Paul and Meyer 2001), and soil (Kalnay and Cai 2003; Trusilova et al. 2008; Song et al. 2014). Urbanization leads to habitats becoming more homogenized and thus many specialist species get lost (loss of natural or specific anthropogenic habitats) to the benefit of generalists and species adapted to urban conditions (Williams et al. 2015). In parallel, species mobility (dispersal ability) plays an important factor in colonizing new urban habitats (Concepción et al. 2015), where those species that are highly mobile respond positively to increases in the proportion of urban habitats. Additionally, alien plant species are very abundant in cities, to which they were mostly deliberately introduced, and these urban and suburban areas provide a range of different local conditions suitable for a variety of species (Lippe and Kowarik 2008). Humans benefit from urban plant species that provide multiple ecosystem services in cities, such as air quality improvement, noise reduction (Bolund and Hunhammar 1999), climate regulation, water regulation and storage, aesthetics and recreation (Gómez-Baggethun and Barton 2013; Potgieter et al. 2017) or food provision (via urban gardening; Barthel et al. 2014). Further, those alien species that become invasive can impact ecosystem services and disservices by affecting local diversity, causing health issues (i.e. allergic reactions) or damaging infrastructure (Shackleton et al. 2018; Vaz et al. 2017).
Biodiversity has been widely studied using many different measures, such as species abundance and species richness (Díaz et al. 2006). However, these measures might not be the most appropriate proxy to determine the effect of biodiversity on ecosystem processes (and vice versa). For example, studying the effect of species abundance on ecosystem processes may divert from the possible impact rare species might have on ecosystem functioning. Further, not only total species richness but also the number of species responding differently to environmental changes is crucial for understanding ecosystem processes. To this end, taking into account species’ functional traits via calculating functional diversity (FD) is considered to be more appropriate (Mcgill et al. 2006; Villéger et al. 2008). Functional diversity accounts for “the range and value of those species and organismal traits that influence ecosystem functioning” (Tilman 2001) and several FD indices relate this to the distribution of species in niche space (e.g. functional richness, evenness, divergence; Mason et al. 2005). Additionally, specific functional traits of plant species can explain different ecosystem processes. For example, leaf traits can be related to drought tolerance and competition, seed traits to life strategies, dispersal and establishment, and root traits or species height help in understanding invasibility and community competitiveness (Laughlin 2014; Funk et al. 2017). Further, SLA (specific leaf area) underlines strategies of nutrient acquisition and competition, while pollination type and flowering period relate to species dispersal. Using functional traits (rather than only species richness or abundance) is therefore particularly important in communities with an abundance of alien and invasive species because higher FD indicates a higher resistance to invasion (Funk et al. 2008).
The numbers of alien plant species introduced into Europe steadily rose from the fifteenth century with a more intensive increase in the nineteenth century (Pyšek et al. 2009). Many of them have been introduced to cities due to their prospective provision of ecosystem services, and since then, many have spread into natural areas where they modify ecosystem properties (Potgieter et al. 2019). Additionally, it is expected that alien plant invasions will intensify in the future depending on socioeconomic scenarios (particularly in arable and urban landscapes; Chytrý et al. 2012), and due to the phenomenon of invasion debt (Essl et al. 2011). Thus, having a record of invasive species through time (decades or centuries) presents a unique opportunity and can be important for studying invasiveness and assessing future trends. So far, comparisons of historical urban floras with the present state were made for Leipzig (1867–1989 Klotz and Gutte 1992; Scholz 2008), Halle/Saale (1848–1983, Klotz 1984; 1687–2005, Knapp et al. 2010), Zürich (1839–1998, Landolt 2000), Plzeň (1880–1990s Chocholoušková and Pyšek 2003; Pyšek et al. 2004a), Brussels (Godefroid 2001), Turnhout (Van der Veken et al. 2004), Bologna (1894–2018, Salinitro et al. 2019), New York City (DeCandido et al. 2004), Indianapolis (Dolan et al. 2011), and Adelaide (Tait et al. 2005), as well as for selected parts of a city (e.g. Pelham Bay Park, New York City (DeCandido 2004), Central Park New York City (DeCandido et al. 2007)). Still, none of these studies focused on functional diversity.
In our paper, we followed the classification of alien species based on their residence time in Germany, including archaeophytes (alien species introduced before 1500 CE), and neophytes (introduced after 1500 CE) with the addition of invasive species (invasive neophytes; see (Richardson et al. 2000; Pyšek et al. 2004b) for definitions). We studied how functional diversity changes in the city of Halle (Saale), Germany. We selected relevant functional traits to examine the difference in functional diversity for native and alien plant species. Specifically, we studied: (1) functional diversity changes occurring during a 320-year period in the urban setting; (2) differences in the trajectories of functional diversity between native species and the three groups of alien species defined above, representing different stages of the invasion process; (3) functional diversity changes for specific functional traits related to urbanization processes (e.g. stress, competitiveness, and ecophysiology). Determining which plant groups (natives and alien) and functional traits benefit and which ones are disadvantaged by urbanization-related processes is important for understanding the mechanisms shaping the composition of urban floras, and for predicting trends in the future as the human population continues to move from rural to urban areas (DESA 2019).
Materials and methods
Study area
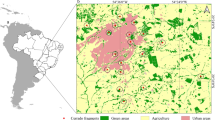
The city of Halle (Saale) is located in central Germany (state of Saxony-Anhalt; Fig. 1b), east of the Harz Mountains (latitude of 51° 28′ N; longitude 11° 58′ E) with a mean elevation of 87 m a.s.l. (range 70–140 m; (Stolle and Klotz 2004). The city is located in the rain shadow of the Harz Mountains, with mean annual precipitation of only 497 mm and a mean annual temperature of 9.1 °C. Halle is traversed by the river Saale (for 27 km) which divides into several branches, forming small islets. Soils are highly heterogeneous, and the non-residential areas within the city predominantly consist of agricultural/horticultural land (~ 21%), forests (~ 10%), industrial and commercial areas (~ 9%), herbaceous flora/grasslands (~ 8%) and green urban areas (green and open spaces in the built-up area, 3%; Arnold et al. 2018). The original natural vegetation types in the area today covered by Halle (Saale) were mainly forests: alder, alder-ash, ash-elm, willow-poplar, oak-hornbeam, dry forests (Stolle and Klotz 2004). The current vascular flora comprises approximately 1400 species (Stolle and Klotz 2004). Areas for nature conservation mainly comprise alluvial forests and meadows as well as dry lawns on porphyritic rock (rhyolite). Halle (Saale) covers an area of about 135 km2 and a population of 240 900 inhabitants (in 2019, Fig. 1a). There was a steady increase (since the beginning of recording in 1871) in population until the late 1980ies and a pronounced drop in the 1990ies due to the political changes in Germany. In recent years, population numbers stabilized.
Historical data
First known records of Halle date from 806. The city started to extensively develop during the twelfth century. During the twentieth century, Halle expanded by incorporating several municipalities. In our analysis, we used historical floristic data on vascular plant species occurrences for the period 1687–2008. Historical records were related to the area within the present administrative borders of Halle with the help of site references given in historical publications. Historical data was composed of published species records performed by over 20 botanists since the late seventeenth century, as well as manuscripts and herbarium records (Table 1). The accuracy of species occurrences was evaluated by Jens Stolle, a competent local botanist who made the following modifications. Species were excluded if (a) it is unlikely that the species occurred in the study area; (b) they occurred casually for a very short period; (c) they were merged into “superspecies”. Further, we included rare species with low dispersal capacity if they occur in Halle today but were not recorded by earlier botanists, assuming that these species must have been overlooked in former times (for further details see Supplementary material S1a and Knapp et al. 2010).
We determined the total number of species and the number of unique species of every status group per time step (status groups categorized according to BiolFlor database, (Klotz et al. 2002), where time steps were distinguished based on publication dates of historical records (Fig. 2). We further distinguished invasive neophytes according to their negative impact, following the KORINA (The Coordination Centre for Invasive Plants in protected areas of Saxony-Anhalt) blacklist of neophytes in Saxony-Anhalt (http://www.korina.info).
Trait data
The traits were obtained from the BiolFlor and LEDA databases (Table 2). Standards of trait measurement are explained in Kleyer et al. (2008) for LEDA and Klotz et al. (2002) for BiolFlor. Regarding alien status, plant species were divided into native, archaeophytes, neophytes and invasive neophytes (Kühn et al. 2004; http://www.ufz.de/biolflor).
Data analysis
All data analyses were performed using R, version 3.6.1 (R Core Team 2017). Functional diversity was calculated for all selected functional traits (listed in Table 2) using the occurrences of every species. Occurrence matrices included the presence and absence of every species for each time step, while functional trait matrices included the categorical and standardized (zero mean, unit standard deviation) numerical trait values for every species. Among the various indices for calculating FD, we chose RaoQ (Rao’s quadratic entropy; Botta‐Dukát 2005) as one of the most versatile and unbiased metrics for each trait per time step and status (Ricotta and Moretti 2011), using function dbFD() in the FD package (Laliberté and Shipley 2011). We selected multivariate RaoQ because it is suitable for multiple traits and it allows a mixture of categorical and continuous variables (Schleuter et al. 2010). RaoQ accounts for both functional richness and divergence (Mason and Bello 2013) as it includes species occurrences and the pairwise functional differences between species (Botta‐Dukát 2005). RaoQ values increase with dissimilarity between traits of species and present abundance-weighted differences between species using their functional traits (Pavoine 2020.). We checked for the independence of RaoQ among periods (temporal autocorrelation) using the correlog() function (ncf package, Bjornstad and Cai 2019). FD indices were not significantly autocorrelated and thus we did not need to account for temporal autocorrelation in the model.
To test whether FD significantly changes over time for each status group and for which functional traits, we used linear regression with RaoQ as a response and status, time, all of the traits and interactions of the previous three groups as predictors. We also performed a test for normality for the transformed and untransformed dependent variable. Untransformed RaoQ values yielded the best model fits and were used in the analysis. For model selection, we used a multi-model inference approach (Burnham and Anderson 2002), employing the function dredge (package MuMIn, Barton 2015). For multiple posthoc comparisons between status groups, we used estimated marginal mean of linear trends (function emtrends from the package emmeans; Lenth et al. 2018), where all possible pairwise contrasts were tested. We preformed post-hoc test (Tukey HSD pairwise comparisons for variable), comparing all 13 functional traits (’stats’ package, R Core Team 2017). For graphical visualization, we used packages ggplot2 (Wickham et al. 2016) and maps (Becker et al. 2018).
Results
Functional diversity for different status groups
The recorded species numbers across time steps are: native 833, archaeophytes 141, neophytes 13, and invasive neophytes 40. Functional diversity (RaoQ) values range from 4 × 10−7 to 3.104. Average values of functional diversity (RaoQ) were highest for native species (Fig. 3), followed by archaeophytes, neophytes, with the lowest average functional diversity for invasive neophytes. The best model included RaoQ ~ Status + Time + Trait + Status:Time + Status:Trait + Time:Trait, and AICc weight of 0.963 which means that it is the most likely model of the ones tested; see S2 in Supplementary material). The second-best model had AICc weight 0.019 (with a delta AICc of 7.81). The best linear model had a high proportion of variation explained (78%), with FD of non-invasive and invasive neophytes differing significantly from native species (Table 3).
Comparison of functional diversity (RaoQ) values (y-axis) for different status groups (native and alien plant species, x-axis). In the box plots, the black line presents median, vertical lines above and below the box indicate the 10th and 90th percentiles. Points above the lines indicate outliers outside the 10th and 90th percentiles
Functional diversity changes over time for different status groups
The functional diversity (RaoQ) of native species did not significantly change over the seven time periods (Table 3b). Compared to native species, FD of neophytes and invasive neophytes increased significantly over time (Table 3d, Fig. 4). Pairwise comparisons showed that there was no significant difference between native species and archaeophytes or between alien groups (Table 4).
Pairwise interaction plot (based on the fitted model) for the estimated marginal means of linear trends. The figure shows linear predictions of the response variable (RaoQ) depending on the predictor variable (time) for native species (black) and alien plant species (archaeophytes in yellow, neophytes in green and invasive neophytes in blue) at different stages of the invasion process with the 95% confidence interval
Changes in FD for different traits for different status groups and over time
Except for the negative relationship of functional diversity (RaoQ) with height, there were no significant relationships for other functional traits using default settings, i.e. duration of flowering as reference trait (Table 3c). Performing a Tukey HSD post-hoc test, comparing all the traits, showed that almost two thirds of the 78 pairs of functional diversity values (RaoQ) of traits differ significantly from each other (see for details in Supplementary material S4). Compared to native species, archaeophytes did not show any significant difference in FD related to traits. Functional diversity (RaoQ) values for neophytes (compared to native species) significantly decreased for height and seed mass (Table 3e). Invasive neophytes showed a significant increase in FD, compared to natives, for the duration of flowering, life form, life span, pollination vector, SLA, and storage organs, and a significant decrease for seed mass (Table 3e).
Height was the only functional trait significantly increasing over time compared to the reference trait (beginning of flowering), while leaf anatomy, life form, life span, ploidy, pollination vector, and storage organs significantly decreased over time (Table 3f, Fig. 5).
Discussion
The species richness of the urban flora of Halle (Saale) increased over three centuries, however FD did not change over time. This suggests that on average species that colonized urban habitats as newcomers and those that went extinct were neither more nor less similar to the resident species. Compared with rural areas, urban areas in Europe are often hotspots of high plant diversity and particularly alien plant species (Haeupler 1974; Kühn et al. 2004; Kowarik 2011). We recorded an increase in native species (that are most likely locally introduced from surrounding regions) and an influx of neophytes, while there was a decrease in archaeophytes, probably as a result of arable land reduction on the account of urbanization (Jarošik et al. 2011). The disappearance of distinctive types of habitats resulted in the loss of some native species, while others might have been introduced or immigrated into the city because of novel environmental conditions. Incoming native and alien species, as well as species remaining present across all time steps, possess traits that make them well adapted to these conditions (the strong filtering effect of urban environmental conditions; Williams et al. 2009; Aronson et al. 2016). The urbanization process begins with habitat transformation via loss and gain of novel habitats. Compared to rural areas, cities contain higher diversity of habitats, communities, and species (Sukopp 1998). For example, under an environmental gradient (e.g. temperature, precipitation, soil characteristics) species occupying a habitat will have a suite of functional traits that allow them to exist along the whole gradient represented in that habitat. In the case of environmental changes associated with urbanization, functional trait values might shift along the corresponding environmental gradient and thus some trait states can be lost or gained (Williams et al. 2015). As the proportion of urban areas increased over time, semi-natural, nutrient-poor, and wet habitats were replaced by those habitats that were dry and nutrient-rich (typical for urban conditions; Pyšek et al. 2004b; Kowarik 2011). These novel habitats still foster a similar amount of variation of functional trait states as before, however, trait values or trait states change due to the presence of new species with different trait values. Accordingly, despite species turnover, overall FD does not change because new traits (which replaced previous traits) occupy a different part of the environmental gradient. For example, in Germany, novel ecosystems emerging from urbanization proved to be beneficial for alien plants (such as neophytes) but lead to homogenization in natives and archaeophytes (Kühn and Klotz 2006). Novel ecosystems are those ecosystems which are originally modified by humans and exhibit historically different abiotic and biotic properties (Hobbs et al. 2006). These novel environments influence interaction between species (or individuals) or directly affect organisms (Heger et al. 2019). Knapp et al. (2010) showed that trait composition in Halle shifted while here we show that variation stayed the same which might be due to loss and gain of environmental conditions at different ends of the cline. The results hence may imply that urbanization processes do not directly lead to a decrease in FD but rather a functional shift, by providing conditions for the same amount of functional variation as previous habitats. Further, habitats get more fragmented which leads to species extinction and introduction (emerging of new habitats and edge effect instigate introduction of new species; Fahrig 2003), as well as narrow functional trait range.
Environmental changes and human preferences are shaping urban flora (Williams et al. 2009), where functional traits either get confined or shift. Worldwide, 55% of people live in urban areas (DESA 2019). The level and rate of urbanization might influence species and functional diversity. Studies so far showed that moderate urbanization promotes species diversity (McKinney 2008), yet across the globe urban floras tend to share species, making urbanization a driver of biotic homogenization (McKinney 2006; Kühn and Klotz 2006). As the human population continues to rise, we can expect that the preference for certain species (Marco et al. 2008) will become more pronounced (e.g. preference for woody and ornamental species) and functionally more similar communities will appear. For example, the size and colour of flowers and leaves as well as the selection of traits for ecological reasons such as tolerance to drought or “exoticness” of cultivated floras were shown to be important in urban landscapes (Kendal et al. 2012). Rapidly growing tree species introduced in the cities may alter ecosystem properties, e.g., carbon sequestration, erosion, water cycle, and soil organic matter (Castro‐Díez et al. 2019). Further, in the urban areas of the United States, Dolan et al. (2011) found species turnover (loss of species preferring natural habitat, i.e., wetlands) to correlate with a decrease in native and an increase in alien species during the last 70 years. The increase in species diversity recorded in Halle can be attributed to an increase in the number of native species (immigrating from the regional species pool to Halle) and alien species (mainly neophytes) which were able to establish and spread after introduction (e.g. to abandoned urban areas; Bello et al. 2006).
Changes in functional diversity for different status groups
The increase in overall neophyte species richness and in that of invasive neophytes parallels the increase in their FD between 1687 and 2008, although FD for both groups remains lower compared to native species. Across all time steps, native species have both the highest species numbers and FDs while for invasive neophytes we recorded an increase in FD for multiple traits. Archaeophytes and non-invasive neophytes showed no difference or decrease in FD compared to the native group.
First, alien species responded with an increase in FD to urbanization, as they may be less limited by typical urban environmental conditions (fragementation, urban heat island etc.) than native species, as many alien species are well adapated to these conditions and many of them possess high dispersal ability or are very fecund (Williams et al. 2009). The loss of natural habitats such as bogs, the gain of typical urban habitats (e.g. roadside vegetation, parks, modern residential areas, industrial estates) and competition between native and alien species over the last three centuries is likely to be the driver behind 22% species turnover in Halle (presence/absence of species between different time periods, previously reported by Knapp et al. (2010) as well as changes in FD.
Second, functional trait values for both continuous and categorical traits are getting similar over time which can be associated with environmental filtering (due to fragmentation or destruction of habitats, pollution, environmental stressors) and the limited number of potential niches. For example, land-use changes (e.g. transformation of natural or agricultural areas to urban) may promote the expansion of previously non-dominant native or alien species [e.g. woody species; (Díaz et al. 2007)].
Third, because of the marked increase in the number of established neophytes in Halle during the last centuries (and especially during the 1900s, where neophytes increased from 38 to 127 species, Fig. 2), there is a tendency for trait homogenization and communities are getting more functionally similar.
Alien species were shown to promote the homogenization of urban floras as themselves, being the drivers of change, or they adapt to and benefit from novel conditions and thus indicate habitat homogenization resulting from a process in which they act as passengers (MacDougall and Turkington 2005; HilleRisLambers et al. 2010). Species as drivers can become dominant (successful alien and native generalists) and alter environmental conditions. However, if the habitat conditions become more homogenized, species that are introduced and become naturalized have more similar functional traits, contrary to invasive species tend to be successful due to their dissimilarities (e.g. greater height compared to natives, archaeophytes and non-invasive neophytes; Divíšek et al. 2018). In Halle, both overall FD and trait FD remained constant over time for natives and species introduced more than five centuries ago (i.e. archaeophytes). Due to their long residence time, these alien species tend to stay functionally similar and thrive in homogenous habitats such as arable fields (Kühn and Klotz 2006).
Functional diversity of individual functional traits
As we observed a decrease in FD for many of the traits investigated, we can expect further filtering of species in the future and uniformity for traits such as life form, pollination vector, or life span. Our analysis demonstrated shifts in certain traits over time (counts of trait states per categorical trait and time step shown in Supplementary material S3), such as phenology expressed as the beginning of flowering. However, focusing on the FD for specific groups, only invasive neophytes showed an increase in half of the studied traits, while archaeophytes and non-invasive neophytes did not differ from natives. As population growth and urbanization intensified in Halle from the seventeenth century onward, these developments affected species richness, composition, and functional traits. These effects are significant mainly because the changes due to urbanization are dramatic (great changes in the relatively short period) and usually encompass large areas (McKinney 2006). For most of the traits a meta-analysis by Williams et al. (2015) did not find consistent responses to disturbances in urban areas, with the exceptions of increasing values of plant height and seed mass. However, we found that height was the only trait showing an increase in FD over time and this might be because urban areas can promote a wide range of different plant heights. Over the last 5000 years, forest cover decreased, and forests were particularly reduced in the eighteenth century (at the beginning of our study period). Prussians started reforestation of the area in the late nineteenth century (Albrecht et al. 1993). This indicates that during the first time steps only species with specific heights were dominant (either tall or shorter species) and in proceeding periods (following replanting of the forest) we can observe an increase in height heterogeneity. Additionally, a wide range of heights in the urban area can be attributed to habitat transformations where smaller and short-lived ruderal species (growing along roads, railway or on brownfields) can increase in abundance, and to competition as taller species can be successful in competing with shorter species and human preference and cultivation. Taller species (trees and shrubs) are introduced to parks and gardens, and some of these species escape and establish—this process takes decades to centuries, depending on species (Kowarik 1995b). Since cities provide different types of habitats many species can grow spontaneously and colonize these areas.
Further, we found a decrease in FD for life form and life span over time, which might be related to habitat loss and environmental changes as a result of urbanization. Extinction of many species which leads to homogenization of functional traits is associated with the disappearance of specific habitats (Pykälä 2019) and conditions (e.g. bogs, wet meadows; Knapp et al. 2010) which lead to loss of respective traits. Furthermore, many phanerophytes (Table 2) are successful in urban areas since they are favoured for their ornamental value and often cultivated in parks and gardens. Certain life forms i.e. therophytes (abundant in the cities due to high soil fertility and habitats with high disturbance, Table 2) often compete with other groups.
Functional diversity of SLA (specific leaf area) did not change over time, possibly because extreme values (both low and high) of SLA may prove to be beneficial in cities (Thompson and McCarthy 2008). Climatic limitations are particularly favourable for plant species characterized by idiosyncratic trait states (or extreme values) rather than facilitating heterogeneity for most functional traits. For example, higher temperature and aridity (characteristic for urban areas and aridity particularly prominent in Halle) promotes a decrease of SLA, while high nutrient availability in the soil leads to an increase in SLA values. Changes in precipitation primarily affect species with different leaf types. Halle’s dry climate promotes plant species adapted to drought, i.e. species with scleromorphic or mesomorphic leaf types are prevailing over other types. Further, urban areas with lower air moisture are advantageous for wind-pollinated species, considering that under these conditions chances of pollen reaching flower stigma are higher (Knapp 2010). Genetic traits (i.e. ploidy level) may have the potential in explaining species establishment and colonization of new habitats. Polyploidy (high ploidy levels) is an important trait of alien and invasive species, affecting the probability of invasion success (Te Beest et al. 2012) and they might be more flexible and able to grow in different habitats (genetic variability leads to adaptation under new environmental conditions; Winter et al. 2008). Winter et al. (2009) showed that homogenization resulted from losing native species with unique traits and gaining alien and native generalist species. Functional homogenization can be further promoted in disturbed urban areas, as generalist species increase in numbers and potentially replace specialist species.
Our results showed that over the past 320 years, functional diversity increased for invasive and non-invasive neophytes, while overall, it remained constant. Parallel to the development of an urban area, functional diversity of native and alien species homogenized for the majority of traits investigated. As worldwide, many cities are growing, outcomes of studying historical data can be widely applicable. Further, integrating historical data and environmental parameters would provide a rigorous representation of past floras and support the prediction of future biodiversity changes due to urbanization.
Data availability statement
The traits were obtained from the BiolFlor (http://www.ufz.de/biolflor) and LEDA (https://uol.de/en/landeco/research/leda/data-files) databases. List of invasive species for Saxony-Anhalt was obtained from KORINA project (http://www.korina.info). The list of species used here is available as supporting information to Knapp et al. (2017), J. Appl. Ecol. 54 (4), 1152–1160 at https://doi.org/10.1111/1365-2664.12826.
References
Albrecht T, Buschendorf J, Drechsler K, Geissler H, Gluch W, Gnielka R, Grosse E, Kaufmann D, Klotz S, Körnig G, Krumbiegel G, Kugler H, Piechocki W, Schauer W, Schmidt V, Schwarze-Neuss E, Utech L, Weidlich M (1993) Die Dölauer Heide - Waldidylle in Großstadtnähe. Magistrat der Stadt Halle, Dezernat Umwelt- und Naturschutz
Arnold J, Kleemann J, Fürst C (2018) A differentiated spatial assessment of urban ecosystem services based on land use data in halle. Germany Land 7:101. https://doi.org/10.3390/land7030101
Aronson MFJ, Nilon CH, Lepczyk CA et al (2016) Hierarchical filters determine community assembly of urban species pools. Ecology 97:2952–2963. https://doi.org/10.1002/ecy.1535
Barthel S, Parker J, Folke C, Colding J (2014) Urban Gardens: pockets of social-ecological memory. In: Tidball KG, Krasny ME (eds) Greening in the red zone: disaster, resilience and community greening. Springer, Netherlands, pp 145–158
Barton K, MK B (2015) Package “MuMIn”
Becker RA, Wilks AR, Brownrigg R et al (2018) maps: Draw geographical maps. R package version 3.3. 0. The Comprehensive R Archive Network (CRAN)
Bello FD, Lepš J, Sebastià M-T (2006) Variations in species and functional plant diversity along climatic and grazing gradients. Ecography 29:801–810. https://doi.org/10.1111/j.2006.0906-7590.04683.x
Bjornstad ON, Cai J (2019) Package ‘ncf’: spatial covariance functions. R package version 1.2–8
Bolund P, Hunhammar S (1999) Ecosystem services in urban areas. Ecol Econ 29:293–301. https://doi.org/10.1016/S0921-8009(99)00013-0
Botta-Dukát Z (2005) Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. J Veg Sci 16:533–540. https://doi.org/10.1111/j.1654-1103.2005.tb02393.x
Burnham KP, Anderson DR (2002) Model Selection and multimodel inference, 2nd edn. Springer, New York, pp 49–97
Castro-Díez P, Vaz AS, Silva JS et al (2019) Global effects of non-native tree species on multiple ecosystem services. Biol Rev 94:1477–1501. https://doi.org/10.1111/brv.12511
Chocholoušková Z, Pyšek P (2003) Changes in composition and structure of urban flora over 120 years: a case study of the city of Plzeň. Flora-Morphol Distrib Funct Ecol Plants 198:366–376
Chytrý M, Wild J, Pyšek P et al (2012) Projecting trends in plant invasions in Europe under different scenarios of future land-use change. Glob Ecol Biogeogr 21:75–87. https://doi.org/10.1111/j.1466-8238.2010.00573.x
Concepción ED, Moretti M, Altermatt F et al (2015) Impacts of urbanisation on biodiversity: the role of species mobility, degree of specialisation and spatial scale. Oikos 124:1571–1582. https://doi.org/10.1111/oik.02166
DeCandido R (2004) Recent changes in plant species diversity in urban Pelham Bay Park, 1947–1998. Biol Cons 120:129–136
DeCandido R, Calvanese N, Alvarez RV et al (2007) The naturally occurring historical and extant flora of Central Park, New York City, New York 1857–2007. J Torrey Bot Soc 134:552–569
DeCandido R, Muir AA, Gargiullo MB (2004) A first approximation of the historical and extant vascular flora of New York City: implications for native plant species conservation. J Torrey Bot Soc 243–251
DESA U (2019) United Nations, Department of Economic and Social Affairs, Population Division. World Population Prospects 2019: Highlights
Díaz S, Fargione J, Iii FSC, Tilman D (2006) Biodiversity loss threatens human well-being. PLoS Biol 4:e277. https://doi.org/10.1371/journal.pbio.0040277
Díaz S, Lavorel S, de Bello F et al (2007) Incorporating plant functional diversity effects in ecosystem service assessments. PNAS 104:20684–20689. https://doi.org/10.1073/pnas.0704716104
Divíšek J, Chytrý M, Beckage B et al (2018) Similarity of introduced plant species to native ones facilitates naturalization, but differences enhance invasion success. Nat Commun 9:4631. https://doi.org/10.1038/s41467-018-06995-4
Dolan RW, Moore ME, Stephens JD (2011) Documenting effects of urbanization on flora using herbarium records. J Ecol 99:1055–1062. https://doi.org/10.1111/j.1365-2745.2011.01820.x
Essl F, Dullinger S, Rabitsch W et al (2011) Socioeconomic legacy yields an invasion debt. Proc Natl Acad Sci 108:203–207
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Funk JL, Cleland EE, Suding KN, Zavaleta ES (2008) Restoration through reassembly: plant traits and invasion resistance. Trends Ecol Evol 23:695–703. https://doi.org/10.1016/j.tree.2008.07.013
Funk JL, Larson JE, Ames GM et al (2017) Revisiting the Holy Grail: using plant functional traits to understand ecological processes: plant functional traits. Biol Rev 92:1156–1173. https://doi.org/10.1111/brv.12275
Godefroid S (2001) Temporal analysis of the Brussels flora as indicator for changing environmental quality. Landsc Urban Plan 52:203–224
Gómez-Baggethun E, Barton DN (2013) Classifying and valuing ecosystem services for urban planning. Ecol Econ 86:235–245. https://doi.org/10.1016/j.ecolecon.2012.08.019
Haeupler H (1974). Statistische Auswertung von Punktrasterkarten der Gefäßpflanzenflora Süd-Niedersachsens. Goltze
Heger T, Bernard-Verdier M, Gessler A et al (2019) Towards an integrative, eco-evolutionary understanding of ecological novelty: studying and communicating interlinked effects of global change. Bioscience 69:888–899. https://doi.org/10.1093/biosci/biz095
HilleRisLambers J, Yelenik SG, Colman BP, Levine JM (2010) California annual grass invaders: the drivers or passengers of change? J Ecol 98:1147–1156. https://doi.org/10.1111/j.1365-2745.2010.01706.x
Hobbs RJ, Arico S, Aronson J et al (2006) Novel ecosystems: theoretical and management aspects of the new ecological world order. Glob Ecol Biogeogr 15:1–7. https://doi.org/10.1111/j.1466-822X.2006.00212.x
Hua L, Shao G, Zhao J (2017) A concise review of ecological risk assessment for urban ecosystem application associated with rapid urbanization processes. Int J Sust Dev World 24:248–261. https://doi.org/10.1080/13504509.2016.1225269
Jarošik V, Pyšek P, Kadlec T (2011) Alien plants in urban nature reserves: From red-list species to future invaders? NeoBiota 10:27
Kalnay E, Cai M (2003) Impact of urbanization and land-use change on climate. Nature 423:528–531. https://doi.org/10.1038/nature01675
Kendal D, Williams KJH, Williams NSG (2012) Plant traits link people’s plant preferences to the composition of their gardens. Landsc Urban Plan 105:34–42. https://doi.org/10.1016/j.landurbplan.2011.11.023
Kleyer M, Bekker RM, Knevel IC et al (2008) The LEDA Traitbase: a database of life-history traits of the Northwest European flora. J Ecol 96:1266–1274
Klotz S (1984) Phytookologische Beitrage zur Charakterisierung und Gliederung urbaner Okosysteme, dargestellt am Beispiel der Stadte Halle und Halle-Neustadt, Ph.D. Thesis, Martin-Luther-University Halle-Wittenberg
Klotz S, Gutte P (1992) Biologisch-ökologische Daten zur Flora von Leipzig–ein Vergleich. Acta Acad Sci 1:94–97
Klotz S, Kühn I, Durka W (2002) BIOLFLOR–Eine Datenbank zu biologisch-ökologischen Merkmalen der Gefäßpflanzen in Deutschland-Schriftenreihe für Vegetationskunde. Bundesamt für Naturschutz, Bonn
Knapp S (2010) Urbanization Causes Shifts of Species’ Trait State Frequencies–a Large Scale Analysis. In: Plant Biodiversity in Urbanized Areas. Springer 13–29
Knapp S, Kühn I, Stolle J, Klotz S (2010) Changes in the functional composition of a Central European urban flora over three centuries. Perspect Plant Ecol Evol Syst 12:235–244. https://doi.org/10.1016/j.ppees.2009.11.001
Kowarik I (1995a) On the role of alien species in urban flora and vegetation. Plant Invasions: General Aspects and Special Problems SPB Academic Publishing, Amsterdam, The Netherlands, pp 85–103
Kowarik I (2011) Novel urban ecosystems, biodiversity, and conservation. Environ Pollut 159:1974–1983. https://doi.org/10.1016/j.envpol.2011.02.022
Kowarik I (1995b) Time lags in biological invasions with regard to the success and failure of alien species. Plant invasions: general aspects and special problems 15–38
Kühn I, Durka W, Klotz S (2004) BiolFlor: a new plant-trait database as a tool for plant invasion ecology. Divers Distrib 10:363–365
Kühn I, Klotz S (2006) Urbanization and homogenization: comparing the floras of urban and rural areas in Germany. Biol Cons 127:292–300. https://doi.org/10.1016/j.biocon.2005.06.033
Laliberté E, Shipley B (2011) FD: measuring functional diversity (FD) from multiple traits, and other tools for functional ecology, R Package
Landolt E (2000) Some results of a floristic inventory within the city of Zurich (1984–1998). Preslia 72:441–456
Laughlin DC (2014) The intrinsic dimensionality of plant traits and its relevance to community assembly. J Ecol 102:186–193. https://doi.org/10.1111/1365-2745.12187
Lenth R, Singmann H, Love J et al (2018) emmeans: Estimated marginal means, aka least-squares means. R package. See http://www.CRANR-projectorg/package=emmeans
MacDougall AS, Turkington R (2005) Are invasive species the drivers or passengers of change in degraded ecosystems? Ecology 86:42–55
Marco A, Dutoit T, Deschamps-Cottin M et al (2008) Gardens in urbanizing rural areas reveal an unexpected floral diversity related to housing density. CR Biol 331:452–465
Mason NWH, de Bello F (2013) Functional diversity: a tool for answering challenging ecological questions. J Veg Sci 24:777–780. https://doi.org/10.1111/jvs.12097
Mason NWH, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111:112–118. https://doi.org/10.1111/j.0030-1299.2005.13886.x
Mcgill B, Enquist B, Weiher E, Westoby M (2006) Rebuilding community ecology from functional traits. Trends Ecol Evol 21:178–185. https://doi.org/10.1016/j.tree.2006.02.002
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176. https://doi.org/10.1007/s11252-007-0045-4
McKinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Cons 127:247–260. https://doi.org/10.1016/j.biocon.2005.09.005
Pandit MK, White SM, Pocock MJO (2014) The contrasting effects of genome size, chromosome number and ploidy level on plant invasiveness: a global analysis. New Phytol 203:697–703. https://doi.org/10.1111/nph.12799
Paul MJ, Meyer JL (2001) Streams in the urban landscape. Annu Rev Ecol Syst 32:333–365
Pavoine S (2020) adiv: an r package to analyse biodiversity in ecology. Methods Ecol Evol. https://doi.org/10.1111/2041-210X.13430
Potgieter LJ, Gaertner M, O’Farrell PJ, Richardson DM (2019) Perceptions of impact: invasive alien plants in the urban environment. J Environ Manage 229:76–87
Potgieter LJ, Gaertner M, Kueffer C et al (2017) Alien plants as mediators of ecosystem services and disservices in urban systems: a global review. Biol Invasions 19:3571–3588. https://doi.org/10.1007/s10530-017-1589-8
Pykälä J (2019) Habitat loss and deterioration explain the disappearance of populations of threatened vascular plants, bryophytes and lichens in a hemiboreal landscape. Glob Ecol Conserv. https://doi.org/10.1016/j.gecco.2019.e00610
Pyšek P, Chocholousková Z, Pyšek A et al (2004) Trends in species diversity and composition of urban vegetation over three decades. J Veg Sci 15:781–788
Pyšek P, Jarošík V, Pergl J et al (2009) The global invasion success of Central European plants is related to distribution characteristics in their native range and species traits. Divers Distrib 15:891–903. https://doi.org/10.1111/j.1472-4642.2009.00602.x
Pyšek P, Richardson DM, Rejmánek M et al (2004) Alien plants in checklists and floras: towards better communication between taxonomists and ecologists. Taxon 53:131–143
Richardson DM, Pyšek P, Rejmánek M et al (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Ricotta C, Moretti M (2011) CWM and Rao’s quadratic diversity: a unified framework for functional ecology. Oecologia 167:181–188. https://doi.org/10.1007/s00442-011-1965-5
Salinitro M, Alessandrini A, Zappi A, Tassoni A (2019) Impact of climate change and urban development on the flora of a southern European city: analysis of biodiversity change over a 120-year period. Sci Rep 9:9464. https://doi.org/10.1038/s41598-019-46005-1
Schleuter D, Daufresne M, Massol F, Argillier C (2010) A user’s guide to functional diversity indices. Ecol Monogr 80:469–484
Scholz A (2008) Historical development of the ora of Leipzig (including Markkleeberg)
Shackleton RT et al (2018) Explaining people’s perceptions of invasive alien species: a conceptual framework. J Environ Manage 229:10–26
Song X, Zhang J, AghaKouchak A et al (2014) Rapid urbanization and changes in spatiotemporal characteristics of precipitation in Beijing metropolitan area. J Geophys Res Atmosp 119:11250–11271. https://doi.org/10.1002/2014JD022084
Stolle J, Klotz S (2004) Flora der Stadt Halle (Saale), Calendula, hallesche Umweltblatter, Halle (Saale).
Sukopp H (2008) On the early history of urban ecology in Europe. Urban Ecology. Springer, Boston, MA 79–97
Sukopp H (1998) Urban ecology—scientific and practical aspects. In: Urban ecology. Springer, 3–16
Syphard AD, Clarke KC, Franklin J et al (2011) Forecasts of habitat loss and fragmentation due to urban growth are sensitive to source of input data. J Environ Manage 92:1882–1893. https://doi.org/10.1016/j.jenvman.2011.03.014
Tait CJ, Daniels CB, Hill RS (2005) Changes in species assemblages within the Adelaide metropolitan area, Australia, 1836–2002. Ecol Appl 15:346–359
te Beest M, Le Roux JJ, Richardson DM et al (2012) The more the better? The role of polyploidy in facilitating plant invasions. Ann Bot 109:19–45
Thompson K, McCarthy MA (2008) Traits of British alien and native urban plants. J Ecol 96:853–859. https://doi.org/10.1111/j.1365-2745.2008.01383.x
Tilman D (2001) Functional diversity. Encyclopedia Biodivers 3:109–120
Trusilova K, Jung M, Churkina G et al (2008) Urbanization impacts on the climate in europe: numerical experiments by the PSU–NCAR mesoscale model (MM5). J Appl Meteor Climatol 47:1442–1455. https://doi.org/10.1175/2007JAMC1624.1
Van der Veken S, Verheyen K, Hermy M (2004) Plant species loss in an urban area (Turnhout, Belgium) from 1880 to 1999 and its environmental determinants. Flora-Morphol Distrib Funct Ecol Plants 199:516–523
Vaz AS, Kueffer C, Kull CA, Richardson DM, Vicente JR, Kühn I, Schröter M, Hauck J, Bonn A, Honrado JP (2017) Integrating ecosystem services and disservices: insights from plant invasions. Ecosyst Serv 23:94–107
Villéger S, Mason NW, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301
von der Lippe M, Kowarik I (2008) Do cities export biodiversity? Traffic as dispersal vector across urban–rural gradients. Divers Distrib 14:18–25. https://doi.org/10.1111/j.1472-4642.2007.00401.x
Wickham H, Chang W, Wickham MH (2016) Package ‘ggplot2.’ Create Eleg Data Vis Using Grammar Graph Version 3(2):1
Williams NSG, Hahs AK, Vesk PA (2015) Urbanisation, plant traits and the composition of urban floras. Perspect Plant Ecol Evol Syst 17:78–86. https://doi.org/10.1016/j.ppees.2014.10.002
Williams NSG, Schwartz MW, Vesk PA et al (2009) A conceptual framework for predicting the effects of urban environments on floras. J Ecol 97:4–9. https://doi.org/10.1111/j.1365-2745.2008.01460.x
Winter M, Kühn I, Nentwig W, Klotz S (2008) Spatial aspects of trait homogenization within the German flora. J Biogeogr 35:2289–2297. https://doi.org/10.1111/j.1365-2699.2008.01967.x
Winter M, Schweiger O, Klotz S et al (2009) Plant extinctions and introductions lead to phylogenetic and taxonomic homogenization of the European flora. Proc Natl Acad Sci 106:21721–21725. https://doi.org/10.1073/pnas.0907088106
Acknowledgements
Marija Milanović was funded by ESCALTE VH-KO-613. Petr Pyšek was supported by EXPRO Grant No. 19-28807X (Czech Science Foundation) and long-term research development project RVO 67985939 (Czech Academy of Sciences). Ingolf Kühn ackowledges support from BiodivERsA project AlienScenarios (BMBF/PT DLR 01LC1807C). We thank Jens Stolle for assembling the floristic dataset, ‘translating’ pre-Linnean into Linnean nomenclature, and for checking the plausibility of historical floristic records. We would like to thank the reviewers for their comments and suggestions for the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
MM, IK and SK developed the main ideas. MM performed the analysis and first manuscript draft. All authors provided critical feedback and contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Milanović, M., Kühn, I., Pyšek, P. et al. Functional diversity changes in native and alien urban flora over three centuries. Biol Invasions 23, 2337–2353 (2021). https://doi.org/10.1007/s10530-021-02509-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-021-02509-4