Abstract
The germination behavior of a plant influences its fitness, persistence, and evolutionary potential, as well as its biotic environment. This can have major effects on the invasive potential of a species. We review the findings of four types of experimental studies comparing basic germination characteristics of invasive versus non-invasive congeners, in their non-native or native distribution range; invasive alien versus native species; and invasive species in their native versus non-native distribution range. Early and/or rapid germination is typical of invasive species rather than their non-invasive congeners, and represents a pre-adaptation from which many invasive and naturalized species benefit. It also occurs more often in invasive than native species, suggesting that competition mitigation or avoidance in the early stages of a plant’s life, via the exploitation of vacant germination niches, might be more useful than a superior competitive ability in novel environments. This is further supported by a tendency of invasive species to germinate earlier and/or faster and have broader germination cues in their non-native than in their native range. It is also supported by broader germination requirements being reported for invasive species than their non-invasive or native congeners. In contrast, high percentage germination is not a consistent predictor of invasiveness, suggesting that the incorporation of a larger fraction of seed production into the soil seed bank rather than high germination is a better (or safer) strategy in novel environments. These patterns indicate that differences in the germination behavior of alien and native species contribute to the invasiveness of many species, although evidence under natural conditions is needed. The role of such differences in the establishment and spread of invasive species in novel environments and their long-term impact on community dynamics requires further study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The identification of suites of traits that can be used to predict the invasiveness of species introduced outside their native distribution range is one of the major quests in invasion science (Rejmánek and Richardson 1996; Alpert et al. 2000; Pyšek and Richardson 2007; Pyšek et al. 2015). While the context-dependent nature of plant invasions has often been stressed, especially in relation to stochastic events and/or disturbance that may either promote or hamper the establishment and spread of alien plants (Williamson 1996; Alpert et al. 2000), traits such as large seed production and high germination, as well as a capacity to germinate under a broad range of environmental conditions characterize many invasive and weedy species (Baker 1974; Colautti et al. 2006; Grime et al. 2007; Pyšek and Richardson 2007; Moravcová et al. 2010, 2015).
As the earliest major developmental transition in a plants’ life (Donohue et al. 2010), germination determines the environmental conditions experienced by seedlings and thus has major effects on the fitness, survival, persistence, ecological niches, distribution ranges, and evolutionary potential of plants (Ross and Harper 1972; Harper 1977; Simons and Johnston 2000; Donohue 2003, 2005a; Donohue et al. 2010). The timing and speed of germination influence the competitive environment (Benech-Arnold et al. 2000; Forbis 2010), including competitive interactions between native and alien species (Gioria and Osborne 2014; Gioria et al. 2016) and indirect competitive interactions (Gioria and Osborne 2014). The germination behavior of a species can thus substantially affect the probability of its establishment (Baker 1974; Crawley 1997; Gerlach and Rice 2003; Colautti et al. 2006; Willis et al. 2014) and its capacity to become invasive in novel environments (Pyšek and Richardson 2007; Luo and Cardina 2012; Gioria et al. 2016).
The importance of germination traits in plant invasions has been typically examined in experimental studies by using measures of germination success (e.g., final percentage germination at the end of a germination experiment, typically after a few weeks), timing (mainly time to germination of the first seedling, hereafter time to germination), speed (germination rate or time to different percentiles of germination), and by estimating the range of environmental conditions under which germination can occur. These traits are useful to estimate the dynamics and outcomes of the germination process (Ranal and Santana 2006), are relatively easy to measure, and allow the collection of information for many species over a relatively short period (Dürr et al. 2015). The role of germination traits as determinants of species invasiveness is highlighted in recent reviews indicating that ‘germination success/rate’ is positively associated with the ‘establishment/spread’ and/or ‘abundance/impact’ of invasive species (Colautti et al. 2006) and that many invasive/alien species germinate earlier, better, or over a broader range of environmental conditions than non-invasive or native species (Pyšek and Richardson 2007).
Here we review the recent literature comparing four trait categories (percentage germination, time to germination, germination speed, and breadth of germination conditions) in several types of comparison: (1) invasive versus non-invasive congeners in their non-native distribution range, (2) invasive versus non-invasive congeners in their native distribution range, (3) invasive alien versus native species/congeners, and (4) invasive species in their native versus non-native distribution ranges. We describe the main trends identified in each type of comparison and discuss their significance in predicting future invasions and for developing sustainable control and restoration measures. We highlight some major methodological issues and practical difficulties associated with germination studies and their limits in predicting germination patterns under natural conditions. We conclude by indicating some promising research directions in this field that can improve our understanding of the role of differences in the germination behavior of native and alien species in the invasion process. The potential long-term implications of such differences under a changing climate are discussed. We follow Richardson et al. (2000) in defining alien and invasive species.
Every picture tells a story: experimental comparisons of germination patterns in plant invasions
Invasive versus non-invasive alien congeners in their non-native distribution ranges
Comparing closely related invasive and non-invasive species, ideally congeners, in their non-native distribution range is useful to identify traits that explain the success of species that have become invasive in novel environments compared to those that have not (Maillet and Lopez-Garcia 2000; Hamilton et al. 2005; van Kleunen et al. 2010). With regard to germination traits, several studies have adopted this approach (Table 1). These studies indicate that percentage germination is not a good predictor of invasiveness (Table 1). In contrast, a short time to germination (or requirement for stratification) and/or rapid germination characterize many invasive species compared to their non-native or less invasive congeners (Table 1), suggesting that germination timing and speed play an important role in determining the invasiveness of many species/congeners.
Invasive species also tend to germinate over a broader range of temperatures than their non-invasive congeners; however, there are only few comparisons of this type (Table 1). For instance, Forcella et al. (1986) examined the germination of three alien species differing in their distribution range in Australia (Echium plantagineum, common (invasive); E. vulgare, uncommon; and E. italicum, rare) and found that E. plantagineum germinated under a broader range of alternating temperatures and more rapidly than E. vulgare and E. italicum (twice and three times, respectively). In contrast, percentage germination was lower in the invader than in both uncommon species, supporting the general view that high percentage germination per se is not a critical factor promoting invasiveness.
Clearly, these patterns are strongly affected by the conditions provided over the duration of germination experiments (Table 1). For instance, percentage germination of seeds of two alien species in Taiwan, Bidens pilosa var. radiata (invasive) and B. bipinnata (non-invasive), is similar at 28 °C, corresponding to a summer temperature regime in the region. However, at 18 °C, corresponding to a local winter temperature, the percentage germination of B. bipinnata is very low (12%) compared to that of the invader (85%) (Hsu and Kao 2014). This suggests that the invasiveness of Bidens pilosa in Taiwan could be associated with its capacity to germinate at times when other species are not active and that it benefits from a low resource competition.
Evidence of the importance of phenotypic plasticity in discriminating between invasive and non-invasive congeners, with respect to germination traits, is weak and further study is required. Ruprecht et al. (2014) recorded plasticity in a range of performance and functional traits for 10 invasive and 10 naturalized but non-invasive alien plants (some of them congeners), in a common garden experiment, under various combinations of water, light, and nutrient availability, and found that the germination ratio and time to germination of invasive and non-invasive species were similar under unfavorable environmental conditions, with low resource availability delaying germination in both species groups.
Some species, however, are more sensitive to stressful conditions than their non-invasive congeners. Complex patterns of germination are reported for two alien species in Ohio, USA, invasive Taraxacum officinale and non-invasive T. laevigatum (Luo and Cardina 2012). While the invader germinates well under a broader range of alternating temperatures compared to the non-invasive species, it is more sensitive to stress, with a significantly lower percentage germination recorded at a low water potential or in darkness than for its non-invasive congener. A broader range of germination temperatures could be beneficial both in the early and later part of the growing season. A capacity to germinate at low temperatures could be important in the early establishment of this species by allowing it to germinate earlier than other species and thus gain a competitive advantage. A capacity to germinate at high temperatures would also allow this invader to germinate throughout the growing season, potentially increasing the probability of germination at ‘safe sites’ (sensu Harper 1977). However, lower germination of T. officinale at low water potential suggests that germination would be low under natural conditions in summer, and that life stages other than germination could be more important in determining the invasiveness of this species (Luo and Cardina 2012).
The invasiveness of many species is often attributed to their ruderal strategy, which consists of producing large numbers of small, persistent seeds that require light for germination and that can germinate rapidly soon after disturbance (Grime et al. 1981; Thompson et al. 1993; Grime 2001; Fenner and Thompson 2005; Grime et al. 2007; Gioria and Osborne 2010). This is evident in some congeneric comparisons between invasive and non-invasive alien congeners, although further evidence is needed. A light requirement for germination was the only predictor of invasiveness among germination traits of 15 Oenothera species that are alien to the Czech Republic and vary in their degree of invasiveness (Mihulka et al. 2003), and has been reported for the invasive T. officinale but not for non-invasive T. laevigatum (Luo and Cardina 2012).
Invasive versus non-invasive congeners in their native distribution ranges
Comparing the traits of species that have become invasive or naturalized following their arrival in a new area with those that have not become invasive or naturalized, using plant material collected from their native distribution range, is useful to assess whether a preadaptation in certain traits acquired at home plays a role in determining the naturalization or invasiveness of a species abroad (Hamilton et al. 2005; Pyšek and Richardson 2007). Although comparative studies of this type are scarce, a preadaptation in germination timing is typical of many naturalized and invasive species. Time to germination is an important trait in the naturalization of species of South African Iridaceae, being on average 15% shorter in 30 native South-African species that have become naturalized elsewhere than in their congeners that did not (van Kleunen and Johnson 2007). Comparisons of 14 congeneric pairs of invasive and non-invasive herbaceous species that were introduced into North America also show that time to germination was significantly shorter (by 10 days, on average) in invasive than in non-invasive species (Schlaepfer et al. 2010).
In contrast, evidence of the importance of high percentage germination as a useful preadaptation acquired in the native range for promoting naturalization or invasiveness in the non-native range is weak. Hock et al. (2015) examined germination patterns of invasive and non-invasive species pairs on the South Island, New Zealand (Echium vulgare vs E. plantagineum; Verbascum thapsus vs V. nigrum), using seeds from European native populations, and found that invasive congeners germinated in higher percentages and more rapidly than non-invasive congeners. In contrast, comparisons of germination patterns using seeds collected from native Spanish populations of three Centaurea species alien to California, invasive C. solstitialis and non-invasive C. calcitrapa and C. sulphurea, show that the percentage germination of the invader is intermediate between that of C. calcitrapa and C. sulphurea (García et al. 2013), indicating that other traits contribute to the invasiveness of this species.
Invasive versus native species
This type of comparison is useful to identify traits that can provide alien species with a competitive advantage over native species (Crawley 1997; Lake and Leishman 2004; Hamilton et al. 2005; Tecco et al. 2010). This approach is more widely used than comparisons between invasive and non-invasive species with respect to germination traits. In general, invasive or naturalized species germinate earlier or more rapidly than native species, although exceptions are reported (Danuso et al. 2012; Moore and Lacey 2009).
Many invasive species also germinate in higher percentages than native congeners. However, patterns in percentage germination are strongly dependent on the environmental conditions provided over the duration of experimental studies (Table 2). This means that only under certain environmental conditions an alien species may benefit from a competitive advantage arising from high germination. For instance, a light requirement is reported for the invasive alien Ruellia nudiflora in Yucatan, Mexico, where percentage germination for this species was significantly lower at 20% ambient light than that of the native R. pereducta, but higher at 80% ambient light (Cervera and Parra-Tabla 2009). However, the invader germinated under a much broader range of temperatures and water potentials than the native species, suggesting that a capacity to germinate under sub-optimal conditions for growth is more important than high germination in promoting the invasiveness of this species in open disturbed areas.
Native species are considered to be better adapted to local environmental conditions (Tecco et al. 2010) and often have more constrained germination cues than alien or invasive species (see Marushia et al. 2010 and references therein). In contrast, invasive species are expected to respond more strongly than native species to stressful conditions (Funk 2013). This could increase the differences in the germination behavior of invasive or alien species with native species. For instance, Pérez-Fernández et al. (2000) showed that low water availability increased the differences in germination timing between seven alien and seven native species that co-occur in southwestern Australia. Low water availability delayed germination of alien species less than that of natives (by 2 and 7 days, respectively), suggesting that a drought event is likely to increase the duration of the germination advantage of alien species and thus their competitive advantage.
Wainwright and Cleland (2013) also found more constrained germination cues in native than alien species. These authors recorded robustness (measured in terms of percentage germination under a variety of conditions) and plasticity (increase in percentage germination in response to favorable conditions) in response to environmental cues (temperature, day length, and soil moisture) in 12 alien and 12 native species that occur commonly in coastal sage scrub in California. They found that both germination plasticity and robustness are greater in alien species, particularly annuals, which germinate earlier than native species, in consistently higher percentages, and respond more strongly to favorable conditions (warm temperatures and high soil moisture).
The importance of phenotypic plasticity in germination traits as a determinant of the success of invasive species over natives is also reported by Paudel and Battaglia (2013). These authors assessed the effects of elevated salinity on the initial recruitment of the invasive woody species Triadica sebifera and two native woody species, Baccharis halimifolia and Morella cerifera, across five soil types characterizing the dominant vegetation zones in coastal Mississippi. Although the invader was in general more sensitive to salinity than the native species, percentage germination in inland soils did not differ across salinity levels, suggesting that plasticity in this trait under increasing levels of salinity could contribute to the spread of this species inland along coastal transition ecosystems (Paudel and Battaglia 2013).
Chrobock et al. (2011) argue that differences in the germination behavior of invasive aliens and native species could be due to an introduction bias resulting from the selective introduction of plants with high and rapid germination. To test this, they compared percentage germination in 42 species of plants native to Switzerland and 47 cultivated alien species, including 26 cultivars, at two light levels. The results of this study support this hypothesis, with cultivated alien species germinating, on average, significantly earlier and to greater percentages than related native species at both light levels. Moreover, alien cultivars germinated significantly earlier and in greater percentages than alien non-cultivars, with the responses to shading not significantly different between these two groups of cultivars (Table 2).
Invasive species in their native and non-native distribution ranges
Examining differences in the behavior of a species in its native and non-native distribution ranges can provide important insights into ecological and evolutionary processes associated with plant invasions (Hierro et al. 2005). However, relatively few studies have adopted this approach with respect to germination traits. These studies report a tendency for invasive species to germinate earlier and/or faster, and at greater percentages, in their non-native than their native distribution ranges, and/or over a broader range of germination conditions (Table 3).
These patterns are attributed to the fact that the germination strategy (in terms of percentage, timing, and speed) of a species or a population is often associated with the degree of predictability of environmental conditions in the native range, which derives from variation in climatic conditions and in the disturbance regime (Venable and Brown 1988; Fenner and Thompson 2005; Venable 2007; Volis and Bohrer 2013). For instance, seeds from native Chinese populations of the invasive Ulmus pumila were found to be more strongly affected by climatic conditions than seeds from non-native North American populations (Hirsch et al. 2012). Seeds from non-native populations germinated significantly faster than those from native populations, while no significant difference in the percentage of germination was reported for seed from both areas at moderate and warm temperatures. Mean time to germination in both ranges was significantly negatively correlated with annual precipitation, and this relationship was stronger for seed from native populations, while mean time to germination for non-native populations was less affected by climatic conditions (Hirsch et al. 2012).
More constrained germination cues in the native than in the non-native range are also reported for the invasive herbaceous plant Ambrosia artemisiifolia. Leiblein-Wild et al. (2014) found that seed from non-native European populations of this species germinated over a broader range of temperatures, at higher percentages and faster (expressed as the number of days required to achieve 50% germination) than seed from native North American populations. The extension of the germination temperature niche of Ambrosia artemisiifolia in non-native populations was combined with an improved resistance to subzero temperatures. Moreover, frost tolerance of seedlings was strongly positively correlated with the probability of spring frosts in the native distribution range, while no correlation with environmental parameters in the non-native range was found for this variable. These findings suggest that invasiveness in A. artemisiifolia is associated with a competitive advantage arising from a capacity to germinate early and exploit an early window of opportunity under which the competition from native species is low, despite the increased risk of post-germination frosts.
Similar findings are reported for Rhododendron ponticum by Erfmeier and Bruelheide (2005). In this study, germination rates (but not percentage germination) differed significantly with respect to the origin of the populations examined, with seeds of invasive Irish populations requiring significantly fewer days to reach 10 and 50% germination than seeds of native Georgian and Spanish populations. These patterns were strongly affected by temperature, with seeds from non-native populations germinating faster than those from native populations at the coldest and warmest temperatures, while at intermediate temperatures, the rate of germination recorded for non-Irish populations was similar to that of Georgian populations and higher than for Spanish populations. These germination patterns are consistent with the lower degree of variability in post-germination environmental conditions experienced by non-native populations, for which the temperature range during the growing season is lower than in the native range, as is the risk of drought (Erfmeier and Bruelheide 2005).
Some invasive species, however, display stricter germination cues in the non-native range and contrasting patterns for percentage germination in the native and non-native range are reported (Table 3). For instance, invasive Japanese strains of Cardamine hirsuta are characterized by strong initial seed dormancy and little germination at high temperatures unlike native European strains (Kudoh et al. 2007). Comparisons of germination patterns for the invasive species Centaurea solstitialis using seed collected from native (Turkey: summer drought; Georgia: summer rain) and non-native populations (California: summer drought; Argentina: summer rain) show that percentage germination of seed from non-native populations is strongly associated with variation in winter precipitation (Hierro et al. 2009). Percentage and rate of germination of seed from non-native genotypes in Argentina (summer rain) were lower than those of seeds from native populations, regardless of the seasonality of the rainfall in the region where they occur, and from non-native populations in regions characterized by summer droughts (Hierro et al. 2009). In contrast, García et al. (2013) found that percentage germination for this species and two non-invasive congeners was higher for non-native Californian populations than for native Spanish populations (C. solstitialis: +63%; C. calcitrapa: +24%; C. sulphurea: +123%; Table 3), suggesting that high germination is important for the establishment of these species in their non-native range.
While differences in germination patterns between native and non-native populations are often interpreted as evidence of the importance of rapid adaptation in the success of invasive species outside their native distribution range (Erfmeier and Bruelheide 2005; García et al. 2013), other processes, including plastic changes in germination traits, can play a key role in the establishment and persistence of alien species, and their capacity to spread and persist in new areas (Grime et al. 1981; Reynolds et al. 2001; Wainwright et al. 2012; Wainwright and Cleland 2013). Plasticity in germination traits represents a means of habitat selection and niche construction (Odling-Smee et al. 1996; Donohue 2003, 2005b), which thereby influence the evolution of plasticity in post-germination traits (Donohue 2003) and consequently the invasive potential of a species.
Methodological issues: do not compare apples with oranges
A number of methodological issues can produce under- or overestimates of the importance of germination traits in plant invasions. Comparative germination studies provide useful information on potential germination-related mechanisms underlying successful invasion but suffer the common limitation of experimental work in not accounting for variation in the biotic and abiotic conditions experienced by seeds in the field. Because these results may be very different from those occurring in nature (Verdú and Traveset 2005; Gioria and Osborne 2013; Gioria et al. 2016), the findings of experimental studies comparing the germination behavior of invasive and non-invasive or native species should be interpreted with caution.
Limitation of experimental studies: experimental conditions
Germination patterns strongly depends on temperature and other environmental conditions, including water potential, light, and nutrient levels (Benech-Arnold et al. 2000; Forcella et al. 2000; Donohue et al. 2010; Baskin and Baskin 2014). The germination responses of a species to environmental factors are complex and interactions among these factors complicate the task of assessing the role of germination traits in plant invasions. For instance, light is known to influence the temperature range for germination (Probert 2000; Dillon and Forcella 1984), with potential contrasting effects on the germination behavior of the compared species. The conditions provided during germination experiments can thus have major effects on the magnitude and direction of differences in the germination behavior of the compared species. This is evident in studies comparing germination patterns under different environmental conditions (e.g. Cervera and Parra-Tabla 2009; Hsu and Kao 2014) or in laboratory versus garden experiments (Perglová et al. 2009; Skálová et al. 2011).
Dormancy-related biases
Seed dormancy hampers our capacity to predict the timing and extent of germination under natural conditions (Benech-Arnold et al. 2000). Seed dormancy is a common attribute of weedy and invasive species, often resulting in the formation of large soil seed-banks (Thompson and Grime 1979; Cavers and Benoit 1989; Benech-Arnold et al. 2000; Forcella et al. 2000; Fenner and Thompson 2005; Gioria and Pyšek 2016). Germination patterns are closely associated with the dynamics of dormancy release (Benech-Arnold et al. 2000; Forcella et al. 2000; Batlla and Benech-Arnold 2007; Baskin and Baskin 2014). As the environmental conditions that are needed to break dormancy differ from those required for germination, dormancy and germination patterns should be predicted separately (Benech-Arnold et al. 2000).
Despite its importance in regulating germination, dormancy is often filtered out in experimental studies, which report germination patterns after dormancy has been broken for the tested seeds. Moreover, dormancy release is a gradual process: as dormancy is released, the temperature range for germination widens until it is maximal, while as dormancy is induced, the range of temperatures over which germination can proceed narrows, until full dormancy is reached (Forcella et al. 2000). Also, under natural conditions, the depth of seed burial has major effects on the breaking of dormancy and on germination patterns, so that studies testing germination under conditions resembling the conditions experienced by seeds on the soil surface (full light), might produce erroneous estimates of the timing and extent of seed germination in the field (e.g. Gioria and Osborne 2013).
Seed heteromorphism, defined as the production of different types of seed by a single individual, which is a common characteristic of weedy species (Cavers and Harper 1966; Harper 1977), can also bias the results because the different seeds vary in dormancy-breaking requirements and germination (Crawley 1997; Gerlach and Rice 2003; García et al. 2013; Table 2). Seed heteromorphism occurs in 18 families, most frequently in the Asteraceae and Chenopodiaceae (Imbert 2002). Whether seed heteromorphism contributes to the invasiveness of members of these two families, which include many invasive species (Daehler 1998), and those of other families with heteromorphic seeds deserves further study.
Maternal effects
As ‘the pathway’ that completes the life cycle of a plant (Donohue 2009), the maternal environment strongly influences seed dormancy and germination patterns (Cavers and Harper 1966; Steadman et al. 2004; Donohue 2005a, b; Campbell et al. 2015). Probert et al. (1985) report a negative relationship between temperature during seed development and maturation, and the degree of dormancy. Conditions during seed maturation also interact with post-dispersal environmental conditions in determining germination phenology (Donohue 2005b). Such interactions have a genetic basis (Donohue 2005b), and their effects extend beyond one generation (Kendall and Penfield 2012). Moreover, maternal effects on seed traits can affect generation time and projected population growth rates (Donohue 2009). For the invasive annual species Raphanus raphanistrum, Campbell et al. (2015) reports significant differences in size and fecundity among offspring generated from parents grown under different soil moisture conditions and suggest that shifts in maternal environments may alter the invasion potential of this species.
The use of seed collected from different habitats/ecosystems or from regions with different climatic conditions can thus differ in the degree of dormancy and in their germination requirements regardless of their native (native vs alien) or invasive (invasive vs non-invasive) status (Fenner and Thompson 2005; Gioria and Osborne 2009a, 2013). Despite their significance in determining the colonization ability of a species (Campbell et al. 2015), maternal effects on germination traits are rarely included in comparative studies assessing the role of germination in plant invasions. Studies accounting for these effects have done so by using seeds produced by plants grown under controlled conditions from seeds collected in the field (e.g., Kudoh et al. 2007; Hierro et al. 2009).
Storage conditions and sampling effects
Storage conditions can strongly influence seed germination. Physiological changes during dry storage are known to make germination requirements less specific and decrease the level of primary dormancy (Probert et al. 1985). Prolonged chilling is reported to widen the range of temperatures suitable for germination (Nikolaeva 1977), facilitate germination at suboptimal temperatures (Roberts and Totterdell 1981), and reduce dependence on other environmental factors (Probert et al. 1985), although it can also induce dormancy (Baskin and Baskin 1988). A strong effect of storage conditions on germination timing and percentage germination is evident in a comparison of germination patterns of the invasive Impatiens glandulifera and less invasive or native congeners (Perglová et al. 2009; Tables 1, 3). Additional experimental artifacts potentially affecting germination patterns and assessments of their importance in plant invasions include whether seeds are collected from one or more individuals in a population or whether there are substantial differences in the timing of seed collection from different populations.
Role of the species compared in plant communities
Studies comparing the germination behavior of invasive and native species should state whether the latter are dominant in native communities and how wide or restricted their distribution is in their native range. If the aim is to identify traits that can explain differences in abundance between closely related invasive and native congeners, invasive (dominant) species should be compared with native species that are dominant in such communities prior to an invasion (Hamilton et al. 2005; Gioria and Osborne 2014; Hejda et al. 2016). For meaningful comparisons between invasive and non-invasive or less invasive species, information on residence time and propagule pressure is also necessary, as the differences in the invasive status (invasive vs non-invasive) of such species could be due to differences in their introduction history and not in their germination traits (Hamilton et al. 2005; Colautti et al. 2006).
Discussion: emerging patterns and global change
Experimental studies indicate that invasive species tend to germinate earlier (shorter time to germination) and/or more rapidly (higher degree of germination synchrony) than non-invasive and native species. This is consistent with studies showing that a germination advantage or priority is a key factor facilitating many plant invasions, especially those by annual plants, in many ecosystems (Grime et al. 1981; Reynolds et al. 2001; Abraham et al. 2009; Godoy et al. 2009; Stevens and Fehmi 2011; Dickson et al. 2012; Wainwright et al. 2012; Kardol et al. 2013; Wainwright and Cleland 2013; see Gioria et al. 2016 for a review). Early-germinating species may benefit from reduced competition and begin to use resources before other species, often with detrimental effects on the establishment and diversity of later-germinating species (Ross and Harper 1972; Seabloom et al. 2003; Abraham et al. 2009; Grman and Suding 2010; Fig. 1), through size-asymmetric competition (Weiner 1990; Abraham et al. 2009), resource pre-emption (Ross and Harper 1972), and soil legacies (Grman and Suding 2010), mechanisms by which they can exert a founder control over native species that emerge later in the season (Körner et al. 2008). This strategy is particularly important in ensuring the persistence of annual plants in communities dominated by perennials (Rees and Long 1992; Verdú and Traveset 2005).
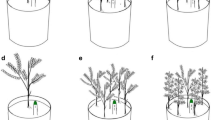
Conceptual diagram representing potential germination niches or windows of opportunity during which competition for resources is null or low, for four groups of species. Species A, which germinates asynchronously, under a broader range of conditions than species B, can take advantage of both early and late windows of opportunity as well as of potential vacant niches arising from differences in germination timing to gain a competitive advantage over species B. Species C are species that germinate earlier and/or later in the growing season than species B and D, and can thus benefit from early and/or later germination niches or windows of opportunity. The germination strategy of species C carries the risks that those conditions promoting germination do not reflect the start of the growing season, or that germination occurs past when conditions for growth and development are no longer suitable. Low or null establishment will follow germination of the majority of seeds if this occurs too early or too late in the growing season. In contrast, asynchronous germination mitigates mortality risks associated with too early and/or too late germination. Climate change resulting in shorter winters and/or extended growing season could favor the successful establishment of species A and species C, although factors other than low competition may affect seedling establishment, such as herbivory or disturbance. Many invasive species that belong to species A and C germinate earlier than their native and/or non-invasive congeners (species C and D), and exploit germination niches under which competition for resources is low (marked by arrows). Germination during early or vacant germination niches could result in potential invasions if they lead to the successful establishment of adult individuals
Early germination can thus be highly beneficial to the initial establishment of invasive species (Gioria and Osborne 2014). However, this is a risky strategy because those conditions that have promoted early germination (e.g., pre-growing season rainfall events or warm spells) might not last until the actual start of the growing season (Lambrinos 2006; Wainwright et al. 2012). Moreover, early germination might increase seedling mortality by herbivores or pathogens (Lambrinos 2006; Ruxton and Schaefer 2012; Wainwright et al. 2012; Gioria et al. 2016). Despite the benefits derived from early access to available resources, early germination does not necessarily result in the establishment of a large proportion of seedlings; other factors such as the competitive ability of an alien species and resource availability in later phases of seedling development can be more important in determining its establishment in novel environments (Sans et al. 2004). The long-term implications of early germination in the persistence of invasive plants also remain unclear (Corbin and D’Antonio 2004; Gioria et al. 2016).
A capacity for rapid germination is a useful strategy to mitigate the effects of inter-specific competition (Grime et al. 1981; Miller et al. 1994; Dyer et al. 2000; Verdú and Traveset 2005; Dürr et al. 2015), as it can suppress the germination or establishment of later germinating seedlings of neighboring species (Fenesi et al. 2014; Gioria et al. 2016). Highly synchronous germination is beneficial in more predictable environments, where it increases the probability of establishment (Fenner and Thompson 2005; Venable 2007), and is thought to play an important role in determining invasiveness of many annual and perennial invasive herbaceous plants (e.g. Beerling and Perrins 1993; Navie et al. 2004; Gioria and Osborne 2009b; Pucheta et al. 2011). However, this is a high-risk strategy in unpredictable environments (Crawley 1997; Gioria et al. 2016), especially when occurring early in the season. In such environments, asynchronous germination might be more beneficial to the persistence of a species in a community, as it spreads the risks associated with unfavorable post-germination environmental conditions and increases the probability of recruitment from the seed bank at times when competition from other species with seasonal germination patterns is low (Venable 2007; Donohue et al. 2010; Gioria et al. 2016). Asynchronous germination is regarded as a major determinant of the ecological niche and distribution range of different plant species (Donohue et al. 2010), including weedy (Brändle et al. 2003) and invasive species (Gioria and Osborne 2013).
The breadth of germination requirements is associated with the ecological demands and geographical distributions of plants, and limits the occupied niche and establishment in novel environments (Donohue et al. 2010). A capacity to germinate under a broad range of environmental conditions reported for invasive species compared to their non-invasive or native counterparts supports the notion that this is an important feature promoting weediness (Baker 1974) or invasiveness (Pyšek and Richardson 2007).
Not surprisingly, percentage germination is not a reliable predictor of invasiveness. Contrasting patterns for this trait occur in each comparison category, suggesting that the germination of a large fraction of seeds produced in a given season is not a requirement (or a safe strategy) for success in a novel environment. High percentage germination in a given season can be regarded as a dimension of propagule pressure, which is known to have important implications for species’ invasiveness and/or the invasibility of the recipient communities (Von Holle and Simberloff 2005; Simberloff 2009). This strategy can result in invasive plants preventing the germination of the seed of native plants, especially when they germinate earlier or later than the seed of native species (Gioria et al. 2016), and/or outcompeting seedlings of native plants (Gioria et al. 2012). However, high germination increases a species’ vulnerability to unfavorable post-germination conditions, especially if occurring early or rapidly (Lambrinos 2006; Wainwright et al. 2012; Gioria et al. 2016).
In unpredictable environments, the incorporation of a larger fraction of seeds into the soil seed bank might be a better strategy for ensuring the persistence of a species, as it mitigates the effects of unfavorable environmental conditions for growth and inter- and intra-specific competition (Thompson and Grime 1979; Venable and Brown 1988; Gioria et al. 2012). This is evident in studies that show that the level of dormancy recorded for certain invasive species is higher than that of non-invasive congeners (Gerlach and Rice 2003; Mandák 2003) or in their non-native compared to their native range (Kudoh et al. 2007). These studies suggest that, for certain species, lower percentage germination in their non-native distribution ranges and potentially resulting in the formation of a larger seed bank might be a more successful strategy for dealing with a novel environment and competing with the native vegetation. These patterns are consistent with recent findings that a persistent soil seed bank (sensu Thompson et al. 1997) is an important determinant of naturalization (Pyšek et al. 2015) and invasiveness in many species (Gioria and Pyšek 2016).
Climate change can strongly affect germination patterns and population dynamics (Walck et al. 2011; Johnson 2014) of native and invasive alien species, and thus may alter the role of germination in the invasion process. Climate change is expected to favor species with broad germination requirements (Walck et al. 2011). It is predicted that the species that detect and respond more rapidly to such changes, at the ecological level, including plastic changes within existing populations or changes in a species’ distribution range (Nicotra et al. 2010; Volis et al. 2015), or at the genetic level, such as adaptation (e.g. Davis et al. 2005; Wolkovich and Cleland 2014; Volis et al. 2015), will have a selective advantage over slowly-responding species (Willis et al. 2010; Dickson et al. 2012).
Information on the germination responses of invasive (and potentially invasive) species and native species to variation in environmental conditions, including climate change, could greatly improve our ability to predict future invasions. Shorter winters, with spring temperatures occurring earlier in the year in many regions (e.g. Walther et al. 2002; Schwartz et al. 2006), could promote the establishment and spread of alien species with shorter stratification requirements for breaking dormancy (Walck et al. 2011; Fig. 1). In a recent experimental study on the effects of chilling and photoperiod on the spring phenology of 36 woody species, Laube et al. (2014) show that invasive and pioneer species have lower chilling requirements than climax species, suggesting that invasive species could profit from increasing winter temperatures, provided that late spring frosts occur earlier.
That many of the invasive species included in this study germinated earlier and/or more rapidly, over a broader range of conditions, and required shorter period of stratification than non-invasive or native species or congeners indicates that shorter winters and less predictable rainfall due to changing climate could promote the establishment of many invasive species. However, the long-term benefits of such changes remain unclear. Moreover, short winters could delay rather than advance germination in species with stricter chilling requirements (Walck et al. 2011). It is possible that narrow germination requirements, which are more often reported for native species compared to invasive alien species, including invasive species in their native range (Table 3), could be a more successful strategy in determining the long-term persistence of invasive species in novel environments.
Whether the germination response of invasive species to variation in environmental conditions is important only in the initial stages of establishment or over the entire invasion process also remains unclear. Naturalized but non-invasive species differ from invasive species in many comparisons of their germination in their native distributions, suggesting that certain germination traits are important also in the invasion phase (Schlaepfer et al. 2010; Hock et al. 2015). That invasive species tend to germinate earlier and/or more rapidly than their non-invasive congeners in their non-native range also suggests that a capacity for early or rapid germination plays an important role not only in the establishment phase but also in the invasion phase. Clearly, the generality of these patterns should be tested for a large number of species belonging to different functional groups reared under a broad range of environmental conditions that closely resemble those encountered by a species in the field, any potential effect of experimental artifacts being minimized.
Future research directions
Despite the importance of the germination behavior of a species in determining its colonization potential (Beckmann et al. 2011), comparative studies examining its importance in plant invasions are scarce. Below we highlight some promising research avenues that could improve our understanding of the role of germination as a critical stage promoting species invasiveness.
-
Research under natural conditions There are virtually no field studies aimed at assessing germination dynamics of seeds of native, invasive alien, and non-invasive alien species under natural conditions (Gioria et al. 2016). There is a great potential for field studies to build up on the results of experimental studies and explore the importance of the germination stage in the invasion process. Because a range of environmental factors and species interactions are known to modify the outcomes of germination and early population development, insights into the importance of germination traits in plant invasions relative to other factors can be obtained only by manipulating conditions in the field. Assessments of the seed banks of invaded communities based on the seedling emergence approach (Thompson et al. 1997), using soil extracted under the sampled communities and grown under conditions that resemble those encountered in the field, are also useful to reveal the degree of germination synchrony and identify germination peaks as well as potential early or late windows of opportunity that can arise from differences in dormancy and germination behavior between alien and native species (Gioria and Pyšek 2016). While field germination patterns are known for many weeds in agricultural systems, many of which are invasive in their non-native range (e.g. Benech-Arnold et al. 2000; Batlla and Benech-Arnold 2007; Gardarin et al. 2011; Guillemin et al. 2013; Dürr et al. 2015), rigorous dormancy and germination studies are needed for species that invade natural and semi-natural ecosystems, as they can have strong effects on biodiversity and ecosystem functioning. These studies should be complemented by theoretical research aimed at predicting field germination dynamics of species differing in their native and invasive status.
-
Accounting for biases As the differences in germination patterns of the groups of interest (invasive vs non-invasive; invasive vs native; invasive in invaded vs native distribution ranges) are often subtle and difficult to detect, it is necessary to take into account potentially biasing factors such as maternal effects, seed collection biases, seed origin, residence time, dominance status, and the other factors dealt with in Section III. While these problems cannot be completely avoided, their potential effects on germination patterns and dormancy must be minimized in order to arrive at unbiased conclusions. To account for these biases, studies comparing the germination behavior of invasive and native species should include information on residence time, on whether native species are dominant in native communities, and how wide or restricted their distribution is in their native range.
-
Context-dependence of plant invasions To account for the context-dependence of the role of species traits in plant invasions (Pyšek and Richardson 2007; Pyšek et al. 2009), germination traits should be explored in relation to other biological traits of invasive species. This would improve our understanding of how the relative importance of germination traits varies at different stages of an invasion process. Complex models are needed to link seed germination dynamics of alien and invasive species in their native and non-native distributions with physiological traits, such as efficiency of water-use and relative growth rate (Kimball et al. 2011), morphological traits, mode of reproduction (sexual vs combination of sexual and asexual), plasticity, and genetic variation, in order to improve our predictions based on the interaction of species traits with other factors (Pyšek et al. 2015). For species reproducing both sexually and asexually, information on the relative importance of these strategies at different stages of the invasion process is needed. Clearly, the importance of recruitment from the seed bank (and thus of dormancy and germination traits) versus vegetative propagation for a species will depend on the environmental conditions in the novel range (Gioria and Osborne 2013). Under stressful conditions, many species rely on vegetative propagation rather than seed production for maintaining their population size at a locality or for rapid expansion (see Gioria and Osborne 2010 and references therein). Differences in the germination responses of alien and native species to such conditions can strongly affect the invasive potential or persistence of alien species (Pérez-Fernández et al. 2000; Cervera and Parra-Tabla 2009; Luo and Cardina 2012).
-
Interactions with other trophic levels The effects of indirect interactions on the invasiveness of alien plants via their effects on germination have been little explored, yet they could have major implications for the establishment or the long-term persistence of invasive species in novel environments. Organisms such as herbivores or pathogens can limit the benefits of a species’ capacity for early, rapid, or high percentage germination (Lambrinos 2006; Ruxton and Schaefer 2012). Animal dispersers can affect the timing and extent of germination of invasive or native species (Jordaan and Downs 2012; see Ruxton and Schaefer 2012 for a review). Reported effects of breeding system and pollen vectors (wind- vs animal-pollinated species) on genetic variation (Berge et al. 1998) could also influence germination patterns.
-
Age- and genetic structure of seed banks as a predictive tool Soil seed banks are composed of seeds in varying degrees of dormancy (Benech-Arnold et al. 2000). The age structure of seeds in the seed bank also affects the requirements for germination (Rice and Dyer 2001; Gioria and Osborne 2013). Knowledge of seed production, seed dormancy, and of the density, type (transient vs persistent, sensu Thompson et al. 1997), and age structure of the seed bank can greatly improve our ability to predict the size of the seed pool that can germinate when certain environmental conditions occur (Gioria and Pyšek 2016). If such information is available from both the native and non-native distribution ranges of a species, it can provide important insights into the germination strategies that are more successful in novel environments and adaptive processes. As potential reservoirs of genetic diversity, seed banks can play an important role in maintaining or even increasing genetic variation over time (Levin 1990; Baskin and Baskin 2014), and those with a persistent seed bank are expected to be capable of more rapid or substantial responses in their timing of germination to year-to-year variation, climatic, or other global or local changes (Gioria and Pyšek 2016).
-
Temperature and water potential thresholds Minimum temperature and water potential for germination are key variables for assessing population dynamics (Bewley and Black 1994; Gardarin et al. 2011; Guillemin et al. 2013; Dürr et al. 2015) and are essential for predicting the timing and extent of seed germination as factors affecting the potential distribution of a species (Benech-Arnold et al. 2000; Gardarin et al. 2011). However, there is surprisingly little information on such thresholds for species that are invasive in non-agricultural systems (Dürr et al. 2015). Knowledge of these thresholds for a large number of species is also necessary to compare the results of different studies on the same species, as they would not depend on the specific conditions provided in each study.
Conclusions
Comparative examinations of germination traits reveal that differences in the germination behavior of alien and native species represent a mechanism that has likely contributed to the naturalization and invasiveness of many alien species. That differences in germination timing and speed are more pronounced in the invasive rather than in the native range of invasive species further indicates the importance of these germination traits as a post-introduction invasion mechanism. A capacity to germinate under a broad range of environmental conditions, allowing the exploitation of germination niches during which competition for resources is low, is more pronounced in invasive than native or non-invasive species. In contrast, the germination of a large component of seeds produced in a given season within a relatively short period is not a good indicator of invasiveness.
This knowledge has important implications for predicting the identity of alien species that could become invasive in future, as well as for identifying the conditions that are more likely to promote the germination of such species. It is also crucial for the development of sustainable control strategies of invasive species and restoration measures that prevent germination of seeds of invasive and alien species, while promoting that of seeds of desirable native species. Given rapid climatic changes, knowledge of the germination behavior of native and alien species under natural conditions is crucial for predicting future plant community dynamics. In this respect, invasion ecology would greatly benefit from rigorous studies of the dormancy and germination patterns of invasive species and from theoretical approaches that have been developed to improve weed management and crop protection. Long-term monitoring programs are required to assess the actual benefits and risks associated with different germination strategies and their long-term impact on plant community dynamics.
References
Abraham JK, Corbin JD, D’Antonio CM (2009) California native and exotic perennial grasses differ in their response to soil nitrogen, exotic annual grass density, and order of emergence. Plant Ecol 201:445–456. doi:10.1007/978-90-481-2798-6_7
Alpert P, Bone E, Holzapfel C (2000) Invasiveness, invasibility and the role of environmental stress in the spread of non-native plants. Perspect Plant Ecol Evol Syst 3:52–66. doi:10.1078/1433-8319-00004
Baker HG (1974) The evolution of weeds. Annu Rev Ecol Evol Syst 5:1–24. doi:10.1146/annurev.es.05.110174.000245
Baskin CC, Baskin JM (1988) Germination ecophysiology of herbaceous plant species in temperate region. Am J Bot 75:286–305. doi:10.2307/2443896
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography, and evolution of dormancy and germination, 2nd edn. Academic/Elsevier, San Diego
Batlla D, Benech-Arnold RL (2007) Predicting changes in dormancy level in weed seed soil banks: implications for weed management. Crop Prot 26:189–197. doi:10.1016/j.cropro.2005.07.014
Beckmann M, Bruelheide H, Erfmeier A (2011) Germination responses of three grassland species differ between native and invasive origins. Ecol Res 26:763–771. doi:10.1007/s11284-011-0834-3
Beerling DJ, Perrins JM (1993) Impatiens glandulifera Royle (Impatiens roylei Walp). J Ecol 81:367–382. doi:10.2307/2261507
Benech-Arnold RL, Sánchez RA, Forcella F, Kruk BC, Ghersa CM (2000) Environmental control of dormancy in weed seed banks in soil. Field Crops Res 67:105–122. doi:10.1016/S0378-4290(00)00087-3
Berge G, Nordal I, Hestmark G (1998) The effect of inbreeding systems and pollination vectors on the genetic variation of small plant populations within an agricultural landscape. Oikos 81:17–29. doi:10.2307/3546463
Bewley JD, Black M (1994) Seeds: physiology of development and germination. Plenum Publishing Corporation, New York
Blair AC, Wolfe LM (2004) The evolution of an invasive plant: an experimental study with Silene latifolia. Ecology 85:3035–3042. doi:10.1890/04-0341
Brändle M, Stadler J, Klotz S, Brandl R (2003) Distributional range size of weedy plant species is correlated to germination patterns. Ecology 84:136–144. doi:10.1890/0012-9658(2003)084[0136:DRSOWP]2.0.CO;2
Campbell LG, Parker RJ, Blakelock G, Pirimova N, Mercer KL (2015) Maternal environment influences propagule pressure of an invasive plant, Raphanus raphanistrum (Brassicaceae). Int J Plant Sci 176:393–403. doi:10.1086/680683
Cavers PB, Benoit DL (1989) Seed banks in arable land. In: Leck MA, Parker VT, Simpson RL (eds) Ecology of soil seed banks. Academic Press Inc., New York, pp 309–328
Cavers PB, Harper J (1966) Germination polymorphism in Rumex crispus and Rumex obtusifolius. J Ecol 54:367–382. doi:10.2307/2257955
Cervera JC, Parra-Tabla V (2009) Seed germination and seedling survival traits of invasive and non-invasive cogeneric Ruellia species (Acanthaceae) in Yucatan, Mexico. Plant Ecol 205:285–293. doi:10.1007/s11258-009-9617-0
Chrobock T, Kempel A, Fischer M, van Kleunen M (2011) Introduction bias: cultivated alien plant species germinate faster and more abundantly than native species in Switzerland. Basic Appl Ecol 12:244–250. doi:10.1016/j.baae.2011.03.001
Colautti R, Grigorovich I, MacIsaac H (2006) Propagule pressure: a null model for biological invasions. Biol Invasions 8:1023–1037. doi:10.1007/s10530-005-3735-y
Corbin JD, D’Antonio CM (2004) Competition between native perennial and exotic annual grasses: implications for an historical invasion. Ecology 85:1273–1283
Crawley MJ (1997) Plant ecology. Blackwell Science Publications, Cambridge
Daehler CC (1998) The taxonomic distribution of invasive angiosperm plants: ecological insights and comparison to agricultural weeds. Biol Conserv 4:167–180. doi:10.1016/S0006-3207(97)00096-7
Danuso F, Zanin G, Sartorato I (2012) A modelling approach for evaluating phenology and adaptation of two congeneric weeds (Bidens frondosa and Bidens tripartita). Ecol Model 243:33–41. doi:10.1016/j.ecolmodel.2012.06.009
Davis MB, Shaw RG, Etterson JR (2005) Evolutionary responses to changing climate. Ecology 86:1704–1714. doi:10.1890/03-0788
Dickson T, Hopwood J, Wilsey B (2012) Do priority effects benefit invasive plants more than native plants? An experiment with six grassland species. Biol Invasions 14:2617–2624. doi:10.1007/s10530-012-0257-2
Dillon P, Forcella F (1984) Germination, emergence, growth and flowering of two silvergrasses, Vulpia bromoides and V. myuros. Austr J Bot 32:165–175. doi:10.1071/BT9840165
Donohue K (2003) Setting the stage: plasticity as habitat selection. Int J Plant Sci 164(S3):S79–S92. doi:10.1086/368397
Donohue K (2005a) Seeds and seasons: interpreting germination timing in the field. Seed Sci Res 15:175–187. doi:10.1079/SSR2005208
Donohue K (2005b) Niche construction through phenological plasticity: life history dynamics and ecological consequences. New Phytol 166:83–92. doi:10.1111/j.1469-8137.2005.01357.x
Donohue K (2009) Completing the cycle: maternal effects as the missing link in plant life histories. Philos Trans R Soc Lond B Biol Sci 364:1059–1074. doi:10.1098/rstb.2008.0291
Donohue K, Rubio de Casas R, Burghardt L, Kovach K, Willis CG (2010) Germination, post-germination adaptation, and species ecological ranges. Annu Rev Ecol Evol Syst 41:293–319. doi:10.1146/annurev-ecolsys-102209-144715
Dreyer GD, Baird LM, Fickler C (1987) Celastrus scandens and Celastrus orbiculatus: comparisons of reproductive potential between a native and an introduced woody vine. Bull Torr Bot Club 114:260–264. doi:10.2307/2996463
Dürr C, Dickie J, Yang X-Y, Pritchard HW (2015) Range of critical temperature and water potential values for the germination of species worldwide. Agric For Meteorol 200:222–232. doi:10.1016/j.agrformet.2014.09.024
Dyer AR, Fenech A, Rice KJ (2000) Accelerated seedling emergence in interspecific competitive neighbourhoods. Ecol Lett 3:523–529. doi:10.1111/j.1461-0248.2000.00187.x
Erfmeier A, Bruelheide H (2005) Invasive and native Rhododendron ponticum populations: is there evidence for genotypic differences in germination and growth? Ecography 28:417–428. doi:10.1111/j.0906-7590.2005.03967.x
Fenesi A, Albert Á-J, Ruprecht E (2014) Fine-tuned ability to predict future competitive environment in Ambrosia artemisiifolia seeds. Weed Res 54:58–69. doi:10.1111/wre.12048
Fenner M, Thompson K (2005) The ecology of seeds, 2nd edn. Cambridge University Press, Cambridge
Ferreras AE, Galetto L (2010) From seed production to seedling establishment: important steps in an invasive process. Acta Oecol 36:211–218. doi:10.1016/j.actao.2009.12.005
Forbis TA (2010) Germination phenology of some Great Basin native annual forb species. Plant Spec Biol 25:221–230. doi:10.1111/j.1442-1984.2010.00289.x
Forcella F, Wood JT, Dillon P (1986) Characteristics distinguishing invasive weeds within Echium (Bugloss). Weed Res 26:351–364. doi:10.1111/j.1365-3180.1986.tb00718.x
Forcella F, Benech-Arnold RL, Sánchez R, Ghersa CM (2000) Modeling seedling emergence. Field Crops Res 67:123–139. doi:10.1016/S0378-4290(00)00088-5
Funk JL (2013) The physiology of invasive plants in low-resource environments. Conserv Physiol. doi:10.1093/conphys/cot026
García Y, Callaway RM, Diaconu A, Montesinos D (2013) Invasive and non-invasive congeners show similar trait shifts between their same native and non-native ranges. PLoS ONE 8(12):e82281. doi:10.1371/journal.pone.0082281
Gardarin A, Dürr C, Colbach N (2011) Prediction of germination rates of weed species: relationships between germination speed parameters and species traits. Ecol Model 222:626–636. doi:10.1016/j.ecolmodel.2010.10.005
Gerlach JD, Rice KJ (2003) Testing life history correlates of invasiveness using congeneric plant species. Ecol Appl 13:167–179. doi:10.1890/1051-0761(2003)013[0167:TLHCOI]2.0.CO;2
Gioria M, Osborne B (2009a) The impact of Gunnera tinctoria Molina (Mirbel) on soil seed bank communities. J Plant Ecol 2:153–167. doi:10.1093/jpe/rtp013
Gioria M, Osborne B (2009b) Assessing the impact of plant invasions on soil seed bank communities: use of univariate and multivariate statistical approaches. J Veg Sci 20:547–556. doi:10.1111/j.1654-1103.2009.01054.x
Gioria M, Osborne B (2010) Similarities in the impact of three large invasive plant species on soil seed bank communities. Biol Invasions 12:1671–1683. doi:10.1007/s10530-009-9580-7
Gioria M, Osborne B (2013) Biological flora of the British Isles: Gunnera tinctoria. J Ecol 101:243–264. doi:10.1111/1365-2745.12022
Gioria M, Osborne B (2014) Resource competition in plant invasions: emerging patterns and research needs. Front Plant Sci. doi:10.3389/fpls.2014.00501
Gioria M, Pyšek P (2016) The legacy of plant invasions: changes in the soil seed bank of invaded plant communities. Bioscience 66:40–53. doi:10.1093/biosci/biv165
Gioria M, Pyšek P, Moravcová L (2012) Soil seed banks in plant invasions: promoting species invasiveness and long-term impact on plant community dynamics. Preslia 84:327–350
Gioria M, Pyšek P, Osborne BA (2016) Timing is everything: does early and late germination favor invasions by alien plants? J Plant Ecol. doi:10.1093/jpe/rtw105
Godoy O, Castro-Diez P, Valladares F, Costa-Tenorio M (2009) Different flowering phenology of alien invasive species in Spain: evidence for the use of an empty temporal niche? Plant Biol 11:803–811. doi:10.1111/j.1438-8677.2008.00185.x
Goergen E, Daehler CC (2001) Reproductive ecology of a native Hawaiian grass (Heteropogon contortus; Poaceae) versus its invasive alien competitor (Pennisetum setaceum; Poaceae). Int J Plant Sci 162:317–326. doi:10.1086/319587
Grime JP (2001) Plant strategies, vegetation processes, and ecosystem properties, 2nd edn. Wiley, Chichester
Grime JP, Mason G, Curtis AV, Rodman J, Band SR (1981) A comparative study of germination characteristics in a local flora. J Ecol 69:1017–1059. doi:10.2307/2259651
Grime JP, Hodgson JG, Hunt R (2007) Comparative plant ecology: a functional approach to common British species, 2nd edn. Castlepoint Press, London
Grman E, Suding KN (2010) Within-year soil legacies contribute to strong priority effects of exotics on native California grassland communities. Restor Ecol 18:664–670. doi:10.1111/j.1526-100X.2008.00497.x
Gruberová H, Bendová K, Prach K (2001) Seed ecology of alien Bidens frondosa in comparison with native species of the genus. In: Brundu G, Brock JH, Camarda I, Child L, Wade M (eds) Plant invasions: species ecology and ecosystem management. Backhuys Publishers, Leiden, pp 99–104
Guillemin JP, Gardarin A, Granger S, Reibel C, Munier-Jolain N, Colbach N (2013) Assessing potential germination period of weeds with base temperatures and base water potentials. Weed Res 53:76–87. doi:10.1111/wre.12000
Hamilton MA, Murray BR, Cadotte MW, Hose GC, Baker AC, Harris CJ, Licari D (2005) Life-history correlates of plant invasiveness at regional and continental scales. Ecol Lett 8:1066–1074. doi:10.1111/j.1461-0248.2005.00809.x
Harper J (1977) The population biology of plants. Academic Press, London
Hejda M, Štajerová K, Pyšek P (2016) Dominance has a biogeographical component: do plants tend to exert stronger impacts in their invaded rather than native range? J Biogeogr. doi:10.1111/jbi.12801
Hierro JL, Maron JL, Callaway RM (2005) A biogeographical approach to plant invasions: the importance of studying exotics in their introduced and native range. J Ecol 93:5–15. doi:10.1111/j.0022-0477.2004.00953.x
Hierro JL, Eren Ö, Khetsuriani L, Diaconu A, Török K, Montesinos D, Andonian K, Kikodze D, Janoian L, Villarreal D, Estanga-Mollica ME, Callaway RM (2009) Germination responses of an invasive species in native and non-native ranges. Oikos 118:529–538. doi:10.1111/j.1600-0706.2008.17283.x
Hirsch H, Wypior C, von Wehrden H, Wesche K, Renison D, Hensen I (2012) Germination performance of native and non-native Ulmus pumila populations. NeoBiota 15:53–68. doi:10.3897/neobiota.15.4057
Hock M, Beckmann M, Hofmann RW, Bruelheide H, Erfmeier A (2015) Effects of UV-B radiation on germination characteristics in invasive plants in New Zealand. NeoBiota 26:21–37. doi:10.3897/neobiota.26.4405
Hsu H-M, Kao W-Y (2014) Vegetative and reproductive growth of an invasive weed Bidens pilosa L. var. radiata and its noninvasive congener Bidens bipinnata in Taiwan. Taiwania 59:119–126. doi:10.6165/tai.2014.59.119
Imbert E (2002) Ecological consequences and ontogeny of seed heteromorphism. Perspect Plant Ecol Evol Syst 5:13–36. doi:10.1078/1433-8319-00021
Johnson S (2014) Climate-induced changes in the germination phenology of Boechera stricta. Master’s thesis. University of South Carolina, Columbia. http://scholarcommons.sc.edu/etd/3020
Jordaan LA, Downs CT (2012) Comparison of germination rates and fruit traits of indigenous Solanum giganteum and invasive Solanum mauritianum in South Africa. S Afr J Bot 80:13–20. doi:10.1016/j.sajb.2012.01.007
Kardol P, Souza L, Classen AT (2013) Resource availability mediates the importance of priority effects in plant community assembly and ecosystem function. Oikos 122:84–94. doi:10.1111/j.1600-0706.2012.20546.x
Kendall S, Penfield S (2012) Maternal and zygotic temperature signalling in the control of seed dormancy and germination. Seed Sci Res 22(S1):S23–S29. doi:10.1017/S0960258511000390
Kimball S, Angert AL, Huxman TE, Venable DL (2011) Differences in the timing of germination and reproduction relate to growth physiology and population dynamics of Sonoran Desert winter annuals. Am J Bot 98:1773–1781. doi:10.3732/ajb.1100034
Körner C, Stöcklin J, Reuther-Thiébaud L, Pelaez-Riedl S (2008) Small differences in arrival time influence composition and productivity of plant communities. New Phytol 177:698–705. doi:10.1111/j.1469-8137.2007.02287.x
Kudoh H, Nakayama M, Lihová J, Marhold K (2007) Does invasion involve alternation of germination requirements? A comparative study between native and introduced strains of an annual Brassicaceae, Cardamine hirsuta. Ecol Res 22:869–875. doi:10.1007/s11284-007-0417-5
Lake J, Leishman M (2004) Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol Conserv 117:215–226. doi:10.1016/S0006-3207(03)00294-5
Lambrinos JG (2002) The variable invasive success of Cortaderia species in a complex landscape. Ecology 83:518–529. doi:10.2307/2680032
Lambrinos JG (2006) Spatially variable propagule pressure and herbivory influence invasion of chaparral shrubland by an exotic grass. Oecologia 147:327–334. doi:10.1007/s00442-005-0259-1
Laube J, Sparks TH, Estrella N, Höfler J, Ankerst DP, Menzel A (2014) Chilling outweighs photoperiod in preventing precocious spring development. Glob Change Biol 20:170–182. doi:10.1111/gcb.12360
Laube J, Sparks TH, Bässler C, Menzel A (2015) Small differences in seasonal and thermal niches influence elevational limits of native and invasive Balsams. Biol Conserv 191:682–691. doi:10.1016/j.biocon.2015.08.019
Leiblein-Wild MC, Kaviani R, Tackenberg O (2014) Germination and seedling frost tolerance differ between the native and invasive range in common ragweed. Oecologia 174:739–750. doi:10.1007/s00442-013-2813-6
Levin D (1990) The seed bank as a source of genetic novelty in plants. Am Nat 135:563–572. doi:10.1086/285062
Luo J, Cardina J (2012) Germination patterns and implications for invasiveness in three Taraxacum (Asteraceae) species. Weed Res 52:112–121. doi:10.1111/j.1365-3180.2011.00898.x
Maillet J, Lopez-Garcia C (2000) What criteria are relevant for predicting the invasive capacity of a new agricultural weed? The case of invasive American species in France. Weed Res 40:11–26. doi:10.1046/j.1365-3180.2000.00171.x
Mandák B (2003) Germination requirements of invasive and non-invasive Atriplex species: a comparative study. Flora 198:45–54. doi:10.1078/0367-2530-00075
Marushia RG, Cadotte MW, Holt JS (2010) Phenology as a basis for management of exotic annual plants in desert invasions. J Appl Ecol 47:1290–1299. doi:10.1111/j.1365-2664.2010.01881.x
Mattana E, Daws MI, Bacchetta G (2010) Comparative germination ecology of the endemic Centranthus amazonum (Valerianaceae) and its widespread congener Centranthus ruber. Plant Spec Biol 25:165–172. doi:10.1111/j.1442-1984.2010.00280.x
Meiners SJ (2005) Seed and seedling ecology of Acer saccharum and Acer platanoides: a contrast between native and exotic congeners. Northeast Nat 12:23–32. doi:10.1656/1092-6194(2005)012[0023:SASEOA]2.0.CO;2
Mihulka S, Pyšek P, Martínková J (2003) Invasiveness of Oenothera congeners in Europe related to seed characteristics. In: Child L, Brock JH, Brundu G, Prach K, Pyšek P, Wade M, Williamson M (eds) Plant invasions: ecological threats and management solutions. Backhuys Publishers, Leiden, pp 213–225
Miller T, Winn A, Schemske D (1994) The effects of density and spatial distribution on selection for emergence time in Prunella vulgaris (Lamiaceae). Am J Bot 81:1–6. doi:10.2307/2445555
Moore JE, Lacey EP (2009) A comparison of germination and early growth of four early successional tree species of the Southeastern United States in different soil and water regimes. Am Mid Nat 162:388–394. doi:10.1674/0003-0031-162.2.388
Moravcová L, Pyšek P, Jarošík V, Havlíčková V, Zákravský P (2010) Reproductive characteristics of neophytes in the Czech Republic: traits of invasive and non-invasive species. Preslia 82:365–390
Moravcová L, Pyšek P, Jarošík V, Pergl J (2015) Getting the right traits: reproductive and dispersal characteristics predict the invasiveness of herbaceous plant species. PLoS ONE 10:e0123634. doi:10.1371/journal.pone.0123634
Navie SC, Panetta FD, McFadyen RE, Adkins SW (2004) Germinable soil seedbanks of central Queensland rangelands invaded by the exotic weed Parthenium hysterophorus L. Weed Biol Manag 4:154–167. doi:10.1111/j.1445-6664.2004.00132.x
Nicotra AB, Atkin OK, Bonser SP, Davidson AM, Finnegan EJ, Mathesius U, Poot P, Purugganan MD, Richards CL, Valladares F, van Kleunen M (2010) Plant phenotypic plasticity in a changing climate. Trends Plant Sci 15:684–692. doi:10.1016/j.tplants.2010.09.008
Nikolaeva M (1977) Factors controlling the seed dormancy pattern. In: Kahn A (ed) The physiology and biochemistry of seed dormancy and germination. North-Holland Publishing Company, Amsterdam, pp 51–74
Odling-Smee FJ, Laland KN, Feldman MW (1996) Niche construction. Am Nat 147:641–648. doi:10.1086/285870
Paudel S, Battaglia LL (2013) Germination responses of the invasive Triadica sebifera and two co-occurring native woody species to elevated salinity across a gulf coast transition ecosystem. Wetlands 33:527–535. doi:10.1007/s13157-013-0410-4
Pérez-Fernández MA, Lamont BB, Marwick AL, Lamont WG (2000) Germination of seven exotic weeds and seven native species in south-western Australia under steady and fluctuating water supply. Acta Oecol 21:323–336. doi:10.1016/S1146-609X(00)01084-5
Perglová I, Pergl J, Skálová H, Moravcová L, Jarošík V, Pyšek P (2009) Differences in germination and seedling establishment of alien and native Impatiens species. Preslia 81:357–375
Perrins J, Fitter A, Williamson M (1993) Population biology and rates of invasion of three introduced Impatiens species in the British Isles. J Biogeogr 20:33–44. doi:10.2307/2845737
Probert R (2000) The role of temperature in the regulation of seed dormancy and germination. In: Fenner M (ed) Seeds: the ecology of regeneration in plant communities. CAB International, Wallingford, pp 261–292
Probert R, Smith R, Birch P (1985) Germination responses to light and alternating temperatures in European populations of Dactylis glomerata L. I. Variability in relation to origin. New Phytol 99:305–316. doi:10.1111/j.1469-8137.1985.tb03658.x
Pucheta E, García-Muro VJ, Rolhauser AG, Quevedo-Robledo L (2011) Invasive potential of the winter grass Schismus barbatus during the winter season of a predominantly summer-rainfall desert in Central-Northern Monte. J Arid Environ 75:390–393. doi:10.1016/j.jaridenv.2010.11.010
Pyšek P, Richardson DM (2007) Traits associated with invasiveness in alien plants: where do we stand? Biol Invasions 193:97–125. doi:10.1007/978-3-540-36920-2_7
Pyšek P, Křivánek M, Jarošík V (2009) Planting intensity, residence time, and species traits determine invasion success of alien woody species. Ecology 90:2734–2744. doi:10.1890/08-0857.1
Pyšek P, Manceur A, Alba C, McGregor K, Pergl J, Štajerová K, Chytrý M, Danihelka J, Kartesz J, Klimešová J, Lučanová M, Moravcová L, Nishino M, Sádlo J, Suda J, Tichý L, Kühn I (2015) Naturalization of central European plants in North America: species traits, habitats, propagule pressure, residence time. Ecology 96:762–774. doi:10.1890/14-1005.1
Radford IJ, Cousens RD (2000) Invasiveness and comparative life-history traits of exotic and indigenous Senecio species in Australia. Oecologia 125:531–542. doi:10.1007/s004420000474
Ranal MA, Santana DG (2006) How and why to measure the germination process? Rev Bras Bot 29:1–11. doi:10.1590/S0100-84042006000100002
Rees M, Long MJ (1992) Germination biology and the ecology of annual plants. Am Nat 139:484–508. doi:10.1086/285340
Rejmánek M, Richardson DM (1996) What attributes make some plant species more invasive? Ecology 77:1655–1661. doi:10.2307/2265768
Reynolds SA, Corbin JD, D’Antonio CM (2001) The effects of litter and temperature on the germination of native and exotic grasses in a coastal California grassland. Madroño 48:230–235
Rice KJ, Dyer AR (2001) Seed aging, delayed germination and reduced competitive ability in Bromus tectorum. Plant Ecol 155:237–243. doi:10.1023/A:1013257407909
Richardson DM, Van Wilgen BW, Mitchell DT (1987) Aspects of the reproductive ecology of four Australian Hakea species (Proteaceae) in South Africa. Oecologia 71:345–354. doi:10.1007/BF00378706
Richardson DM, Pyšek P, Rejmánek M, Barbour M, Panetta F, West C (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107. doi:10.1046/j.1472-4642.2000.00083.x
Roberts E, Totterdell S (1981) Seed dormancy in Rumex species in response to environmental factors. Plant Cell Environ 4:97–106. doi:10.1111/j.1365-3040.1981.tb01044.x
Ross MA, Harper JL (1972) Occupation of biological space during seedling establishment. J Ecol 60:77–88. doi:10.2307/2258041
Ruprecht E, Fenesi A, Nijs I (2014) Are plasticity in functional traits and constancy in performance traits linked with invasiveness? An experimental test comparing invasive and naturalized plant species. Biol Invasions 16:1359–1372. doi:10.1007/s10530-013-0574-0
Ruxton GD, Schaefer HM (2012) The conservation physiology of seed dispersal. Philos Trans R Soc Lond B Biol Sci 367:1708–1718. doi:10.1098/rstb.2012.0001
Sans FX, Garcia-Serrano H, Afán I (2004) Life-history traits of alien and native Senecio species in the Mediterranean region. Acta Oecol 26:167–178. doi:10.1016/j.actao.2004.04.001
Schlaepfer DR, Glaettli M, Fischer M, van Kleunen M (2010) A multi-species experiment in their native range indicates pre-adaptation of invasive alien plant species. New Phytol 185:1087–1099. doi:10.1111/j.1469-8137.2009.03114.x
Schwartz MD, Ahas R, Aasa A (2006) Onset of spring starting earlier across the Northern Hemisphere. Glob Change Biol 12:343–351. doi:10.1111/j.1365-2486.2005.01097.x
Seabloom EW, Harpole WS, Reichman OJ, Tilman D (2003) Invasion, competitive dominance, and resource use by exotic and native California grassland species. Proc Natl Acad Sci USA 100:13384–13389. doi:10.1073/pnas.1835728100
Simberloff D (2009) The role of propagule pressure in biological invasions. Annu Rev Ecol Evol Syst 40:81–102. doi:10.1146/annurev.ecolsys.110308.120304
Simons AM, Johnston MO (2000) Variation in seed traits of Lobelia inflata (Campanulaceae): sources and fitness consequences. Am J Bot 87:124–132. doi:10.2307/2656690
Skálová H, Moravcová L, Pyšek P (2011) Germination dynamics and seedling frost resistance of invasive and native Impatiens species reflect local climatic conditions. Perspect Plant Ecol Evol Syst 13:173–180. doi:10.1016/j.ppees.2011.03.005
Steadman KJ, Ellery AJ, Chapman R, Moore A, Turner NC (2004) Maturation temperature and rainfall influence seed dormancy characteristics of annual ryegrass (Lolium rigidum). Austr J Agric Res 55:1047–1057. doi:10.1071/AR04083
Stevens JM, Fehmi JS (2011) Early establishment of a native grass reduces the competitive effect of a non-native grass. Restor Ecol 19:399–406. doi:10.1111/j.1526-100X.2009.00565.x
Tecco PA, Diaz S, Cabido M, Urcelay C (2010) Functional traits of alien plants across contrasting climatic and land-use regimes: do aliens join the locals or try harder than them? J Ecol 98:17–27. doi:10.1111/j.1365-2745.2009.01592.x
Thompson K, Grime PJ (1979) Seasonal variation in seed banks of herbaceous species in ten contrasting habitats. J Ecol 67:893–921. doi:10.2307/2259220
Thompson K, Band SR, Hodgson JG (1993) Seed size and shape predict persistence in soil. Funct Ecol 7:236–241. doi:10.2307/2389893
Thompson K, Bakker JP, Bekker RM (1997) Soil seed banks of NW Europe: methodology, density and longevity. Cambridge University Press, Cambridge
van Kleunen M, Johnson SD (2007) South African Iridaceae with rapid and profuse seedling emergence are more likely to become naturalized in other regions. J Ecol 95:674–681. doi:10.1111/j.1365-2745.2007.01250.x
van Kleunen M, Dawson W, Schlaepfer D, Jeschke JM, Fischer M (2010) Are invaders different? A conceptual framework of comparative approaches for assessing determinants of invasiveness. Ecol Lett 13:947–958. doi:10.1111/j.1461-0248.2010.01503.x
Venable DL (2007) Bet hedging in a guild of desert annuals. Ecology 88:1086–1090. doi:10.1890/06-1495
Venable DL, Brown JS (1988) The selective interactions of dispersal, dormancy and seed size as adaptations for reducing risk in variable environments. Am Nat 131:360–384. doi:10.1086/284795
Verdú M, Traveset A (2005) Early emergence enhances plant fitness: a phylogenetically controlled meta-analysis. Ecology 86:1385–1394. doi:10.1890/04-1647
Volis S, Bohrer G (2013) Joint evolution of seed traits along an aridity gradient: seed size and dormancy are not two substitutable evolutionary traits in temporally heterogeneous environment. New Phytol 197:655–667. doi:10.1111/nph.12024
Volis S, Ormanbekova D, Yermekbayev K (2015) Role of phenotypic plasticity and population differentiation in adaptation to novel environmental conditions. Ecol Evol 5:3818–3829. doi:10.1002/ece3.1607
Von Holle B, Simberloff D (2005) Ecological resistance to biological invasion overwhelmed by propagule pressure. Ecology 86:3212–3218. doi:10.1890/05-0427
Wainwright CE, Cleland EE (2013) Exotic species display greater germination plasticity and higher germination rates than native species across multiple cues. Biol Invasions 15:2253–2264. doi:10.1007/s10530-013-0449-4
Wainwright CE, Wolkovich EM, Cleland EE (2012) Seasonal priority effects: implications for invasion and restoration in a semi-arid system. J Appl Ecol 49:234–241. doi:10.1111/j.1365-2664.2011.02088.x
Walck J, Hidayati SN, Dixon KW, Thompson K, Poschlod P (2011) Climate change and plant regeneration from seed. Glob Change Biol 17:2145–2161. doi:10.1111/j.1365-2486.2010.02368.x
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395. doi:10.1038/416389a
Weiner J (1990) Asymmetric competition in plant populations. Trends Ecol Evol 5:360–364. doi:10.1016/0169-5347(90)90095-U
Williamson M (1996) Biological invasions. Chapman and Hall, London
Willis CG, Ruhfel BR, Primack RB, Miller-Rushing AJ, Losos JB, Davis CC (2010) Favorable climate change response explains non-native species’ success in Thoreau’s woods. PLoS ONE 5(1):e8878. doi:10.1371/journal.pone.0008878
Willis CG, Baskin CC, Baskin JM, Auld JR, Venable DL, Cavender-Bares J, Donohue K, Rubio de Casas R, NESCent Germination Working Group (2014) The evolution of seed dormancy: environmental cues, evolutionary hubs, and diversification of the seed plants. New Phytol 203:300–309. doi:10.1111/nph.12782
Wolkovich EM, Cleland EE (2014) Phenological niches and the future of invaded ecosystems with climate change. AoB Plants 6:plu013. doi:10.1093/aobpla/plu013
Acknowledgements
We thank John Cardina, Wen-Yuan Kao, James Moore, Eszter Ruprecht, and Hana Skálová for providing additional information on their studies, and Tony Dixon (Norwich) for improving our English. We also thank Dan Simberloff and Laura Meyerson for valuable comments on the manuscript. MG and PP were funded by Projects No. 15-13491S (Czech Science Foundation), 14-36079G Centre of Excellence PLADIAS (Czech Science Foundation), and long-term research development Project RVO 67985939 (The Czech Academy of Sciences). PP acknowledges support of a Praemium Academiae award from The Czech Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Gioria, M., Pyšek, P. Early bird catches the worm: germination as a critical step in plant invasion. Biol Invasions 19, 1055–1080 (2017). https://doi.org/10.1007/s10530-016-1349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-016-1349-1