Abstract
Non-indigenous invasive aquatic plants affect macrophyte community composition, structure and abundance, and are recognised as one of the major causes of biodiversity loss in aquatic ecosystem. Their mass development poses serious ecological, sociological and economical threats in many countries. We investigated biological, physicochemical, hydromorphological and geographical data from 447 Polish lowland lakes to recognise the phenomenon of invasive macrophyte species and to identify environmental factors that determine their occurrence and abundance. We focused on Elodea canadensis (EC) as the most common and widespread alien hydrophyte in Poland. Phytocoenoses of EC were present in 40 % of analysed lakes. The area occupied by EC ranged from 2.3 to 5.5 % of the total vegetated area per year, on average, with a maximum of 37.8 %. A comparison between two sub-periods (2005–2009 and 2010–2013) revealed a lack of increase in EC mean abundance within the analysed period. Likewise, no evidence of EC effects on the native flora, its richness and diversity and no effects on the ecological status of the lakes were found. In the analysed period, EC did not demonstrate invasive character in Polish lakes. The habitat preference of EC was biased towards larger and deeper lakes located at higher altitudes, with longer water retention times, better water quality and better ecological status based on macrophytes compared with non-invaded lakes. We found altitude and water quality as the main determinants of EC occurrence within the sampled area. We suggest that both climate warming and accelerated eutrophication may contribute to reducing EC spread in Europe observed during recent decades.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The problem of the appearance of alien species and their impact on native biological assemblages has become especially weighty during the past century, as the development of transport and international trade has contributed to the rapid and pervasive spread of species beyond their natural range of occurrence (Hussner et al. 2010; Lenda et al. 2014). In the past two decades, there has been a marked increase in the number of scientific papers concerning the growing threat to native fauna and flora from invasive alien species (Thomaz et al. 2015). One of the most common definitions used in many ecological studies, though relatively vague, describes alien invasive species as those that colonise and spread into new territories where they have never occurred before (definition overview in Pyšek 1995). In their new territories, alien species can expand their populations either in terms of geographical area and/or in terms of increasing frequency and density. These species also can be referred to as aggressive invaders, as defined by Simberloff and Rejmánek (2011). According to recent European Union legislation, invasive alien species are defined as those that are, firstly, outside their natural distribution area, and that, secondly, threaten or adversely impact biodiversity and related ecosystem services (EU 2014).
The rates of geographical spread and successful establishment of species that are non-native to certain regions are determined by an array of biotic and abiotic factors that include, among others, the number and size of release events, i.e. propagule pressure (Lockwood et al. 2005; Redekop et al. 2016), the availability of vectors and pathways for propagule dispersal (Panov et al. 2009; Redekop et al. 2016), proper habitat conditions such as nutrient and light availability (Thiébaut 2005) or climate (Chytrý et al. 2009), as well as favourable ecological interactions (overview in Fleming and Dibble 2015). The introduction of submerged weeds into a waterbody largely depends on human activities (Lockwood et al. 2005; Hussner et al. 2010), and the more propagules are introduced, the more likely the dispersal of the species within and between waterbodies and the higher the likelihood of successful invasion and establishment (Lockwood et al. 2005; Redekop et al. 2016). Moreover, ecosystems disturbed by human pressure are more prone to invasions than stable, undisturbed ecosystems (Hussner et al. 2010), because the ecological niche of lost macrophytes can be easily colonised by other species (Bakker et al. 2013), including exotic ones (Zehnsdorf et al. 2015). Thus, waterbodies more exposed to anthropogenic influences (trade, tourism pressure, increase in population density and increased eutrophication) are more likely to be invaded than less affected ones. The dispersal of species between waterbodies is strongly related to hydrological connectivity (Panov et al. 2009), with rivers and canals serving as invasion corridors. Thus, it can be assumed that flowing waters (including open lakes) would be more prone to invasion than, for example, more isolated lakes with closed basins.
The ecological impact posed by alien species includes an array of mechanisms and scenarios, of which competition is widely regarded as one of the most important for aquatic plant communities (Fleming and Dibble 2015). Mass development of a successfully established plant invaders modifies macrophyte community composition, structure and abundance, and leads to biodiversity loss that is recognised as one of the major negative environmental outcomes of invasion. Apparently, not all alien species have a negative impact on local populations or ecosystems. Some of them may become naturalised and remain low in abundance, while the others (aggressive invaders) may outcompete native vegetation and cause substantial problems related to ecosystem functioning (Hussner et al. 2010). While invasive alien species may affect the taxonomic composition and functioning of ecosystems, they are also one of the subjects of interest in the Water Framework Directive (WFD; EU 2000) and must be taken into account when assessing the ecological status of surface waters, including lakes.
In a comprehensive overview of the appearance of alien plant species in surface waters in Europe, Hussner (2012) reported the highest numbers of alien aquatic plant species in Italy and France, followed by Germany, Belgium, Hungary and the Netherlands, while in northern and eastern Europe the problem of invasive species seems to be marginal. In Polish waters, the number of alien plant species was negligible (Hussner 2012), especially compared with neighbouring countries, and the most common and widespread alien hydrophyte is E. canadensis (Tokarska-Guzik 2005b). Thus, the question appears: if alien species are common in neighbouring countries, why are they not so in Poland?
The low number of alien aquatic macrophyte species in Poland (Tokarska-Guzik 2005a; Hussner 2012) may suggest that some of the conditions favouring successful invasion may not be met. To resolve this, we performed a comprehensive overview of the occurrence and abundance of invasive macrophyte species in Polish lakes using a relatively long-term and geographically widespread dataset. We investigated biological, physicochemical, hydromorphological and geographical data from 447 Polish lowland lakes (571 lake-years when including repeated surveys) surveyed for macrophytes during much of the past decade (in the years 2005–2013) within the lake monitoring programme and other national research projects. All of the analysed lakes are natural and are considered significant waterbodies sensu WFD (i.e. they have an area greater than 0.5 km2), and constitute almost half (43 %) of the significant lake waterbodies in Poland (Kolada et al. 2005).
The primary goal of our study was to explore whether alien plant arrivals have an impact on the structure and functioning of macrophytes in Poland with a special focus on: (1) E. canadensis as the most common invader in inland waters in the country and (2) lakes as a separate water category according to the WFD. We aimed at: (1) defining the range of invasion of E. canadensis in Polish lakes, its geographical spread and abundance; (2) determining the habitat requirements of E. canadensis, to identify the environments that are most likely to be invaded; (3) exploring the effects of E. canadensis on the taxonomic richness and diversity of lacustrine flora, particularly in the context of the threat that it poses to the lake ecological status, as required by the WFD.
Materials and methods
Study system
At the beginning of the millennium, Canadian waterweed (Elodea canadensis Michaux) was reported as the only hydrophyte of among more than 300 plant species that are considered alien arrivals since the fifteenth century and are naturalised in Poland (Tokarska-Guzik 2005a). Since then, reports of the occurrence of other alien hydrophytes (i.e., Azolla filiculoides Lam., Hygrophila polysperma (Roxb.) T. Anderson, Lemna turionifera Landolt and Vallisneria spiralis L.) from Polish waters have also become available in the recent literature (Gąbka and Owsianny 2009; Szczęśniak et al. 2009; Tokarska-Guzik et al. 2014). Moreover, to our knowledge, in the lake monitoring database during the period of 2007–2013, at least one record of Elodea nuttallii St John (Lake Ryńskie, surveyed in 2010) and one record of Vallisneria spiralis (Lake Pątnowskie, surveyed in 2013) were noted (A. Kolada unpubl. obs.). The observations of the other alien hydrophyte species in Polish waters seem to be fortuitous at the present time, however, while E. canadensis remains the most common and widespread alien hydrophyte in Poland (Tokarska-Guzik 2005b).
Canadian waterweed (Elodea canadensis Michaux, hereafter referred to as EC) is a North American native species that became invasive in Europe, Asia and Australia. EC was first recorded in Europe in 1836 in Great Britain (Moore and More 1866, reviewed in Simpson 1984), and since then it has greatly expanded and colonised many running and standing waters. Recently, it has become the most widespread alien aquatic plant, reported in 41 European countries (Hussner 2012), and is among the most common alien species in the waters of north-western Europe (Hilt and Gross 2008; Hussner et al. 2010). In France and Germany, a mass development of EC has been observed (Hilt et al. 2006; Hussner et al. 2010), posing serious ecological, sociological and economical threats (Zehnsdorf et al. 2015).
The first report of EC in Poland dates to 1867 (Abromeit et al. 1898, after Tokarska-Guzik 2005b), and since then it has spread almost throughout the entire country (with the exception of mountainous areas), inhabiting all types of surface waters (Tokarska-Guzik 2005b). Since the early 1950s, EC has been recognised as a common component of aquatic flora in Poland (Kornaś and Medwecka-Kornaś 1957; Bernatowicz 1960; Rejewski 1984; Kłosowski 2006; Jabłońska and Kłosowski 2015) and is currently a naturalised species throughout the country. Despite its widespread appearance in Polish waters, to our knowledge, no comprehensive overview of EC frequency, abundance and invasion in lakes has been carried out. Neither the effects of EC on species richness and diversity nor its effects on lake ecological status have been evaluated until recently.
Lakes and lake-years
Data on aquatic vegetation, morphology and water physicochemical properties from 447 Polish lowland lakes surveyed during the period 2005–2013 were analysed. Each year a different set of lakes was surveyed (from 19 lakes in 2005 to 91 in 2011; 63 lakes per year on average). Of the 447 lakes, 345 were surveyed once during the analysed period; 83 lakes were studied in 2 years, 16 lakes were studied in three years and for three lakes surveys of 4 years (with 1–7 years between surveys) were available. The final dataset was composed of complete data from 571 lake-years (surveys). All of the studied lakes are natural, lowland (<250 m a.s.l.), with non-coloured medium- to high-alkalinity waters (>0.3 meq L−1), but they differ in morphometry and trophy (Table 1).
Biological and environmental data
Data on macrophytes were collected within the national lake monitoring programme (513 lake surveys from the period of 2007–2013), the Polish–Norwegian Research Fund (PNRF) Project ‘deWELopment’ (11 lakes surveyed in 2009; Kolada et al. 2014) and other national research projects (47 lakes surveyed in 2005–2006; Ciecierska 2008; Kolada 2010).
The aquatic vegetation of all the lakes was surveyed once a year during the peak of the vegetation season (June–September) using a unified field survey procedure based on the transect method (Ciecierska and Kolada 2014). The observations of aquatic vegetation were made along 30 m wide belt transects set perpendicularly to the shoreline and covering the whole vegetated zone, from the shoreline to the outer limit of macrophyte growth. The observations were made by wading and boating with the use of a rake and a bathyscope. At each transect the maximum colonisation depth, the mean vegetation coverage and the relative coverage of all the submerged, floating-leaved and emergent plant communities (syntaxa) were determined. Data collected within transects were then readjusted to the whole-lake scale. For the identification and classification of aquatic plant communities, the phytosociological approach was applied (Braun-Blanquet 1964). The plant communities were identified on the basis of their dominant species, and the syntaxonomic systems established by Brzeg and Wojterska (2001) and Matuszkiewicz (2002) were adopted.
Data on the water quality of 11 lakes were collected within the PNRF project ‘deWELopment’ (Soszka and Ochocka 2011), while those for all the other lakes used in the study were collected within the national lake monitoring programme; the latter data are owned by the Chief Inspectorate for Environmental Protection in Poland. The lakes were sampled for water physicochemistry and chlorophyll a in the same year as the vegetation surveys were conducted, four times during the vegetation season, from March to October. In this study, the seasonal means of pH, alkalinity, water temperature at 1 m depth, concentrations of total phosphorus, total nitrogen, chlorophyll a and water transparency as measured with a Secchi disk were analysed (Table 1). Morphological, hydrological and geographical data for all of the analysed lakes were derived from the national monitoring database. In this analysis, data on lake surface area, maximum and mean depths, water volume, water retention time (time needed for complete water exchange), latitude, longitude and altitude of the lake centre point were used (Table 1).
Statistical analyses
The spatial distribution, relative abundance and habitat requirements of EC in lakes were analysed. The spatial distribution of EC in lakes throughout the country was explored using GIS tools (ArcGIS for Desktop Advanced version 10.2). The average area occupied by EC as a proportion of the total vegetated area (hereafter referred to as %area_EC) in subsequent years was compared using the Kruskal–Wallis test.
Links between environmental parameters and the taxonomic composition of lakes invaded or not invaded by EC were tested with the use of canonical community ordination techniques in the CANOCO 4.5 software (ter Braak and Šmilauer 2002). Seventy-eight plant communities of submerged and floating-leaved vegetation identified in all the lakes (Table S1 in Online Resources) were used to estimate the length of a gradient in the standard deviation of species (or plant communities in this case) turnover using detrended correspondence analysis (DCA). As the gradient of biological data tested was sufficiently long (4.27 SD), the unimodal context was assumed and canonical correspondence analysis (CCA) with untransformed species data was further applied to identify environmental variables shaping the vegetation pattern in the analysed lakes (ter Braak and Šmilauer 2002).
Pre-selection of the environmental variables was performed by applying principal component analysis (PCA). Fifteen abiotic parameters, including geographical distribution, lake morphology, hydrography and selected water quality indicators were tested. The parameters are listed in Table 1. The data were tested for normal distribution and data on TP, TN and SD were log-transformed. The PCA produced five significant principal components (eigenvalues >1.0), with the first three components together explaining 57.5 % of the total lake variance (26.1, 18.4 and 12.9 %, respectively). The variables which were not inter-correlated (Spearman’s rho |<0.7|) and were best correlated with the first three principal components (factor loadings |>0.6|), were selected and used in the CCA. The distribution of lakes invaded or not invaded by EC in an ordination space described by the pre-selected environmental determinants was compared. To identify plant communities concomitant with EC phytocoenoses, the distribution of plant communities in the CCA ordination space (inter-species distance) was analysed.
To characterise EC habitat requirements in a more quantitative manner, the environmental factors listed in Table 1 were compared between EC invaded and non-invaded lakes using the non-parametric Mann–Whitney U-test, with the median as a measure of the central tendency. Moreover, the ecological status of EC invaded and non-invaded lakes was compared according to the Ecological State Macrophyte Index (ESMI; Ciecierska and Kolada 2014). The ESMI is the WFD-compliant multimetric for assessing the ecological status of lakes exposed to eutrophication pressure applied in lake monitoring in Poland. It evaluates the state of macrophyte composition and structure in relation to the state expected in undisturbed (reference) conditions. The values of the ESMI range from 0 to 1, where 1 describes almost pristine and 0 the most disturbed conditions. Values below 0.410 indicate lakes in unacceptable conditions (lakes requiring restoration measures), and values above 0.680 indicate lakes with very good ecological status based on their macrophytes.
To investigate the current trends in EC performance in Polish lakes, of the 102 lakes surveyed more than once during the analysed period, 45 were selected for which at least one survey during the first (2005–2009) and the second sub-period (2010–2013) was available and where EC was noted at least once. For those of the 45 lakes that were surveyed more than once during one or both sub-periods, for the first sub-period the oldest and for the second sub-period the most recent survey was used. For each of the selected 45 pairs of surveys, comparisons of the share of the area inhabited by EC and the main richness, diversity and evenness measures (Heip et al. 1998) as well as the Ecological State Macrophyte Index (Ciecierska and Kolada 2014) between the two sub-periods were conducted using paired sample t tests (repeated measures with normal distribution; for all indices in both sub-periods Shapiro–Wilk W > 0.05). The analysed measures included the total numbers of hydrophyte communities (syntaxa; S), the Shannon–Wiener diversity index (H′ with natural logarithm as a base; Shannon and Weaver 1949), the Simpson’s index of diversity (1 − λ; Simpson 1949; Pielou 1975) and the Pielou’s index of evenness (Pielou 1975). Statistical analyses were performed using STATISTICA version 10.0 software (StatSoft Inc. 2011).
Results
Distribution and abundance
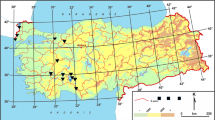
The distribution of EC in the analysed lakes was limited to the central-north and lowland (≤242 m a.s.l.) part of the country (Fig. 1; Table 1) as all of the Polish lakes with an area greater than 0.5 km2 are located there, within the range of the Baltic glaciation (Kolada et al. 2005). The phytocoenoses of EC were present in 178 of 447 lakes (40 %) and in 212 of 571 surveys (37 %) (Fig. 1). The number of lakes surveyed each year ranged from 19 in 2005 to 91 in 2011, and the number of lakes where EC was present ranged from 9 in 2006 to 35 in 2010 (Fig. 2a). The proportion of lakes with EC in a set of lakes surveyed in subsequent years was relatively stable and ranged from 30 % in 2007 to 58 % in 2005, most frequently being around 36 % (90th percentile). The proportion of the total vegetated area occupied by EC phytocoenoses (%area_EC) was relatively low, ranging between 0.005 and 37.8 % of the total area covered by hydrophyte communities (3.5 % on average in total; Fig. 2b). Among the surveys where EC was noted (n = 212), no statistically significant differences in %area_EC among subsequent years (Kruskal–Wallis H (8;212) = 2.50, p = 0.962; Fig. 2b) were found.
The total number of lakes surveyed (grey bars) and the number of lakes where the phytocoenoses of E. canadensis were noted (black bars) in subsequent years of the analysed period (upper panel, a) and the area occupied by phytocoenoses of E. canadensis as proportion of the total vegetated area (%area_EC) in subsequent years of the analysed period (lower panel, b); the whisker-plots in the lower panel (b) present the quartile range with the median (boxes), non-outlier range (whiskers), outlying (circles) and extreme values (stars)
The lakes where EC was found, although located within lowlands, were clustered in areas at considerably higher altitudes (120 m a.s.l. on average) than those not invaded by EC (85 m a.s.l. on average; Fig. 3; Table 1). Of the 241 lakes located below 100 m a.s.l., EC was found in 63 (26 %), whereas of the 206 lakes located above 100 m a.s.l., 115 (56 %) were invaded by EC. Lakes invaded by EC were also more often located in northern and eastern areas compared with lakes where EC was not identified (Table 1).
Habitat requirements
The nine environmental variables used in the CCA (listed in Fig. 4a) explained 7 % of the total variance in the lacustrine vegetation of the analysed lakes (total inertia 12.813, sum of all canonical eigenvalues 0.842). The first two axes of the CCA ordination accounted for 45 % of the variation in the 78 plant communities with respect to the environmental variables tested. The first axis was most strongly correlated with water transparency (R = 0.61), TN (−0.44), TP (−0.40), maximum depth (0.35) and water retention time (0.34), while the second axis was correlated with altitude (−0.41) and longitude (−0.11; Fig. 4a). In the CCA ordination space, the lakes invaded by EC (Fig. 4a, black circles) were clearly shifted towards higher altitudes and were characterised by greater depth, greater water volume, longer retention time and higher water transparency compared to the non-invaded lakes (Fig. 4a, open circles).
The CCA ordination of the lakes invaded (black circles) and not invaded by E. canadensis (open circles) (left panel, a) and 78 plant communities identified in the analysed lakes (right panel, b); the eigenvalues of axes 1 and 2 (CC1 and CC2) were 0.25 and 0.13, respectively, and the total inertia was 12.8; key to the short codes of the environmental factors in the left panel (a) is included in Table 1 and to the plant community abbreviations in the right panel (b) is included in Table S1 in Online Resources; different fonts in the right panel indicate ecological groups of plant communities: italic underlined—communities of stoneworts, italic bold—isoetids, regular—bryophytes, italic—angiosperms (excluding isoetids), EC (ELOD CANA) is marked with regular bold font
Likewise, the comparison of hydromorphological and water quality properties between invaded and non-invaded lakes confirmed that, in our study, EC was more likely to inhabit larger and deeper lakes with longer water retention times, lower seasonal mean water temperatures and better water quality than non-invaded lakes (statistically highly significant differences in almost all of the features analysed except for pH; Table 1).
Syntaxonomic composition
In the 571 lake-years analysed, 78 hydrophyte communities (syntaxa) were identified, while in 212 surveys where EC was identified, 70 hydrophyte communities were recorded (Table S1 in Online Resources). The CCA revealed a distinct horizontal gradient in the distribution of the plant communities, which was related to water trophy, with syntaxa more sensitive to eutrophication located on the right side of the ordination diagram and more tolerant syntaxa clustered on the left side. Moreover, the positions of syntaxa preferring higher altitudes were shifted towards the lower part of the diagram (Fig. 4b).
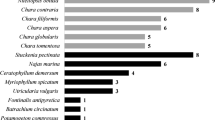
Phytocoenoses of EC were most frequently accompanied by the phytocoenoses of submerged angiosperms typical of mesotrophy and meso-eutrophy (Myriophyllum spicatum, M. verticillatum, Najas marina, Ranunculus circinatus, R. trichophyllus, Potamogeton natans, P. nodosus, Hippuris vulgaris, Callitriche cophocarpa and Lemna trisulca) and more tolerant species of stoneworts (e.g., Nitellopsis obtusa, Chara fragilis, Ch. tomentosa, Ch. aspera and Ch. vulgaris) (Table S1).
The lakes where EC was found had better ecological status based on their macrophyte index ESMI (the median value of ESMI was 0.507, which corresponds to good ecological status; n = 172) compared with lakes where EC was not identified (the median ESMI was 0.360, which corresponds to moderate ecological status; n = 307; Z = − 8.27, p < 0.0001, based on the Mann–Whitney U-test).
Effect on native flora
The comparison of the %area_EC in 45 lakes that were surveyed at least once in each sub-period (2005–2009 and 2010–2013) revealed that the EC mean abundance did not change significantly between the sub-periods (Table 2). Likewise, the syntaxonomic diversity (H′, 1 − λ) and evenness (J′) measures as well as ecological status based on the ecological state macrophyte index (ESMI) did not differ significantly between the sub-periods, though the syntaxonomic richness (S) was significantly higher in the second than in the first sub-period (Table 2).
Discussion
What is the scale of the spread and abundance of EC in Polish lakes?
The appearance of most of the alien invasive species in many European countries is fairly well documented (presence/absence data), but for the vast majority of plant invasions, studies measuring the rate of invasion in quantitative terms are scarce or absent (Pyšek 1995). The spread of EC in Poland during the last half-century and its current distribution are well documented (Tokarska-Guzik 2005a, b; Tokarska-Guzik et al. 2008; Zając et al. 2011), and these studies indicate a rapid and wide proliferation of EC in Polish waters, suggesting a serious environmental problem. The species has been classified as a transformer, i.e. an invasive plant that changes the character, condition, form or nature of ecosystems (Richardson et al. 2000). It is considered invasive on the national and international scales, with a large increase in localities, and an increase in abundance in existing localities and in colonised new localities (Tokarska-Guzik 2005a, b).
Our study provides the first comprehensive overview of the appearance of EC and its impact on native flora in a specific category of surface waters—lakes constituting significant waterbodies sensu WFD. This overview has been made on the basis of actual field surveys conducted using a standardised sampling procedure (Ciecierska and Kolada 2014) from almost five hundred lakes during a period of almost a decade (2005–2013). Concerning this specific water category, our results do not fully support the observations of other authors about the invasiveness of EC in Poland. Although we do not question the extensive geographical spread of the species throughout the country, we did not observe its mass development nor its threat to plant diversity in lakes. Therefore, in our opinion, the species cannot be considered an aggressive invader sensu Simberloff and Rejmánek (2011) nor a transformer sensu Richardson et al. (2000), at least not in lakes. This inconsistency between ours other authors’ findings may be explained as follows. Firstly, our study addressed significant lake waterbodies (i.e. lakes with a surface area greater than 0.5 km2) and we cannot make conclusions about EC invasiveness in smaller lakes or in any other type of surface waters where EC may be much more aggressive. Secondly, most overviews of EC distribution in Poland have been made on the basis of the ATPOL database (Atlas of distribution of vascular plants in Poland; Zając 1978) where the basic plant mapping units are 10 × 10 km squares (Tokarska-Guzik 2005a, b; Tokarska-Guzik et al. 2008; Zając et al. 2011). This cartogram method provides information about whether the species was present or absent within a unit, but based on this information, little can be said about the species occurrence in any given water category (whether the species was found in a river, stream, canal, ditch, lake or pond) or about its abundance in a particular waterbody. Hence, the results of previous works on EC performance in Poland and ours are difficult to compare, as EC may be more aggressive in certain water categories than in others.
The hydrological connectivity of a waterbody strongly determines its exposure to invasion (Panov et al. 2009). We expected that lakes with shorter retention times (more river-like, hence, more exposed to flushing and influences of tributaries) would be more exposed to invasion due to higher propagule dispersal and the greater probability of release events compared with more isolated lakes with low rates of water exchange. Surprisingly, EC was more likely to inhabit lakes with longer water retention time (2 years on average; Table 1) compared with the non-invaded lakes (approximately ten months, on average). Lakes with longer water retention times are usually located away from the main waterway networks and are less exposed to human activities, and hence to potential release events. Intense human pressure can largely explain the invasion success of EC in western Europe (Hussner et al. 2010). Polish waters are less affected by anthropogenic influences (Hussner et al. 2010). Therefore they can most likely be assumed to be relatively stable ecosystems, with high redundancy and high functional, phylogenetic and species diversity (Fleming and Dibble 2015), thus being less susceptible to invasion. Lower anthropogenic pressure could be one of the explanations for the generally low numbers of non-indigenous species in Poland and the lower invasiveness of those alien species that have already arrived.
Which environmental factors promote EC occurrence in Polish lakes?
The environmental factors determining aggressive or non-aggressive behaviour of EC in different geographical areas are not fully understood (Mjelde et al. 2014). Light and water temperature as well as nutrition, sediment composition and inorganic carbon availability are foremost among the factors affecting the productivity of submerged macrophytes such as EC (Barko et al. 1986). Chytrý et al. (2009) pointed to the climatic reasons as one of the major factors that explain the clear differences in the number of aquatic plant species between different parts of Europe. This was also emphasised by Hussner (2012). These authors pointed in particular to a longer growing season and milder winter temperatures favouring the invasion of exotic species in countries of the south-western region, in contrast to countries of the north-eastern region with harsher climate conditions. Based on a mesocosm study, Zhang et al. (2015) demonstrated the enhanced growth of EC with increasing temperature in autumn, suggesting that EC will be a better competitor in a warmer climate. Riis et al. (2012) found that EC exhibited high plasticity in its responses to either low or high temperatures and light availability, which allowed it to be an early coloniser and generalist, thus promoting its invasiveness. In our study, the distribution of EC clearly related to altitude (Fig. 3), and, therefore, the thermal conditions. Lakes invaded by EC were located at higher altitudes (120 m a.s.l., on average, Table 1) and had longer periods with sub-zero temperatures (more than 60 days; Lorenc 2005) than those where EC was not found (85 m a.s.l. and <60 days, on average, respectively). Additionally, phytocoenoses of EC occurred in lakes with lower average seasonal surface water temperatures than non-invaded lakes (Table 1). All of the abovementioned characteristics indicate a preference of EC for lower temperatures within the sampled areas. Our findings are consistent with those reported by Greulich and Trémolières (2006), who demonstrated that in the Alsatian Rhine floodplain, EC occurred in waters with temperatures that were significantly lower than those inhabited by two other alien species of Elodea (E. nuttallii and E. ernstiae).
Accelerated eutrophication is considered one of the crucial factors favouring the invasion of alien species (Hussner et al. 2010). Canadian waterweed is known to have a wide ecological amplitude, colonising a wide range of aquatic environments, extending from peaty to calcareous sites and from mesotrophic to eutrophic conditions (Pokorný et al. 1984; Madsen et al. 1991; Thiébaut 2005; Hérault et al. 2008). In our study, although all of the sampled waterbodies were more or less eutrophic (Table 1), the occurrence of EC was clearly associated with waters of better quality, lower nutrient concentrations (concentrations of TP = 43 µg L−1 and TN = 1.1 mg L−1, on average; Table 1), higher visibility (2.5 m of Secchi disk visibility; Table 1) and better ecological status (ESMI = 0.507, on average) than waters not invaded by EC (respectively, TP = 64 µg L−1, TN = 1.7 mg L−1, SD = 1.3 m and ESMI = 0.360, on average). Likewise, in the Alsatian Rhine floodplain, EC was demonstrated to prefer waters with low to medium nutrient concentrations, significantly less rich in phosphate and ammonium than waters inhabited by other alien invaders, E. nuttallii and E. ernstiae (Greulich and Trémolières 2006). Requirements concerning water quality may also explain the EC preference for larger and deeper lakes observed in our study (Table 1), as such ecosystems are usually less eutrophied due to their larger water volume acting as a solvent for contaminants. The EC preference for less eutrophied waters observed in our study enabled us to suggest that global eutrophication may contribute to the reduction of EC proliferation in European waters and can explain to a certain extent the stabilisation of its invasion or even its withdrawal from some areas that has been observed in recent decades (Hussner 2012; Mjelde et al. 2012; Brundu 2015). It might be that in Poland EC previously inhabited more lakes but has withdrawn from some of them due to the eutrophication process. This hypothesis, however, is difficult to verify due to the lack of comparable data from the past.
Does EC affect taxonomic richness and diversity of native flora in Polish lakes?
Phytocoenoses of EC appeared to share habitat requirements with and, therefore, were most frequently accompanied by the phytocoenoses of stoneworts and submerged angiosperms (Fig. 4b, Table S1), which in general are considered relatively sensitive to eutrophication, and hence are treated as ‘valuable’ (usually found in un-impacted ecosystems, typical of lakes in good ecological condition; Ciecierska and Kolada 2014). This implies that, when highly invasive, EC could pose a potential threat to the most valuable elements of Polish aquatic flora. Although EC was identified in 40 % of the analysed lakes, in none of these lakes the species predominated over phytocoenoses of native species. Usually it occupied a few per cent of the total vegetated area, while a maximum share of 37.8 % was noted incidentally (Fig. 2b). The comparison of the syntaxonomic diversity and richness measures between the two sub-periods of the analysed period in the lakes invaded by EC showed no negative effects of the alien species on native flora, its richness, diversity and ecological status (Table 2). Because a relatively small subset of the lakes in our study were surveyed for macrophytes in both sub-periods (n = 45) and over a relatively short period (9 years), the study may be considered to be too limited to adequately mirror general long-term tendencies in EC behaviour. However, examples from other countries show that EC can dominate lake phytolittoral within a few years of invasion [e.g., Lake Steinsfjord in Norway (Mjelde et al. 2012)]. Thus, we assumed that if EC were going to develop dramatically, as seen in other studies, it probably would have done so during the nine-year period; hence, our findings can be considered illustrative to some extent.
The behaviour of EC varies across European regions (Mjelde et al. 2014). While in some countries, such as Germany, France and Norway, this species is seen as aggressive, rapidly expanding and displacing native species (Thiébaut 2005; Hilt and Gross 2008; Hussner et al. 2010; Mjelde et al. 2012; Zehnsdorf et al. 2015), in others, such as Slovenia, it remains non-aggressive and, even if recorded, it does not increase in abundance (Greulich and Trémolières 2006; Kuhar et al. 2010; Mjelde et al. 2014). Our results do not confirm the invasive nature of EC in Polish lakes and point to its unaggressive incorporation into native plant assemblages.
To conclude, our study indicates that populations of EC in Polish lakes are well established and stable, without evidence for further proliferation or significant negative impacts on native lacustrine flora. We found altitude and water quality to be the main determinants of EC occurrence within the sampled area. Among the lake habitats analysed in our study, EC seemed to prefer these with harsher climatic conditions and waters with lower temperatures and nutrient concentrations. Thus, we suggest that both climate warming and accelerated eutrophication may contribute to reduced EC spread and invasion, as well as to displacement of EC by the later North American arrival, Elodea nuttallii, observed in other European countries in recent decades (Simpson 1990; Barrat-Segretain 2001; Greulich and Trémolières 2006). The latter species is known to be more resistant to nutrient enrichment and temperature increases, hence more competitive and of higher invasive ability than EC (Greulich and Trémolières 2006). The fact that in more eutrophied lakes no evidence for invasion by other alien hydrophyte species was found may be explained, among other factors, by lower human pressure affecting Polish waters (hence, fewer opportunities for propagule dispersal), compared to waters in western Europe.
References
Bakker ES, Sarneel JM, Gulati R, Liu Z, Van Donk E (2013) Restoring macrophyte diversity in shallow temperate lakes: biotic versus abiotic constraints. Hydrobiologia 710:23–37
Barko JW, Adams MS, Clesceri NL (1986) Environmental factors and their consideration in the management of submersed aquatic vegetation: a review. J Aquat Plant Manage 24:1–10
Barrat-Segretain M-H (2001) Invasive species in the Rhone River floodplain (France): replacement of Elodea canadensis Michaux by E. nuttallii St. John in two former river channels. Arch Hydrobiol 152:237–251
Bernatowicz S (1960) Charakterystyka jezior na podstawie roślin naczyniowych. Roczniki Nauk Rolniczych 77:79–99
Braun-Blanquet J (1964) Pflanzensoziologie. Springer, Wien-New York
Brundu G (2015) Plant invaders in European and Mediterranean inland waters: profiles, distribution, and threats. Hydrobiologia 746:61–79
Brzeg A, Wojterska M (2001) Plant communities in Wielkopolska: the state of knowledge and threats. In: Wojterska M (ed) Flora and vegetation of Wielkopolska and South-Pomerania Lakeland, guide BOOK of 52. Reunion of Polish Botanical Society, Poznan, pp 39–110
Chytrý M, Pyšek P, Wild J, Pino J, Maskell LC, Vilà M (2009) European map of alien plant invasions based on the quantitative assessment across habitats. Divers Distrib 15:98–107
Ciecierska H (2008) Macrophyte based indices of the ecological state of lakes. Dissertations and Monographs, 139, University of Warmia and Mazury in Olsztyn, Poland (in Polish with English summary)
Ciecierska H, Kolada A (2014) ESMI: a macrophyte index for assessing the ecological status of lakes. Environ Monit Assess 186:5501–5517
EU (2000) Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. OJEC L 327/1
EU (2014) Regulation (EU) no 1143/2014 of the European Parliament and of the council of 22 October 2014 on the prevention and management of the introduction and spread of invasive alien species. OJEC L 317/35
Fleming JP, Dibble ED (2015) Ecological mechanisms of invasion success in aquatic macrophytes. Hydrobiologia 746:23–37
Gąbka M, Owsianny PM (2009) First records of the Hygrophila polysperma Roxb T. Anderson (Acanthaceae) in Poland. Botanika Steciana 13:9–14
Greulich S, Trémolières M (2006) Present distribution of the genus Elodea in the Alsatian upper Rhine floodplain (France) with a special focus on the expansion of Elodea nuttallii St. John during recent decades. Hydrobiologia 570:249–255
Heip CHR, Herman PMJ, Soeraert K (1998) Indices of diversity and evenness. Océanis 24(4):61–87
Hérault B, Bornet A, Trémolières M (2008) Redundancy and niche differentiation among the European invasive Elodea species. Biol Invasions 10:1099–1107
Hilt S, Gross EM (2008) Can allelopathically active submerged macrophytes stabilise clear-water states in shallow eutrophic lakes? Basic Appl Ecol 9:422–432
Hilt S, Gross EM, Hupfer M, Morscheid H, Mahlmann J, Melzer A, Poltz J, Sandrock S, Scharf E-M, Schneider S, van de Weyer K (2006) Restoration of submerged vegetation in shallow eutrophic lakes—a guideline and state of the art in Germany. Limnologica 36:155–171
Hussner A (2012) Alien aquatic plant species in European countries. Weed Res 52:297–306
Hussner A, van de Weyer K, Gross EM, Hilt S (2010) Comments on increasing number and abundance of non-indigenous aquatic macrophyte species in Germany. Weed Res 50:519–526
Jabłońska E, Kłosowski S (2015) Ecology of rare water plant communities in lakes of north-eastern Poland. Acta Soc Bot Pol 81(1):3–9
Kłosowski S (2006) The relationships between environmental factors and the submerged Potametea associations in lakes of north-eastern Poland. Hydrobiologia 560:15–29
Kolada A (2010) The use of aquatic vegetation in the lake assessment: testing the sensitivity of macrophyte metrics to anthropogenic pressures and water quality. Hydrobiologia 656:133–147
Kolada A, Soszka H, Cydzik D, Golub M (2005) Abiotic typology of Polish lakes. Limnologica 35:145–150
Kolada A, Ciecierska H, Ruszczyńska J, Dynowski P (2014) Sampling techniques and inter-surveyor variability as sources of uncertainty in Polish macrophyte based metric for lake ecological status assessment. Hydrobiologia 737:256–279
Kornaś J, Medwecka-Kornaś A (1957) Zapiski florystyczne z Sądecczyzny. Fragm Flor et Geobot 3(1):3–9
Kuhar U, Germ M, Gaberščik A (2010) Habitat characteristics of an alien species Elodea canadensis in Slovenian watercourses. Hydrobiologia 656:205–212
Lenda M, Skórka P, Knops JMH, Moroń D, Sutherland WJ, Kruszewska K, Woyciechowski M (2014) Effect of the internet commerce in dispersal modes of invasive alien species. PLoS one 9(6):e99786
Lockwood JL, Cassey P, Blackburn TM (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228
Lorenc H (2005) Atlas of climate in Poland. Institute of Meteorology and Water Management, Warsaw (in Polish)
Madsen JD, Sutherland JW, Bloomfield JA, Eichler LW, Boylen CW (1991) The decline of native vegetation under dense Eurasian watermilfoil canopies. J Aquat Plant Manage 29:94–99
Matuszkiewicz W (2002) A guide book to identification plant communities in Poland. PWN, Warsaw, Poland (in Polish)
Mjelde M, Lombardo P, Berge D, Johansen SW (2012) Mass invasion of Elodea canadensis Michx. in a large, clear-water, species-rich Norwegian lake—impact on macrophyte biodiversity. Ann Limnol Int J Lim 48:225–240
Mjelde M, Bolpagni R, Lombardo P, Azzella MM (2014) Aggressiveness of the non-native macrophyte Elodea canadensis is closely associated with local environmental factors in European lakes. In: 8th Shallow lakes conference (12–17 October, Antalya, Turkey), E-book of abstracts: 124
Panov VE, Alexandrov B, Arbačiauskas K, Binimelis R, Copp GH, Grabowski M, Lucy F, Leuven RSEW, Nehring S, Paunović M, Semenchenko V, Son MO (2009) Assessing the risks of aquatic species invasions via European inland waterways: from concepts to environmental indicators. Integr Environ Assess Manag 5:110–126
Pielou EC (1975) Ecological diversity. Wiley, New York
Pokorný J, Kvĕt J, Ondok JP, Toul Z, Ostrý I (1984) Production—ecological analysis of a plant community dominated by Elodea canadensis Michx. Aquat Bot 19:263–292
Pyšek P (1995) On the terminology used in plant invasion studies. In: Pyśek P, Prach K, Rejmánek M, Wade M (eds) Plant invasions – general aspects and special problems. SPB Academic Publishing, Amsterdam, pp 71–81
Redekop P, Hofstra D, Hussner A (2016) Elodea canadensis shows a higher dispersal capacity via fragmentation than Egeria densa and Lagarosiphon major. Aquat Bot 130:45–49
Rejewski M (1984) Biomass and production of macrophytes in the lakes of Laski region in Tuchola Forests. Acta Universitatis Nicolai Copernici 57(14):97–119
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Riis T, Olesen B, Clayton JS, Lambertini C, Brix H, Sorrell BK (2012) Growth and morphology in relation to temperature and light availability during the establishment of three invasive aquatic plant species. Aquat Bot 102:56–64
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois Press, Urbana
Simberloff D, Rejmánek M (eds) (2011) Encyclopaedia of biological invasions. University of California Press, Berkeley
Simpson EH (1949) Measurement of diversity. Nature 163:688
Simpson DA (1984) A short history of the introduction and spread of Elodea Michx in the British Isles. Watsonia 17:121–132
Simpson DA (1990) Displacement of Elodea canadensis Michx by Elodea nuttallii (Planch.) H. St. John in the British Isles. Watsonia 18:173–177
Soszka H, Ochocka A (2011) Lakes. Supporting physicochemical elements. In: Soszka H (ed) Ecological status assessment of the waters in the Wel river catchment. Guidelines for integrated assessment of ecological status of rivers and lakes to support river basin management plans. Institute of Inland Fisheries, Olsztyn, pp 249–256 (in Polish with English summary)
StatSoft, Inc. (2011) STATISTICA (data analysis software system), version 10. www.statsoft.com
Szczęśniak E, Błachuta J, Krukowski M, Picińska-Fałtynowicz J (2009) Distribution of Azolla filiculoides Lam. (Azollaceae) in Poland. Acta Soc Bot Pol 78(3):241–246
ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for Windows User’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Thiébaut G (2005) Does competition for phosphate supply explain the invasion pattern of Elodea species? Water Res 39:3385–3393
Thomaz SM, Kovalenko KE, Havel JE, Kats LB (2015) Aquatic invasive species: general trends in the literature and introduction to the special issue. Hydrobiologia 746:1–12
Tokarska-Guzik B (2005a) Invasive ability of kenophytes occurring in Poland: a tentative assessment. In: Nentwig W, Bacher S, Cock MJW, Dietz H, Gigon A, Wittenberg R (eds) Biological invasions—from ecology to control, vol 6. NEOBIOTA, pp 47–65
Tokarska-Guzik B (2005b) The establishment and spread of Alien plant species (Kenophytes) in the Flora of Poland. The University of Silesia Press, Katowice
Tokarska-Guzik B, Zając M, Zając A (2008) Geographical and ecological aspects of the spread of alien plant species in Poland. In: Rabitsh W, Essl F, Klingenstein F (eds) Biological Invasions—from ecology to conservation, vol 7. NEOBIOTA, pp 143–152
Tokarska-Guzik B, Bzdęga K, Nowak T, Lewandowska A, Gancarek M, Frelich M (2014) Alien plants in Poland: research directions and putting the results into practice. Biodiv Res Conserv 35:57–74
Zając A (1978) Atlas of distribution of vascular plants in poland (ATPOL). Taxon 27:481–484
Zając A, Tokarska-Guzik B, Zając M (2011) The role of rivers and streams in the migration of alien plants into the Polish Carpathians. Biodiv Res Conserv 23:43–56
Zehnsdorf A, Hussner A, Eismann F, Rönicke H, Melzer A (2015) Management options of invasive Elodea nuttallii and Elodea canadensis. Limnologica 51:110–117
Zhang X, Odgaard R, Olesen B, Lauridsen TL, Liboriussen L, Søndergaard M, Liu Z, Jeppesen E (2015) Warming shows differential effects on late-season growth and competitive capacity of Elodea canadensis and Potamogeton crispus in shallow lakes. Inland Waters 5:421–432
Acknowledgments
The Chief Inspectorate for Environmental Protection and Voivodeship Inspectorates for Environmental Protection in Poland are kindly acknowledged as providers of monitoring data on water quality and aquatic vegetation of a substantial portion of the lakes analysed in this study. Special thanks go to Prof. Hanna Ciecierska and Dr Joanna Ruszczyńska from the University of Warmia and Mazury who supported us in the field survey during our joined research projects and who provided some of the biological data used in this study. The study was supported by the Polish Ministry of Science and Higher Education. Biological and physicochemical data on 11 lakes were acquired from the Project ‘deWELopment’ supported by a Grant from Norway through the Norwegian Financial Mechanism within the framework of the Polish–Norwegian Research Fund (PNRF-220-AI-1/07).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10530_2016_1212_MOESM1_ESM.pdf
Online Resources: Table S1. The list of species identified in lakes surveyed during the period 2005-2013 analysed in the study, where phytocoenoses of Elodea canadensis (EC) were present (n = 212) or absent (n = 359). Supplementary material 1 (PDF 409 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kolada, A., Kutyła, S. Elodea canadensis (Michx.) in Polish lakes: a non-aggressive addition to native flora. Biol Invasions 18, 3251–3264 (2016). https://doi.org/10.1007/s10530-016-1212-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-016-1212-4