Abstract
Objective
Its eps gene cluster, the antioxidant activity and monosaccharide composition of exopolysaccharides, the expression levels of related genes at different fermentations were analyzed for clarifying the exopolysaccharide biosynthesis mechanism of Lactobacillus delbrueckii subsp. bulgaricus LDB-C1.
Results
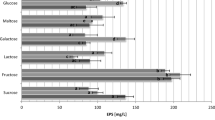
The comparison analysis of eps gene clusters indicated that the gene clusters present diversity and strain specificity. The crude exopolysaccharides from LDB-C1 exhibited a good antioxidant activity. Compared with glucose, fructose, galactose, and fructooligosaccharide, inulin significantly improved the exopolysaccharide biosynthesis. The structures of EPSs were significantly different under different carbohydrate fermentation conditions. Inulin obviously increased the expressions of most EPS biosynthesis related genes at fermentation 4 h.
Conclusion
Inulin accelerated the beginning of the exopolysaccharide production in LDB-C1, and the enzymes promoted by inulin was beneficial for the accumulation of exopolysaccharide at the whole fermentation process.








Similar content being viewed by others
References
Adolfsson O, Meydani SN, Russell RM (2004) Yogurt and gut function. Am J Clin Nutr 2:245–256. https://doi.org/10.1093/ajcn/80.2.245
Ali K, Mehmood MH, Iqbal MA, Masud T et al (2019) Isolation and characterization of exopolysaccharide-producing strains of Lactobacillus bulgaricus from curd. Food Sci Nutr 7:1207–1213. https://doi.org/10.1002/fsn3.905
Bourret RB, Silversmith RE (2010) Two-component signal transduction. Curr Opin Microbiol 13:113–115. https://doi.org/10.1016/j.mib.2010.02.003
Bouzar F, Cerning J, Desmazeaud M (1996) Exopolysaccharide production in milk by Lactobacillus delbrueckii ssp. bulgaricus CNRZ 1187 and by two colonial variants. J Dairy Sci 79:205–211. https://doi.org/10.3168/jds.S0022-0302(96)76352-X
Buntin N, Hongpattarakere T, Ritari J, Douillard FP, Paulin L, Boeren S, Shetty SA, de Vos WM (2017) An inducible operon is involved in inulin utilization in Lactobacillus plantarum strains, as revealed by comparative proteogenomics and metabolic profiling. Appl Environ Microbiol 83:e02402-e2416. https://doi.org/10.1128/AEM.02402-16
Cui YH, Qu XJ (2021) Genetic mechanisms of prebiotic carbohydrate metabolism in lactic acid bacteria: Emphasis on Lacticaseibacillus casei and Lacticaseibacillus paracasei as flexible, diverse and outstanding prebiotic carbohydrate starters. Trends Food Sci Technol 115:486–499. https://doi.org/10.1016/j.tifs.2021.06.058
Cui YH, Liu W, Qu XJ, Chen ZT, Zhang X, Liu T, Zhang LW (2012) A two component system is involved in acid adaptation of Lactobacillus delbrueckii subsp bulgaricus. Microbiol Res 167:253–261. https://doi.org/10.1016/j.micres.2011.11.003
Cui YH, Jiang X, Hao MY, Qu XJ, Hu T (2017) New advances in exopolysaccharides production of Streptococcus thermophilus. Arch Microbiol 199:799–809. https://doi.org/10.1007/s00203-017-1366-1
Cui YH, Wang MH, Zheng YK, Miao K, Qu XJ (2021) The carbohydrate metabolism of Lactiplantibacillus plantarum. Int J Mol Sci 22:13452. https://doi.org/10.3390/ijms222413452
Darilmaz DO, Aslım B, Suludere Z, Akca G (2011) Influence of gastrointestinal system conditions on adhesion of exopolysaccharide-producing Lactobacillus delbrueckii subsp. bulgaricus strains to caco-2 cells. Braz Arch Biol Technol 54:917–926. https://doi.org/10.1590/S1516-89132011000500009
Faber EJ, Kamerling JP, Vliegenthart JFG (2001) Structure of the extracellular polysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus 291. Carbohydr Res 331:183–194. https://doi.org/10.1016/s0008-6215(01)00012-x
Farias DD, de Araujo FF, Neri-Numa IA, Pastore GM (2019) Prebiotics: trends in food, health and technological applications. Trends Food Sci Tech 93:23–35. https://doi.org/10.1016/j.tifs.2019.09.004
Ganesan P, Kumar CS, Bhaskar N (2008) Antioxidant properties of methanol extract and its solvent fractions obtained from selected Indian red seaweeds. Bioresource Technol 99:2717–2723. https://doi.org/10.1016/j.biortech.2007.07.005
Gibson GR, Hutkins R, Sanders ME, Prescott SL et al (2017) Expert consensus document: The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastro Hepatol 14:491–502. https://doi.org/10.1038/nrgastro.2017.75
Goh KKT, Haisman DR, Singh H (2005) Development of an improved procedure for isolation and purification of exopolysaccharides produced by Lactobacillus delbrueckii subsp. bulgaricus NVFB 2483. Appl Microbiol Biot 67:202–208. https://doi.org/10.1007/s00253-004-1739-7
Grobben GJ, Sikkema J, Smith MR, de Bont JAM (1995) Production of extracellular polysaccharides by Lactobacillus delbrueckii ssp. bulgaricus NCFB 2772 grown in a chemically defined medium. J Appl Bacteriol 79:103–107. https://doi.org/10.1111/j.1365-2672.1995.tb03130.x
Grobben GJ, van Casteren WHM, Schols HA, Oosterveld A, Sala G, Smith MR, Sikkema J, deBont JAM (1997) Analysis of the exopolysaccharides produced by Lactobacillus delbrueckii subsp. bulgaricus NCFB 2772 grown in continuous culture on glucose and fructose. Appl Microbiol Biot 48:516–521. https://doi.org/10.1007/s002530051089
Gruter M, Leeflang BR, Kuiper J, Kamerling JP, Vliegenthart JFG (1993) Structural characterisation of the exopolysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus RR grown in skimmed milk. Carbohydr Res 239:209–226. https://doi.org/10.1016/0008-6215(93)84216-s
Guan YX, Cui YH, Qu XJ, Jing K (2021) Safety and robustness aspects analysis of Lactobacillus delbrueckii ssp. bulgaricus LDB-C1 based on the genome analysis and biological tests. Arch Microbiol 203:3955–3964. https://doi.org/10.1007/s00203-021-02383-7
Hao P, Zheng HJ, Yu Y, Ding GH et al (2011) Complete sequencing and pan-genomic analysis of Lactobacillus delbrueckii subsp. bulgaricus reveal its genetic basis for industrial yogurt production. PLoS ONE 6:e15964. https://doi.org/10.1371/journal.pone.0015964
Harding LP, Marshall VM, Hernandez Y, Gu Y, Maqsood M, McLay N, Laws AP (2005) Structural characterisation of a highly branched exopolysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus NCFB 2074. Carbohydr Res 340:1107–1111. https://doi.org/10.1016/j.carres.2005.01.038
Hu T, Cui YH, Qu XJ (2021) Analysis of the proteolytic system of Streptococcus thermophilus strains CS5, CS9, CS18 and CS20. Int Dairy J 118:105025. https://doi.org/10.1016/j.idairyj.2021.105025
Kanmani P, Albarracin L, Kobayashi H, Hebert EM et al (2018) Genomic characterization of Lactobacillus delbrueckii TUA4408L and evaluation of the antiviral activities of its extracellular polysaccharides in porcine intestinal epithelial cells. Front Immunol 9:2178. https://doi.org/10.3389/fimmu.2018.02178
Lamothe G, Jolly L, Mollet B, Stingele F (2002) Genetic and biochemical characterization of exopolysaccharide biosynthesis by Lactobacillus delbrueckii subsp. bulgaricus. Arch Microbiol 178:218–228. https://doi.org/10.1007/s00203-002-0447-x
Li SQ, Shah NP (2014) Antioxidant and antibacterial activities of sulphated polysaccharides from Pleurotus eryngii and Streptococcus thermophilus ASCC 1275. Food Chem 165:262–270. https://doi.org/10.1016/j.foodchem.2014.05.110
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu YQ, Song SC, Tian HX, Yu HY, Zhao JX, Chen C (2018) Functional analysis of the role of CcpA in Lactobacillus plantarum grown on fructooligosaccharides or glucose: a transcriptomic perspective. Microb Cell Fact 17:201. https://doi.org/10.1186/s12934-018-1050-4
Makino S, Ikegami S, Kano H, Sashihara T, Sugano H, Horiuchi H, Saito T, Oda M (2006) Immunomodulatory effects of polysaccharides produced by Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. J Dairy Sci 89:2873–2881. https://doi.org/10.3168/jds.S0022-0302(06)72560-7
Marshall VM, Laws AP, Gu Y, Levander F, Rådström P, De Vuyst L, Degeest B, Vaningelgem F, Dunn H, Elvin M (2001) Exopolysaccharide-producing strains of thermophilic lactic acid bacteria cluster into groups according to their EPS structure. Lett Appl Microbiol 32:433–437. https://doi.org/10.1046/j.1472-765x.2001.00937.x
Nagai T, Makino S, Ikegami S, Itoh H, Yamada H (2011) Effects of oral administration of yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 and its exopolysaccharides against influenza virus infection in mice. Int Immunopharmacol 11:2246–2250. https://doi.org/10.1016/j.intimp.2011.09.012
Nishimura-Uemura J, Kitazawa H, Kawai Y, Itoh T, Oda M, Saito T (2003) Functional alteration of murine macrophages stimulated with extracellular polysaccharides from Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Food Microbiol 20:267–273. https://doi.org/10.1016/S0740-0020(02)00177-6
Petry S, Furlan S, Waghorne E, Saulnier L, Cerning J, Maguin E (2003) Comparison of the thickening properties of four Lactobacillus delbrueckii subsp. bulgaricus strains and physicochemical characterization of their exopolysaccharides. Fems Microbiol Lett 221:285–291. https://doi.org/10.1016/S0378-1097(03)00214-3
Sánchez-Medina I, Gerwig GJ, Urshev ZL, Kamerling JP (2007) Structural determination of a neutral exopolysaccharide produced by Lactobacillus delbrueckii ssp. bulgaricus LBB.B332. Carbohydr Res 342:2735–2744. https://doi.org/10.1016/j.carres.2007.09.005
Sánchez-Medina I, Frank M, von der Lieth CW, Kamerling JP (2009) Conformational analysis of the neutral exopolysaccharide produced by Lactobacillus delbrueckii ssp. bulgaricus LBB.B26. Org Biomol Chem 7:280–287. https://doi.org/10.1039/b810468a
Settachaimongkon S, Nout M, Fernandes E, Hettinga KA, Valenberg H (2014) Influence of different proteolytic strains of Streptococcus thermophilus in co-culture with Lactobacillus delbrueckii subsp. bulgaricus on the metabolite profile of set-yoghurt. Int J Food Microbiol 177:29–36. https://doi.org/10.1016/j.ijfoodmicro.2014.02.008
Swanson KS, Gibson GR, Hutkins R, Reimer RA et al (2020) The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat Rev Gastro Hepatol 17:687–701. https://doi.org/10.1038/s41575-020-0344-2
Tang WZ, Dong MS, Wang WL, Han S, Rui X, Chen XH, Jiang M, Zhang QQ, Wu JJ, Li W (2017) Structural characterization and antioxidant property of released exopolysaccharides from Lactobacillus delbrueckii ssp. bulgaricus SRFM-1. Carbohydr Polym 173:654–664. https://doi.org/10.1016/j.carbpol.2017.06.039
Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3-new capabilities and interfaces. Nucleic Acids Res 40:e115. https://doi.org/10.1093/nar/gks596
Van Calsteren MR, Gagnon F, Nishimura J, Makino S (2015) Structure determination of the neutral exopolysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus OLL1073R-1. Carbohydr Res 413:115–122. https://doi.org/10.1016/j.carres.2015.05.015
Van de Guchte M, Penaud S, Grimaldi C, Barbe V et al (2006) The complete genome sequence of Lactobacillus bulgaricus reveals extensive and ongoing reductive evolution. Proc Natl Acad Sci USA 103:9274–9279. https://doi.org/10.1073/pnas.0603024103
Warner JB, Lolkema JS (2003) CcpA-dependent carbon catabolite repression in bacteria. Microbiol Mol Biol R 67:475–490. https://doi.org/10.1128/MMBR.67.4.475-490.2003
Welman AD, Maddox IS (2003) Exopolysaccharides from lactic acid bacteria: perspectives and challenges. Trends Biotechnol 21:269–274. https://doi.org/10.1016/S0167-7799(03)00107-0
Zeidan AA, Poulsen VK, Janzen T, Buldo P, Derkx PMF, Oregaard G, Neves AR (2017) Polysaccharide production by lactic acid bacteria: from genes to industrial applications. FEMS Microbiol Rev 41:S168–S200. https://doi.org/10.1093/femsre/fux017
Zhang L, Liu CH, Li D, Zhao YJ, Zhang X, Zeng XP, Yang ZN, Li SY (2013) Antioxidant activity of an exopolysaccharide isolated from Lactobacillus plantarum C88. Int J Biol Macromol 54:270–275. https://doi.org/10.1016/j.ijbiomac.2012.12.037
Zhou Y, Cui Y, Qu X (2019) Exopolysaccharides of lactic acid bacteria: structure, bioactivity and associations: a review. Carbohyd Polym 207:317–332. https://doi.org/10.1016/j.carbpol.2018.11.093
Zhou Y, Cui YH, Suo C, Wang Q, Qu XJ (2021) Structure, physicochemical characterization, and antioxidant activity of the highly arabinose-branched exopolysaccharide EPS-M2 from Streptococcus thermophilus CS6. Int J Biol Macromol 192:716–727. https://doi.org/10.1016/j.ijbiomac.2021.10.047
Supporting information
Supplementary Table 1: The information of L. delbrueckii subsp. bulgaricus strains used in this study.
Supplementary Table 2: The primers used in the qPCR analysis.
Supplementary Table 3: Carbohydrate utilization of L. delbrueckii ssp. bulgaricus strains.
Supplementary Table 4: The EPS yield of L. delbrueckii ssp. bulgaricus strain LBD-C1 under mediums containing different sugars at different fermentation stages.
Supplementary Table 5: The OD and pH of L. delbrueckii ssp. bulgaricus strain LBD-C1 under mediums containing different sugars at different fermentation stages.
Supplementary Table 6: The expressions of eps gene clusters and regulation genes of L. delbrueckii ssp. bulgaricus strain LBD-C1 under mediums containing different sugars at different fermentation stages.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guan, Y., Cui, Y., Wang, Q. et al. Inulin increases the EPS biosynthesis of Lactobacillus delbrueckii ssp. bulgaricus LDB-C1. Biotechnol Lett 45, 639–654 (2023). https://doi.org/10.1007/s10529-023-03365-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-023-03365-z