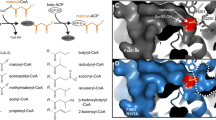
Abstract
Core biochemical pathways such as Fatty-acid synthesis II (FAS II) is ascribed to the synthesis of fatty-acids, biotin and lipoic acid in prokaryotes. It has two dehydrogenases namely, FabG and FabI which interact with the fatty-acid chain bound to Acyl-carrier protein (ACP), a well-studied enzyme which binds to substrates of varying lengths. This protein–protein interaction ‘broadens’ the active site of these dehydrogenases thus, contributing to their flexible nature. This property is exploited for catalysing numerous chiral synthons, alkanes, long-chain alcohols and secondary metabolites in industries especially with FabG. FASI relegates FASII in eukaryotes making it a ‘relic gene pool’ and an antibacterial drug target with diverse inhibitor and substrate markush. FabG often substitutes other dehydrogenases for producing secondary metabolites in nature. This redundancy is probably due to gene duplication or addition events possibly making FabG, a progenitor to some of the complex short-chain dehydrogenases used in organisms and industries today.





Similar content being viewed by others
Change history
17 May 2019
In the original publication of the article, under section Polyketide synthesis: a pathway similar to fatty acid synthesis, the sentences “Phylogeny of KS domains and proteins of FAS and PKS, inferred by Bayesian estimation. Numbers above branches indicate posterior clade probability values.” and “Branch length indicates number of inferred amino acid changes per position.” in the first paragraph were included inadvertently.
References
Andersson A, Jordan D, Schneider G, Lindqvist Y (1997) A flexible lid controls access to the active site in 1,3,8-trihydroxynaphthalene reductase. FEBS Lett 400:173–176. https://doi.org/10.1016/s0014-5793(96)01382-8
Beld J, John Lee D, Burkart MD (2015) Fatty acid biosynthesis revisited: structure elucidation and metabolic engineering. Mol BioSyst 11:38–59. https://doi.org/10.1039/c4mb00443d
Beste Dany J V, Espasa M, Bonde B, Kierzek AM, Stewart GR, McFadden J (2009) The genetic requirements for fast and slow growth in mycobacteria. PLoS ONE 4:e5349. https://doi.org/10.1371/journal.pone.0005349
Biswas R, Singh BK, Dutta D, Basak A, Das AK (2018) Domain swapping between FabGs deciphers the structural determinant for in-solution oligomerization and substrate binding. Biophys Chem 237:9–21. https://doi.org/10.1016/j.bpc.2018.03.003
Brinster S, Lamberet G, Staels B, Trieu-Cuot P, Gruss A, Poyart C (2009) Type II fatty acid synthesis is not a suitable antibiotic target for Gram-positive pathogens. Nature 458:83–86. https://doi.org/10.1038/nature07772
Bukhari Habib ST, Jakob RP, Maier T (2014) Evolutionary origins of the multienzyme architecture of giant fungal fatty acid synthase. Structure 22:1775–1785. https://doi.org/10.1016/j.str.2014.09.016
Carbone V, Schofield LR, Zhang Y, Sang C, Dey D, Hannus IM, Martin WF, Sutherland-Smith AJ, Ronimus RS (2015) Structure and evolution of the archaeal lipid synthesis enzymesn-glycerol-1-phosphate dehydrogenase. J Biol Chem 290:21690–21704. https://doi.org/10.1074/jbc.m115.647461
Chan DI, Vogel HJ (2010) Current understanding of fatty acid biosynthesis and the acyl carrier protein. Biochem J 430:1–19. https://doi.org/10.1042/bj20100462
Chen Z, Kastaniotis AJ, Miinalainen IJ, Rajaram V, Wierenga RK, Kalervo Hiltunen J (2009) 17β-Hydroxysteroid dehydrogenase type 8 and carbonyl reductase type 4 assemble as a ketoacyl reductase of human mitochondrial FAS. FASEB J 23:3682–3691. https://doi.org/10.1096/fj.09-133587
Cheng J, Ma J, Lin J, Fan Z-C, Cronan JE, Wang H (2011) Only one of the five ralstonia solanacearum long-chain 3-ketoacyl-acyl carrier protein synthase homologues functions in fatty acid synthesis. Appl Environ Microbiol 78:1563–1573. https://doi.org/10.1128/aem.07335-11
Cronan JE (2008) Biotin and lipoic acid: synthesis, attachment, and regulation. EcoSal Plus. https://doi.org/10.1128/ecosalplus.3.6.3.5
Cronan JE, Thomas J (2009) Bacterial fatty acid synthesis and its relationships with polyketide synthetic pathways. Methods Enzymol 459:395–433. https://doi.org/10.1016/s0076-6879(09)04617-5
Darouiche RO, Mansouri MD, Gawande PV, Madhyastha S (2009) Antimicrobial and antibiofilm efficacy of triclosan and DispersinB(R) combination. J Antimicrob Chemother 64:88–93. https://doi.org/10.1093/jac/dkp158
Das D, Eser BE, Han J, Sciore A, Neil E, Marsh G (2011) Oxygen-independent decarbonylation of aldehydes by cyanobacterial aldehyde decarbonylase: a new reaction of diiron enzymes. Angew Chem Int Ed 50:7148–7152. https://doi.org/10.1002/anie.201101552
Debajyoti D, Bhattacharyya S, Mukherjee S, Saha B, Das AK (2011) Crystal structure of FabG4 from mycobacterium tuberculosis reveals the importance of C-terminal residues in ketoreductase activity. J Struct Biol 174:147–155. https://doi.org/10.1016/j.jsb.2010.11.012
Dibrova DV, Galperin MY, Mulkidjanian AY (2014) Phylogenomic reconstruction of archaeal fatty acid metabolism. Environ Microbiol 16:907–918. https://doi.org/10.1111/1462-2920.12359
Favia AD, Nobeli I, Glaser F, Thornton JM (2008) Molecular docking for substrate identification: the short-chain dehydrogenases/reductases. J Mol Biol 375:855–874. https://doi.org/10.1016/j.jmb.2007.10.065
Freund GS, O’Brien TE, Vinson L, Carlin DA, Yao A, Mak WS, Tagkopoulos I, Facciotti MT, Tantillo DJ, Siegel JB (2017) Elucidating substrate promiscuity within the fabi enzyme family. ACS Chem Biol 12:2465–2473. https://doi.org/10.1021/acschembio.7b00400
Ghatak A, Bharatham N, Shanbhag AP, Datta S, Venkatraman J (2017) Delineating substrate diversity of disparate short-chain dehydrogenase reductase from Debaryomyces hansenii. PLOS ONE 12:e0170202. https://doi.org/10.1371/journal.pone.0170202
Gurvitz A (2009a) The essential mycobacterial genes, fabG1 and fabG4, encode 3-oxoacyl-thioester reductases that are functional in yeast mitochondrial fatty acid synthase type 2. Mol Genet Genom 282:407–416. https://doi.org/10.1007/s00438-009-0474-2
Gurvitz A (2009b) Caenorhabditis elegans F09E10.3 encodes a putative 3-oxoacyl-thioester reductase of mitochondrial type 2 fatty acid synthase FAS II that is functional in yeast. J Biomed Biotechnol 2009:1–6. https://doi.org/10.1155/2009/235868
Haq SF, Shanbhag AP, Karthikeyan S, Hassan I, Thanukrishnan K, Ashok A, Sukumaran S et al (2018) A strategy to identify a ketoreductase that preferentially synthesizes pharmaceutically relevant (S)-alcohols using whole-cell biotransformation. Microb Cell Fact 17(1):192. https://doi.org/10.1186/s12934-018-1036-2
Heath RJ, Rock CO (1995) Enoyl-Acyl carrier protein reductase (fabI) plays a determinant role in completing cycles of fatty acid elongation in Escherichia coli. J Biol Chem 270:26538–26542. https://doi.org/10.1074/jbc.270.44.26538
Heath RJ, Yuen-Tsu Yu, Shapiro MA, Olson E, Rock CO (1998) Broad spectrum antimicrobial biocides target the FabI component of fatty acid synthesis. J Biol Chem 273:30316–30320. https://doi.org/10.1074/jbc.273.46.30316
Heath RJ, Nancy S, Murphy CK, Rock CO (2000) The enoyl-[acyl-carrier-protein] reductases FabI and FabL from Bacillus subtilis. J Biol Chem 275:40128–40133. https://doi.org/10.1074/jbc.m005611200
Heath R, White S, Rock C (2002) Inhibitors of fatty acid synthesis as antimicrobial chemotherapeutics. Appl Microbiol Biotechnol 58:695–703. https://doi.org/10.1007/s00253-001-0918-z
Heimer G, Kerätär J, Riley L, Balasubramaniam S, Eyal E, Pietikäinen L et al (2016) MECR mutations cause childhood-onset dystonia and optic atrophy, a mitochondrial fatty acid synthesis disorder. Am J Hum Genet 99(6):1229–1244. https://doi.org/10.1016/j.ajhg.2016.09.021
Hoang TT, Sullivan SA, Schweizer HP, Cusick JK (2002) β-Ketoacyl acyl carrier protein reductase (FabG) activity of the fatty acid biosynthetic pathway is a determining factor of 3-oxo-homoserine lactone acyl chain lengths. Microbiology 148:3849–3856. https://doi.org/10.1099/00221287-148-12-3849
Hölsch K, Weuster-Botz D (2010) New oxidoreductases from cyanobacteria: exploring nature’s diversity. Enzyme Microbial Technol 47:228–235. https://doi.org/10.1016/j.enzmictec.2010.06.006
Holsch K, Havel J, Haslbeck M, Weuster-Botz D (2008) Identification, cloning, and characterization of a novel ketoreductase from the cyanobacterium Synechococcus sp. strain PCC 7942. Appl Environ Microbiol 74:6697–6702. https://doi.org/10.1128/aem.00925-08
Hou J, Wojciechowska K, Zheng H, Chruszcz M, Cooper DR, Cymborowski M, Skarina T et al (2012) Structure of a short-chain dehydrogenase/reductase from Bacillus anthracis. Acta Crystallogr Sect F 68:632–637. https://doi.org/10.1107/s1744309112017939
Huang H, Dan W, Tian W-X, Ma X-F, Xiao-Dong W (2008) Antimicrobial effect by extracts of rhizome of Alpinia officinarum may relate to its inhibition of β-ketoacyl-ACP reductase. J Enzyme Inhib Med Chem 23:362–368. https://doi.org/10.1080/14756360701622099
Javidpour P, Pereira JH, Goh E-B, McAndrew RP, Ma SM, Friedland GD, Keasling JD, Chhabra SR, Adams PD, Beller HR (2013) Biochemical and structural studies of NADH-dependent FabG used to increase the bacterial production of fatty acids under anaerobic conditions. Appl Environ Microbiol 80:497–505. https://doi.org/10.1128/aem.03194-13
Jenke-Kodama H, Dittmann E (2009) Evolution of metabolic diversity: insights from microbial polyketide synthases. Phytochemistry 70:1858–1866. https://doi.org/10.1016/j.phytochem.2009.05.021
Jenke-Kodama H, Sandmann A, Müller R, Dittmann E (2005) Evolutionary Implications of bacterial polyketide synthases. Mol Biol Evol 22:2027–2039. https://doi.org/10.1093/molbev/msi193
Jez JM, Bennett MJ, Schlegel BP, Lewis M, Penning TM (1997) Comparative anatomy of the aldo–keto reductase superfamily. Biochem J 326:625–636. https://doi.org/10.1042/bj3260625
Kanesaki Yu, Suzuki I, Allakhverdiev SI, Mikami K, Murata N (2002) Salt stress and hyperosmotic stress regulate the expression of different sets of genes in Synechocystis sp. PCC 6803. Biochem Biophys Res Commun 290:339–348. https://doi.org/10.1006/bbrc.2001.6201
Karchmer AW, Hafkin B (2013) Is there a future for FabI inhibitors as antibacterial agents. Clin Invest 3:707–709. https://doi.org/10.4155/cli.13.65
Kavanagh KL, Jörnvall H, Persson B, Oppermann U (2008) Medium- and short-chain dehydrogenase/reductase gene and protein families. Cell Mol Life Sci 65:3895–3906. https://doi.org/10.1007/s00018-008-8588-y
Keatinge-Clay AT (2016) The structural relationship between iterative and modular PKSs. Cell Chem Biol 23:540–542. https://doi.org/10.1016/j.chembiol.2016.05.005
Khosla C, Gokhale RS, Jacobsen JR, Cane DE (1999) Tolerance and specificity of polyketide synthases. Ann Rev Biochem 68:219–253. https://doi.org/10.1146/annurev.biochem.68.1.219
Kim K-H, Ha BH, Kim SJ, Hong SK, Hwang KY, Kim EE (2011) Crystal structures of Enoyl-ACP reductases I (FabI) and III (FabL) from B. subtilis. J Mol Biol 406:403–415. https://doi.org/10.1016/j.jmb.2010.12.003
Kohli GS, John U, Van Dolah FM, Murray SA (2016) Evolutionary distinctiveness of fatty acid and polyketide synthesis in eukaryotes. The ISME J 10:1877–1890. https://doi.org/10.1038/ismej.2015.263
Kristan K, Bratkovič T, Sova M, Gobec S, Preželj A, Urleb U (2009) Novel inhibitors of β-ketoacyl-ACP reductase from Escherichia coli. Chem-Biol Interact 178:310–316. https://doi.org/10.1016/j.cbi.2008.09.030
Leibundgut M, Maier T, Jenni S, Ban N (2008) The multienzyme architecture of eukaryotic fatty acid synthases. Curr Opin Struct Biol 18:714–725. https://doi.org/10.1016/j.sbi.2008.09.008
Liang Y, Tian W, Ma X (2013) Inhibitory effects of grape skin extract and resveratrol on fatty acid synthase. BMC Complement Altern Med 13(1):361. https://doi.org/10.1186/1472-6882-13-361
Liu J, Wu J, Li Z (2014) Enoyl acyl carrier protein reductase (FabI) catalyzed asymmetric reduction of the C=C double bond of α, β-unsaturated ketones: preparation of (R)-2-alkyl-cyclopentanones. Chem Commun 50:9729–9732. https://doi.org/10.1039/c4cc04150j
Liu J-S, Yi-Chia Kuan Yu, Tsou T-YL, Hsu W-H, Yang M-T, Lin J-Y, Wang W-C (2018) Structure-guided design of Serratia marcescens short-chain dehydrogenase/reductase for stereoselective synthesis of (R)-phenylephrine. Sci Rep 8(1):2316. https://doi.org/10.1038/s41598-018-19235-y
Lombard J, López-García P, Moreira D (2012) An ACP-independent fatty acid synthesis pathway in archaea: implications for the origin of phospholipids. Mol Biol Evol 29:3261–3265. https://doi.org/10.1093/molbev/mss160
Marrakchi H, Zhang Y-M, Rock CO (2002) Mechanistic diversity and regulation of Type II fatty acid synthesis. Biochem Soc Trans 30:1050–1055. https://doi.org/10.1042/bst0301050
Massengo-Tiassé RP, Cronan JE (2009) Diversity in enoyl-acyl carrier protein reductases. Cell Mol Life Sci 66:1507–1517. https://doi.org/10.1007/s00018-009-8704-7
Mistry TL, Truong L, Ghosh AK, Johnson ME, Mehboob S (2016) Benzimidazole-based FabI inhibitors: a promising novel scaffold for anti-staphylococcal drug development. ACS Infect Dis 3:54–61. https://doi.org/10.1021/acsinfecdis.6b00123
Movahed E, Tan GMY, Munusamy K, Yeow TC, Tay ST, Wong WF, Looi CY (2016) Triclosan demonstrates synergic effect with amphotericin B and fluconazole and induces apoptosis-like cell death in cryptococcus neoformans. Front Microbiol 7:360. https://doi.org/10.3389/fmicb.2016.00360
Mulkidjanian A, Galperin M, Makarova K, Wolf Y, Koonin E (2008) Evolutionary primacy of sodium bioenergetics. Biol Direct 3:13. https://doi.org/10.1186/1745-6150-3-13
Ohlrogge J (1995) Lipid biosynthesis. Plant Cell Online 7:957–970. https://doi.org/10.1105/tpc.7.7.957
Parish T, Roberts G, Laval F, Schaeffer M, Daffe M, Duncan K (2007) Functional complementation of the essential gene fabG1 of mycobacterium tuberculosis by mycobacterium smegmatis fabG but not Escherichia coli fabG. J Bacteriol 189:3721–3728. https://doi.org/10.1128/jb.01740-06
Ren Q, Sierro N, Witholt B, Kessler B (2000) FabG, an NADPH-dependent 3-ketoacyl reductase of Pseudomonas aeruginosa, provides precursors for medium-chain-length poly-3-hydroxyalkanoate biosynthesis in Escherichia coli. J Bacteriol 182:2978–2981. https://doi.org/10.1128/jb.182.10.2978-2981.2000
Rock CO, Jackowski S (2002) Forty years of bacterial fatty acid synthesis. Biochem Biophys Res Commun 292:1155–1166. https://doi.org/10.1006/bbrc.2001.2022
Roy N, Mandal S, Mahanti B, Dasgupta S (2018) Antimicrobial activity of green tea: a comparative study with different green tea extract. PharmaTutor 6:23. https://doi.org/10.29161/pt.v6.i1.2018.23
Schirmer A, Rude MA, Li X, Popova E, del Cardayre SB (2010) Microbial biosynthesis of alkanes. Science 329:559–562. https://doi.org/10.1126/science.1187936
Sul SH, Smith S (2008) Fatty acid synthesis in eukaryotes. In: Hei SS, Smith S (eds) Biochemistry of lipids, lipoproteins and membranes. Elsevier, Amsterdam, pp 155–190
Sun X-B, Tian W-X (2009) inhibitory effects of allium vegetable extracts on fatty acid synthase. Food Sci Technol Res 15:343–346. https://doi.org/10.3136/fstr.15.343
Taguchi Kazunori, Aoyagi Yoshihiro, Matsusaki Hiromi, Fukui Toshiaki, Doi Yoshiharu (1999) Co-expression of 3-ketoacyl-ACP reductase and polyhydroxyalkanoate synthase genes induces PHA production in Escherichia coli HB101 strain. FEMS Microbiol Lett 176:183–190. https://doi.org/10.1111/j.1574-6968
Voelker TA, Davies HM (1994) Alteration of the specificity and regulation of fatty acid synthesis of Escherichia coli by expression of a plant medium-chain acyl-acyl carrier protein thioesterase. J Bacteriol 176:7320–7327. https://doi.org/10.1128/jb.176.23.7320-7327.1994
Wang Haihong, Cronan John E (2004) Only one of the two annotated lactococcus lactis fabG genes encodes a functional β-ketoacyl-acyl carrier protein reductase†. Biochemistry 43:11782–11789. https://doi.org/10.1021/bi0487600
Woodley John M (2008) New opportunities for biocatalysis: making pharmaceutical processes greener. Trends Biotechnol 26:321–327. https://doi.org/10.1016/j.tibtech.2008.03.004
Wright H Tonie (2004) Cofactors in fatty acid biosynthesis—active site organizers and drug targets. Structure 12:358–359. https://doi.org/10.1016/j.str.2004.02.027
Yamamoto H, Matsuyama A, Kobayashi Y (2003) Synthesis of ethyl (S)-4-chloro-3-hydroxybutanoate using fabG-homologues. Appl Microbiol Biotechnol 61:133–139. https://doi.org/10.1007/s00253-002-1188-0
Yu X, Liu T, Zhu F, Khosla C (2011) In vitro reconstitution and steady-state analysis of the fatty acid synthase from Escherichia coli. Proc Natl Acad Sci USA 108:18643–18648. https://doi.org/10.1073/pnas.1110852108
Zhang Yong-Mei, Rock Charles O (2004) Evaluation of epigallocatechin gallate and related plant polyphenols as inhibitors of the FabG and FabI reductases of bacterial type II fatty-acid synthase. J Biol Chem 279:30994–31001. https://doi.org/10.1074/jbc.m403697200
Zhang Yong-Mei, White Stephen W, Rock Charles O (2006) Inhibiting Bacterial Fatty Acid Synthesis. J Biol Chem 281:17541–17544. https://doi.org/10.1074/jbc.r600004200
Acknowledgements
Dr. Santanu Datta helped in conceiving the thought process for this study. Special thanks to Prof. Ramaswamy S., Dr. Nainesh Katagihallimath, Dr. Nagakumar Bharatham and Arindam Ghatak for introducing the structural and industrial aspects of these enzymes. Thanks to Prof. Sudip Kundu for helping me pursue this in an academic setup.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shanbhag, A.P. FabG: from a core to circumstantial catalyst. Biotechnol Lett 41, 675–688 (2019). https://doi.org/10.1007/s10529-019-02678-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02678-2