Abstract
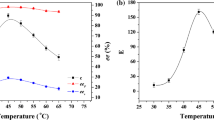
Isooctane was the best reaction medium for the enantioselective esterification of (R,S)-2-methylalkanoic acid with n-butanol using Carica papaya lipase as catalyst. Increasing linear alkyl-chain length of racemic 2-methylalkanoic acids from ethyl to hexyl increased the enantioselectivity (E) from 2.1 to 98.2 for the esterification of racemic 2-methylalkanoic acids with n-butanol at 35°C. Decreasing reaction temperature from 40 to 20°C increased the enantioselectivity (E) from 14 to 33 for the esterification of racemic 2-methylhexanoic acids with n-butanol. We obtained a maximum enantioselectivity, of E = 24.3, for the enantioselective esterification of racemic 2-methylhexanoic acids with n-butanol in isooctane at water activity 0.33, and at 35°C.


Similar content being viewed by others
References
Ahmed SN, Kazlauskas RJ, Morinville AH, Grochulski P, Schrag JD, Cygler M (1994) Enantioselectivity of Candida rugosa lipase towards carboxylic acids: a predictive rule from substrate mapping and X-ray crystallography. Biocatalysis 9:209–225
Berglund P, Holmquist M, Hedenström E, Hult K, Högberg HE (1993) 2-Methylalkanoic acids resolved by esterification catalysed by lipase from Candida rugosa: alcohol chain length and enantioselectivity. Tetrahedron Asymmetr 4:1869–1878
Berglund P, Holmquist M, Hult K, Högberg HE (1995) Alcohols as enantioselective inhibitors in a lipase catalysed esterification of a chiral acyl donor. Biotechnol Lett 17:55–60
Caro Y, Pina M, Turon F, Guilbert S, Mougeout E, Fetsch DV, Attwool P, Graille J (2002) Plant lipases: biocatalyst aqueous environment in relation to optimal catalytic activity in lipase-catalyzed synthesis reactions. Biotechnol Bioeng 77:693–703
Chen CC, Tsai SW (2005) Carica papaya lipase: a novel biocatalyst for the enantioselective hydrolysis of (R,S)-naproxen 2,2,2-trifluoroethyl ester. Enzyme Microb Technol 36:127–132
Cheng YC, Tsai SW (2004) Enantioselective esterification of (R,S)-2-(4-chlorophenoxy)-propionic acid via Carica papaya lipase in organic solvents. Tetrahedron Asymmetr 15:2917–2920
Domínguez de María P, Sinisterra JV, Tsai SW, Alcántara AR (2006) Carica papaya lipase: an emerging and versatile biocatalyst. Biotechnol Adv 24:493–499
Engel KH (1991) Lipase-catalyzed enantioselective esterification of 2-methylalkanoic acids. Tetrahedron Asymmetr 2:165–168
Fitzpatrick PA, Klibanov AM (1991) How can the solvent affect enzyme enantioselectivity? J Am Chem Soc 113:3166–3171
Heinsman NWJT, Franssen MCR, Van Der Padt A, Boom RM, Van’t Riet K, De Groot AE (2002) Lipase-mediated resolution of branched chain fatty acids. Biocatal Biotransform 20:297–309
Högberg HE, Edlund H, Berglund P, Hedenström E (1993) Water activity influences enantioselectivity in a lipase-catalysed resolution by esterification in an organic solvent. Tetrahedron Asymmetr 4:2123–2126
Holmberg E, Hoimquist M, Hedenström E, Berglund P, Norin T, Högberg HE, Hult K (1991) Reaction conditions for the resolution of 2-methylalkanoic acids in esterification and hydrolysis with lipase from Candida cylindracea. Appl Microbiol Biotechnol 35:572–578
Krishna SH, Karanth NG (2002) Lipases and lipase-catalyzed esterification reactions in nonaqueous media. Catal Rev 44:499–591
Kwon DY, Hong YJ, Yoon SH (2000) Enantiomeric synthesis of (S)-2-methylbutanoic acid methyl ester, apple flavor, using lipases in organic solvent. J Agric Food Chem 48:524–530
Laane C, Boeren S, Vos K, Veeger C (1987) Rules for optimization of biocatalysis in organic solvents. Biotechnol Bioeng 30:81–87
Maciel MI, Hansen TJ, Aldinger SB, Labows JN (1986) Flavor chemistry of cashew apple juice. J Agric Food Chem 34:923–927
Macleod AJ, Pieris NM (1981) Volatile flavor components of beli fruit (Aegle marmelos) and a processed product. J Agric Food Chem 29:49–53
Miyazawa T, Onishi K, Murashima T, Yamada T, Tsai SW (2005) Resolution of non-protein amino acids via Carica papaya lipase-catalyzed enantioselective transesterification. Tetrahedron Asymmetr 16:2569–2573
Nakamura K, Kinoshita M, Ohno A (1995) Structure of solvent affects enantioselectivity of lipase-catalyzed transesterification. Tetrahedron 51:8799–8808
Ng IS, Tsai SW (2005) Partially purified Carica papaya lipase: a versatile biocatalyst for the hydrolytic resolution of (R,S)-2-arylpropionic thioesters in water-saturated organic solvents. Biotechnol Bioeng 91:106–113
Nguyen BV, Hedenström E (1999) Candida rugosa lipase as an enantioselective catalyst in the esterification of methyl branched carboxylic acids: resolution of rac-3,7-dimethyl-6-octenoic acid (citronellic acid). Tetrahedron Asymmetr 10:1821–1826
Parida S, Dordick JS (1991) Substrate structure and solvent hydrophobicity control lipase catalysis in organic media. J Am Chem Soc 113:2253–2259
Pleiss J, Fischer M, Schmid RD (1998) Anatomy of lipase binding sites: the scissile fatty acid binding site. Chem Phys Lipids 93:67–80
Sabbani S, Hedenström E (2009) Control of water activity in lipase catalysed esterification of chiral alkanoic acids. J Mol Catal B: Enzym 58:6–9
Sabbani S, Hedenström E, Nordin O (2006) The enantioselectivity of Candida rugosa lipase is influenced by the particle size of the immobilising support material Accurel. J Mol Catal B: Enzym 42:1–9
Um PJ, Drueckhammer DG (1998) Dynamic enzymatic resolution of thioesters. J Am Chem Soc 120:5605–5610
Villeneuve P (2003) Plant lipases and their applications in oils and fats modification. Eur J Lipid Sci Technol 105:308–317
Acknowledgments
The authors would like to thank the National Science Council of the Republic of China, Taiwan, for financially supporting this research under Contract No. NSC- 96-2221-E-218 -029.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chang, CS., Ho, SC. Enantioselective esterification of (R,S)-2-methylalkanoic acid with Carica papaya lipase in organic solvents. Biotechnol Lett 33, 2247–2253 (2011). https://doi.org/10.1007/s10529-011-0692-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-011-0692-7