Abstract
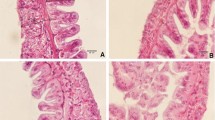
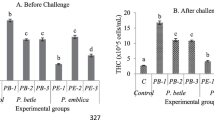
Shrimp is a popular commodity due to its fast growth and high economic value. However, stress-related disease outbreaks caused by intensifying agricultural practices and environmental factors have led to significant financial losses globally. Using chemical compounds to combat infectious pathogens has also led to antibiotic resistance and accumulation of residues in tissues and the environment, posing consumer risks. This study investigated the potential benefits of Terminalia catappa leaves (TCL) as a natural plant product-based dietary supplement for Litopenaeus vannamei postlarvae against Vibrio parahaemolyticus infection. Experimental diets with TCL extract were prepared and the feeding trial was carried out for 28 days with L. vannamei postlarvae (PL10). The growth parameters and digestive enzymes were measured. Vibrio challenge test was conducted for 48 h. Oxidative stress, immunomodulation and histopathology of hepatopancreases were analysed after vibrio challenge in PL given with/without TCL diet. The study revealed that TCL diet has an increased body weight and survival rate of the shrimp than basal diet via increasing digestive enzyme activity. TCL diet also increases the survival of shrimp against the vibrio challenge. It increases antioxidant enzymes and eliminates oxidative stress. TCL acts as an immunostimulant by increasing prophenoloxidase, crustin, lysozyme and penaeid 3a genes. Moreover, the cellular damage caused by oxidative stress to hepatopancreases of the shrimp was reduced. The findings in this study suggest that the TCL diet has a dual property of growth promoter and immunostimulant and can be a sustainable and cost-effective alternative to traditional aquaculture practices.








Similar content being viewed by others
Data availability
Data will be made available from the corresponding author on reasonable request.
Code availability
Not applicable
Abbreviations
- AHPND:
-
Acute hepatopancreatic necrosis disease
- df:
-
Dilution factor
- DNSA:
-
Dinitrosalicylic acid
- FBW:
-
Final wet body weight
- FCR:
-
Feed conversion rate
- H&E:
-
Haematoxylin and eosin
- IBW:
-
Initial wet body weight
- L-DOPA:
-
L-3,4-Dihydroxyphenylalanine
- MDA:
-
Malondialdehyde
- PL:
-
Postlarvae
- pNPP:
-
p-Nitrophenyl palmitate
- PO:
-
Phenoloxidase
- SGR:
-
Specific growth ratio
- SOD:
-
Superoxide dismutase activity
- T-AOC:
-
Total antioxidant capacity
- TCBS:
-
Thiosulfate-citrate-bile salt-sucrose agar
- TCE:
-
Terminalia catappa leaf extract
- TCL:
-
Terminalia catappa leaves
- TCP:
-
Terminalia catappa leaf powder
- WG:
-
Weight gain
- WSSV:
-
White spot syndrome virus
References
Aguilar Diaz De Leon J, Borges CR (2020) Evaluation of oxidative stress in biological samples using the thiobarbituric acid reactive substances assay. J Vis Exp 2020. https://doi.org/10.3791/61122
Andriantahina F, Liu X, Huang H et al (2012) Comparison of reproductive performance and offspring quality of domesticated Pacific white shrimp, Litopenaeus vannamei. Aquaculture 324:194–200. https://doi.org/10.1016/j.aquaculture.2011.10.026
Ardi I, Zamroni M, Setiadi E (2020) Utility of almond leaf (Terminalia cattapa) for improving growth and color quality of neon tetra fish (Paracheirodon innesi). IOP Conf Ser Earth Environ Sci 441:012136. https://doi.org/10.1088/1755-1315/441/1/012136
Azhar N, Yudiati E (2023) Outbreak simulation of Litopenaeus vannamei recovery rate with oral alginate and spirulina diet supplementation against Vibrio parahaemolyticus AHPND. Aquac Int 31:1659–1676. https://doi.org/10.1007/s10499-023-01050-6
Champasri C, Phetlum S, Pornchoo C (2021) Diverse activities and biochemical properties of amylase and proteases from six freshwater fish species. Sci Rep 11:5727. https://doi.org/10.1038/s41598-021-85258-7
Chanda S, Rakholiya K, Nair R (2011) Antimicrobial activity of Terminalia catappa L. leaf extracts against some clinically important pathogenic microbial strains. Chin Med 02:171–177. https://doi.org/10.4236/cm.2011.24027
Chen M, Chen XQ, Tian LX et al (2020) Improvement of growth, intestinal short-chain fatty acids, non-specific immunity and ammonia resistance in Pacific white shrimp (Litopenaeus vannamei) fed dietary water-soluble chitosan and mixed probiotics. Comp Biochem Physiol Part - C Toxicol Pharmacol 236:108791. https://doi.org/10.1016/j.cbpc.2020.108791
Chen YT, Kuo CL, Wu CC et al (2023) Effects of Panax notoginseng water extract on immune responses and digestive enzymes in white shrimp Litopenaeus vannamei. Animals 13:1131. https://doi.org/10.3390/ani13071131
Deris ZM, Iehata S, Ikhwanuddin M et al (2020) Immune and bacterial toxin genes expression in different giant tiger prawn, penaeus monodon post-larvae stages following AHPND-causing strain of vibrio parahaemolyticus challenge. Aquac Rep 16:100248. https://doi.org/10.1016/j.aqrep.2019.100248
Di Pinto A, Terio V, Novello L, Tantillo G (2011) Comparison between thiosulphate-citrate-bile salt sucrose (TCBS) agar and CHROMagar Vibrio for isolating Vibrio parahaemolyticus. Food Control 22:124–127. https://doi.org/10.1016/j.foodcont.2010.06.013
Dong M, Feng L, Kuang SY et al (2013) Growth, body composition, intestinal enzyme activities and microflora of juvenile Jian carp (Cyprinus carpio var. Jian) fed graded levels of dietary valine. Aquac Nutr 19:1–14. https://doi.org/10.1111/j.1365-2095.2011.00926.x
Haridevamuthu B, Guru A, Velayutham M et al (2023a) Long non-coding RNA, a supreme post-transcriptional immune regulator of bacterial or virus-driven immune evolution in teleost. Rev Aquac 15:163–178. https://doi.org/10.1111/raq.12709
Haridevamuthu B, Manjunathan T, Guru A et al (2022) Hydroxyl containing benzo[b]thiophene analogs mitigates the acrylamide induced oxidative stress in the zebrafish larvae by stabilizing the glutathione redox cycle. Life Sci 298:120507. https://doi.org/10.1016/j.lfs.2022.120507
Haridevamuthu B, Raj D, Kesavan D et al (2023b) Trihydroxy piperlongumine protects aluminium induced neurotoxicity in zebrafish: behavioral and biochemical approach. Comp Biochem Physiol Part C Toxicol Pharmacol 268:109600. https://doi.org/10.1016/j.cbpc.2023.109600
Hassan SAH, Sharawy ZZ, El Nahas AF et al (2022) Modulatory effects of various carbon sources on growth indices, digestive enzymes activity and expression of growth-related genes in Whiteleg shrimp, Litopenaeus vannamei reared under an outdoor zero-exchange system. Aquac Res 53:5594–5605. https://doi.org/10.1111/are.16041
Havanapan PO, Taengchaiyaphum S, Paemanee A et al (2021) Caspase-3, a shrimp phosphorylated hemocytic protein is necessary to control YHV infection. Fish Shellfish Immunol 114:36–48. https://doi.org/10.1016/j.fsi.2021.04.007
Hong NTX, Linh NTH, Baruah K et al (2022) The combined use of Pediococcus pentosaceus and Fructooligosaccharide improves growth performance, immune response, and resistance of whiteleg shrimp Litopenaeus vannamei against Vibrio parahaemolyticus. Front Microbiol 13. https://doi.org/10.3389/fmicb.2022.826151
IMARC Group (2023) Shrimp market: Global industry trends, share, size, growth, Opportunity and Forecast 2023–2028. https://www.imarcgroup.com/prefeasibility-report-shrimp-processing-plant
Jahromi ST, Pourmozaffar S, Jahanbakhshi A et al (2021) Effect of different levels of dietary Sargassum cristaefolium on growth performance, hematological parameters, histological structure of hepatopancreas and intestinal microbiota of Litopenaeus vannamei. Aquaculture 533:736130. https://doi.org/10.1016/j.aquaculture.2020.736130
Letchumanan V, Chan KG, Lee LH (2014) Vibrio parahaemolyticus: a review on the pathogenesis, prevalence, and advance molecular identification techniques. Front Microbiol 5. https://doi.org/10.3389/fmicb.2014.00705
Li M, Wei D, Huang S et al (2022) Medicinal herbs and phytochemicals to combat pathogens in aquaculture. Aquac Int 30:1239–1259. https://doi.org/10.1007/s10499-022-00841-7
Liu L, Shan LP, Zhou Y, Chen J (2021) Small molecule inhibitors of white spot syndrome virus: promise in shrimp seedling culture. Int J Mol Sci 22:3450. https://doi.org/10.3390/ijms22073450
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Marković NS (2018) Feed formulation for sustainable aquaculture. In: M. R. R. R-E, F. D. P-E, N. D. S, M. J. H. L-R (eds) Resource enhancement and sustainable aquaculture practices in Southeast Asia: challenges in responsible production of aquatic species 2014. Tigbauan, Iloilo, Philippines: Aquaculture Department, Southeast Asian Fisheries Development Center., pp 223–230
Miandare HK, Mirghaed AT, Hosseini M et al (2017) Dietary Immunogen® modulated digestive enzyme activity and immune gene expression in Litopenaeus vannamei post larvae. Fish Shellfish Immunol 70:621–627. https://doi.org/10.1016/j.fsi.2017.09.048
Murugan R, Guru A, Haridevamuthu B et al (2022) Lantibiotics: an antimicrobial asset in combating aquaculture diseases. Aquac Int 30:2365–2387. https://doi.org/10.1007/s10499-022-00908-5
Mutalipassi M, Di Natale M, Mazzella V, Zupo V (2018) Automated culture of aquatic model organisms: shrimp larvae husbandry for the needs of research and aquaculture. Animal 12:155–163. https://doi.org/10.1017/S1751731117000908
Muthu B, Guru A, Sudhakaran G et al (2022) Double-edged sword role of shrimp miRNA explains an evolutionary language between shrimp-pathogen interactions that unties the knot of shrimp infection. Rev Aquac 14:578–593. https://doi.org/10.1111/raq.12613
Muttharasi C, Gayathri V, Muralisankar T et al (2021) Growth performance, digestive enzymes and antioxidants activities in the shrimp Litopenaeus vannamei fed with Amphiroa fragilissima crude polysaccharides encapsulated Artemia nauplii. Aquaculture 545:737263. https://doi.org/10.1016/j.aquaculture.2021.737263
NavinChandran M, Iyapparaj P, Moovendhan S et al (2014) Influence of probiotic bacterium Bacillus cereus isolated from the gut of wild shrimp Penaeus monodon in turn as a potent growth promoter and immune enhancer in P. monodon. Fish Shellfish Immunol 36:38–45. https://doi.org/10.1016/j.fsi.2013.10.004
Nguyen TV, Alfaro A, Arroyo BB et al (2021) Metabolic responses of penaeid shrimp to acute hepatopancreatic necrosis disease caused by Vibrio parahaemolyticus. Aquaculture 533:736174. https://doi.org/10.1016/j.aquaculture.2020.736174
Nugroho RA, Aryani R, Anggraini WDC et al (2019) Dietary Terminalia catappa leaves reduced growth performance but increased hematological profiles and survival rate of Pangasianodon hypophthalmus. IOP Conf Ser Earth Environ Sci 348:012033. https://doi.org/10.1088/1755-1315/348/1/012033
Nugroho RA, Manurung H, Nur FM, Prahastika W (2017) Terminalia catappa L. extract improves survival, hematological profile and resistance to Aeromonas hydrophila in Betta sp. extract. Arch Polish Fish 25:103–115. https://doi.org/10.1515/aopf-2017-0010
Nurhafizah WWI, Lee KL, Laith A. AR, et al. (2021) Virulence properties and pathogenicity of multidrug-resistant Vibrio harveyi associated with luminescent vibriosis in Pacific white shrimp, Penaeus vannamei. J Invertebr Pathol 186:107594. https://doi.org/10.1016/j.jip.2021.107594
Prathiviraj R, Rajeev R, Fernandes H et al (2021) A gelatinized lipopeptide diet effectively modulates immune response, disease resistance and gut microbiome in Penaeus vannamei challenged with Vibrio parahaemolyticus. Fish Shellfish Immunol 112:92–107. https://doi.org/10.1016/j.fsi.2021.02.018
Soonthornchai W, Chaiyapechara S, Jarayabhand P et al (2015) Interaction of vibrio spp. with the inner surface of the digestive tract of penaeus monodon. PLoS One 10:e0135783. https://doi.org/10.1371/journal.pone.0135783
Sowmya RKA (2021) Antibacterial activity and time-kill assay of Terminalia catappa L. and Nigella sativa L. and selected human pathogenic bacteria. J Pure Appl Microbiol 15:285–299. https://doi.org/10.22207/JPAM.15.1.22
Sritunyalucksana K, Soderhall K (2000) The proPO and clotting system in crustaceans. Aquaculture 191:53–69. https://doi.org/10.1016/S0044-8486(00)00411-7
Syahidah A, Saad CR, Daud HM, Abdelhadi YM (2015) Status and potential of herbal applications in aquaculture: a review. Iran J Fish Sci 14:27–44
Tassanakajon A, Rimphanitchayakit V, Visetnan S et al (2018) Shrimp humoral responses against pathogens: antimicrobial peptides and melanization. Dev Comp Immunol 80:81–93. https://doi.org/10.1016/j.dci.2017.05.009
Tassanakajon A, Somboonwiwat K, Supungul P, Tang S (2013) Discovery of immune molecules and their crucial functions in shrimp immunity. Fish Shellfish Immunol 34:954–967. https://doi.org/10.1016/j.fsi.2012.09.021
Velázquez-Lizárraga AE, Juárez-Morales JL, Racotta IS et al (2019) Transcriptomic analysis of Pacific white shrimp (Litopenaeus vannamei, Boone 1931) in response to acute hepatopancreatic necrosis disease caused by Vibrio parahaemolyticus. PLoS One 14:e0220993. https://doi.org/10.1371/journal.pone.0220993
Wangkahart E, Wachiraamonloed S, Lee P-T et al (2022) Impacts of Aegle marmelos fruit extract as a medicinal herb on growth performance, antioxidant and immune responses, digestive enzymes, and disease resistance against Streptococcus agalactiae in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol 120:402–410. https://doi.org/10.1016/j.fsi.2021.11.015
Wei H, Tan B, Yang Q et al (2023) Growth, nonspecific immunity, intestinal flora, hepatopancreas, and intestinal histological results for Litopenaeus vannamei fed with diets supplement with different animal by-products. Aquac Rep 29:101521. https://doi.org/10.1016/j.aqrep.2023.101521
Yakubu Y, Talba AM, Chong CM et al (2020) Effect of Terminalia catappa methanol leaf extract on nonspecific innate immune responses and disease resistance of red hybrid tilapia against Streptococcus agalactiae. Aquac Rep 18:100555. https://doi.org/10.1016/j.aqrep.2020.100555
Yu Q, Han F, Huang M et al (2023a) The effects of dietary myo-inositol on growth and physiological, biochemical, and molecular responses in the Pacific white shrimp Litopenaeus vannamei. Aquaculture 568:739323. https://doi.org/10.1016/j.aquaculture.2023.739323
Yu Q, Liu J, Yang J et al (2023b) Postharvest preservation technologies for marine-capture shrimp: a review. Food Bioprocess Technol. https://doi.org/10.1007/s11947-023-03049-6
Yu Y, Ding P, Qiao Y et al (2023c) The feces of sea urchins as food improves survival, growth, and resistance of small sea cucumbers Apostichopus japonicus in summer. Sci Rep 13:5361. https://doi.org/10.1038/s41598-023-32226-y
Yuan Y, Lawrence AL, Chehade SB, et al (2021) Feed intake as an estimation of attractability in Pacific white shrimp Litopenaeus vannamei. Aquaculture 532:736041. https://doi.org/10.1016/j.aquaculture.2020.736041
Yunus K, Mardhiah Jaafar A, John A (2019) Acute-lethal toxicity (LC50) effect of Terminalia catappa Linn. leaves extract on Oreochromis niloticus (red Nile tilapia) juveniles under static toxicity exposure. Orient J Chem 35:270–274. https://doi.org/10.13005/ojc/350132
Funding
The authors gratefully acknowledge the Researchers Supporting Project Number (RSP2023R414), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by B. Haridevamuthu, Abhirami Chandran and David Raj. Contributed reagents/materials/analysis tools by Bader O. Almutairi, Selvaraj Arokiyaraj, M. Dhanaraj, S, Seetharaman. Conceptualisation, formal analysis, methodology, project administration, resources, supervision, visualisation, writing—original draft and editing by Jesu Arockiaraj. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
No approval of research ethics committees was required to accomplish the goals of this study because experimental work was conducted with an unregulated invertebrate species.
Consent to participate
Not applicable
Competing interest
The authors declare no competing interests.
Additional information
Handling editor: Amany Abbass
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haridevamuthu, B., Chandran, A., Raj, D. et al. Growth performance and immunomodulatory effect of Terminalia catappa L. diet on Litopenaeus vannamei against Vibrio parahaemolyticus challenge. Aquacult Int (2023). https://doi.org/10.1007/s10499-023-01284-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-023-01284-4