Abstract
Quantifying basic biological data, such as the effects of variable temperatures on development and survival, is crucial to predicting and monitoring population growth rates of pest species, many of which are highly invasive. One of the most globally important pests of cereals is the eriophyoid wheat curl mite (WCM), Aceria tosichella, which is the primary vector of several plant viruses. The aim of this study was to evaluate temperature-dependent development and survival of WCM at a wide range of constant temperatures in the laboratory (17–33 °C). The development time of each stage depended significantly on temperature and it was negatively correlated with temperature increase. At high temperatures (27–33 °C), individuals had shorter developmental times, with the shortest (6 days) at 33 °C, whereas at the lowest tested temperatures (17–19 °C), developmental time was almost 3× longer. Moreover, temperature had a clear effect on survival: the higher the temperature, the lower the survival rate. These data provide information promoting more efficient and effective manipulation of WCM laboratory colonies, and further our understanding of the ramifications of temperature change on WCM physiology and implications for the growth and spread of this globally invasive pest.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Basic biological data, such as the effect of variable temperature on development, survival, and reproduction, are especially crucial for predicting and monitoring population growth rates of pest species, many of which are highly invasive. The increasing spread of non-indigenous or alien species to non-native regions is a globally important problem of increasing urgency (Dukes and Mooney 1999; Butchart et al. 2010; Hu et al. 2011; Cini et al. Cini et al., 2014; Seebens et al. 2017; Bianchi et al. 2019; Liu et al. 2020), as their spread is facilitated by the inexorable rise of globalization in travel and trade (Navia et al. 2010; Bertelsmeier et al. 2017; Vanbergen et al. 2018). The establishment of invasive species depends on their ability to reach new areas and the environmental conditions they encounter in new sites, including ambient temperatures. Environmental changes, such as elevated temperatures may increase the suitability of a given region to invaders from warmer climates (Walther et al. 2009). Increasing temperatures in particular may influence the development time and survival of pests and invasive species, allowing them to rapidly reach adult stages and produce offspring in a shorter time (Hanselmann et al. 2011; Salum et al. 2014; Ju et al. 2015, 2017; Xie et al. 2018). To address the issue of temporal and spatial dynamics and the invasive potential of non-indigenous species, it is critical to understand how temperature affects their development, survival, and other life-history traits.
Mites in the superfamily Eriophyoidea, numbering over 5000 species and characterized by their minute size (< 300 µm), relatively simple morphology, and obligate herbivory, are becoming widely recognized as globally adventive or invasive species (Navia et al. 2010). Their importance in the fields of agriculture and ecology has grown in the wake of recent significant advancements in microscopy and molecular biology that facilitate improved taxonomic (Navajas and Navia 2009; Monfreda et al. 2010; de Lillo et al. 2010; Chetverikov et al. 2015; Skoracka et al. 2015; Laska et al. 2018), behavioral (Skoracka et al. 2007; Michalska et al. 2010; Kiedrowicz et al. 2017; Laska et al. 2019), and other studies of these tiny, fascinating beasts. Many eriophyoid species have been recorded as agricultural pests, either due to their own feeding or as vectors of plant diseases (Navia et al. 2010; de Lillo et al. 2018). As such they have been employed as study subjects to test a variety of ecological hypotheses (Thomas and Hein 2003; Michalska et al. 2010; Navia et al. 2010; Oliveira-Hofman et al. 2015; Wosula et al. 2016; Kuczyński et al. 2016; Laska et al. 2017, 2019; Kiedrowicz et al. 2017). With this increased attention, there is a growing need for fundamental biological data from important eriophyoid species, such as data on their developmental responses to variations in ambient temperature and other abiotic factors that are essential to understanding their life histories and population dynamics.
One of the most globally important pests of cereals is the eriophyoid wheat curl mite (WCM), Aceria tosichella, which is the primary vector of Wheat streak mosaic virus, and several other plant viruses. It is a well-studied mite that is known to be a complex of cryptic species consisting of genotypes differing in several ecological traits (e.g., host plant range, thermal optima, ability to transmit viruses) (Skoracka et al. 2013, 2018a, b; McMechan et al. 2014; Kuczyński et al. 2016). The most pestiferous WCM biotypes (in Europe and South America known as MT-1 and MT-8, and in Australia and North America known as type 2 and type 1, respectively; Skoracka et al. 2018b) are the most widespread, occurring in major areas of cereal cultivation worldwide (North and South America, Africa, Europe, Asia, and Oceania) (Navia et al. 2013; Skoracka et al. 2014b). A positive correlation between temperature and population growth rate has been detected in these pestiferous biotypes, including intraspecific variation for this trait (Kuczyński et al. 2016). For example, the temperature ranges within which biotypes’s populations were able to increase were: 12.2–40.0 °C for MT-1 and 10.4–35.7 °C for MT-8, with the highest temperatures representing the upper thresholds for survival, and the optimal temperature for population growth were 35.1 and 31.9 °C for MT-1 and MT-8, respectively (Kuczyński et al. 2016).
The broad range of WCM’s thermal tolerance also improves its colonization and invasive potential (Navia et al. 2013; Kuczyński et al. 2016). Moreover, its small size enables WCM, like many other eriophyoids, to escape detection when infesting commodities and to disperse on light wind currents (Navia et al. 2013). Given the aforementioned and other characteristics (e.g., easy laboratory manipulation, relatively short generation time, and well-established laboratory rearing protocols), WCM represents a useful model species for the study of Eriophyoidea and other invasive arthropods; as such the benefits of accumulating basic biological data for WCM are ultimately multiplied.
Whereas some life-history parameters of WCM (sensu lato) have been observed under specific temperatures (e.g., egg development, mite survival) (del Rosario and Sill 1965; Boczek and Chyczewski 1975; Sabelis and Bruin 1996; Skare et al. 2003) there is currently lack of information on the survival and development time of specific WCM biotypes while inhabiting their hosts over a wide range of constant temperatures. Wosula et al. (2015) tested the effect of temperature on off-host survival of WCM biotypes 1 and 2 and showed that length of survival of both biotypes decreased with increasing temperature. Such data are essential for the development of phenological models and in the case of pestiferous biotypes, effective management plans and preventive strategies.
The primary goal of this study was a comprehensive assessment of the effects of temperature on ontogenetic development and survival of WCM genotype MT-1 (also known as type 2). Temperature-dependent development and survival was evaluated at a wide range of constant temperatures in the laboratory (17–33 °C) within its biological limits, which were tested previously (Kuczyński et al. 2016). These results were then used to establish optimal temperatures for rearing WCM under laboratory conditions and to determine the relationship between temperature and generation time. This information is extremely important for designing experiments involving this species and for long-term maintenance of laboratory colonies. Additionally, these results aid assessments of the invasive potential of WCM MT-1 under changing climatic conditions, and provide a starting point for analogous studies of other Eriophyoidea.
Materials and methods
Study system
The subject of the study was A. tosichella, genotype MT-1 (Skoracka et al. 2014a). This genotype was chosen due to its high invasive potential compared to other lineages within the WCM complex. The MT-1 lineage is distributed worldwide, having been recorded in the Nearctic, the Palearctic and the Australasian realms (Skoracka et al. 2014b). MT-1 inhabits wild grasses (Poaceae), including smooth brome, tall oat-grass, wall-barley, and quackgrass; and cultivated grasses, i.e., cereals, including wheat, triticale, barley. MT-1 can also survive on plant species in the family Amaryllidaceae (e.g., onion and garlic) (Skoracka et al. 2013).
A mite stock colony was maintained in the Population Ecology Lab, Faculty of Biology, Adam Mickiewicz University. WCM MT-1 specimens used to establish this stock colony were collected in July 2009 from wheat in Choryń, Poland (52.0433°, 16.7672°; GenBank acc. no. JF920077). The MT-1 genotype was identified by sequencing the mitochondrial cytochrome c oxidase subunit I gene fragment (COI) (Skoracka et al. 2013). All experimental animals were reared on bread wheat plants, Triticum aestivum L., variety ‘Muszelka’, grown in pots from commercially available seeds. Infested plants were kept in cages consisting of metal frames wrapped in fine nylon mesh (micron size 44) to avoid contamination, under laboratory conditions (22–24 °C, ca. 45% RH, L12:D12).
Testing WCM developmental time and survival at various temperatures
To test WCM development and survival at various temperatures we used the Murashige and Skoog (MS) medium (Murashige and Skoog 1962) modified by Karpicka-Ignatowska et al. 2019. This is the only method allowing for monitoring daily eriophyid mites development. Due to WCM microscopic and hidden life style, it is not possible to monitor daily development of these mites on the whole plants. Modified MS medium consisted of a solution of basic chemical nutrients and phytohormones to maintain leaf fragment turgidity and prevent deterioration, prepared according to Karpicka-Ignatowska et al. (2019). We transferred single females of WCM from the stock colony to 5 × 5 mm wheat leaf fragments placed on modified MS medium. Due to the minute size of WCM, it may be difficult to determine the sex without examination under a phase-contrast microscope. However, females are generally longer (ca. 30%) than males and on this basis we chose females for this experiment. Afterwards, mites were incubated in growth chamber at 10 constant temperatures ranging from 17 to 33 °C, at 2-°C intervals, 80 ± 5% RH (the optimal humidity conditions for maintaining good condition of leaf fragments and WCM development according to preliminary observations) and L16:D8 photoperiod. There were 10 replications for each temperature treatment. Females were monitored daily and removed from the experimental arena after laying their first egg. Only individuals that laid eggs (thus females) were included in the experiment. Mite development was monitored daily and the time needed for reaching each stage was recorded (larva, quiescent larva, nymph, quiescent nymph, adult). Female’s progeny was incubated and controlled until the F1 adult deposited the first egg or until the observed individual died. The time of individuals’ death was recorded and included in the estimation of WCM survival for the given temperature treatment. A complete generation of mite development was defined as the time of oviposition by an experimental female until the first egg of the next generation. Females’ survival was assessed to their first oviposition. After the experiment, adult individuals were mounted on microscope slides in modified Berlese medium (Monfreda et al. 2010) and their sex was determined using a phase-contrast microscope (Olympus BX41).
Statistical analysis
To test how temperature influences the development time of each WCM MT-1 stage, as well as cumulative development time (the entire life cycle: from egg to next generation egg), a generalized additive model (GAM) was used (Wood 2017). The Gamma distribution with the log-link function was used for the response variable.
An Additive Cox Proportional Hazard Model (Hastie and Tibshirani 1990; Wood et al. 2016; Wood 2017) was used to test the influence of temperature and sex on WCM survival. At first, two candidate models were generated: the interaction model, which included sex as a parametric term and smoothers for the temperature (fitted separately for each sex), and the main effects model, with a common smooth fit for both sexes. Then, candidate models were compared based on the Akaike information criterion (AIC), with the model having the lowest AIC value being considered the best. Statistical analysis was performed in R v.4.0 (R Core Team 2019) using the mgcv 1.8 package (Wood 2017).
Results
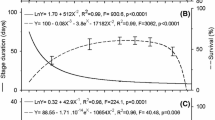
WCM MT-1 showed successful egg-to-egg development across the whole range of temperatures tested, from 17 to 33 °C (Table 1, Fig. 1). The development time of each stage depended significantly on temperature (Table 1) and the general pattern of development was consistent during ontogeny (Fig. 1). The rate of development of WCM MT-1 individuals accelerated with increase in temperature. At high temperatures (27–33 °C), individuals had shorter developmental times, with the shortest (average < 6 days) at ca. 33 °C, whereas at the lowest tested temperatures (17–19 °C) egg-to-egg development time was almost 3 × longer (Fig. 1, Table 2). Mean cumulative development time ranged from 17.7 days at 17 °C to 5.7 days at 33 °C (Table 2).
There were no signs that the pattern of temperature influence on hazard rate differed between the sexes (there was 6:1 female:male ratio, 85.7% females in the population). Based on AIC values, the main effects model was the best fit, given the data (main effects model AIC = 474.8 vs. interaction model AIC = 475.9). Moreover, in the main effects model, sex had no clear effect on a hazard rate (P = 0.84), suggesting that the sexes did not differ in their baseline hazards. Thus, in the final model, sex was excluded, and the only predictor remaining in this model was temperature, which had a clear effect on survival: the higher the temperature, the lower the survival rate (GAM results: λ2 = 84.5, edf = 2.4, p < 0.0001, n = 275; Fig. 2). However, the relationship between temperature and hazard rate is non-linear: the risk of death increases as the temperature rises, but above ca. 29 °C there is no further change (Fig. 3).
Discussion
The accumulation of basic biological data represents an essential resource for comprehensive studies of species like wheat curl mite. In addition to being an economically important pest species, WCM is the de facto model for the study of Eriophyoidea, due to the relative ease with which it can be collected from the field, on at least four continents (Carew et al. 2009; Skoracka et al. 2012, 2014b, 2018a; Miller et al. 2013; Karpicka-Ignatowska et al. 2019; Khalaf et al. 2020), and with which it can be cultured in the laboratory (e.g., Karpicka-Ignatowska et al. 2019), as well as its well-documented genetic and physiological diversity (Carew et al. 2009; Hein et al. 2012; Navia et al. 2013; Szydło et al. 2015; Skoracka et al. 2018a). Such studies have yielded important data on passive dispersal in microscopic eukaryotes (Laska et al. 2019; Karpicka-Ignatowska et al. 2019), virus transmission by herbivorous arthropods (Seifers et al. 2002; Schiffer et al. 2009; Navia et al. 2013; McMechan and Hein 2015, 2017; Wosula et al. 2018; Singh et al. 2018; Tatineni and Hein 2018), physiology (Wosula et al. 2015), genetics (Miller et al. 2013; Skoracka et al. 2018a), ecology and behavior (Kiedrowicz et al. 2017; Laska et al. 2019), distribution (Schiffer et al. 2009; Navia et al. 2010, 2013; Skoracka et al. 2014b; Khalaf et al. 2020), as well as specific assessments of its impact on crop losses (Navia et al. 2013; McMechan et al. 2014; Singh et al. 2018; Skoracka et al. 2018b). In this study we have broadened the knowledge about this important plant parasite by providing basic information on the functional relationship between temperature and ontogenetic development and survival of WCM MT-1 biotype over a wide range of temperatures, comprising the first such report for this species.
The current research enables more effective manipulation of WCM laboratory colonies to streamline studies investigating the effects of specific mite life stages on host plants (e.g., dispersal, demographics, disease transmission), and furthers our understanding of the ramifications of temperature change on WCM physiology and implications for the growth and spread of this globally invasive pest and other non-model eriophyoid species. This enhanced knowledge also creates the possibility of manipulating colony developmental time, increasing or reducing the number of mite generations during a given period according to the goals of the project. For example, fewer generations may be desired for experimentally selected lines being reared in the laboratory, to reduce the incidence of random genetic mutations. By contrast, shorter generation times may be desired when the goal is to quickly produce a large number of individuals from a given population or in genetic studies of adaptation to environmental conditions imposed in the laboratory (e.g., experimental evolution, artificial selection). This precise information about temperature-dependent life-history traits informs planning for complex, long-term studies and should thus accelerate progress. In the field, such data enable improved estimation of intra-specific demographics for ecological and host-pest synchrony studies (Pulatov et al. 2016; Huang and Hao 2020). It is important to note that the study presented here was performed under laboratory conditions designed specifically to maintain minute phytophagous arthropods; in this case, mites were reared on plant fragments placed on an artificial medium. In this way, we were able to precisely estimate development and survival. It would be valuable in the future to validate these data under more natural conditions.
The negative correlation we found between temperature and development time (Table 2, Fig. 2) is a commonly observed phenomenon in invertebrates (Schwartzberg et al. 2014; Soroye et al. 2020). However, it adds substantial evidence to previous studies showing that the effect of rising temperature on the development and survival of WCM presents important agricultural and environmental challenges resulting from its accelerated development and invasion potential (Navia et al. 2013; Wosula et al. 2015; Kuczyński et al. 2016). As rising global temperatures exacerbate challenges posed by ectothermic pests (Cannon 1998; Bebber et al. 2013; Lehmann et al. 2020), information is needed to anticipate how these changes will affect specific taxa. Combining laboratory data, such as those presented here, with those collected in the field facilitates the creation of models to predict population growth rate and population dynamics under natural conditions (Fand et al. 2015; Kuczyński et al. 2016). As such, the improved fundamental understanding of thermal effects on phenology and survival of the model species WCM will enable the design of tools to predict mite outbreaks in the field and develop strategies for integrated pest management of WCM and other eriophyoid pests.
From the data presented here it is clear that the higher the temperature the shorter the developmental time of MT-1 (Fig. 1, Table 2), and that survival of MT-1 decreases as temperature rises, but above 29 °C the hazard rate does not change (Fig. 3). The shorter the developmental time, the faster adults start to reproduce. Interactions between these parameters (development, survival, and reproduction) determine the population growth rate. Although we did not measure reproduction directly, we can conclude that the shorter the time to sexual maturity, the sooner adult individuals will reproduce, thus population growth will be higher, provided that hazard rate will not substantially increase, as was shown in our results for temperatures above 29 °C. Shorter generation times, combined with higher proportions of individuals reproducing at higher temperature strongly suggest that rising temperature will result in increased population growth of MT-1 as temperature approaches the previously estimated optimum for MT-1 population growth of 35.1 °C (Kuczyński et al. 2016). Therefore, the implications for rising temperatures in cereal growing regions of the world are clear.
It is also important to note that the greatest gains in rate of WCM development are found at the lower end of the tested temperature range (see Table 2). For example, under a hypothetical scenario of a local rise of 4 °C, a relatively cool cereal-growing region (17–21 °C during the growing season) that warms to more moderate temperature (21–25 °C) would face a decrease in WCM development time of 28.9–35.6% (from a range of 11.4–17.7 to 8.1–11.4 days per generation), increasing the number of generations during a 30-day period from approximately 2–3 to 3–4 generations, with a concomitant increase in population size. By comparison, under a similar 4 °C rise, a region changing from a warm (25–29 °C) to hot (29–33 °C) climate would experience only a 14.9–17.3% decrease in WCM development time (from 6.7–8.1 to 5.7–6.7 days per generation); thus, whereas the development rate would increase, the number of generations expected in a 30-day period would remain roughly between four and five generations. Ergo, whereas warmer regions foster more generations of WCM than cooler regions, it can be postulated that there is great potential for increased WCM pest status in regions where temperatures would rise from cool to moderate levels. Increased pest status under warming climates has also been predicted for other ectothermic pest species (Pulatov et al. 2016; Gu et al. 2018; Iwamura et al. 2020) and the data presented here add to these global concerns.
Availability of data and materials
The datasets generated and analyzed during the current study are available in the Zenodo repository under: https://doi.org/10.5281/zenodo.4008542.
Code availability
Custom code used in this study is available in the Zenodo repository under: https://doi.org/10.5281/zenodo.4008542.
References
Bebber DP, Ramotowski MAT, Gurr SJ (2013) Crop pests and pathogens move polewards in a warming world. Nat Clim Change 3:985–988. https://doi.org/10.1038/nclimate1990
Bertelsmeier C, Ollier S, Liebhold A, Keller L (2017) Recent human history governs global ant invasion dynamics. Nat Ecol Evol 1:1–8. https://doi.org/10.1038/s41559-017-0184
Bianchi CN, Azzola A, Bertolino M et al (2019) Consequences of the marine climate and ecosystem shift of the 1980–90s on the Ligurian Sea biodiversity (NW Mediterranean). Eur Zool J 86:458–487. https://doi.org/10.1080/24750263.2019.1687765
Boczek J, Chyczewski J (1975) Beobachtungen zur Biologie einiger Gallmilbenarten (Eriophyoidea) der Gräser. Tag Verlag 134:83–90
Butchart SHM, Walpole M, Collen B et al (2010) Global biodiversity: indicators of recent declines. Science 328:1164–1168. https://doi.org/10.1126/science.1187512
Cannon RJCC (1998) The implications of predicted climate change for insect pests in the UK, with emphasis on non-indigenous species. Glob Change Biol 4:785–796. https://doi.org/10.1046/j.1365-2486.1998.00190.x
Carew M, Schiffer M, Umina P et al (2009) Molecular markers indicate that the wheat curl mite, Aceria tosichella Keifer, may represent a species complex in Australia. Bull Entomol Res 99:479. https://doi.org/10.1017/S0007485308006512
Chetverikov PE, Cvrković T, Makunin A et al (2015) Basal divergence of Eriophyoidea (Acariformes, Eupodina) inferred from combined partial COI and 28S gene sequences and CLSM genital anatomy. Exp Appl Acarol 67:219–245. https://doi.org/10.1007/s10493-015-9945-9
Cini A, Anfora G, Escudero-Colomar LA et al (2014) Tracking the invasion of the alien fruit pest Drosophila suzukii in Europe. J Pest Sci (2004) 87:559–566. https://doi.org/10.1007/s10340-014-0617-z
de Lillo E, Craemer C, Amrine JW, Nuzzaci G (2010) Recommended procedures and techniques for morphological studies of Eriophyoidea (Acari: Prostigmata). Exp Appl Acarol 51:283–307. https://doi.org/10.1007/s10493-009-9311-x
de Lillo E, Pozzebon A, Valenzano D, Duso C (2018) An intimate relationship between eriophyoid mites and their host plants—a review. Front Plant Sci 9:1786. https://doi.org/10.3389/fpls.2018.01786
del Rosario MS, Sill WH (1965) Physiological strains of Aceria tulipae and their relationships to transmission of wheat streak mosaic virus. Phytopathology 55:1168–1175
Dukes JS, Mooney HA (1999) Does global change increase the success of biological invaders? Trends Ecol Evol 14:135–139. https://doi.org/10.1016/S0169-5347(98)01554-7
Fand BB, Sul NT, Bal SK, Minhas PS (2015) Temperature impacts the development and survival of common cutworm (Spodoptera litura): simulation and visualization of potential population growth in India under warmer temperatures through life cycle modelling and spatial mapping. PLoS ONE 10:e0124682. https://doi.org/10.1371/journal.pone.0124682
Gu S, Han P, Ye Z et al (2018) Climate change favours a destructive agricultural pest in temperate regions: late spring cold matters. J Pest Sci (2004) 91:1191–1198. https://doi.org/10.1007/s10340-018-1011-z
Hanselmann AJ, Gergs R, Rothhaupt K-O (2011) Embryonic development time of the freshwater mysid Limnomysis benedeni Czerniavsky as a function of water temperature. Aquat Ecol 45:539–546. https://doi.org/10.1007/s10452-011-9374-8
Hastie T, Tibshirani R (1990) Generalized additive models. Chapman and Hall, Boca Raton
Hein GL, French R, Siriwetwiwat B, Amrine JW (2012) Genetic characterization of North American populations of the wheat curl mite and dry bulb mite. J Econ Entomol 105:1801–1808. https://doi.org/10.1603/ec11428
Hu J, de Barro P, Zhao H et al (2011) An extensive field survey combined with a phylogenetic analysis reveals rapid and widespread invasion of two alien whiteflies in China. PLoS ONE 6:e16061. https://doi.org/10.1371/journal.pone.0016061
Huang J, Hao HF (2020) Effects of climate change and crop planting structure on the abundance of cotton bollworm, Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae). Ecol Evol 10:1324–1338. https://doi.org/10.1002/ece3.5986
Iwamura T, Guzman-Holst A, Murray KA (2020) Accelerating invasion potential of disease vector Aedes aegypti under climate change. Nat Commun 11:1–10. https://doi.org/10.1038/s41467-020-16010-4
Ju RT, Zhu HY, Gao L et al (2015) Increases in both temperature means and extremes likely facilitate invasive herbivore outbreaks. Sci Rep 5:1–10. https://doi.org/10.1038/srep15715
Ju RT, Gao L, Wei SJ, Li B (2017) Spring warming increases the abundance of an invasive specialist insect: links to phenology and life history. Sci Rep 7:1–12. https://doi.org/10.1038/s41598-017-14989-3
Karpicka-Ignatowska K, Laska A, Kuczyński L et al (2019) A novel experimental approach for studying life-history traits of phytophagous arthropods utilizing an artificial culture medium. Sci Rep 9:20327. https://doi.org/10.1038/s41598-019-56801-4
Khalaf L, Timm A, Chuang W-P et al (2020) Modeling Aceria tosichella biotype distribution over geographic space and time. PLoS ONE 15:e0233507. https://doi.org/10.1371/journal.pone.0233507
Kiedrowicz A, Kuczyński L, Lewandowski M et al (2017) Behavioural responses to potential dispersal cues in two economically important species of cereal-feeding eriophyid mites. Sci Rep 7:3890. https://doi.org/10.1038/s41598-017-04372-7
Kuczyński L, Rector BG, Kiedrowicz A et al (2016) Thermal niches of two invasive genotypes of the wheat curl mite Aceria tosichella: congruence between physiological and geographical distribution data. PLoS ONE 11:e0154600. https://doi.org/10.1371/journal.pone.0154600
Laska A, Rector BG, Kuczyński L, Skoracka A (2017) Is body size important? Seasonal changes in morphology in two grass-feeding Abacarus mites. Exp Appl Acarol 72:317–328. https://doi.org/10.1007/s10493-017-0159-1
Laska A, Majer A, Szydło W et al (2018) Cryptic diversity within grass-associated Abacarus species complex (Acariformes: Eriophyidae), with the description of a new species, Abacarus plumiger n. sp. Exp Appl Acarol 76:1–28. https://doi.org/10.1007/s10493-018-0291-6
Laska A, Rector BG, Skoracka A, Kuczyński L (2019) Can your behaviour blow you away? Contextual and phenotypic precursors to passive aerial dispersal in phytophagous mites. Anim Behav 155:141–151. https://doi.org/10.1016/J.ANBEHAV.2019.07.003
Lehmann P, Ammunét T, Barton M et al (2020) Complex responses of global insect pests to climate warming. Front Ecol Environ 18:141–150. https://doi.org/10.1002/fee.2160
Liu X, Blackburn TM, Song T et al (2020) Animal invaders threaten protected areas worldwide. Nat Commun 11:1–9. https://doi.org/10.1038/s41467-020-16719-2
McMechan AJ, Hein GL (2015) Planting date and variety selection for management of viruses transmitted by the wheat curl mite (Acari: Eriophyidae). J Econ Entomol 109:70–77. https://doi.org/10.1093/JEE/TOV311
McMechan AJ, Hein GL (2017) Population dynamics of the wheat curl mite (Acari: Eriophyidae) during the heading stages of winter wheat. J Econ Entomol 110:355–361. https://doi.org/10.1093/JEE/TOX028
McMechan AJ, Tatineni S, French R, Hein GL (2014) Differential transmission of Triticum mosaic virus by wheat curlmite populations collected in the great plains. Plant Dis 98:806–810. https://doi.org/10.1094/PDIS-06-13-0582-RE
Michalska K, Skoracka A, Navia D (2010) Behavioural studies on eriophyoid mites: an overview. Exp Appl Acarol 51:31–59. https://doi.org/10.1007/s10493-009-9319-2
Miller AD, Skoracka A, Navia D et al (2013) Phylogenetic analyses reveal extensive cryptic speciation and host specialization in an economically important mite taxon. Mol Phylogenet Evol 66:928–940. https://doi.org/10.1016/j.ympev.2012.11.021
Monfreda R, Lekveishvili M, Petanovic R, Amrine JW (2010) Collection and detection of eriophyoid mites. Exp Appl Acarol 51:273–282. https://doi.org/10.1007/s10493-009-9315-6
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Navajas M, Navia D (2009) DNA-based methods for eriophyoid mite studies: review, critical aspects, prospects and challenges. Eriophyoid mites: progress and prognoses. Springer Netherlands, Dordrecht, pp 257–271
Navia D, Ochoa R, Welbourn C, Ferragut F (2010) Adventive eriophyoid mites: a global review of their impact, pathways, prevention and challenges. Exp Appl Acarol 51:225–255. https://doi.org/10.1007/s10493-009-9327-2
Navia D, de Mendonça RS, Skoracka A et al (2013) Wheat curl mite, Aceria tosichella, and transmitted viruses: an expanding pest complex affecting cereal crops. Exp Appl Acarol 59:95–143. https://doi.org/10.1007/s10493-012-9633-y
Oliveira-Hofman C, Wegulo SN, Tatineni S, Hein GL (2015) Impact of Wheat streak mosaic virus and Triticum mosaic virus coinfection of wheat on transmission rates by wheat curl mites. Plant Dis 99:1170–1174. https://doi.org/10.1094/PDIS-08-14-0868-RE
Pulatov B, Jönsson AM, Wilcke RAI et al (2016) Evaluation of the phenological synchrony between potato crop and Colorado potato beetle under future climate in Europe. Agric Ecosyst Environ 224:39–49. https://doi.org/10.1016/j.agee.2016.03.027
R Core Team (2019) R: A Language and environment for statistical computing
Sabelis MW, Bruin J (1996) Evolutionary ecology: life history patterns, food plant choice and dispersal. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites—their biology, natural enemies and control. Elsevier, Amsterdam, pp 329–366
Salum JK, Mwatawala MW, Kusolwa PM, Meyer MD (2014) Demographic parameters of the two main fruit fly (Diptera: Tephritidae) species attacking mango in Central Tanzania. J Appl Entomol 138:441–448. https://doi.org/10.1111/jen.12044
Schiffer M, Umina P, Carew M et al (2009) The distribution of wheat curl mite (Aceria tosichella) lineages in Australia and their potential to transmit wheat streak mosaic virus. Ann Appl Biol 155:371–379. https://doi.org/10.1111/j.1744-7348.2009.00349.x
Schwartzberg EG, Jamieson MA, Raffa KF et al (2014) Simulated climate warming alters phenological synchrony between an outbreak insect herbivore and host trees. Oecologia 175:1041–1049. https://doi.org/10.1007/s00442-014-2960-4
Seebens H, Blackburn TM, Dyer EE et al (2017) No saturation in the accumulation of alien species worldwide. Nat Commun 8:14435. https://doi.org/10.1038/ncomms14435
Seifers DL, Harvey TL, Louie R et al (2002) Differential transmission of isolates of the High Plains virus by different sources of Wheat curl mites. Plant Dis 86:138–142. https://doi.org/10.1094/PDIS.2002.86.2.138
Singh K, Wegulo SN, Skoracka A, Kundu JK (2018) Wheat streak mosaic virus: a century old virus with rising importance worldwide. Mol Plant Pathol 19:2193–2206. https://doi.org/10.1111/mpp.12683
Skare JM, Wijkamp I, Rezende J et al (2003) Colony establishment and maintenance of the eriophyid wheat curl mite Aceria tosichella for controlled transmission studies on a new virus-like pathogen. J Virol Methods 108:133–137
Skoracka A, Kuczyński L, Rector BG (2007) Divergent host acceptance behavior suggests host specialization in populations of the polyphagous mite Abacarus hystrix (Acari: Prostigmata: Eriophyidae). Environ Entomol 36:899–909. https://doi.org/10.1603/0046-225x(2007)36[899:dhabsh]2.0.co;2
Skoracka A, Kuczyński L, Santos de Mendonça R et al (2012) Cryptic species within the wheat curl mite Aceria tosichella (Keifer) (Acari : Eriophyoidea), revealed by mitochondrial, nuclear and morphometric data. Invertebr Syst 26:417–433. https://doi.org/10.1071/IS11037
Skoracka A, Kuczyński L, Szydło W, Rector BG (2013) The wheat curl mite Aceria tosichella (Acari: Eriophyoidea) is a complex of cryptic lineages with divergent host ranges: evidence from molecular and plant bioassay. Biol J Linn Soc 109:165–180. https://doi.org/10.1111/bij.12024
Skoracka A, Kuczyński L, Rector B, Amrine JW (2014a) Wheat curl mite and dry bulb mite: untangling a taxonomic conundrum through a multidisciplinary approach. Biol J Linn Soc 111:421–436. https://doi.org/10.1111/bij.12213
Skoracka A, Rector B, Kuczyński L et al (2014b) Global spread of wheat curl mite by its most polyphagous and pestiferous lineages. Ann Appl Biol 165:222–235. https://doi.org/10.1111/aab.12130
Skoracka A, Magalhães S, Rector BG, Kuczyński L (2015) Cryptic speciation in the Acari: a function of species lifestyles or our ability to separate species? Exp Appl Acarol 67:165–182. https://doi.org/10.1007/s10493-015-9954-8
Skoracka A, Lopes LF, Alves MJ et al (2018a) Genetics of lineage diversification and the evolution of host usage in the economically important wheat curl mite, Aceria tosichella Keifer, 1969. BMC Evol Biol 18:122. https://doi.org/10.1186/s12862-018-1234-x
Skoracka A, Rector BG, Hein GL (2018b) The interface between wheat and the wheat curl mite, Aceria tosichella, the primary vector of globally important viral diseases. Front Plant Sci 9:1098. https://doi.org/10.3389/fpls.2018.01098
Soroye P, Newbold T, Kerr J (2020) Climate change contributes to widespread declines among bumble bees across continents. Science 367:685–688. https://doi.org/10.1126/science.aax8591
Szydło W, Hein G, Denizhan E, Skoracka A (2015) Exceptionally high levels of genetic diversity in wheat curl mite (Acari: Eriophyidae) populations from Turkey. J Econ Entomol 108:2030–2039. https://doi.org/10.1093/jee/tov180
Tatineni S, Hein GL (2018) Genetics and mechanisms underlying transmission of Wheat streak mosaic virus by the wheat curl mite. Curr Opin Virol 33:47–54. https://doi.org/10.1016/j.coviro.2018.07.012
Thomas JA, Hein GL (2003) Influence of volunteer wheat plant condition on movement of the wheat curl mite, Aceria tosichella, in winter wheat. Exp Appl Acarol 31:253–268. https://doi.org/10.1023/B:APPA.0000010384.12678.46
Vanbergen AJ, Espíndola A, Aizen MA (2018) Risks to pollinators and pollination from invasive alien species. Nat Ecol Evol 2:16–25. https://doi.org/10.1038/s41559-017-0412-3
Walther G-R, Roques A, Hulme PE et al (2009) Alien species in a warmer world: risks and opportunities. Trends Ecol Evol 24:686–693. https://doi.org/10.1016/J.TREE.2009.06.008
Wood SN (2017) Generalized additive models: an introduction with R, 2nd edn. Chapman and Hall, Boca Raton
Wood SN, Pya N, Säfken B (2016) Smoothing parameter and model selection for general smooth models. J Am Stat Assoc 111:1548–1563. https://doi.org/10.1080/01621459.2016.1180986
Wosula EN, McMechan AJ, Hein GL (2015) The effect of temperature, relative humidity, and virus infection status on off-host survival of the wheat curl mite (Acari: Eriophyidae). J Econ Entomol 108:1545–1552. https://doi.org/10.1093/jee/tov185
Wosula EN, McMechan AJ, Oliveira-Hofman C et al (2016) Differential transmission of two isolates of Wheat streak mosaic virus by five wheat curl mite populations. Plant Dis 100:154–158. https://doi.org/10.1094/PDIS-03-15-0342-RE
Wosula EN, McMechan AJ, Knoell E et al (2018) Impact of timing and method of virus inoculation on the severity of wheat streak mosaic disease. Plant Dis 102:645–650. https://doi.org/10.1094/PDIS-08-17-1227-RE
Xie H, Wu S, Yu L et al (2018) Effects of elevated CO2 and temperature on twospotted spider mite (Acari: Tetranychidae) feeding on maize. J Entomol Sci 53:205–218. https://doi.org/10.18474/JES17-59.1
Acknowledgements
The authors give special thanks to Jacek Radwan from the Evolutionary Biology Group, Adam Mickiewicz University, for lending a growing chamber, Jarosław Raubic from the Population Ecology Lab, AMU, for his help in conducting experiments, and the company DANKO Hodowla Roślin for providing the Triticum aestivum seeds. The study was supported by the National Science Centre, Poland (NSC), Research Grant No. 2016/21/B/NZ8/00786. AL scholarships was funded by European Social Funds POWR.03.02.00–00–I006/17, NSC (PhD Scholarship No. 2019/32/T/NZ8/00151) and Adam Mickiewicz University Foundation (Awarded in 2019/2020).
Funding
The study was supported by the National Science Centre, Poland (NSC); Research Grant No. 2016/21/B/NZ8/00786. AL scholarships was funded by European Social Funds POWR.03.02.00–00–I006/17, NSC (PhD Scholarship No. 2019/32/T/NZ8/00151) and Adam Mickiewicz University Foundation (Awarded in 2019/2020).
Author information
Authors and Affiliations
Contributions
KKI, AL, LK, AS designed the study; KKI, AL performed experiments and gathered the data; LK analyzed the data; LK, KKI, BR, AL, AS, interpreted the results; AS, LK, provided all necessary reagents and infrastructure to perform the experiments and led the work; KKI, LK, AS, AL designed the concept of the manuscript; KKI, BR, AS, LK, AL wrote the manuscript and accepted its final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
No ethical approval is required.
Research involving human and/or animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karpicka-Ignatowska, K., Laska, A., Rector, B.G. et al. Temperature-dependent development and survival of an invasive genotype of wheat curl mite, Aceria tosichella. Exp Appl Acarol 83, 513–525 (2021). https://doi.org/10.1007/s10493-021-00602-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-021-00602-w