Abstract
Predatory mites of the Phytoseiidae family are important biological control agents. Many species of this family are omnivores, i.e., besides on prey, they can feed on plant resources such as nectar and pollen. It has been shown that the addition of alternative food for predators to a crop enhances biological control. However, factors such as food availability and quality can also affect interactions such as cannibalism, and thus influence biological control. We investigated the role of quality of the alternative food in the tendency of Amblyseius herbicolus to engage in cannibalism, a common ecological interaction in many phytoseiid mite species. Cannibalism on eggs by A. herbicolus was significantly reduced in the presence of high-quality food (cattail pollen) compared to egg cannibalism without alternative food, whereas this was not the case in the presence of low-quality food (cotton pollen). This suggests that cattail pollen is a high-quality alternative food, not only because it results in increased development and reproduction of predators, but also because it can minimize cannibalism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Providing alternative sources of food to natural enemies has become an important strategy in biological control. It allows the introduction of the biological control agent before the pest is present in crops and enables their persistence when the target pest is scarce (McMurtry and Scriven 1966; Altieri and Letourneau 1982; Landis et al. 2000; Gurr et al. 2017). Different methods can be used to provide alternative foods in crops; one is the use of non-crop plants that provide food or prey to natural enemies (Ramakers and Voet 1995; Frank 2010; Amaral et al. 2013; van Rijn et al. 2013; Avery et al. 2014; Kumar et al. 2015; Fonseca et al. 2017). Another way is to directly supply pollen or other food sources on the crop plant (van Rijn et al. 2002; Duso et al. 2004; González-Fernández et al. 2009; van Maanen et al. 2010; Adar et al. 2014; Duarte et al. 2015). Many predatory arthropods feed on pollen as alternative food, i.e., predatory bugs (Wong and Frank 2013), ladybugs (Amaral et al. 2013), lacewings (Venzon et al. 2006), hoverflies (van Rijn et al. 2013) and predatory mites (McMurtry and Scriven 1966; Nomikou et al. 2002; van Rijn et al. 2002).
The addition of alternative food such as pollen to a crop can enhance the efficacy of pest control because it increases in the densities of natural enemies, which subsequently causes a reduction in pest populations (van Rijn et al. 2002; Nomikou et al. 2010; Delisle et al. 2015; Lee and Zhang 2018). However, the nutritional value may differ among alternative food types. For instance, pollen morphology may affect its edibility and thus affect the reproductive success and population growth of natural enemies (Yue et al. 1994; van Rijn and Tanigoshi 1999; Goleva and Zebitz 2013). Furthermore, providing alternative food to natural enemies can directly affect the interactions with conspecifics (e.g., cannibalism) and with other biocontrol agents (e.g., intraguild predation) (Lucas et al. 1998; Shakya et al. 2009; Frank et al. 2010; Calabuig et al. 2018). Here, we investigate the effects of the quality of alternative food on the occurrence of cannibalism. Reduction of cannibalism can result in changes in the densities of natural enemies, and consequently, in changes of pest densities.
Cannibalism consists of killing and at least partially consuming conspecific individuals and has been recorded in more than 1300 animal taxa (Fox 1975; Elgar and Crespi 1992). Cannibalism significantly affects population dynamics (Persson et al. 2003; Rudolf 2007), but its occurrence, strength and effect varies within and among species (Fox 1975; Leonardsson 1991; Rudolf 2008). Cannibalism may result in population extinction, but also in persistence if juveniles and adults feed on different resources and the adult food source is scarce (van den Bosch et al. 1988). One group of natural enemies that is known for cannibalism are predatory mites (Schausberger 2003; Revynthi et al. 2018). These phytoseiids feed on arthropod prey of different families and many species can also feed on plant-provided food sources (e.g., pollen, nectar, plant exudates) (McMurtry and Croft 1997; van Rijn and Tanigoshi 1999; Nomikou et al. 2003; Croft et al. 2004). The use of pollen as food for generalist predatory mites in biological control has a long history, mainly related to mass rearing under laboratory conditions (McMurtry and Scriven 1965), but also as supplemental food in crops (Ramakers 1990; Nomikou et al. 2002; van Rijn et al. 2002; Montserrat et al. 2013; Adar et al. 2014; Lee and Zhang 2018).
The omnivorous predatory mite Amblyseius herbicolus (Chant) (Acari: Phytoseiidae) is a generalist predator of phytophagous mites and insects, capable of reproducing and developing when feeding on pollen alone (Reis et al. 2007; Rodríguez-Cruz et al. 2013; Duarte et al. 2015). Supplementing pepper plants with pollen resulted in higher densities of this predator and better control of broad mite populations (Duarte et al. 2015). These higher predator densities were obviously caused by the addition of food, but possibly also because of lower levels of cannibalism. However, the effect of pollen on the cannibalistic behaviour of this species has not been investigated before. The objective of the present study was therefore to assess the juvenile development and survival, and the oviposition rate of A. herbicolus on five pollen species as alternative foods. Furthermore, we investigated the effect of pollen quality on the tendency of this predatory mite to engage in cannibalism.
Materials and methods
Rearing of Amblyseius herbicolus
A rearing of A. herbicolus was started with mites collected from tomato plants in gardens in the urban and rural areas of Prados (Minas Gerais, Brazil, 21º 03′ 0″ S, 44º 04′ 47″ W) by Cardoso (2019). The mites were reared on arenas made of PVC sheets (15 × 10 cm) on top of foam pads (h = 4 cm), which were kept in plastic trays (29 × 14 × 4 cm) filled with water. To avoid mite escapes, the edges of the arenas were wrapped in wet tissue paper, which also served as a water source (van Rijn and Tanigoshi 1999). Six small pieces of tent-shaped PVC sheet were placed randomly on the arena to serve as shelters. A small piece of cotton wool was placed below each tent as an oviposition site and cattail pollen was offered as food. These arenas were kept in a climate-controlled room (25 ± 1 ºC, 70 ± 10% RH, 12:12 L:D).
Pollen sources
Five pollen species were tested as food source for A. herbicolus: palm (Syagrus romanzoffiana [Cham.] Glassman, Arecaceae), castor bean (Ricinus communis L., Euphorbiaceae), corn (Zea mays L., Poaceae), cattail (Typha sp., Typhaceae) and cotton pollen (Gossypium hirsutum L., Malvaceae). They were all obtained from pesticide-free and non-transgenic plants on the campus of the Federal University of Viçosa (Minas Gerais, Brazil). From palm and castor bean, flowering bunches and branches were collected, respectively. They were cut, and their stems were placed in buckets filled with water to maintain turgidity until the flowers opened. After 72 h, paper was placed under the reproductive structures to collect falling pollen, which was transferred to Petri dishes (Ø = 10 cm, h = 1 cm). Corn and cattail pollen were obtained by collecting male reproductive structures of the plants and shaking them above a Petri dish. Cotton pollen was taken from the flowers of the plants with a fine brush and was transferred to a Petri dish. All pollen were dried in an oven (50 ºC) for 24 h and subsequently transferred to plastic tubes (Ø = 1 cm, h = 5 cm) and stored in a refrigerator at 4 °C until further use.
Experimental arenas
The experimental units used for all experiments consisted of black plastic Petri dishes (Ø = 5 cm, h = 1.5 cm) closed with transparent lids. The black color of the Petri dishes allowed better observation of the mites. A small piece of tissue paper soaked in water was placed in each arena as a source of water. All adults used in the experiments were gravid females derived from the rearing arenas, aged between 10 and 12 days after egg eclosion (5–7 days old since becoming adult). All experiments were conducted in the same climate-controlled room as above.
Juvenile development and survival
To evaluate the suitability of different diets as a food source for phytoseiid mites, it is common to estimate the intrinsic growth rate (rm), performing a full life table study. However, this method requires frequent evaluations for extensive periods (Van Dinh et al. 1988). Indeed, a simpler method to compare the performance of phytoseiids among diets, including prey, is to measure the peak rate of oviposition, developmental rate and juvenile survival (Janssen and Sabelis 1992; Nomikou et al. 2001).
Newly hatched larvae of A. herbicolus were removed from the stock colony and each individual was placed in an experimental unit. Ample amounts of one of the five species of pollen were offered on small pieces of plastic PVC (1 cm2). To avoid depletion and loss of quality of the pollen, new pollen was offered daily on new pieces of plastic PVC; those of the previous day were removed to avoid contamination with fungi. Twenty replicates were carried out for each pollen species. Juvenile development and survival were monitored daily until the individuals reached adulthood or had died. Data on the effect of different species of pollen on development time and survival were analysed with Cox proportional hazards model of the ‘survival’ package (Therneau 2013) in R v.3.6.0 (R Core Team 2019).
Oviposition
For each replicate, a single adult female of A. herbicolus was taken from the rearing and placed on an experimental unit as above. Oviposition was recorded daily for 5 days, but the oviposition of the first day was not included in the analysis because of the possible effect of the diet of the preceding days (Sabelis 1990). The eggs were counted daily while removing them from the arenas. The effect of pollen species on the oviposition rate was assessed with a linear mixed-effects model with treatment and time as fixed factors and individuals as a random factor to correct for repeated measures. Contrasts among treatments were assessed with the Tukey method with the package lsmeans (Lenth 2016).
Effect of diet quality on cannibalism
Based on the experiments above, cattail pollen (Typha sp.) and cotton pollen (G. hirsutum) were selected as the best and worst diet, respectively. We subsequently investigated the effect of pollen quality on the tendency of A. herbicolus to engage in cannibalism. Amblyseius herbicolus eggs were used as the stage to be cannibalized by adult females.
Adult females were placed singly on the experimental units and were starved for 24 h before being used in the experiment to avoid possible effects of the previous diet. Then, they were placed in the Petri dishes together with six conspecific eggs (< 24 h old). Ample amounts of cattail or cotton pollen were placed in the experimental units, except for control treatment, which had no pollen. The numbers of cannibalized eggs were assessed after 1, 3, 5, 20 and 24 h using a stereoscopic microscope (Zeiss Stemi 2000-c). Cannibalized eggs were recognized by the shell from which the internal egg content was removed (adapted from Yao and Chant 1989). No eggs hatched during the experiment. Twenty replicates were carried out for each diet. Data on the effect of diet on cannibalism were analysed with a Cox mixed effects model (coxme) of the ‘survival’ package (Therneau 2015). Diets (cattail pollen, cotton pollen and no food) were analysed as fixed factors and replicate as random factor to correct for repeated measures. All analyses were performed with the statistical software R (R Core Team 2019).
Results
Juvenile development and survival
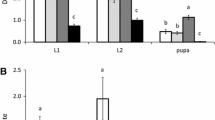
Diets of different species of pollen significantly affected the juvenile development of the predator (Fig. 1, Cox proportional hazards: likelihood ratio = 37.3, df = 4, P < 0.0001). Juvenile development was shorter on diets consisting of cattail pollen and palm pollen. Feeding on cotton pollen resulted in the longest juvenile development. There was no significant effect of predator diet on juvenile survival (Fig. 1, Cox proportional hazards: likelihood ratio = 6.34, df = 4, P = 0.17).
Development and survival of Amblyseius herbicolus juveniles fed on pollen from cattail, palm, corn, castor bean and cotton. Shown are the mean (± SE) cumulative proportions of individuals that reached adulthood as a function of time. Survival is given by the final proportion of adults. Juvenile development in treatments with different letters (see the key entries) differed significantly (contrasts after survival analysis, P < 0.05)
Oviposition
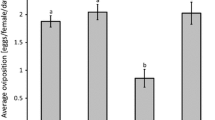
There was a significant effect of pollen species on the oviposition of A. herbicolus (Fig. 2, LME: \({{\rm X}}_{4}^{2}\)= 12.9, P = 0.012). Feeding on cattail and palm tree pollen resulted in the highest oviposition rates, but oviposition on palm pollen differed significantly among days (Fig. 2, cattail: F1,50 = 0.38, P = 0.54; palm: F1,53 = 4.49, P = 0.039). Oviposition on corn pollen was intermediate between palm and castor bean pollen but was not significantly different among days (Fig. 2, F1,41 = 0.06, P = 0.79). The oviposition rate with castor bean pollen decreased significantly among days (Fig. 2, F1,50 = 25.37, P < 0.001). Cotton pollen resulted in the lowest oviposition rate (Fig. 2, F1,47 = 13.75, P < 0.001). The sharp drop in oviposition with castor bean and cotton during the last days of evaluation may indicate longer-term negative effects of these pollens.
Mean (± SE) oviposition rate of Amblyseius herbicolus on the 2nd–5th day of feeding on a diet consisting of cattail pollen (17 replicates), palm pollen (18), corn pollen (14), castor bean pollen (17) and cotton pollen (16). Different letters following the key entries indicate significant differences among treatments (contrasts through model simplification after LME; P < 0.05). Asterisks indicate a significant difference in oviposition rates among days for each treatment. *0.01 < P < 0.05; ***P < 0.001; n.s not significant
Effect of diet quality on cannibalism
No eggs were cannibalized per 24 h in the presence of cattail pollen, and less than 20% of the eggs were cannibalized in the treatment with cotton pollen and in the control (Fig. 3). Because the survival analysis requires at least some mortality in all treatments, we assumed that one egg of the treatment with cattail pollen was cannibalized at the maximum duration of the experiment (24 h). Notice that this will result in underestimating the difference among treatments. The resulting statistical analysis showed that there was a significant effect of diet quality on cannibalism (Fig. 3, Cox mixed effects: Log likelihood ratio = 17.7, df = 2, P < 0.001) and the cattail treatment differed significantly from the two other treatments, showing that cannibalism was affected by diet quality.
Mean (± SE) cumulative proportion of cannibalized eggs of Amblyseius herbicolus as a function of time in the presence of a conspecific adult predator (cannibal). Besides on the eggs, the cannibals could feed on cattail pollen (high-quality food), cotton pollen (low-quality), or had no food (control). Treatments with different letters (see the key entries) were significantly different (contrasts after a Cox mixed-effects proportional hazards model, P < 0.05)
Discussion
The predatory mite A. herbicolus was able to develop and reproduce when fed each of the five species of pollen under laboratory conditions, showing that they fed on all pollen species. However, the pollen clearly affected oviposition rates and juvenile development differentially; hence, they differed in quality (Figs. 1 and 2). We also show that egg cannibalism by A. herbicolus adults was affected by the quality of the alternative diet offered. Mites fed with cattail pollen, the superior food source, did not display cannibalistic behaviour; adult females that fed on cotton pollen, the inferior diet, and mites without food cannibalized significantly more (Fig. 3).
One of the main benefits of cannibalism is the acquisition of nutrients, especially when food availability is low (Fox 1975; van den Bosch et al. 1988). However, there is common consensus that there is no nutritional benefit of cannibalism when optimal conditions are present (Via 1999). Thus, cannibalism is expected to be more prevalent with nutritionally inferior food sources (Vangansbeke et al. 2014), in agreement with the results obtained here (Fig. 3). Vangansbeke et al. (2014) showed that females of Amblydromalus limonicus fed with Typha pollen cannibalized less than when feeding on other alternative food sources. Ferreira et al. (2008) also found that previous feeding with cattail pollen decreased cannibalism in Iphiseius degenerans. Typha sp. was also found to be a high-quality diet in other studies (van Rijn and Tanigoshi 1999; Nomikou et al. 2003; Messelink et al. 2008; Goleva and Zebitz 2013; Lee and Zhang 2018; Ajila et al. 2019). Performance of this strain of A. herbicolus soon after the establishment of the culture in the lab was lower than the performance found here, showing that the predators had adapted to cattail pollen as a food source.
In contrast, cannibalism was observed when cotton pollen was present, and this treatment did not differ significantly from that without food. Previous studies have also shown that cotton pollen is low-quality food for predatory mites (Elbadry and Elbenhawy 1968; Zaher and Shehata 1971). This may be due to low concentrations of some nutrients in this pollen. The absence—or reduced concentration—of specific factors in food, such as protein, vitamins or minerals, have been implicated as stimuli for cannibalism, especially related to the nitrogen content (Wolcott and Wolcott 1984). Cannibalism, then, may be an ideal strategy for N-limited predators to meet nutritional requirements when prey is scarce or when alternative foods are of low quality (Denno and Fagan 2003). Another reason for the low quality of pollen may be the presence of defensive compounds. Although concentrations of secondary metabolites in pollen are possibly too low to be harmful to predatory mites (Ranabhat et al. 2014), negative effects may occur. The sharp drop in oviposition with cotton pollen, also observed for castor bean during the last days of the experiment (Fig. 2), may indicate long-term accumulation of these secondary metabolites in the mites. Furthermore, there is possibly a relationship between the ability of mites to feed and develop on certain species of pollen with differences in mite morphology (e.g., feeding apparatus, sensory organs), physiology (digestive system) and behaviour (e.g., feeding preferences) (van Rijn and Tanigoshi 1999). These relationships require further investigation for A. herbicolus.
Proximate mechanisms may also explain why hunger and decreased diet quality promote cannibalism. First, hunger triggers foraging behaviour, increasing the likelihood of intraspecific contact and hence cannibalism (Charnov 1976). Second, foraging theory predicts that consumers should expand their diet during periods of hunger or low levels of food (Charnov 1976). Under this scenario, the new diet will include items previously ignored due to low energy gain and/or high acquisition costs, such as the risk of trying to cannibalize (Polis 1981). The lower nutrient content of cotton compared to cattail pollen probably caused reduced selectivity of the adult females, and hence engagement in cannibalism.
In nature, resources are often ephemeral and organisms will therefore commonly encounter situations without food, and thus have to adapt to feed on other available resources. The presence of alternative food can ensure the longer persistence of these organisms when food is scarce (Altieri and Letourneau 1982; Landis et al. 2000; Duarte et al. 2015; Gurr et al. 2017). Several papers have shown the potential of pollen as alternative food for predatory mites (van Rijn and Tanigoshi 1999; Nomikou et al. 2003; Goleva and Zebitz 2013; Montserrat et al. 2013; Duarte et al. 2015; Ajila et al. 2019) and how it can be used to improve biocontrol (van Rijn et al. 2002; Duso et al. 2004; González-Fernández et al. 2009; Nomikou et al. 2010; Delisle et al. 2015; Duarte et al. 2015). However, only a few studies have investigated whether pollen quality can affect interactions between natural enemies and thus influence biological control. For example, Pina et al. (2012) have shown that low-quality pollen resulted in satisfactory control of Tetranychus urticae by a combination of two phytoseiid mites because this pollen does not support high densities of the species Euseius stipulatus, which is the superior intraguild predator, thus reducing lethal and non-lethal interactions with the intraguild prey Neoseiulus californicus. Calabuig et al. (2018) found that feeding E. stipulatus with high-quality cattail pollen decreased larval cannibalism and intraguild predation by this mite. Other studies with different alternative foods have also shown that the quality of the resource can affect the levels of cannibalism and intraguild predation for various species of natural enemies (Snyder et al. 2000; Lucas et al. 2009). Here, we demonstrate that egg cannibalism by adults of A. herbicolus did not occur when cattail pollen was provided. Therefore, cattail pollen probably provided the necessary nutrients for A. herbicolus, thereby decreasing the engagement of this predator mite in cannibalism.
In conclusion, our study shows that providing high-quality alternative food will result in higher densities of the biocontrol agent A. herbicolus, not only because of increased development and reproduction, but also because the presence of pollen decreased cannibalism. Together, this may explain the higher densities of A. herbicolus on plants with pollen observed by Duarte et al. (2015). Factors affecting the densities of natural enemies will also indirectly determine the densities of the target pest species and therefore the efficacy of biocontrol. Thus, population-dynamical experiments on plants are needed to verify whether the addition of alternative food is beneficial for biological control, especially for natural enemies that display cannibalistic behaviour, simply because food is added, or also because it reduces cannibalism.
References
Adar E, Inbar M, Gal S et al (2014) Pollen on-twine for food provisioning and oviposition of predatory mites in protected crops. Biocontrol 59:307–317
Altieri MA, Letourneau DK (1982) Vegetation management and biological control in agroecosystems. Crop Prot 1:405–430
Amaral DSSL, Venzon M, Duarte MVA et al (2013) Non-crop vegetation associated with chili pepper agroecosystems promote the abundance and survival of aphid predators. Biol Control 64:338–346
Avery PB, Kumar V, Xiao Y et al (2014) Selecting an ornamental pepper banker plant for Amblyseius swirskii in floriculture crops. Arthropod Plant Interact 8:49–56
Calabuig A, Pekas A, Wäckers FL (2018) The quality of nonprey food affects cannibalism, intraguild predation, and hyperpredation in two species of phytoseiid mites. J Econ Entomol 111:72–77
Cardoso AC (2019) Search for predatory mites to control tomato pests. MSc Dissertation, Federal University of Viçosa
Charnov EL (1976) Optimal foraging theory: the marginal value theorem. Theor Popul Biol 9:129–136
Croft BA, Blackwood JS, McMurtry JA (2004) Classifying life-style types of phytoseiid mites: diagnostic traits. Exp Appl Acarol 33:247–260
Delisle JF, Shipp L, Brodeur J (2015) Apple pollen as a supplemental food source for the control of western flower thrips by two predatory mites, Amblyseius swirskii and Neoseiulus cucumeris (Acari: Phytoseiidae), on potted chrysanthemum. Exp Appl Acarol 65:495–509
Denno RF, Fagan WF (2003) Might nitrogen limitation promote omnivory among carnivorous arthropods? Ecology 84:2522–2531
Duarte MV, Venzon M, de Bittencourt MC et al (2015) Alternative food promotes broad mite control on chilli pepper plants. Biocontrol 60:817–825
Duso C, Malagnini V, Paganelli A et al (2004) Pollen availability and abundance of predatory phytoseiid mites on natural and secondary hedgerows. Biocontrol 49:397–415
Elbadry E, Elbenhawy E (1968) The effect of non-prey food, mainly pollen, on the development, survival, and fecundity of Amblyseius gossipi (Acarina: Phytoseiidae). Entomol Exp Appl 11:269–272
Elgar MA, Crespi BJ (1992) Cannibalism. Ecology and evolution among diverse taxa. Oxford University Press, New York
Ferreira JAM, Eshuis B, Janssen A, Sabelis MW (2008) Domatia reduce larval cannibalism in predatory mites. Ecol Entomol 33:374–379
Fonseca MM, Lima E, Lemos F et al (2017) Non-crop plant to attract and conserve an aphid predator (Coleoptera: Coccinellidae) in tomato. Biol Control 115:129–134
Fox LR (1975) Cannibalism in natural populations. Annu Rev Ecol Syst 6:87–106
Frank SD (2010) Biological control of arthropod pests using banker plant systems: past progress and future directions. Biol Control 52:8–16
Frank SD, Shrewsbury PM, Denno RF (2010) Effects of alternative food on cannibalism and herbivore suppression by carabid larvae. Ecol Entomol 35:61–68
Goleva I, Zebitz CPW (2013) Suitability of different pollen as alternative food for the predatory mite Amblyseius swirskii (Acari, Phytoseiidae). Exp Appl Acarol 61:259–283
González-Fernández J, De la Peña F, Hormaza JI et al (2009) Alternative food improves the combined effect of an omnivore and a predator on biological pest control. A case study in avocado orchards. Bull Entomol Res 99:433–444
Gurr GM, Wratten SD, Landis DA, You M (2017) Habitat management to suppress pest populations: progress and prospects. Annu Rev Entomol 62:91–109
Janssen A, Sabelis MW (1992) Phytoseiid life-histories, local predator-prey dynamics, and strategies for control of tetranychid mites. Exp Appl Acarol 14:233–250
Kumar V, Xiao Y, McKenzie C, Osborne L (2015) Early establishment of the phytoseiid mite Amblyseius swirskii (Acari: Phytoseiidae) on pepper seedlings in a Predator-in-First approach. Exp Appl Acarol 65:465–481
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201
Lee MH, Zhang Z-Q (2018) Assessing the augmentation of Amblydromalus limonicus with the supplementation of pollen, thread, and substrates to combat greenhouse whitefly populations. Sci Rep 8:1–14
Lenth R (2016) Least-squares means: the R package lsmeans. J Stat Softw 69:1–33
Leonardsson K (1991) Effects of cannibalism and alternative prey on population dynamics of Saduria entomon (Isopoda). Ecology 72:1273–1285
Lucas E, Coderre D, Brodeur J (1998) Intraguild predation among aphid predators: characterization and influence of extraguild prey density. Ecology 79:1084–1092
Lucas É, Fréchette B, Alomar O (2009) Resource quality, resource availability, and intraguild predation among omnivorous mirids. Biocontrol Sci Technol 19:555–572
McMurtry J, Scriven G (1965) Insectary production of phytoseiid mites. J Econ Entomol 58:282–284
McMurtry J, Scriven G (1966) The influence of pollen and prey density on the number of prey consumed by Amblyseius hibisci (Acarina: Phytoseiidae). Ann Entomol Soc Am 59:147–149
McMurtry JA, Croft BA (1997) Life-styles of phytoseiid mites and their role in biological control. Annu Rev Entomol 42:291–321
Messelink GJ, van Maanen R, van Steenpaal SEF, Janssen A (2008) Biological control of thrips and whiteflies by a shared predator: two pests are better than one. Biol Control 44:372–379
Montserrat M, Guzman C, Sahun RM et al (2013) Pollen supply promotes, but high temperatures demote, predatory mite abundance in avocado orchards. Agric Ecosyst Environ 164:155–161
Nomikou M, Janssen A, Schraag R, Sabelis MW (2001) Phytoseiid predators as potential biological control agents for Bemisia tabaci. Exp Appl Acarol 25:271–291
Nomikou M, Janssen A, Schraag R, Sabelis MW (2002) Phytoseiid predators suppress populations of Bemisia tabaci on cucumber plants with alternative food. Exp Appl Acarol 27:57–68
Nomikou M, Janssen A, Sabelis MW (2003) Phytoseiid predators of whiteflies feed and reproduce on non- prey food sources. Exp Appl Acarol 31:15–26
Nomikou M, Sabelis MW, Janssen A (2010) Pollen subsidies promote whitefly control through the numerical response of predatory mites. Biocontrol 55:253–260
Persson L, De Roos AM, Claessen D et al (2003) Gigantic cannibals driving a whole-lake trophic cascade. Proc Natl Acad Sci USA 100:4035–4039
Pina T, Argolo PS, Urbaneja A, Jacas JA (2012) Effect of pollen quality on the efficacy of two different life-style predatory mites against Tetranychus urticae in citrus. Biol Control 61:176–183
Polis GA (1981) The evolution and dynamics of intraspecific predation. Annu Rev Ecol Syst 12:225–251
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramakers PMJ (1990) Manipulation of phytoseiid thrips predators in the absence of thrips. IOBC-WPRS Bull 13:169–172
Ramakers PMJ, Voet SJP (1995) Use of castor bean, Ricinus communis, for the introduction of the thrips predator Amblyseius degenerans on glasshouse-grown sweet pepper. Meded Fac Landbouwwet Rijksuniv Gent 60:885–891
Ranabhat NB, Goleva I, Zebitz CP (2014) Life tables of Neoseiulus cucumeris exclusively fed with seven different pollens. Biocontrol 59:195–203
Reis PR, Teodoro AV, Pedro Neto M, da Silva EA (2007) Life history of Amblyseius herbicolus (Chant) (Acari: Phytoseiidae) on coffee plants. Neotrop Entomol 36:282–287
Revynthi AM, Janssen A, Egas M (2018) Gender-specific differences in cannibalism between a laboratory strain and a field strain of a predatory mite. Exp Appl Acarol 74:239–247
Rodríguez-Cruz FA, Venzon M, Pinto CMF (2013) Performance of Amblyseius herbicolus on broad mites and on castor bean and sunnhemp pollen. Exp Appl Acarol 60:497–507
Rudolf VHW (2007) The interaction of cannibalism and omnivory: consequences for community dynamics. Ecology 88:2697–2705
Rudolf VHW (2008) Impact of cannibalism on predator-prey dynamics: size-structured interactions and apparent mutualism. Ecology 89:1650–1660
Sabelis MW (1990) How to analyze prey preference when prey density varies? A new method to discriminate between effects of gut fullness and prey type composition. Oecologia 82:289–298
Schausberger P (2003) Cannibalism among phytoseiid mites: a review. Exp Appl Acarol 29:173–191
Shakya S, Weintraub PG, Coll M (2009) Effect of pollen supplement on intraguild predatory interactions between two omnivores: the importance of spatial dynamics. Biol Control 50:281–287
Snyder WE, Joseph SB, Preziosi RF, Moore AJ (2000) Nutritional benefits of cannibalism for the lady beetle Harmonia axyridis (Coleoptera: Coccinellidae) when prey quality is poor. Environ Entomol 29:1173–1179
Therneau TM (2013) A package for survival analysis in S
Therneau TM (2015) COXME: mixed effects Cox models
Vacacela Ajila HE, Colares F, Lemos F et al (2019) Supplementary food for Neoseiulus californicus boosts biological control of Tetranychus urticae on strawberry. Pest Manag Sci 75:1986–1992
van den Bosch F, de Roos AM, Gabriel W (1988) Cannibalism as a life boat mechanism. J Math Biol 26:619–633
van Dinh N, Janssen A, Sabelis MW (1988) Reproductive success of Amblyseius idaeus and Amblyseius anonymus on a diet of two-spotted spider mites. Exp Appl Acarol 4:41–51
van Maanen R, Vila E, Sabelis MW, Janssen A (2010) Biological control of broad mites (Polyphagotarsonemus latus) with the generalist predator Amblyseius swirskii. Exp Appl Acarol 52:29–34
van Rijn PCJ, Tanigoshi LK (1999) Pollen as food for the predatory mites Iphiseius degenerans and Neoseiulus cucumeris (Acari: Phytoseiidae): dietary range and life history. Exp Appl Acarol 23:785–802
van Rijn PCJ, van Houten YM, Sabelis MW (2002) How plants benefit from providing food to predators even when it is also edible to herbivores. Ecology 83:2664–2679
van Rijn PC, Kooijman J, Wäckers FL (2013) The contribution of floral resources and honeydew to the performance of predatory hoverflies (Diptera: Syrphidae). Biol Control 67:32–38
Vangansbeke D, Nguyen DT, Audenaert J et al (2014) Diet-dependent cannibalism in the omnivorous phytoseiid mite Amblydromalus limonicus. Biol Control 74:30–35
Venzon M, Rosado MC, Euzébio DE et al (2006) Suitability of leguminous cover crop pollens as food source for the green lacewing Chrysoperla externa (Hagen) (Neuroptera: Chrysopidae). Neotrop Entomol 35:371–376
Via S (1999) Cannibalism facilitates the use of a novel environment in the flour beetle, Tribolium castaneum. Heredity 82:267–275
Wolcott DL, Wolcott TG (1984) Food quality and cannibalism in the red land crab, Gecarcinus lateralis. Physiol Zool 57:318–324
Wong SK, Frank SD (2013) Pollen increases fitness and abundance of Orius insidiosus Say (Heteroptera: Anthocoridae) on banker plants. Biol Control 64:45–50
Yao D, Chant D (1989) Population growth and predation interference between two species of predatory phytoseiid mites (Acarina: Phytoseiidae) in interactive systems. Oecologia 80:443–455
Yue B, Childers CC, Fouly AH (1994) A comparison of selected plant pollens for rearing Euseius mesembrinus (Acari: Phytoseiidae). Int J Acarol 20:103–108
Zaher M, Shehata KK (1971) Biological studies on the predator mite Typhlodromus pyri Sch. (Acarina Phytoseiidae) with the effect of prey and non prey substances. Z Für Angew Entomol 67:389–394
Acknowledgements
We thank Dr. Madelaine Venzon, Dr. Rodrigo Soares Ramos and colleagues from the Laboratory of Acarology of the Federal University of Viçosa for suggestions on experimental design and discussions. Constructive comments of the three anonymous reviewers were highly appreciated. We also thank the Brazilian research funding agencies Coordination for the Improvement of Higher Education Personnel (CAPES), Foundation for Research Support of the State of Minas Gerais (FAPEMIG) and the National Council of Scientific and Technological Development (CNPq). IM was supported by CNPq and CAPES. MMF (Process 88887.314241/2019-00) and PAFC were supported by CAPES. AP by FAPEMIG and CNPq.
Author information
Authors and Affiliations
Contributions
IM, AJ, and MMF conceived the ideas and designed the methodology; AC: collected and identified the mites and performed initial oviposition tests; IM, MMF and PAFC collected the data, IM and AJ analysed the data; IM, AP and AJ led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The experiments comply with the current laws of the country in which the experiments were performed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marcossi, Í., Fonseca, M.M., Carbajal, P.A.F. et al. High-quality alternative food reduces cannibalism in the predatory mite Amblyseius herbicolus (Acari: Phytoseiidae). Exp Appl Acarol 81, 189–200 (2020). https://doi.org/10.1007/s10493-020-00500-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-020-00500-7