Abstract
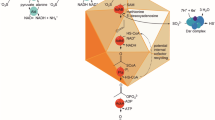
The gram-negative anaerobic gut bacterium Bilophila wadsworthia is the third most common isolate in perforated and gangrenous appendicitis, being also found in a variety of other infections. This organism performs a unique kind of anaerobic respiration in which taurine, a major organic solute in mammals, is used as a source of sulphite that serves as terminal acceptor for the electron transport chain. We show here that molecular hydrogen, one of the major products of fermentative bacteria in the colon, is an excellent growth substrate for B. wadsworthia. We have quantified the enzymatic activities associated with the oxidation of H2, formate and pyruvate for cells obtained in different growth conditions. The cell extracts present high levels of hydrogenase activity, and up to five different hydrogenases can be expressed by this organism. One of the hydrogenases appears to be constitutive, whereas the others show differential expression in different growth conditions. Two of the hydrogenases are soluble and are recognised by antibodies against a [FeFe] hydrogenase of a sulphate reducing bacterium. One of these hydrogenases is specifically induced during fermentative growth on pyruvate. Another two hydrogenases are membrane-bound and show increased expression in cells grown with hydrogen. Further work should be carried out to reveal whether oxidation of hydrogen contributes to the virulence of B. wadsworthia.





Similar content being viewed by others
References
Abbe K, Takahashi S, Yamada T (1982) Involvement of oxygen-sensitive pyruvate formate-lyase in mixed-acid fermentation by Streptococcus mutans under strictly anaerobic conditions. J Bacteriol 152:175–182
Backhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI (2005) Host-bacterial mutualism in the human intestine. Science 307:1915–1920
Baron EJ (1997) Bilophila wadsworthia: a unique gram-negative anaerobic rod. Anaerobe 3:83–86
Baron EJ, Summanen P, Downes J, Roberts MC, Wexler H, Finegold SM (1989) Bilophila wadsworthia, gen. nov. and sp. nov., a unique gram-negative anaerobic rod recovered from appendicitis specimens and human faeces. J Gen Microbiol 135:3405–3411
Baron EJ, Curren M, Henderson G, Jousimiessomer H, Lee K, Lechowitz K, Strong CA, Summanen P, Tuner K, Finegold SM (1992) Bilophila wadsworthia isolates from clinical specimens. J Clin Microbiol 30:1882–1884
Campieri M, Gionchetti P (2001) Bacteria as the cause of ulcerative colitis. Gut 48:132–135
Chabriere E, Vernede X, Guigliarelli B, Charon MH, Hatchikian EC, Fontecilla-Camps JC (2001) Crystal structure of the free radical intermediate of pyruvate:ferredoxin oxidoreductase. Science 294:2559–2563
Claros MC, Schumacher U, Jacob M, Gerardo SH, Kleinkauf N, Goldstein EJC, Finegold SM, Rodloff AC (1999) Characterization of Bilophila wadsworthia isolates using PCR fingerprinting. Anaerobe 5:589–593
Cummings JH, Macfarlane GT (1991) The control and consequences of bacterial fermentation in the human colon. J Appl Bacteriol 70:443–459
de Bok FAM, Roze EHA, Stams AJM (2002) Hydrogenases and formate dehydrogenases of Syntrophobacter fumaroxidans. Antonie Van Leeuwenhoek 81:283–291
Denger K, Laue H, Cook AM (1997) Thiosulfate as a metabolic product: the bacterial fermentation of taurine. Arch Microbiol 168:297–301
Finegold S, Summanen P, Hunt Gerardo S, Baron E (1992) Clinical importance of Bilophila wadsworthia. Eur J Clin Microbiol Infect Dis 11:1058–1063
Gill SR, Pop M, Deboy RT, Eckburg PB, Turnbaugh PJ, Samuel BS, Gordon JI, Relman DA, Fraser-Liggett CM, Nelson KE (2006) Metagenomic analysis of the human distal gut microbiome. Science 312:1355–1359
Guarner F (2005) The intestinal flora in inflammatory bowel disease: normal or abnormal? Curr Opin Gastroenterol 21:414–418
Guarner F (2006) Enteric flora in health and disease. Digestion 73(suppl 1):5–12
Heidelberg JF, Seshadri R, Haveman SA, Hemme CL, Paulsen IT, Kolonay JF, Eisen JA, Ward N, Methe B, Brinkac LM, Daugherty SC, Deboy RT, Dodson RJ, Durkin AS, Madupu R, Nelson WC, Sullivan SA, Fouts D, Haft DH, Selengut J, Peterson JD, Davidsen TM, Zafar N, Zhou LW, Radune D, Dimitrov G, Hance M, Tran K, Khouri H, Gill J, Utterback TR, Feldblyum TV, Wall JD, Voordouw G, Fraser CM (2004) The genome sequence of the anaerobic, sulfate-reducing bacterium Desulfovibrio vulgaris Hildenborough. Nat Biotechnol 22:554–559
Jormakka M, Byrne B, Iwata S (2003) Formate dehydrogenase—a versatile enzyme in changing environments. Curr Opin Struct Biol 13:418–423
Kelly D, Conway S, Aminov R (2005) Commensal gut bacteria: mechanisms of immune modulation. Trends Immunol 26:326–333
Laue H, Cook AM (2000a) Biochemical and molecular characterization of taurine: pyruvate aminotransferase from the anaerobe Bilophila wadsworthia. Eur J Biochem 267:6841–6848
Laue H, Cook AM (2000b) Purification, properties and primary structure of alanine dehydrogenase involved in taurine metabolism in the anaerobe Bilophila wadsworthia. Arch Microbiol 174:162–167
Laue H, Denger K, Cook AM (1997) Taurine reduction in anaerobic respiration of Bilophila wadsworthia RZATAU. Appl Environ Microbiol 63:2016–2021
Laue H, Friedrich M, Ruff J, Cook AM (2001) Dissimilatory sulfite reductase (desulfoviridin) of the taurine-degrading, non-sulfate-reducing bacterium Bilophila wadsworthia RZATAU contains a fused DsrB-DsrD subunit. J Bacteriol 183:1727–1733
Laue H, Smits TH, Schumacher UK, Claros MC, Hartemink R, Cook AM (2006) Identification of Bilophila wadsworthia by specific PCR which targets the taurine:pyruvate aminotransferase gene. FEMS Microbiol Lett 261:74–79
Lie TJ, Clawson ML, Godchaux W, Leadbetter ER (1999) Sulfidogenesis from 2-aminoethanesulfonate (taurine) fermentation by a morphologically unusual sulfate-reducing bacterium, Desulforhopalus singaporensis sp. nov Appl Environ Microbiol 65:3328-3334
Loubinoux J, Bronowicki JP, Pereira IAC, Mougenel JL, Le Faou AE (2002) Sulfate-reducing bacteria in human feces and their association with inflammatory bowel diseases. FEMS Microbiol Ecol 40:107–112
Macdonald TT, Monteleone G (2005) Immunity, inflammation, and allergy in the gut. Science 307:1920–1925
Maier RJ (2005) Use of molecular hydrogen as an energy substrate by human pathogenic bacteria. Biochem Soc Trans 33:83–85
Matias PM, Pereira IAC, Soares CM, Carrondo MA (2005) Sulphate respiration from hydrogen in Desulfovibrio bacteria: a structural biology overview. Progr Biophys Mol Biol 89:292–329
Mazmanian SK, Kasper DL (2006) The love-hate relationship between bacterial polysaccharides and the host immune system. Nat Rev Immunol 6:849–858
McOrist S, Keller L, McOrist AL (2003) Search for Lawsonia intracellularis and Bilophila wadsworthia in malabsorption-diseased chickens. Can J Vet Res 67:232–234
McOrist AL, Warhurst M, McOrist S, Bird AR (2001) Colonic infection by Bilophila wadsworthia in pigs. J Clin Microbiol 39:1577–1579
Narushima S, Itoha K, Miyamoto Y, Park SH, Nagata K, Kuruma K, Uchida K (2006) Deoxycholic acid formation in gnotobiotic mice associated with human intestinal bacteria. Lipids 41:835–843
Olson JW, Maier RJ (2002) Molecular hydrogen as an energy source for Helicobacter pylori. Science 298:1788–1790
Roediger WEW, Moore J, Babidge W (1997) Colonic sulfide in pathogenesis and treatment of ulcerative colitis. Dig Dis Sci 42:1571–1579
Schumacher UK, Eiring P, Hacker FM (1997) Incidence of Bilophila wadsworthia in appendiceal, peritoneal and fecal samples from children. Clin Microbiol Infect 3:134–136
Unden G, Bongaerts J (1997) Alternative respiratory pathways of Escherichia coli: energetics and transcriptional regulation in response to electron acceptors. Biochim Biophys Acta—Bioenerg 1320:217–234
Valente FMA, Almeida CC, Pacheco I, Carita J, Saraiva LM, Pereira IAC (2006) Selenium is involved in regulation of periplasmic hydrogenase gene expression in Desulfovibrio vulgaris Hildenborough. J Bacteriol 188:3228–3235
Vignais PM, Colbeau A (2004) Molecular biology of microbial hydrogenases. Curr Issues Mol Biol 6:159–188
Vignais PM, Billoud B, Meyer J (2001) Classification and phylogeny of hydrogenases. FEMS Microbiol Rev 25:455–501
Vorholt JA, Thauer RK (2002) Molybdenum and tungsten enzymes in C1 metabolism. In: Sigel A, Sigel H (eds) Metal ions in biological systems, vol 39. Taylor & Francis, New York, pp 571–619
Weatherburn MW (1967) Phenol-hypochlorite reaction for determination of ammonia. Anal Chem 39:971–974
Yamamoto Y, Sato Y, Takahashi-Abbe S, Takahashi N, Kizaki H (2000) Characterization of the Streptococcus mutans pyruvate formate-lyase (PFL)-activating enzyme gene by complementary reconstitution of the in vitro PFL-reactivating system. Infect Immun 68:4773–4777
Zoetendal EG, Vaughan EE, de Vos WM (2006) A microbial world within us. Mol Microbiol 59:1639–1650
Acknowledgements
We thank João Carita for growth of B. wadsworthia cells in a bioreactor, Cristina Leitão for HPLC analysis and Isabel Pacheco for GC analysis. This work was supported by the research grant POCTI/ESP/44782/02 funded by Fundação para a Ciência e Tecnologia (FCT, MCES, Portugal) and FEDER program. F.M.A.V. was supported by FCT PhD grant SFRH/BD/9187/2002, S.M.S. by FCT PhD grant SFRH/BD/24312/2005 and S.S.V. by FCT PhD grant SFRH/BD/30648/2006.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
da Silva, S.M., Venceslau, S.S., Fernandes, C.L.V. et al. Hydrogen as an energy source for the human pathogen Bilophila wadsworthia . Antonie van Leeuwenhoek 93, 381–390 (2008). https://doi.org/10.1007/s10482-007-9215-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-007-9215-x