Abstract
The integration of trees within agricultural systems delivers the opportunity to provide multiple benefits over those afforded by agriculture without trees. The use of windbreaks as a form of agroforestry, in water scarce environments, is primarily used to reduce windspeeds in order to decrease evapotranspiration. Quick growing poplar species such as Populus simonii ((Carrière) Wesm.) are frequently utilised within windbreak structures, but to date, few allometric equations are available to quantify biomass production and to make inferences about carbon storage potential of this species, and none outside the forest. To fill this knowledge gap, we destructively sampled 17 P. simonii growing within a windbreak on a wine estate in the Western Cape Provence, South Africa. Power functions were constructed to explain tree height, whole tree aboveground woody biomass, stem and branch biomass as a function of stem diameter at 1.3 m. Additional functions were developed to predict individual branch length and biomass based on branch stub diameter. The presented models explained each variable with high significance. The models could be used to estimate carbon stock per km of windbreak for the given example. Furthermore, bark percentage predicted by stem sectional diameter was modelled to provide a function that can separate wood and bark fractions as a further outlook for the species’ utilisation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Windbreaks often consist of trees planted in a single line with close tree spacing to form a dense barrier in farmed landscapes. Sometimes, multi-row windbreak structures are also used. Windbreaks consisting of trees have both direct and indirect influences on the crop, have rotations of greater than one year and can diversify the products derived from them, which may be crops or non-timber forest product plus timber or firewood.
The primary function of a windbreak is to slow and redirect the wind field over the protected cropping area, with windbreak influence reaching mean values of 15 times windbreak height into the adjacent field (Weninger et al. 2021). Windbreaks are often aligned perpendicular to the prevailing wind or winds that are disruptive at certain times of the year. This deflection of the wind field has been shown to influence multiple environmental variables on the leeward side of the structure, positively influencing air and soil temperatures, humidity and evapotranspiration rates (du Toit et al. 2019; Veste et al. 2020; Podhrázská et al. 2021). The resultant shading effect from trees on the adjacent crop (Bohn Reckziegel et al. 2021, 2022) increases crop yield in water limited systems (Moreno 2008; Campi et al. 2009). But shading may equally present negative effects on growth in other regions due to increased competition for light and other resources dependent on factors such as tree density and individual site conditions (Ivezić et al. 2021). The presence of trees forming windbreaks within farmed landscapes not only provides direct influence on local site climate. It also influences other site attributes and ecosystem services such as soil nutrient content, water redistribution patterns, and impacts the above and belowground carbon sequestration potential of the site, offering a longer-term storage potential than conventional agricultural cropping without trees (Geertsema et al. 2016; Sheppard et al. 2020; Weninger et al. 2021). The utilisation of tree rows as windbreaks is commonplace in the Western Cape Province, South Africa (Veste et al. 2020). A trade-off between productive space and protective function is often a deciding factor in their placement and dimensions, additional benefits derived from such tree rows may influence their utilisation. The establishment and tending of trees within a farmed landscape can be considered a form of agroforestry.
Many tree species are used within windbreaks, shelterbelts and hedgerows. Attributes of trees commonly used for this application including fast growth, dense branching structure with long crowns, low competition (i.e. can be established at high density and do not compete with the intercrop) as well as wind, sun and frost tolerance. One commonly used genus is Populus (Poplars), these are a group of 25 to 30 species belonging to the family Salicaceae. Populus simonii ((Carrière) Wesm.) is part of the Populus section Tacamahaca (balsam poplars) and is commonly known as Simon’s/Simon poplar or as Chinese cottonwood since the species is native to north-eastern China and Mongolia. P. simonii has been introduced into North America, Australasia and southern Africa, in particular, South Africa. Alongside its frequent utilisation in shelterbelts, it is commonly utilised for low quality timber applications such as pallet wood, or for energy wood and for phytoremediation purposes. The genus offers fast growth and easy establishment, and therefore, a quick and efficient formation of a windbreak structure with minimal initial costs and tending. Due to a limited utilisation of a durable timber product, only limited long-term carbon storage potential can be realised from Poplar species planted in windbreaks, nevertheless, the practice allows for an additional degree of carbon uptake and storage over multiple years in contrast to treeless agricultural cropping regimes.
Land under agricultural cultivation has a high potential for the storage of carbon, the integration of trees into treeless landscapes provides an opportunity for the increase and preservation of soil organic carbon stocks (Maier et al. 2023) as well as carbon stored in aboveground biomass (AGB) (Zomer et al. 2016) and belowground biomass (Magalhães 2015). The concept of carbon farming specifically incorporates agroforestry as functional systems that integrate trees with conventional arable and livestock systems resulting in decreased Greenhouse gas emissions and increased carbon storage (Sharma et al. 2021). Zomer et al. (2016) estimated biomass carbon on agricultural land suggesting a mean national value for South Africa of > 10 Mg C ha−1 with little change between 2000 and 2010.
Modelling in order to construct allometric equations is frequently utilised as a mean of exploring parameters in the natural world based on the idea of biological scaling. In this sense, the researcher is able to acquire a hard to obtain metric (i.e. in this case weight of biomass) from easily attainable measurements such as diameter at breast height (DBH; the stem diameter measured at a standard height of 1.3 m) or total tree height. Allometry is based on the sampling of a population and the construction of an allometric equation through regression. The use of allometric equations is commonplace for the biomass and volumetric modelling in trees (Picard et al. 2012; Henry et al. 2013; Morhart et al. 2013a, 2013b, 2016). To date there are few studies focusing on the biomass production of P. simonii. Previous investigations stem exclusively from Chinese literature. For example, Gao et al (2014) investigated the biomass and carbon storage potential of the species in plantations producing two factor equations for leaf, branch, truck root and whole tree fractions. Other works are evident by title but lack an English translation of the main text. It is our understanding that this is the first instance where biomass modelling of P. simonii has been reported specifically for trees sampled from outside the forest. Our aim is to present allometric equations applicable for P. simonii trees grown in windbreak situations as a form of agroforestry under a Mediterranean climate. Allometric equations are to be based on DBH as an independent predictor variable and to use this information to make suggestions for potential carbon storage in the given study system. Beside equations for whole tree biomass estimation, we also provide information on tree components such as stem, total branch, individual branch and bark proportion as well as bark thickness.
Materials and methods
Site description
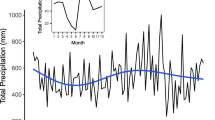
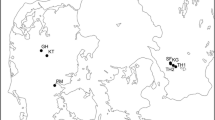
The sample trees were derived from a windbreak located in the vineyards of the Babylonstoren Estate in the Western Cape near Stellenbosch, South Africa (coordinates: 33° 49′ 34.6764ʺ S, 18° 55′ 16.734ʺ E, 198 m a.s.l.; Fig. 1), close to the town of Paarl (Fig. 1). The site experiences a hot summer Mediterranean climate (Köppen-Geiger classification: CSa, c.f. (Peel et al. 2007)) with wet winters and dry summers with an average annual precipitation sum of 770 mm and a mean annual temperature of 17.6 °C (Bargmann 2003; Meadows 2015; Climate-Data.org 2022). Annual temperature and precipitation distribution can be seen in Fig. 2. Topography has been suggested to highly influence local climate, particularly increasing the precipitation sum (Bargmann 2003). For this reason, actual precipitation on the study site may be higher than stated for Paarl.
Location inforation: a Study site location near Paarl, Western Cape, South Africa. Adapted from (d-maps 2023a, 2023b). b High-resolution orthophoto of the study area on the Babylonstoren wine estate, Stellenbosch, South Africa reconstructed from drone images (ground resolution 2.34 cm/pixel), including location detail for the sampled Populus simonii windbreak (blue dashed line) between citrus fields, c the sample windbreak prior to sampling
Climate diagram using data from the 1991 to 2021 climate normal period for the weather station of the town Paarl approximately 10.5 km north east of the study site. Data originated from Climate-Data.org (2022). Bars represent the monthly precipitation sums (mm), the line is the monthly mean temperature, upper ribbon is the monthly maximum temperature, lower ribbon is the monthly minimum temperature (°C)
Located in the Franschhoek wine valley at the foot of the Simonsberg, soils are derived from clays weathered from the sedimentary Malmesbury group formations and sandy loams from the Cape Granite Suite (Wellington 1955; Bargmann 2003). A detailed geological description of the region is given by Bargmann (2003) in relation to the extensive wine cultivation in the region. The site is flat and located within an active wine estate, the windbreak is flanked by vines and citrus plantations (see Fig. 1b and c). For this reason, irrigation, pest management and fertilisation effects on growth cannot be discounted.
This study focuses on P. simonii trees, with a planting date of 2000, in a single row with an average initial spacing of 1 m, forming a windbreak. Management of the windbreak is limited and likely restricted to maintenance of access to the adjacent trackway. Due to the low planting density with limited competition outside the row, we consider the trees to show a more open growth form, particularly in the axis perpendicular to the windbreak row.
Fieldwork
Fieldwork was carried out during January 2021. Prior to felling, tree ID numbers and DBH measurements were recorded, DBH was marked on the stem for each sample tree. Trees were felled at a height of 10 cm. The following measurements were made post felling (see also Fig. 3): total length of the tree, as a proxy for standing tree height; the branch diameter at the cut face (hereafter branch stub diameter), measured in two perpendicular directions as well as its length from stub to tip. Five sample branches per tree were identified from five equal sections in the live crown. After debranching, any leaves on the sample branches were separated from the branches and wet mass for branches without leaves, was recorded. Disks were marked to be removed every metre along the stem, including at the base (0.3 m), at 1.3 m and at the minimum stem diameter of 7 cm. An overview of individual tree sampling strategy is given in Fig. 3. Wet mass for all the disks were recorded in the field using a scale with an accuracy of 1.0 g. The fresh weight of the billets (i.e. the remaining stem wood not sampled as discs) between the disks and tree top were determined in the field using a hanging scale with an accuracy of 0.25 kg.
Laboratory work
Branches were dried at 105 °C according to the ISO 18134–2 standard (ISO 2017) until of constant weight, measured using a scale with an accuracy of 0.1 g. The average over- and under-bark diameter of all disks were measured to calculate the average wet bark thickness. The wet disks were de-barked and wet mass were recorded for disks and bark separately. The water volume displacement was recorded for all disks, utilising the Archimedean principle, for wood density calculation. Disks and bark were then dried separately to 105 °C until of constant weight. An individual tree conversion factor derived from the sampled stem discs was calculated as a ratio of wet weight against dry weight following forced drying. The conversion factor was applied to the fresh field weights for the whole tree, thus, providing whole tree dry weight for each of the sample trees.
Data analyses
All data exploration and analysis were carried out using the R programming environment 4.2.1 (R Core Team 2023). Linear and exponential regressions were fitted to the data using the function lm() (Wilkinson and Rogers 1973; Chambers 1992). The alpha level of significance was assumed to be at 0.05 for all analyses. We followed common practice to apply non-linear allometric equations as this is an efficient method of relating an easily measurable parameter to that of a less obtainable dependent factor, such as dry tree biomass (Parresol 1999; Zianis et al. 2005; Picard et al. 2012; Morhart et al. 2013a, 2016; Cifuentes Jara et al. 2015; Roxburgh et al. 2015). DBH is frequently used as predictor (Zianis et al. 2005; Picard et al. 2012) and is known to be strongly indicative of dry aboveground biomass.
Due to the nature of non-linear regression methods violating the assumption of homoscedasticity when using original measurement scales, log-transformation is necessary (Seifert and Seifert 2014). Where Y is the dependent variable (biomass compartment) and X the independent predictor variable (DBH), the data was log transformed, resulting in the following equation (Eq. 1). Here, a is the intercept, b the slope and ε describes the additive error.
When modelling branch relationships, the data was also log-transformed and a linear mixed effect model procedure was used, where the individual tree was included in the model as a random effect using the R package lme4 (Bates et al. 2015). Potential linear mixed effect models are denoted as “b” models.
This renders a linear relationship and denoting the correct model fit of an allometric power equation (Picard et al. 2012). Nevertheless, log-transformed data introduced a systematic negative bias to the data set (Baskerville 1972; Sprugel 1983; Zeng and Tang 2011). Such bias is generally accepted to be rectified utilising Eq. 2 where σ is the residual standard error of the regression as given in Eq. 2 forming a correction factor (CF) as given in Eq. 3 (Sprugel 1983; Zeng and Tang 2011).
In order to apply the regression for the calculation of AGB in arithmetic form, Eq. 1 must be back-transformed according to Eq. 4 using the regression coefficients derived from the function and the CF as described in Eq. 3.
To provide confidence intervals to our corrected power functions (Eq. 4), we applied bootstrapping of the model coefficients by randomly resampling the data. After bootstrapping the model was based on 1000 resamples, we determined the confidence percentiles (0.025 and 0.975) of all bootstrapped coefficients and applied the correction factor CF to the percentiles.
Outliers were removed if they were identified as being outside 1.5 times the interquartile range above the upper quartile or lower quartile. Goodness of fit was evaluated utilising the adjusted coefficient of determination (R2adj), a metric that considers the number of variables in a data set, or the conditional R2 if a mixed model was used and through the evaluation of relative squared error (RSE). Homoscedasticity of the residuals was checked using the Breusch-Pagan test (Breusch and Pagan 1979; Koenker 1981; Krämer and Sonnberger 1986) in the lmtest package. Normal distribution was assessed utilising the Shapiro–Wilk test (Shapiro and Wilk 1965). A Durbin-Watson test (Durbin and Watson 1950) was applied to assess whether there was autocorrelation between residuals.
The issue of additivity (Seifert and Seifert 2014), in regards to separated tree portions was investigated by applying beta regression (Cribari-Neto and Zeileis 2010; Douma and Weedon 2019). Beta regressions can be used to model the proportions of the constituent biomass compartments of whole trees. The predicted proportions can then be multiplied with the predictions of a model for total tree biomass to derive the biomass of the modelled compartments. Nevertheless, neither the intercept nor slope as output from the beta regression were significant for this dataset. Therefore, we conclude that the model is not stating anything about the relation between dependent and independent variables and can be disregarded.
In order to present a comparison of our biomass model for AGB with other models found in the literature, the GlobAllomeTree database (Henry et al. 2013) was explored alongside internet-based search engines. With a lack of specific P. simonii focused studies, the search was initially expanded to include all Populus species from the Tacamahaca (balsam poplars) section. This also yielded limited results so the search was again broadened to include all Populus studies with biomass models that had a similar DBH range and where possible, similar tree age. Within this modified search criteria, no comparable studies from Mediterranean climate regions were found, therefore, studies with a broader geographical range were included in order to frame the presented data in our study, namely:
-
P. maximowicii × P. trichocarpa hybrids located in southern Germany with a DBH range of 1.8 cm–8.9 cm, n = 30, harvested at 3 years old (Morhart et al. 2013a);
-
A generalised model derived from five Populus hybrids (namely: P. trichocarpa × P. deltoides, P. deltoides × P. nigra, P. canadensis × P. maximowiczii, P. nigra × P. maximowiczii, P. maximowiczii × P. balsamifera), with a DBH range of 4.3 cm–37.3 cm, n = 120, harvested after 13 years in Québec, Canada (Truax et al. 2014);
-
P. canadensis × P. maximowiczii hybrids in Québec, Canada with DBH ranges from 7.5 cm–24.0 cm, n = 36, harvested after 14 years (Fortier et al. 2017);
-
P. balsamifera and P. tremuloides with a DBH range of 5 cm–35 cm in Canada with the equation obtained via the GlobAllomeTree database (Singh and Misra 1979);
-
P. tremula in Sweden including a DBH range of 20 cm–108 cm, n = 111, harvested at ages between 5 and 24 years (Johansson 1999).
The quantification of the carbon storage capacity and the energetic potential of trees in windbreaks requires the knowledge of the carbon content of single trees. To calculate the individual tree’s carbon content, we assumed a dry carbon concentration of 45% (stem and branch fractions) as derived by Gao et al. (2014) from P. simonii plantations in semiarid temperate region of northwest China, the authors give mean carbon content values for different compartments namely leaf, branch, stem and root. Carbon dioxide equivalent (CO2e) was estimated by multiplying the calculated carbon content value by 3.67 (Guest et al. 2013). Root to shoot ratios (RS) may be applied to partially describe biomass partitioning belowground in the absence of sampling. Various root to shoot ratios for hybrid poplar are given and vary according to poplar species, age and tree density alongside site conditions. Mean values range from approximately 0.2 to 0.3 in the literature (Puri et al. 1994; Fang et al. 2007; McIvor et al. 2009; Benomar et al. 2012; Fortier et al. 2015; Oliveira et al. 2018) and in some cases less (Fortier et al. 2015), however, these are hybrid poplars grown on short rotations often at very high density. A generalised ratio supported by the IPCC (1996) suggests a RS of 0.25 for temperate broadleaf species which would encompass P. simonii in much of its distribution range. Nevertheless, in dry climates the RS will increase (Kou et al. 2022), in a meta-analysis of pine and eucalypt allometry, du Toit et al. (2016) found that mean annual precipitation sum was a strong predictor of RS, especially in drier climates. The authors showed that in climates with 700 mm mean annual precipitation sum, such as experienced in Paarl, the RS of pine and eucalypt species was increased. Due to the dry nature of the site respectively, a value of 0.3 was therefore utilised within this study to reflect increased biomass allocation to the root system. The value was combined with the derived biomass function to gain an estimate of belowground biomass and carbon storage therein.
Results
Exploration of tree parameters
The tree biometric parameters are listed in Table 1 for each sampled tree. Tree DBH varied between 6.9 cm and 31 cm (mean 20.3 cm) and measured tree length post felling between 6.20 m and 20.1 m (mean 12.6 m). Approximately 90% of all sampled trees (n = 17) were above 10 cm DBH and 80% of above 10 m in height. Mean windbreak width was 4.9 m as a proxy for mean crown diameter perpendicular to the axis of the windbreak row.
A summary of the applied tree-wise conversion factors plus the wet and dry weights of the sampled woody component can be found in Table 1. The mean of the tree fresh biomass (± the standard error) was 311.42 kg (± 59.59 kg), ranging from 18.78 kg to 803.05 kg with 32% of the biomass representing the branch biomass with a mean biomass weight of 99.65 kg (± 19.23 kg). Figure 4 outlines the data distribution frequency of DBH, tree length, branch stub diameter and individual branch length, each data set displays an approximate bell curve distribution.
Tree and branch allometry
The measured DBH and height of the sampled trees display a strong relation with the fitting of a power regression model (see Fig. 5 and Table 2), all model assumptions were met. Field measurements of tree DBH and height are highly correlated (R2adj = 0.896) with individual branch length similarly demonstrating a high correlation with branch stub diameter (R2adj = 0.802). Likewise, all model assumptions were met.
Biomass estimation
The assessment of dry woody AGB was carried out by regressing tree biomass with measured tree biometric parameters, specifically DBH, defined as a log-transformed predictor (Picard et al. 2015). Height as an independent predictor variable did not significantly improve the models, and was therefore, excluded from the modelling process. The whole tree biomass, the dry tree stem biomass and the branch dry biomass are explained by utilising a power function as is commonplace in biomass studies. DBH is shown to be an excellent predictor of the three dependent variables explaining 95.4%, 95.9% and 88.3% of the variance for each of the latter dependent variables respectively.
Function coefficients and statistical measures of model fit are given in Table 2. P-values derived from the SW and BP tests were greater than 0.05, thus, also indicating normality and homogeneity of the residuals. Plots showing DBH against individual dependent variables are shown in Fig. 6 showing the whole tree biomass alongside stem and branch stem biomass portions. Comparisons were made between the current study and similar studies from the literature. Figure 7 shows the current model for AGB for P. simonii plotted against six other models. Selection of comparison models included models that concerned Populus spp. (Tacamahaca section and hybrids) but also included other Populus sections that had individuals sampled from outside a closed forest environment. The displayed models vary in their range of applicability across all diameters, i.e. the range of tree sizes sampled. This is reflected in the length of each curve shown in Fig. 7 without extrapolation.
The branch biomass is shown to be highly correlated with the branch diameter (R2adj = 0.871). The branch stub diameter explains more than two third of branch biomass variance as shown in Fig. 8. The majority of branches sampled have a branch stub diameter of 40 mm or less (see also Table 2) with larger diameters (> 40 mm) represented by fewer samples.
Bark and wood partitioning
Bark biomass was predicted as a function of sectional diameter using data collected from sample discs collected from the stem. Figure 9 shows the resultant model (model 7b, see also Table 2) which was fitted using a power model. The R2adj suggests that nearly 77% of the variation can be explained by the model. With increasing diameter of the stem sections, bark proportion reduces. Double bark thickness (model 8b, see also Table 2) measured in cm is also displayed in Fig. 9 plotted against sectional diameter; it can be observed that with increasing diameter of the stem the double bark thickness also increases. The R2adj suggests that 76% of the variation can be explained by the presented model.
Carbon stock
The calculated CO2e storage of a tree with increasing diameter is shown in Fig. 10. The mean study tree with a DBH of 20.3 cm will store, estimated by the model using a power function as shown in Eq. 4 (see Fig. 10), on average of 199 kg CO2e. We can further calculate that based on the sampled windbreak 198.5 Mg CO2e km−1 is stored on average in the AGB, or by upscaling for comparison with other studies based on mean windbreak width a total storage of 110.7 Mg C ha−1 or 406.2 Mg CO2e ha−1 can be reported.
Utilising the derived biomass function for whole tree biomass combined with the generalised RS of 0.3 an estimate of belowground biomass and associated carbon storage can be made. For the calculated mean tree with a DBH of 20.3 cm, 59.7 kg CO2e is potentially stored in the belowground portion. On the assumption of 1 m tree spacing, 6.0 Mg CO2e km−1 belowground carbon storage can be estimated to be stored within the example P. simonii windbreak.
Discussion
Few biomass studies that explore the AGB production potential of P. simonii are available in the literature. It was our primary objective to contribute a specific function describing AGB for this species focused on trees outside the forest, in particular with a focus on agroforestry systems. We presented models for tree height and individual branch length alongside biomass models describing total dry AGB, stem biomass, total branch biomass and individual branch biomass. We also provide models for an assessment of bark proportion and bark thickness based on sectional diameter.
Tree parameters and biomass production
The prediction of total tree height could be made with a high degree of accuracy as a function of DBH with nearly 90% of the variation explained by the model. The presented model (model 1a; Table 2) is limited in range to the sampled trees, therefore, accurate in the DBH range of 6.90 cm to 31.0 cm. Site-specific factors such as local climate and soil properties must also be accounted for when applying the model. These factors correspondingly include micro and macro effects of location, competition, and on agricultural sites, indirect fertilisation and irrigation. These results were derived specifically from a single row windbreak located in a wine estate in South Africa, within a Mediterranean climate, for this reason the derived allometric equations should equally only be applied to similar situations. DBH, as is commonly shown (Paul et al. 2016), was a suitable predictor variable demonstrating high R2adj values as a metric of model fit. Height as an independent predictor variable did not significantly improve the models and was therefore excluded from the modelling process, also negating the need for a two-factor function. DBH represents a measurement which is measured quickly and accurately in the field. Height measurements, however, can have a larger variation due to measurement inaccuracies, deteriorating with increasing tree height. While every effort was taken to produce accurate analytical results following a defined and tested protocol, human and accuracy errors in the determination of fresh weight of the whole tree by weighing multiple parts cannot be discounted. The low number of sample trees (n = 17) was limited by the labour-intensive nature and cost effectiveness of the exercise of the sampling technique as is commonplace in biomass studies (Roxburgh et al. 2015), but nonetheless, may provide a source of uncertainty considering between tree variability in growth form (Vorster et al. 2020). Likewise, a relatively narrow range of tree dimensions means that the resultant function is limited in its predictive capacity to trees of similar dimensions and under comparable growing conditions. The accuracy of dried weights is relative to the accuracy of the field scale.
The sample trees were taken from a single row windbreak with a defined between tree spacing of 1 m. Given the location of these trees, we assume that the trees were not as heavily influenced by competition pressure as in forests with higher canopy closure. Therefore, we also assume that the growing habit of the sample trees is more akin to open-grown trees. While the management of individual trees was not evident (i.e. pruning, lopping, clearance from the trackside etc.) their growth may have been affected by the adjacent farming activities and presence of a farm track at the base of the windbreak on the north-eastern side (i.e. possible soil compaction or root disturbance).
Model 2b (Table 2) describing individual branch length predicted by branch stub diameter (explicitly the diameter at the first order divide between the main stem and branch) was also highly significant, a sample size of 84 branches provided a robust basis for the regression. Individual sample sizes ranged from a lower boundary of 7 mm up to 78 mm. The largest branch stub diameters (> 60 mm) were only represented by two branches, this might have influenced the slope at its upper limit. The prediction of individual branch length can be considered to be less accurate than the DBH/height model as branch extension is also influenced by genetics, site conditions, competition, management, damage and shading (Ceulemans et al. 1990; King 1997; Souch and Stephens 1998). Furthermore, we assume that “within” row branch extension would be more heavily influenced by competition than “between” row branch extension. We demonstrated that branch stub diameter is a reliable predictor of individual branch biomass (model 6b), in the same way that DBH is an important predictor for whole tree biomass and other aboveground tree portions. Since branch diameter (n = 84) explains significant variability of branch biomass, a diameter-based generalised model is clearly a representation of branch biomass response to site condition and/or competition.
Comparison between existing studies and our data (Fig. 7) showed differences can be seen, seated between the Aspen studies (Populus section Populus) (Singh and Misra 1979; Johansson 1999) and the presented Tacamahaca studies (Singh and Misra 1979; Morhart et al. 2013a; Truax et al. 2014; Fortier et al. 2017), these displayed a greater biomass production, although this cannot be attributed to species alone. The Tacamahaca studies show similarity in their curves alongside the AGB model presented in this paper. At larger diameters, the studies show increasing divergence. Nevertheless, we can generalise and suggest that the Tacamahaca studies show 10%–20% less biomass production than the P. simonii sample at the given DBH range and the Aspen studies show 20%–30% more. Climate and site conditions cannot be accounted for in such generalisations and are most likely to play highly influential roles on tree growth in each situation.
Bark proportion
Bark thickness varies with tree species, age, rate of growth influenced by climate, competition and position on the tree with thicker bark at the base of the tree and thinnest within the crown (Konôpka et al. 2022). Based on the samples taken which were exclusively from the stem, we modelled bark percentage as a function of sectional diameter. This allows practitioners to gauge the proportion of bark biomass in relation to the amount of woody biomass at tree level based on any measured diameter in the tree. Once again, the suggested function is limited in its predictive capacity to trees of the same species, the measured sectional diameter range and comparable climatic and site conditions. As a rule, sectional diameter is a function of age and, concurring with Konôpka et al. (2022) bark proportion and bark thickness can be described by increasing sectional diameter. While we only sampled trees of one age, inferences can still be made to trees of the same species under similar climatic conditions but of different age by comparing sectional diameter. Generally speaking, larger proportions of nutrients are contained within the bark than in woody portions, with smaller diameters. In previous work, Morhart et al. (2013a) measured dry bark biomass as a function of diameter class when studying short-rotation poplar hybrids. Both models correlate closely, validating our approach. While in closed forest production systems it is more likely that crown material is left on site to decay, returning nutrients to the site (Seifert and Seifert 2014) also increasing soil organic carbon. In agricultural systems where land utilisation is more structured, felled trees or arisings from pruning treatments may be cleared away more completely, hence, representing an export of nutrient and biomass. The appreciation of such biomass functions can be used as a basis for a quantification of such nutrient export.
Biomass determination for utilisation and ecosystem services
The utilisation of woody perennial species as windbreaks in agricultural environments is commonplace. The need to quantify biomass production justifies the sampling and the production of allometric equations as a tool that can be utilised by land owners to estimate the biomass of the growing stock. The Poplar genus as a whole is well represented within allometric equation studies, this can be largely attributed to its utilisation and success within short-rotation coppice systems producing biomass feed stocks for energy generation purposes. Here, the quantification of biomass is essential for comparisons between sites and clonal varieties as an estimation of oven dry yield. Within agricultural settings it is less likely that windbreaks once removed will be utilised for industrial energy conversion, rather for a more informal and local firewood use, especially as the resultant timber is of very low quality and of small diameter. Of greater interest in such situations is the carbon storage potential of longer-lived windbreak structures. As classical energy production systems utilising poplar will be harvested on a 3 to 10-year rotation, windbreaks have the potential to offer a mid-term carbon sink on lands that would otherwise be treeless. The estimation of biomass can be used as a proxy for carbon accounting in such systems with an assumption that carbon constitutes 45% of dry woody biomass (Gao et al. 2014) with further estimation of CO2e using a multiplication factor based on atomic weight (Guest et al. 2013). By applying our models, we have been able to suggest that approximately 198.5 Mg CO2e km−1 could be sequestered in the aboveground portion of our example windbreak, with the potential of a further 6.0 Mg CO2e km−1 of windbreak length for the belowground biomass. Uncertainties in the applied RS ratio connected to variation in species, site conditions, inter and intra species competition and tree age may provide a deviation in this estimate (e.g. a RS of 0.25 would yield approximately 5.0 Mg CO2e km−1 and an RS of 0.2, a yield of 4.0 Mg CO2e km−1). Nevertheless, this constitutes a substantial figure that can be accounted for within a total on-farm carbon storage budget over a tree-less farm and aligns with the concept of ‘carbon-farming’ (Sharma et al. 2021) and carbon storage within agroforestry systems (Zomer et al. 2016). Other contemporary studies have also made estimations of carbon storage within woody biomass within farmed landscapes. Native hedgerows in temperate north-western Europe have also been intensively studied concerning biomass production and carbon storage. Although dimensionally smaller (e.g. 1–4 m wide and up to 3.5 m in height), carbon storage in the range of 35–47 Mg C ha−1 or approximately 20–30 Mg CO2e km−1 was also possible despite a reduced height and width (Axe et al. 2017; Drexler et al. 2021; Biffi et al. 2023; Black et al. 2023) in comparison with the poplar windbreak presented in this study. This emphasises the potential of incorporating woody perennials within a farmed landscape, highlighting the increased carbon storage. Caution should be applied when considering values on a hectare basis for linear structures, especially when comparing with closed canopy forest stands, where carbon storage would be overestimated as it does not account for mortality and self-thinning effects due to high tree density.
Alternative methods have been made to assess the aboveground carbon content of trees; one promising method for biomass assessment as an alternative to destructive sampling is the utilisation of LiDAR (Light Detection and Ranging) in the form of terrestrial laser scanning. Here high-resolution point cloud data can be utilised to construct highly accurate quantitative structural models where tree volume or volumes of defined tree parts can be ascertained for the calculation of carbon content (Schindler et al. 2023a, 2023b). Nevertheless, non-destructive methods still currently rely on the extrapolation of data derived from destructive sampling and subsequent analysis, reinforcing the ongoing necessity for species-wise and location-based traditional biomass studies.
The presence of windbreak trees can increase soil organic carbon stocks and the nutrient status of the surrounding soils through litterfall (Gao et al. 2014). The belowground biomass of trees, i.e. the root portion, has been suggested to range between 20 and 26% by species specific studies (e.g. Boscia Senegalensis (Awé et al. 2021) or Eucalyptus (Kuyah et al. 2013)) or more generalised sources (Santantonio et al. 1977; IPCC 1996; Cairns et al. 1997). Estimations of belowground biomass are hard to obtain due to extensive methods required to excavate root biomass (Magalhães 2015), especially on a scale required to construct meaningful biomass functions. For this reason, RS are frequently applied as a mean to estimate belowground carbon stocks. Within this study we did not make an active assessment of belowground woody biomass and relied on a generalised RS based on dryland ecosystems. Nevertheless, variability due to vegetation type, climate variables and individual tree parameters can influence the biomass partitioning (Pregitzer et al. 1990). For this reason, RS should be more specific towards species, woodland type and region (Magalhães and Seifert 2015). It is common to only find AGB functions, but with the increasing importance of carbon accountancy and the utilisation of trees on agricultural land for multipurpose benefits, efforts should be directed at also empirically qualifying belowground stocks. Upon the removal of the windbreak structure, if the belowground biomass portion remain in-situ, a net increase in residual belowground carbon can be assumed. More accurate estimate of belowground biomass remain in the realms of destructive sampling and at present can only be achieved by root excavation, however methods for example utilising ground penetrating radar may hold promise for the future (Sun et al. 2023).
Conclusions
We present a set of allometric equations for P. simonii based on samples derived from a windbreak located on a wine estate near Stellenbosch, South Africa. Models for total tree height, AGB, stem and total branch biomass are given utilising DBH as a quick and accurate predictor variable. Alongside, functions for individual branch length and individual branch biomass are given as a function of branch stub diameter. Lastly, functions to estimate the bark proportion and bark thickness have also been presented utilising sectional diameter as independent variable.
Data was collected from one site, for one species at one age under specific climatic conditions, therefore, the transferability of results is limited to similar cultivation situations of P. simonii. The utilisation of poplar as a windbreak species in the Western Cape Province is commonplace due to its easy establishment and fast growth. It is likely that our results can be directly transferred to other windbreak structures in the region. The quantification of whole tree biomass and the biomass of separate tree components allows for complete accountability for in-situ (e.g. ecosystem services) and ex-situ (e.g. utilisation of tree biomass for energetic use) benefits, for estimating volume production, or for example, as a basis for a quantification of nutrient export an issue of particular importance within agroforestry systems.
We discussed how such functions can be further applied for the estimation of belowground biomass and whole tree carbon storage, both relevant for the promotion and utilisation of trees in agricultural systems. Windbreaks such as those evaluated within this study are generally longer lived than the crops cultivated between, but are inherently shorter lived than trees located within forest stands, which is largely due to management constraints and perceived negative impacts of large trees on farms. Nevertheless, the opportunity to store carbon within both the above and belowground portions of woody biomass provides an additional and alternative opportunity for mid-term carbon sequestration on land which is frequently utilised in the absence of trees. These benefits can be considered additional to the provision of wind and water erosion reduction benefits alongside the intended, and primary, sheltering effects of windbreaks influencing windspeeds, evapotranspiration as well as air and surface temperatures on the leeward side which are also of high relevance (Sheppard et al. 2020; Veste et al. 2020).
References
Awé VD, Noiha NV, Chimi DC, Ganamé M, Vroh BTA, Zapfack L (2021) Allometric models for predicting biomass and carbon pools of Boscia Senegalensis (Pers.) Lam. Ex Poir. (Capparaceae) populations in Central Africa: a case study from Cameroon. RJEES 1:123–146. https://doi.org/10.31586/rjees.2021.156
Axe MS, Grange ID, Conway JS (2017) Carbon storage in hedge biomass—A case study of actively managed hedges in England. Agr Ecosyst Environ 250:81–88. https://doi.org/10.1016/j.agee.2017.08.008
Bargmann C (2003) Geology and wine 7. Geology and wine production in the Coastal Region, Western Cape Province South Africa. Geocan 30:161–182
Baskerville GL (1972) Use of Logarithmic regression in the estimation of plant biomass. Can J For Res 2:49–53. https://doi.org/10.1139/x72-009
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Soft 67:1–48. https://doi.org/10.18637/jss.v067.i01
Benomar L, DesRochers A, Larocque G (2012) The effects of spacing on growth, morphology and biomass production and allocation in two hybrid poplar clones growing in the boreal region of Canada. Trees Struct Funct 26:939–949. https://doi.org/10.1007/s00468-011-0671-6
Biffi S, Chapman PJ, Grayson RP, Ziv G (2023) Planting hedgerows: biomass carbon sequestration and contribution towards net-zero targets. Sci Total Environ 892:164482. https://doi.org/10.1016/j.scitotenv.2023.164482
Black K, Lanigan G, Ward M, Kavanagh I, hUallacháin DÓ, Sullivan LO (2023) Biomass carbon stocks and stock changes in managed hedgerows. Sci Total Environ 871:162073. https://doi.org/10.1016/j.scitotenv.2023.162073
Bohn Reckziegel R, Larysch E, Sheppard JP, Kahle H-P, Morhart C (2021) Modelling and comparing shading effects of 3D tree structures with virtual leaves. Remote Sens 13:532. https://doi.org/10.3390/rs13030532
Bohn Reckziegel R, Sheppard JP, Kahle H-P, Larysch E, Spiecker H, Seifert T, Morhart C (2022) Virtual pruning of 3D trees as a tool for managing shading effects in agroforestry systems. Agrofor Syst 96:89–104. https://doi.org/10.1007/s10457-021-00697-5
Breusch TS, Pagan AR (1979) A simple test for heteroscedasticity and random coefficient variation. Econometrica 47:1287. https://doi.org/10.2307/1911963
Cairns MA, Brown S, Helmer EH, Baumgardner GA (1997) Root biomass allocation in the world’s upland forests. Oecologia 111:1–11. https://doi.org/10.1007/s004420050201
Campi P, Palumbo AD, Mastrorilli M (2009) Effects of tree windbreak on microclimate and wheat productivity in a Mediterranean environment. Eur J Agron 30:220–227. https://doi.org/10.1016/j.eja.2008.10.004
Ceulemans R, Stettler RF, Hinckley TM, Isebrands JG, Heilman PE (1990) Crown architecture of Populus clones as determined by branch orientation and branch characteristics. Tree Physiol 7:157–167. https://doi.org/10.1093/treephys/7.1-2-3-4.157
Chambers JM (1992) Linear models. In: Hastie TJ (ed) Statistical models in S. Routledge, New York, pp 95–144
Cifuentes Jara M, Henry M, Réjou-Méchain M, Wayson C, Zapata-Cuartas M, Piotto D, Alice Guier F, Castañeda Lombis H, Castellanos López E, Cuenca Lara R, Cueva Rojas K, del Águila Pasquel J, Duque Montoya Á, Fernández Vega J, Jiménez Galo A, López O, Marklund L, Michel Fuentes J, Milla F, Návar Chaidez J, Ortiz Malavassi E, Pérez J, Ramírez Zea C, Rangel García L, Rubilar Pons R, Saint-André L, Sanquetta C, Scott C, Westfall J (2015) Guidelines for documenting and reporting tree allometric equations. Ann For Sci 72:763–768. https://doi.org/10.1007/s13595-014-0415-z
Climate-Data.org (2022) Paarl climate (South Africa): data and graphs for weather & climate in Paarl. https://en.climate-data.org/africa/south-africa/western-cape/paarl-9597/. Accessed 17 Jan 2023
Cribari-Neto F, Zeileis A (2010) Beta regression in R. J. Stat. Soft. 34:1–24. https://doi.org/10.18637/jss.v034.i02
d-maps (2023a) Outline map of Africa. https://d-maps.com/carte.php?num_car=729&lang=en. Accessed 16 June 2023
d-maps (2023b) Outline map of the Western Cape provence, South Africa. https://d-maps.com/carte.php?num_car=89840&lang=en. Accessed 16 June 2023
Douma JC, Weedon JT (2019) Analysing continuous proportions in ecology and evolution: a practical introduction to beta and Dirichlet regression. Methods Ecol Evol 10:1412–1430. https://doi.org/10.1111/2041-210X.13234
Drexler S, Gensior A, Don A (2021) Carbon sequestration in hedgerow biomass and soil in the temperate climate zone. Reg Environ Chang 21:74. https://doi.org/10.1007/s10113-021-01798-8
Durbin J, Watson GS (1950) Testing for serial correlation in least squares regression. I Biom 37:409–428. https://doi.org/10.1093/biomet/37.3-4.409
Fang S, Xue J, Tang L (2007) Biomass production and carbon sequestration potential in poplar plantations with different management patterns: carbon sequestration in China’s forest ecosystems. J Environ Manag 85:672–679
Fortier J, Truax B, Gagnon D, Lambert F (2015) Plastic allometry in coarse root biomass of mature hybrid poplar plantations. Bioenerg Res 8:1691–1704. https://doi.org/10.1007/s12155-015-9621-2
Fortier J, Truax B, Gagnon D, Lambert F (2017) Allometric equations for estimating compartment biomass and stem volume in mature hybrid poplars: general or site-specific? Forests 8:309. https://doi.org/10.3390/f8090309
Gao Y, Cheng J, Ma Z, Zhao Y, Su J (2014) Carbon storage in biomass, litter, and soil of different plantations in a semiarid temperate region of northwest China. Ann For Sci 71:427–435. https://doi.org/10.1007/s13595-013-0355-z
Geertsema W, Rossing WAH, Landis DA, Bianchi FJ, van Rijn PCJ, Schaminée JHJ, Tscharntke T, van der Werf W (2016) Actionable knowledge for ecological intensification of agriculture. Front Ecol Environ 14:209–216. https://doi.org/10.1002/fee.1258
Guest G, Bright RM, Cherubini F, Strømman AH (2013) Consistent quantification of climate impacts due to biogenic carbon storage across a range of bio-product systems. Environ Impact Assess Rev 43:21–30. https://doi.org/10.1016/j.eiar.2013.05.002
Henry M, Bombelli A, Trotta C, Alessandrini A, Birigazzi L, Sola G, Vieilledent G, Santenoise P, Longuetaud F, Valentini R, Picard N, Saint-André L (2013) GlobAllomeTree: International platform for tree allometric equations to support volume, biomass and carbon assessment. iForest Biogeosci For 6:326–330. https://doi.org/10.3832/ifor0901-006
IPCC (1996) Revised 1996 IPCC guidelines for national greenhouse gas inventories: reference manual (Volume 3)
ISO (2017) Solid biofuels determination of moisture content oven dry method. International Organisation for Standardization, Geneva, Switzerland
Ivezić V, Yu Y, van der Werf W (2021) Crop yields in European agroforestry systems: a meta-analysis. Front Sustain Food Syst 5:606631. https://doi.org/10.3389/fsufs.2021.606631
Johansson T (1999) Biomass equations for determining fractions of European aspen growing on abandoned farmland and some practical implications. Biomass Bioenerg 17:471–480. https://doi.org/10.1016/S0961-9534(99)00073-2
King DA (1997) Branch growth and biomass allocation in Abies amabilis saplings in contrasting light environments. Tree Physiol 17:251–258. https://doi.org/10.1093/treephys/17.4.251
Koenker R (1981) A note on studentizing a test for heteroscedasticity. J Econom 17:107–112. https://doi.org/10.1016/0304-4076(81)90062-2
Konôpka B, Pajtík J, Šebeň V, Merganičová K (2022) Modeling bark thickness and bark biomass on stems of four broadleaved tree species. Plants (Basel) 11:1148. https://doi.org/10.3390/plants11091148
Kou X, Han W, Kang J (2022) Responses of root system architecture to water stress at multiple levels: a meta-analysis of trials under controlled conditions. Front Plant Sci 13:1085409. https://doi.org/10.3389/fpls.2022.1085409
Krämer W, Sonnberger H (1986) The linear regression model under test. Springer, Heidelberg
Kuyah S, Dietz J, Muthuri C, van Noordwijk M, Neufeldt H (2013) Allometry and partitioning of above- and below-ground biomass in farmed eucalyptus species dominant in Western Kenyan agricultural landscapes. Biomass Bioenerg 55:276–284. https://doi.org/10.1016/j.biombioe.2013.02.011
Magalhães TM (2015) Allometric equations for estimating belowground biomass of Androstachys johnsonii Prain. Carbon Balanc Manag 10:16. https://doi.org/10.1186/s13021-015-0027-4
Magalhães TM, Seifert T (2015) Tree component biomass expansion factors and root-to-shoot ratio of Lebombo ironwood: measurement uncertainty. Carbon Balanc Manag 10:9. https://doi.org/10.1186/s13021-015-0019-4
Maier R, Schack-Kirchner H, Nyoka BI, Lang F (2023) Gliricidia intercropping supports soil organic matter stabilization at Makoka research station. Malawi Geoderma Reg 35:e00730. https://doi.org/10.1016/j.geodrs.2023.e00730
McIvor IR, Douglas GB, Benavides R (2009) Coarse root growth of Veronese poplar trees varies with position on an erodible slope in New Zealand. Agrofor Syst 76:251–264. https://doi.org/10.1007/s10457-009-9209-y
Meadows ME (2015) The cape winelands. In: Grab SW, Knight J (eds) Landscapes and landforms of South Africa. Springer, Cham Switzerland, pp 103–109
Moreno G (2008) Response of understorey forage to multiple tree effects in Iberian dehesas. Agr Ecosyst Environ 123:239–244. https://doi.org/10.1016/j.agee.2007.04.006
Morhart C, Sheppard JP, Spiecker H (2013a) Above ground leafless woody biomass and nutrient content within different compartments of a P. maximowicii × P. trichocarpa poplar clone. Forests 4:471–487. https://doi.org/10.3390/f4020471
Morhart C, Sheppard JP, Seidl F, Spiecker H (2013b) Influence of different tillage systems and weed treatments in the establishment year on the final biomass production of short rotation coppice poplar. Forests 4:849–867. https://doi.org/10.3390/f4040849
Morhart C, Sheppard JP, Schuler JK, Spiecker H (2016) Above-ground woody biomass allocation and within tree carbon and nutrient distribution of wild cherry (Prunus avium L.)—a case study. Forest Ecosyst 3:1–15. https://doi.org/10.1186/s40663-016-0063-x
Oliveira N, Rodríguez-Soalleiro R, Pérez-Cruzado C, Cañellas I, Sixto H, Ceulemans R (2018) Above- and below-ground carbon accumulation and biomass allocation in poplar short rotation plantations under Mediterranean conditions. Adaptation of forests and forest management to changing climate selected papers from the conference on adaptation of forests and forest management to changing climate with emphasis on forest health: a review of science, policies and practices Umeå, Sweden. For Ecol Manag 428:57–65. https://doi.org/10.1016/j.foreco.2018.06.031
Parresol BR (1999) Assessing tree and stand biomass: a review with examples and critical comparisons. For Sci 45:573–593
Paul KI, Roxburgh SH, Chave J, England JR, Zerihun A, Specht A, Lewis T, Bennett LT, Baker TG, Adams MA, Huxtable D, Montagu KD, Falster DS, Feller M, Sochacki S, Ritson P, Bastin G, Bartle J, Wildy D, Hobbs T, Larmour J, Waterworth R, Stewart HTL, Jonson J, Forrester DI, Applegate G, Mendham D, Bradford M, O’Grady A, Green D, Sudmeyer R, Rance SJ, Turner J, Barton C, Wenk EH, Grove T, Attiwill PM, Pinkard E, Butler D, Brooksbank K, Spencer B, Snowdon P, O’Brien N, Battaglia M, Cameron DM, Hamilton S, McAuthur G, Sinclair J (2016) Testing the generality of above-ground biomass allometry across plant functional types at the continent scale. Glob Change Biol 22:2106–2124. https://doi.org/10.1111/gcb.13201
Peel MC, Finlayson BL, McMahon TA et al (2007) Updated world map of the Köppe–-Geiger climate classification. Hydrol Earth Syst Sci 4:439–473
Picard N, Saint-Andre L, Henry M (2012) Manual for building tree volume and biomass allometric equations: from field measurement to prediction. Food and Agricultural Organization of the United Nations and Centre de Coopération Internationale en Recherche Agronomique pour le Développement, Rome, Montpellier
Picard N, Rutishauser E, Ploton P, Ngomanda A, Henry M (2015) Should tree biomass allometry be restricted to power models? For Ecol Manag 353:156–163. https://doi.org/10.1016/j.foreco.2015.05.035
Podhrázská J, Kučera J, Doubrava D, Doležal P (2021) Functions of windbreaks in the landscape ecological network and methods of their evaluation. Forests 12:67. https://doi.org/10.3390/f12010067
Pregitzer KS, Dickmann DI, Hendrick R, Nguyen PV (1990) Whole-tree carbon and nitrogen partitioning in young hybrid poplars. Tree Physiol 7:79–93. https://doi.org/10.1093/treephys/7.1-2-3-4.79
Puri S, Singh V, Bhushan B, Singh S (1994) Biomass production and distribution of roots in three stands of Populus deltoides. For Ecol Manage 65:135–147. https://doi.org/10.1016/0378-1127(94)90165-1
R Core Team (2023) R: a language and environment for statistical computing. https://www.R-project.org/
Roxburgh SH, Paul KI, Clifford D, England JR, Raison RJ (2015) Guidelines for constructing allometric models for the prediction of woody biomass: how many individuals to harvest? Ecosphere 6:38. https://doi.org/10.1890/ES14-00251.1
Santantonio D, Hermann RK, Overton WS (1977) Root biomass studies in forest ecosystems. Pedobiologia 17:1–31
Schindler Z, Seifert T, Sheppard JP, Morhart C (2023) Allometric models for above-ground biomass, carbon and nutrient content of wild cherry (Prunus avium L.) trees in agroforestry systems. Ann For Sci 80:28. https://doi.org/10.1186/s13595-023-01196-6
Schindler Z, Morhart C, Sheppard JP, Frey J, Seifert T (2023) In a nutshell: Exploring single tree parameters and above-ground carbon sequestration potential of common walnut (Juglans regia L.) in agroforestry systems. Agrofor Syst. https://doi.org/10.1007/s10457-023-00844-0
Seifert T, Seifert S (2014) Modelling and simulation of tree biomass. In: Seifert T (ed) Bioenergy from wood: sustainable production in the tropics. Springer, Netherlands, Dordrecht, pp 43–65
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591. https://doi.org/10.2307/2333709
Sharma M, Kaushal R, Kaushik P, Ramakrishna S (2021) Carbon farming: prospects and challenges. Sustainability 13:11122. https://doi.org/10.3390/su131911122
Sheppard JP, Bohn Reckziegel R, Borrass L, Chirwa PW, Cuaranhua CJ, Hassler SK, Hoffmeister S, Kestel F, Maier R, Mälicke M, Morhart C, Ndlovu NP, Veste M, Funk R, Lang F, Seifert T, du Toit B, Kahle H-P (2020) Agroforestry: an appropriate and sustainable response to a changing climate in Southern Africa? Sustainability 12:6796. https://doi.org/10.3390/su12176796
Singh KP, Misra R (1979) Structure and functioning of natural, modified and silvicultural ecosystems in Eastern Uttar Pradesh. Final Technical Report (1975–1978), MAB research project, Banaras Hindu University, Varanasi, India, 160 p
Souch CA, Stephens W (1998) Growth, productivity and water use in three hybrid poplar clones. Tree Physiol 18:829–835. https://doi.org/10.1093/treephys/18.12.829
Sprugel DG (1983) Correcting for bias in log-transformed allometric equations. Ecology 64:209–210. https://doi.org/10.2307/1937343
Sun D, Jiang F, Wu H, Liu S, Luo P, Zhao Z (2023) Root location and root diameter estimation of trees based on deep learning and ground-penetrating radar. Agronomy 13:344. https://doi.org/10.3390/agronomy13020344
du Toit B, Dovey S, Seifert T, Muyambo P, Malherbe GF (2016) Carbon sequestration in South African plantation forests: Progress report number 1: Techniques to estimate net carbon sequestration carbon at the Tier 2 level using available data from local studies and international literature. https://doi.org/10.13140/RG.2.1.4714.8409
du Toit B, Little KM, Da Costa D, Viero PW, Sheppard JP, Kunneke A, Lambrechts HA, Mbongo W, Malherbe GF (2019) Silvicultural production under climatic conditions of high-water deficit. In: Póvoa de Mattos P et al (eds) XXV IUFRO world congress: forest research and cooperation for sustainable. Pesquisa Florestal Brasileira, Colombo, Brazil, pp 634
Truax B, Gagnon D, Fortier J, Lambert F (2014) Biomass and volume yield in mature hybrid poplar plantations on temperate abandoned farmland. Forests 5:3107–3130. https://doi.org/10.3390/f5123107
Veste M, Littmann T, Kunneke A, du Toit B, Seifert T (2020) Windbreaks as part of climate-smart landscapes reduce evapotranspiration in vineyards Western Cape Province South Africa. Plant Soil Environ 66:119–127. https://doi.org/10.17221/616/2019-PSE
Vorster AG, Evangelista PH, Stovall AEL, Ex S (2020) Variability and uncertainty in forest biomass estimates from the tree to landscape scale: the role of allometric equations. Carbon Balanc Manag 15:8. https://doi.org/10.1186/s13021-020-00143-6
Wellington JH (1955) Southern Africa. A geographical study. Vol. 1. Physical geography. Vol. 2. Economic and human geography. Southern Africa. A geographical study. Vol. 1. Physical geography. Vol 2. Economic and human geography. Cambridge University Press, Cambridge
Weninger T, Scheper S, Lackóová L, Kitzler B, Gartner K, King NW, Cornelis W, Strauss P, Michel K (2021) Ecosystem services of tree windbreaks in rural landscapes—a systematic review. Environ Res Lett 16:103002. https://doi.org/10.1088/1748-9326/ac1d0d
Wilkinson GN, Rogers CE (1973) Symbolic description of factorial models for analysis of variance. Appl Stat 22:392. https://doi.org/10.2307/2346786
Zeng WS, Tang SZ (2011) Bias correction in logarithmic regression and comparison with weighted regression for nonlinear models. Nat Prec. https://doi.org/10.1038/npre.2011.6708.1
Zianis D, Muukkonen P, Mäkipää R, Mencuccini M (2005) Biomass and stem volume equations for tree species in Europe. Silva Fenn Monogr 4(4):1–63
Zomer RJ, Neufeldt H, Xu J, Ahrends A, Bossio D, Trabucco A, van Noordwijk M, Wang M (2016) Global tree cover and biomass carbon on agricultural land: the contribution of agroforestry to global and national carbon budgets. Sci Rep 6:29987. https://doi.org/10.1038/srep29987
Acknowledgements
This research was funded by the German Federal Ministry of Education and Research (BMBF), as part of the project “Agroforestry in Southern Africa—New Pathways of Innovate Land Use Systems Under a Changing Climate” (grant number 01LL1803A). We acknowledge support from the Open Access Publication Fund of the University of Freiburg. We would like to extend our thanks to the Babylonstoren farm manager Ian de Villiers who facilitated access to the site and permitted the removal and processing of the study windbreak. Thank you to Stefan Seifert for providing the high-resolution orthophoto of the study area as shown in Fig. 2, and to the two anonymous reviewers whose comments helped improve the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. Bundesministerium für Bildung und Forschung (01LL1803A).
Author information
Authors and Affiliations
Contributions
JPS conceived and designed the experiment; BdT, CJC, AK and GM collected field data and undertook sample preparation; JPS, EL, ZS and CJC analysed the data; JPS, EL and CJC wrote the initial manuscript draft; in addition, JPS, EL, ZS, BdT, AK, GM, CM, RBR, TS and H-PK provided conceptual and editorial advice and reviewed the manuscript. Project administration by JPS, EL and H-PK; funding acquisition by JPS, CM and H-PK. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sheppard, J.P., Larysch, E., Cuaranhua, C.J. et al. Assessment of biomass and carbon storage of a Populus simonii windbreak located in the Western Cape Province, South Africa. Agroforest Syst 98, 697–714 (2024). https://doi.org/10.1007/s10457-023-00940-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-023-00940-1