Abstract
Pollen allergies are responsible for a considerable global public health burden, and understanding exposure is critical to addressing the health impacts. Atmospheric pollen counts are routinely used as a predictor of risk; however, immune responses are triggered by specific proteins known as allergens, which occur both within and on the surface of the pollen grain. The ratio between atmospheric pollen counts and allergen concentrations (‘pollen potency’) has been shown to be inconsistent, with potentially important implications for pollen monitoring practice. Despite this, there has been no previous synthesis of the literature and our understanding of the factors that influence pollen potency remains poor. We conducted a scoping review with the aim of deriving a current understanding of: (a) the factors that influence pollen potency; (b) its variation through time, between taxa and by location; and (c) the implications for pollen monitoring practice. Our synthesis found that pollen potency is highly variable within and between seasons, and between locations; however, much of this variability remains unexplained and has not been deeply investigated. We found no predictable pollen potency patterns relating to taxon, geography or time, and inconclusive evidence regarding possible driving factors. With respect to human health, the studies in our synthesis generally reported larger associations between atmospheric allergen loads and allergy symptoms than whole pollen counts. This suggests that pollen potency influences public health risk; however, the evidence base remains limited. Further research is needed to better understand both pollen potency variability and its implications for health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Allergic sensitisation to pollen poses a considerable individual and public health burden because it is a major cause of two common conditions: allergic rhinitis (AR, commonly referred to as hay fever) and allergic asthma. These conditions are common and globally pervasive. Asthma alone affects approximately 339 million individuals worldwide (Global Asthma Network, 2018), while it is estimated that AR affects 10–30% of the global adult population (Pawankar et al., 2013). The burden of both diseases is notable: allergic asthma can have serious health consequences including mortality (Thien et al., 2018), while AR symptoms result in reduced productivity, increased absenteeism from work and decreased quality of life (Katelaris et al., 2012). Overall, these impacts create a substantial economic burden. For example, the cost of absenteeism and presenteeism due to allergic diseases in the European Union was estimated to range from EUR€55 to €155 billion per annum (Zuberbier et al., 2014), while the indirect costs of AR and urticaria (allergic hives) in Asian countries have been estimated at USD $105.4 billion per annum (Kulthanan et al., 2018). AR and allergic asthma can be triggered by exposure to many different allergens, including those associated with plant pollen. A changing climate, increasing atmospheric carbon dioxide levels, the spread of exotic aeroallergen-producing species into new areas (Alan et al., 2020) and increasing geographic ranges of allergenic pollen taxa are all anticipated to increase pollen allergen exposure (Damialis et al., 2019), which will likely worsen the burden of these diseases in the future (Beggs, 2016). Thus, effective management and treatment of pollen allergy are a global health priority.
Symptoms of pollen allergies are typically seasonal because they are associated with the temporal patterns of pollen release into the atmosphere. In this context, atmospheric pollen monitoring networks have been established in many countries to provide individuals, clinicians and public health agencies with a means of tracking potential allergy risk (Alcázar et al., 2015; Haberle et al., 2014; Johnston et al., 2018). Pollen monitoring is typically undertaken with a volumetric spore trap, such as a Hirst-type trap (Hirst, 1952), that continuously samples air for periods of up to one week. Airborne bioaerosols and particles, including pollen, impact onto a microscope slide or tape that is covered with an adhesive. The deposited pollen grains are morphologically identified, and the atmospheric concentrations of different pollen taxa are then estimated (Buters et al., 2018). Pollen data can then be used by clinicians and patients to make informed decisions to minimise personal exposure and mitigate their risk. Many studies have supported the utility of providing publicly available atmospheric concentrations of whole pollen grains for this purpose, and they remain the ‘gold standard’ around the world (Buters et al., 2018).
However, one limitation of atmospheric pollen monitoring is that whole pollen grains are not the direct cause of allergy symptoms. Instead, symptoms arise when high molecular weight proteins, known as allergens, trigger an individual’s immune response by the binding of specific class E immunoglobulins (IgE). Allergens are located on the surface and within the cytoplasm of individual pollen grains (e.g. Behrendt et al., 1999; Süring et al., 2016) and are present on fine (< 2 µm) pollen fragments released into the atmosphere (e.g. Buters et al., 2010; Schäppi et al., 1997b; Taylor et al., 2002). Atmospheric allergen detection requires different sampling and laboratory infrastructure to regular pollen monitoring. This includes sampling air with a different device (for example, a High Volume Impactor) and immunochemical detection of the allergen with ELISAs instead of microscopic identification (Buters et al., 2010). Therefore, despite the close theoretical linkage between allergen concentrations and symptom severity, allergen concentrations are not routinely measured by pollen monitoring networks.
Importantly, there is evidence that atmospheric allergen concentrations are not directly related to trends in atmospheric pollen counts (Buters et al., 2012, 2015). There are two potential explanations for this: (a) there is variation in the amount of allergen carried by individual pollen grains and/or (b) there is variable release of allergen-carrying fragments into the atmosphere (Schäppi et al., 1997a, 1997b, 1999). Schäppi et al., (1997a, 1999) were among the first to hypothesise that atmospheric allergen concentrations may be independent of atmospheric pollen counts, based on experiments demonstrating allergen-carrying particles being liberated from grass and birch pollen grains. More recently, the Health Impacts of Airborne Allergen Information (HIALINE) Network project (Buters et al., 2010) set out to investigate the relationship between atmospheric pollen counts and atmospheric allergen across a wide range of European environments. This group defined the ratio of pollen grains to allergen detected via immunoassay as ‘pollen potency’, and we follow this terminology here. The HIALINE project produced some of the first data showing significant variation in potency between taxa, within and between seasons, and between locations (Buters et al., 2010, 2012, 2015). Their findings suggest that the phenomenon of variable pollen potency might be more common than previously estimated.
Given the important role of allergens in precipitating allergy symptoms, variation in pollen potency may have important health implications. We hypothesised that varying pollen potency might be a factor underlying the significant global variation in the threshold of pollen grains that are required to trigger symptoms in sensitised individuals (de Weger et al., 2013). Further, if atmospheric allergen concentrations are decoupled from pollen counts, then atmospheric allergen concentrations could be a better predictor of risk of symptoms and may be more appropriate to monitor than concentrations of whole pollen grains (Cecchi, 2013). Although allergen monitoring implementing molecular and immunological approaches currently remains cost-prohibitive as standard practice, developing a better understanding of the relationship between whole pollen counts and atmospheric allergen loads may enable us to provide allergy patients with enhanced information about their personal risk.
To date, there has been no synthesis of the literature to investigate the characteristics of pollen potency and the implications for pollen monitoring practice and human health. We therefore undertook this scoping review with the aims of understanding: (a) taxonomic, temporal and geographic variation in pollen potency; (b) the factors that influence pollen potency; and (c) the implications for human health and pollen monitoring practice.
2 Methods
We sought peer-reviewed studies that evaluated the quantitative relationship between pollen and allergen (that is, pollen potency), published up to 5 February 2021. Our literature search strategy sought to capture studies that evaluated pollen potency in any way, including studies that considered the atmospheric ratio of allergen to whole pollen, as well as studies that quantified the relationship between pollen and allergen with pollen collected directly from plants. Given the interdisciplinary nature of the topic, we opted for a scoping review methodology as outlined in Munn et al. (2018). Search terms were developed to provide consistent results across three databases: Scopus, Web of Science and Medline via Ovid. The search terms used for each database and the number of results for each are listed in Table 1.
The results of the literature searches were exported into EndNote (Clarivate Analytics, Philadelphia, USA) then into Covidence (Veritas Health Innovation, Melbourne, Australia). Covidence native software detected and removed duplicates. We screened remaining studies for relevance by title and abstract, with two authors (LT and NB) using a key (Supplementary Figure 1) that was designed to only progress publications that investigated either (a) the ratio of atmospheric allergen to atmospheric whole pollen counts or (b) the amount of allergen present on/in a pollen grain or collection of pollen grains. Publications that received a yes or maybe were moved to full-text screening. LT then carried out full-text screening on all publications that were voted yes or maybe. Articles were deemed relevant if they quantified a relationship between allergen and pollen grains in the study design. We excluded review articles, conference proceedings, non-English language, non-peered reviewed articles and other non-article publications or publications that were inaccessible through reasonable searching protocols. We manually screened the reference lists of the final list of publications to obtain additional relevant publications. Publications that were deemed relevant were progressed to extraction. The relevant information was recorded from each study using a pre-designed data collection table: (i) title, (ii) first author, (iii) study location, (iv) study period, (v) taxa studied, (vi) allergen studied, (vii) methods of pollen and allergen sampling and measurement, (viii) the metric used for measurement and (ix) the conclusions about pollen potency.
3 Results
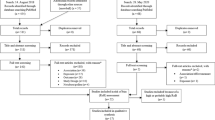
A total of 35 peer reviewed articles met the inclusion criteria (Fig. 1). The majority of the studies were completed in locations across Europe (n = 28). Of the remaining studies, four were completed in Saitama, Japan, and three in Melbourne, Australia. Furthermore, Supplementary Figure 2 shows the number of articles in our extraction published per year. This demonstrates an increase in research frequency in the last decade but no substantial increase in the last few years.
Full-text screening revealed that most studies (n = 32) measured the relationship between atmospheric concentrations of pollen and allergen. Two methods were employed to demonstrate this relationship: (a) average weight of atmospheric allergen in picograms (pg) per pollen grain for a continuous period (e.g. year or season) and/or (b) as a graphical depiction of disaggregated daily data of coupled atmospheric pollen and allergen concentrations through time. Sixteen publications used both methods, two used only the former and 14 used only the latter. Figure 2 summarises the results from all publications that used the first method (n = 16) to provide a quantitative comparison of average pollen potency between taxa, locations and seasons. Figure 2 also summarises the methods used to collect atmospheric allergen and pollen grains, respectively. While all of these studies collected whole pollen grains using Hirst-type samplers, allergens were collected via a mixture of High Volume Cascade Impactors and cyclone samplers.
Reported seasonal average pollen potency measurements by pollen taxon from all studies that provided a measurement in picograms of allergen per pollen grain (n = 16). Year denotes year of measurement. Country denotes the country where sampling occurred. Pollen denotes the method of pollen collection (A = Hirst-type continuous flow volumetric sampler). Allergen denotes the method of allergen collection (C = Cyclone Sampler (Burkard) or D = high-volume Impactor). The bars show the average pollen potency across that season measured in picograms of allergen per pollen grain rounded to two figures. Note that the scale for potency measurements differs by taxon. Records for Ash and Privet are listed with Olive because they share the Ole e 1 antibody used for allergen quantification; Alder records are listed with Birch for the same reason. Within each taxon, measurements from same sampling location are grouped to facilitate comparison of between-season variation. Summary statistics for each taxon are provided in the bottom right; these statistics are based on all reported seasonal average pollen potency measurements for that taxon
The three remaining articles measured the relationship between pollen and allergen using pollen collected from plants rather than atmospheric measurements; these studies included an average measurement of weight of allergen in pg per weight of pollen in micrograms (Beck et al., 2013; Behrendt et al., 1999; Buters et al., 2008). These papers primarily investigated environmental factors that affect the amount of allergen borne by pollen grains, with the denominator being weight of pollen, rather than individual pollen grains. Therefore, results from these studies are not directly comparable with the more common approach of monitoring atmospheric concentrations of pollen or pollen allergens. No studies investigated the specific amount of allergen associated with singular pollen grains, meaning we were not able to investigate pollen potency relating to variable allergen content of individual pollen grains. For a full summary of extracted information, see Supplementary Table 1.
Ten pollen taxa were investigated across the 35 studies, with numerous studies investigating more than one taxon. Thirteen papers studied Betula (birch) pollen, ten studied Poaceae (grass) pollen, nine studied Olea (olive) pollen, four studied Cryptomeria japonica (Japanese cedar) pollen, five studied Platanus (Plane tree) pollen, four studied Urticaceae (pellitory) pollen, two each studied Ambrosia (ragweed), Fraxinus (ash) and Alnus (alder) pollen and one study investigated Ligustrum (privet) pollen. In most instances, studies investigated pollen potency with respect to the allergen most commonly evaluated in clinical practice (for example Bet v 1 for birch). However, several studies investigated potency with respect to additional or alternative allergens for a respective pollen taxon (full details in Supplementary Table 1).
3.1 Temporal variation in pollen potency
Temporal variation in pollen potency was examined in 29 publications, with most exploring temporal variation across a single pollen season. Of these, 19 publications displayed disaggregated daily data graphically; all demonstrated that atmospheric pollen and allergen concentrations became decoupled across one single season. However, fluctuations in potency were irregular, and neither the individual studies themselves, nor our subsequent synthesis of these studies found any predictable patterns as to when in a season potency would be greatest. For example, measurements of olive pollen potency in Córdoba (Spain) from 2012 to 2014 indicated that potency was at its highest at the mid-point of the 2012 season; however, it was moderate or low at the same point of the season in 2013 and 2014 (Plaza et al., 2016). Overall, almost all studies that tracked within-season variation in pollen potency recorded large and often sharp fluctuations. For example, Buters et al. (2010) demonstrated a more than ten-fold difference in birch pollen potency within a single season, while Buters et al. (2015) reported up to 17-fold differences within seasons for grass pollen potency. Overall, we found no consistent patterns of within-season variation across taxa and seasons.
Ten studies compared variation across multiple seasons. One method for evaluating between-season variation is the comparison of average pollen potency values across multiple seasons. As shown in Fig. 2, studies that investigated potency across multiple seasons in a single location found differing magnitudes of variation in season-average values. For example, Buters et al. (2015) found that average grass pollen potency differed little across three seasons at one location in France, recording 2.4 pg/pollen grain in 2009 and 2010 and 2.9 pg/pollen grain in 2011. Conversely, the same study found that average potency varied over threefold over the same three seasons in one location in Italy, with average measurements of 1.5, 2.9 and 0.8 pg/pollen grain, respectively (Buters et al., 2015).
3.2 Taxonomic variation in pollen potency
Pollen potency measurements in pg/pollen grain were reported for nine of the ten taxa investigated across the included studies (only graphical disaggregated daily data were reported for Japanese cedar). Across the nine taxa with measurements in pg/pollen grain, median season-average pollen potency ranged from 0.4 to 2.6 pg of allergen per pollen grain (Fig. 2). The two most widely studied taxa, grass and birch, demonstrated similar medians (2.6 and 2.3 pg/pollen grain, respectively), similar minima (0.7 and 1.0 pg/pollen grain) and similar maxima (15 and 21 pg/pollen grain). Furthermore, grass, birch and olive pollen demonstrate similar magnitudes of variation in season-average potency measurements, with inter-quartile ranges of 2.0–3.6 pg/pollen grain, 1.2–3.1 pg/pollen grain and 0.6–3.8 pg/pollen grain, respectively. Season-average potency measurements for Platanus and Urticaceae have typically been substantially lower (Fig. 2).
Expanding from season averages to comparative trends in temporal pollen potency variation, four publications tracked temporal variation in more than one taxon across one or multiple seasons (Table 2). These studies found that different taxa exhibited substantially different within- and between-season pollen potency trends. For example, Rodríguez-Rajo et al. (2011) displayed seasonal pollen potency trends for grass, Platanus and Urticaeae pollen in three cities in Spain. Each taxon demonstrated distinct pollen potency dynamics within single seasons, which in turn were inconsistent across the three study locations (for details see Supplementary Table 1). Overall, our synthesis did not identify any consistent patterns in within- or between-season variation related to taxon.
3.3 Geospatial variation in pollen potency
Twelve studies compared pollen potency across locations (Table 2). We found that there were sometimes substantial differences in season average measurements across locations, even within the same country. For example, Rodríguez-Rajo et al. (2011) found that in Spain, grass pollen potency was on average almost twice as high (8.7 pg/pollen) in the city of Ourense in 2007 compared to the city of León (4.7 pg/pollen). In addition, some studies explored whether temporal dynamics in pollen potency across a season were similar or different across multiple locations. For example, Buters et al. (2015) graphically displayed the relationship between atmospheric grass pollen and the allergen Phl p 5 for the 2009–2011 seasons for ten separate locations across Europe. They found stark differences in temporal patterns of pollen potency across locations.
3.4 Factors influencing pollen potency
Twenty-five publications investigated potential drivers of pollen potency (Table 3). No single variable was consistently identified as strongly influential. Only one publication investigated the factors that affected pollen potency directly by testing the association between environmental variables and the ratio of pollen allergen to whole pollen counts (Jochner et al., 2015). This study reported a significant negative correlation between pollen potency and humidity (but no correlation between pollen potency and sunshine duration or precipitation). Since there are no other studies that investigate this directly, one can investigate the factors that affect the individual components of pollen and allergen. Thus, the remaining publications made inferences about driving factors by investigating the relationship between environmental variables and one or both of these components.
Twenty-four publications, primarily derived from European network studies, have described associations between meteorological factors and pollen counts, allergen concentrations and/or pollen potency. However, our synthesis found no consistent patterns. For example, three studies found a positive relationship between rainfall and atmospheric allergen concentration (Schäppi et al., 1997a, 1997b; Wang et al., 2013a), while four studies showed the inverse (Beck et al., 2013; De Linares et al., 2010; Schäppi et al., 1997b; Wang et al., 2013b). Two studies demonstrated a negative correlation between rainfall and atmospheric pollen concentration (Jochner et al., 2015; Schäppi et al., 1997b), while one study did not find any correlation (Rodríguez-Rajo et al., 2011). Our synthesis found likewise conflicting results regarding associations between allergen and/or whole pollen concentrations and other possible meteorological drivers, such as humidity (Buters et al., 2015; Fernández-González et al., 2019a) and temperature (Beck et al., 2013; Galan et al., 2013).
Other environmental variables that were associated with one or both components of pollen potency were altitude, proximity to the ocean and prevalence of long-distance dispersed pollen (Table 3). Few studies discussed the potential influence of plant physiology and genetics [for an exception see Buters et al. (2008)]. Aside from climatic conditions, factors such as soil composition and tree health have been identified as likely to influence the quantity of allergen released from pollen (Buters et al., 2008); however, these have not been directly studied. Ozone levels have also been proposed as a possible driving factor, with some support from the very limited data available: Beck et al. (2013) found a weak positive relationship between ozone levels and Bet v1 atmospheric concentrations (Beck et al., 2013; Buters et al., 2008).
3.5 Pollen potency and health
Five publications investigated the relevance of pollen potency for human health by testing the relationship between allergen and pollen with a health outcome related to asthma or allergies (Table 4). Four of these were undertaken in Europe; the other was undertaken in Melbourne, Australia. All but one of the studies found that allergen concentrations had a slightly higher correlation with human health outcomes than whole pollen concentrations; the fifth (Porcel Carreño et al., 2020) reported mixed results, with whole grass and olive pollen sometimes appearing more strongly associated with asthma emergency department attendances than allergen, depending on the lag and allergen metric used (for details see Table 4). However, across all studies, differences were generally not substantive and the correlation between the health proxy and whole pollen counts was still high: for example, Brito et al. (2011) reported a value of r = 0.7 for the relationship between whole olive pollen counts and daily symptom counts, but a value of r = 0.8 for the relationship with Ole e 1 allergen concentrations.
Two additional publications amongst those returned by our search investigated factors other than pollen potency that contributed to the magnitude of the human immune response. Beck et al. (2013) found that birch pollen extracts prepared from environments higher in ozone were more strongly correlated with components of a stronger allergic response (e.g. skin prick test wheal measurements, cytokine secretion and neutrophil migration). This study also described weak positive correlations between higher ozone and higher Bet v 1 concentrations (r = 0.37, p < 0.05); however, it did not directly assess the relationship between Bet v 1 and health outcomes. Behrendt et al. (1999) showed that grass, birch and pine pollen all secrete the eicosanoid prostaglandin E2 and leukotriene B4, which are initiators of innate and inflammatory responses. These two studies suggested that there are factors beyond allergen content that could additionally contribute to the human immune response to pollen.
4 Discussion
The results of this scoping review demonstrate inconsistent findings with respect to all facets of pollen potency across the peer reviewed literature. Consequently, there is insufficient evidence to support clear conclusions about the factors that influence pollen potency, nor explain the large degree of variation between taxa, across time and between locations. However, this review identified several key gaps in the existing literature, and on this basis we generate recommendations to inform the pollen potency research agenda. The implications of the review results are discussed in further detail below, stratified by our research aims: (a) variation in pollen potency, (b) factors influencing pollen potency and (c) pollen potency and health outcomes.
4.1 Variation in pollen potency
Our synthesis revealed a large degree of temporal, taxonomic and geographic variation in pollen potency measurements. With respect to temporal variation, a key implication of the degree and sporadic nature of the variation reported both within and between seasons is that it is difficult to either (a) predict pollen potency at any stage in the season or (b) extrapolate potency trends from 1 year to another. This greatly limits the capacity to forecast pollen potency for a season, let alone days during a season, based on previous observations. One caveat is that the maximum number of seasons over which pollen potency was tracked was three: it is possible that patterns may become evident with longer time series. With respect to variation between taxa, this review identified similar ranges in average pollen potency measurements reported for birch, grass and olive, but substantially lower average measurements for Platanus and Urticaceae. Further research is required to establish whether the lower potency of the latter taxa is a consistent trend or simply an artefact of the low number of studies carried out on these taxa to date. Further, disaggregated daily data [e.g. as reported by Fernández-González et al. (2010) and De Linares et al. (2010)], revealed that different pollen taxa displayed distinct within-season pollen potency dynamics. This further highlights the difficulty of transferring pollen potency research: trends observed in one taxon in a given location cannot be extrapolated to any other. Further, due to the limited taxonomic resolution of current pollen detection methods for grass (Campbell et al., 2020; Jones et al., 2020), overlapping seasons of cross-reactive species and the presence of more than one major allergen (Lol p 1 and Phl p 5) further confound the conclusions that can be drawn about grass pollen potency (Buters et al., 2015; Jochner et al., 2015; Ščevková et al., 2020). Finally, given the substantial and non-systematic geospatial variation in pollen potency, we also conclude that findings cannot be extrapolated geospatially, nor by climatic region. With the caveat that most studies were conducted in Europe, we also found insufficient evidence to conclude that particular locations exhibit more or less potent pollen. This implies that it is not possible at this time to support the hypothesis that pollen potency variation is the driver of different symptom thresholds in different locations (de Weger et al., 2013).
4.2 Factors influencing pollen potency
The results of this review highlight several major gaps in our understanding of the factors that influence pollen potency. Our synthesis suggests that meteorological factors including temperature and humidity may influence temporal and/or geographic variation in pollen potency, as numerous studies have reported associations between pollen potency and meteorological variables (see Table 3). However, no study found a decisive influence of any one variable, the direction and magnitude of effects were inconsistent across studies, and the mechanisms remain unclear. For example, it is unclear whether the effects of meteorological variables relate primarily to their impacts on allergen production and/or free allergen release due to pollen fragmentation. In addition to meteorological variables, a small number of studies also reported associations between pollen potency and geographic factors such as altitude and proximity to coast (Table 3); however, again the predictability and mechanisms behind these associations remain unclear. Our review suggests that pollen potency is most likely determined by a complex web of interacting factors, which we are currently unable to reconcile using available data. A systematic and standardised methodological approach to investigating pollen potency drivers may be required to provide greater clarity (discussed further below).
4.3 Pollen potency and health
Our synthesis suggests that atmospheric allergen counts might be more correlated with allergic symptoms than pollen counts. All but one of the publications that investigated this relationship showed that allergen concentrations had a greater magnitude in the size of the association between allergy symptoms than pollen concentrations, although the differences were not always substantial. This finding suggests that although allergen concentrations may generally better predict health outcomes than whole pollen counts, the substantially higher cost of obtaining information about allergen concentrations might not be justified by a modest improvement in correlations with clinical outcomes.
Importantly, the results also highlight that allergen concentrations do not fully explain trends in health outcome responses as a degree of residual variance remains (Table 4). This suggests that factors other than pollen allergen load impact the prevalence and magnitude of allergic response (Beck et al., 2013; Behrendt et al., 1999). While outside the scope of this review, these factors might include plant fibres and trichomes (Sercombe et al., 2011), other environmental allergen sources that patients may be co-sensitised to [e.g. fungal as shown in Tham et al. (2017)], and/or other compounds associated with pollen, often referred to as non-allergenic pollen-derived compounds (Gilles et al., 2012). With respect to the latter, the most commonly investigated are pollen-associated lipid mediators (PALMs), which are molecules that can interact with the immune system, impacting the immune system response (Beck et al., 2013; Behrendt et al., 1999; Gilles et al., 2012). Resident bacteria on pollen grains are also believed to enhance the immune response, or in some situations, act as an immune sensitiser and activate the immune system (Obersteiner et al., 2016; Oteros et al., 2019). Further potential contributing factors include the immune adjuvant effect of pollution on pollen grains [reviewed in Senechal et al. (2015)], as well as Adenosine and NADPH-oxidase levels in pollen, both of which have been associated with a stronger immune response (Beck et al., 2013; Gilles et al., 2012; Wimmer et al., 2015). We suggest that to achieve a more holistic understanding of pollen allergy risk, the influence of PALMs and other non-allergenic compounds on health outcomes should be further explored in future research.
4.4 Implications for pollen monitoring practice and future research
The results of this review have important implications for pollen monitoring and allergy risk communication. Our results highlight that the understanding of pollen potency remains too rudimentary for its application in public health practice. The degree of unsystematic and unexplained variability in pollen potency diminishes predictive capacity. Further, we are not yet able to measure atmospheric allergen content (and thus pollen potency) routinely and in real time, in part due to the high cost of molecular and immunological approaches to airborne allergen measurement. This means that on a day-to-day basis, we are not able to use pollen potency information to supplement the allergy risk information provided by whole pollen counts.
In relation to future research, these results highlight the need to better understand and characterise pollen potency, as well as the broader range of factors that contribute to an immune response to pollen. While Supplementary Figure 2 suggests an increase in research effort in the last decade, there has not been a considerable increase in the last few years. Consequently, we recommend an increase in research effort and that future studies work towards standardised methodologies that systematically examine pollen potency drivers. Specifically, we suggest the standardisation of approaches to allergen collection and measurement: across the studies included in this review, a diversity of collection and measurement approaches were utilised (details in Supplementary Table 1). Although analysis of Fig. 2 does not suggest a systematic difference in results generated by the two main methods used for atmospheric allergen collection (cyclone and High Volume Impactor samplers), methodological differences could nevertheless potentially contribute to some of the inconsistency in conclusions found here. We were unable to explicitly assess this due to the difficulty of controlling for other differences between studies and suggest the literature would benefit from a direct experimental comparison of results generated by different methods to generate a single recommended sampling approach. In addition to greater methodological homogeneity, longer time series of single taxa across one or multiple locations would also be highly valuable in order to provide data sets with greater power to discern potentially complex patterns and trends. Further, we recommend that future research incorporates size-fractioned sampling to facilitate a better understanding of the relative role of pollen fragmentation versus variation in allergen per original pollen grain in driving pollen potency variation.
Further, in addition to meteorological variables, we highlight the need to consider a broader range of potential pollen potency drivers, such as herbivory (e.g. Lommen et al., 2017), land disturbances (e.g. Kernerman et al., 1992) and pollution (e.g. Beck et al., 2013; Kim et al., 2018; Singer et al., 2005; Ziska, 2020; Ziska et al., 2019). Such factors have been shown to increase allergen content and have high potential to act as strong drivers of pollen potency because some allergens are plant defence proteins (Matricardi et al., 2016), which are commonly upregulated during plant stress (Tashpulatov et al., 2004). Agriculture or other activities that disturb vegetation could also play a role, for example via the re-aerosolisation of deposited pollen (and pollen fragments) (e.g. Kernerman et al., 1992; Taylor et al., 2002). Factors related to plant physiology and genetics also remain under-explored; further investigation of the size distribution of atmospheric allergen could also help resolve the degree to which pollen fragmentation contributes to pollen potency variation (e.g. Buters et al., 2012; De Linares et al., 2010; Wang et al., 2012). Finally, future research should also investigate the role of pollen-derived factors such as PALMs, Adenosine, NADPH-oxidase, bacterial LPS and pollutants in modulating the immune response (Beck et al., 2013; Gilles et al., 2009, 2012; Oeder et al., 2015).
5 Strengths and limitations of the review
To our knowledge, this scoping review represents the first to synthesise published research summarising findings on pollen potency and the implications for human health. This has enabled us to highlight key gaps in the existing literature, supporting an evidence-based agenda for the ongoing characterisation of the relationship between atmospheric pollen and allergen concentrations, and their implications for individual and public health outcomes. It is, however, important to acknowledge certain limitations in our approach. First, we only included studies published in English. Second, as a scoping review, we did not systematically assess studies for data quality or risk of bias. These limitations are unlikely to substantially affect our core finding that there is substantive and frequently un- or poorly explained variability in pollen potency results.
6 Conclusion
The published literature reveals considerable taxonomic, temporal and geographic variation in pollen potency. The inconsistent and largely unexplained variation in pollen potency identified in this review makes it challenging to draw definitive conclusions about the factors that may influence pollen potency and to what extent. Similarly, insufficient evidence exists to be able to predict allergen levels in the air by extrapolating potency data onto pollen counts. Results from this review confirm that further research is needed to better understand what is a highly complex system of factors that determine pollen potency. To aid the comparison of work in this field, we recommend that the field adopts more consistent methodologies to enable standardised characterisation of pollen potency. A better understanding of the factors driving pollen potency will in turn improve our understanding of allergic disease and offer insights to direct effective management options for sufferers in future. We also recommend the future research agenda investigates the characteristics of free allergen caused by pollen fragmentation, methods of detection and non-allergen pollen-derived molecules in order to enhance our understanding of the full range of factors that determine the intensity of human allergic response.
References
Alan, Ş, Sarışahin, T., Acar Şahin, A., Kaplan, A., & Pınar, N. M. (2020). An assessment of ragweed pollen and allergen loads in an uninvaded area in the Western Black Sea region of Turkey. Aerobiologia, 36(2), 183–195. https://doi.org/10.1007/s10453-019-09620-z
Albertini, R., Ugolotti, M., Buters, J., Weber, B., Thibaudon, M., Smith, M., Galán, C., Berger, U., Brandao, R. M., Antunes, C. M., Grewling, L., Rantio-Lehtimäki, A., Cecchi, L., Sauliene, I., & Sofiev, M. (2013). The European project HIALINE (health impacts of airborne allergen information network): Results of pollen and allergen of Betula monitoring in Parma (2009). Review of Allergy and Clinical Immunology, 23(1), 14–20.
Alcázar, P., Galán, C., Torres, C., & Domínguez-Vilches, E. (2015). Detection of airborne allergen (Pla a 1) in relation to Platanus pollen in Córdoba, South Spain. Annals of Agricultural and Environmental Medicine, 22(1), 96–101. https://doi.org/10.5604/12321966.1141376
Álvarez-López, S., Fernández-González, M., González-Fernández, E., Garrido, A., & Rodríguez-Rajo, F. J. (2020). Tree allergen pollen-related content as pollution source in the city of Ourense (NW Spain). Forests, 11(11), 1129. https://doi.org/10.3390/f11111129
Beck, I., Jochner, S., Gilles, S., McIntyre, M., Buters, J. T. M., Schmidt-Weber, C., Behrendt, H., Ring, J., Menzel, A., & Traidl-Hoffmann, C. (2013). High environmental ozone levels lead to enhanced allergenicity of birch pollen. PLoS ONE, 8(11), e80147. https://doi.org/10.1371/journal.pone.0080147
Beggs, P. J. (Ed.). (2016). Impacts of climate change on allergens and allergic diseases. Cambridge University Press.
Behrendt, H., Tomczok, J., Sliwa-Tomczok, W., Kasche, A., von Eschenbach, C. E., Becker, W. M., & Ring, J. (1999). Timothy grass (Phleum pratense L.) pollen as allergen carriers and initiators of an allergic response. International Archives of Allergy and Immunology, 118(2–4), 414–418. https://doi.org/10.1159/000024151
Brito, F. F., Gimeno, P. M., Carnes, J., Martin, R., Fernandez-Caldas, E., Lara, P., López-Fidalgo, J., & Guerra, F. (2011). Olea europaea pollen counts and aeroallergen levels predict clinical symptoms in patients allergic to olive pollen. Annals of Allergy, Asthma & Immunology: Official Publication of the American College of Allergy, Asthma, & Immunology, 106(2), 146–152. https://doi.org/10.1016/j.anai.2010.11.003
Buters, J. T. M., Antunes, C., Galveias, A., Bergmann, K. C., Thibaudon, M., Galan, C., Schmidt-Weber, C., & Oteros, J. (2018). Pollen and spore monitoring in the world. Clinical and Translational Allergy, 8, 9. https://doi.org/10.1186/s13601-018-0197-8
Buters, J. T. M., Kasche, A., Weichenmeier, I., Schober, W., Klaus, S., Traidl-Hoffmann, C., Menzel, A., Huss-Marp, J., Krämer, U., & Behrendt, H. (2008). Year-to-year variation in release of Bet v 1 allergen from birch pollen: Evidence for geographical differences between West and South Germany. International Archives of Allergy and Immunology, 145(2), 122–130. https://doi.org/10.1159/000108137
Buters, J. T. M., Prank, M., Sofiev, M., Pusch, G., Albertini, R., Annesi-Maesano, I., Antunes, C., Behrendt, H., Berger, U., Brandao, R., & Celenk, S. (2015). Variation of the group 5 grass pollen allergen content of airborne pollen in relation to geographic location and time in season. The Journal of Allergy and Clinical Immunology, 136(1), 87–95.e86. https://doi.org/10.1016/j.jaci.2015.01.049
Buters, J. T. M., Thibaudon, M., Smith, M., Kennedy, R., Rantio-Lehtimäki, A., Albertini, R., Reese, G., Weber, B., Galan, C., Brandao, R., & Antunes, C. M. (2012). Release of Bet v 1 from birch pollen from 5 European countries. Results from the HIALINE study. Atmospheric Environment, 55, 496–505. https://doi.org/10.1016/j.atmosenv.2012.01.054
Buters, J. T. M., Weichenmeier, I., Ochs, S., Pusch, G., Kreyling, W., Boere, A. J., Schober, W., & Behrendt, H. (2010). The allergen Bet v 1 in fractions of ambient air deviates from birch pollen counts. Allergy, 65(7), 850–858. https://doi.org/10.1111/j.1398-9995.2009.02286.x
Campbell, B. C., Al Kouba, J., Timbrell, V., Noor, M., Massel, K., Gilding, E. K., Angel, N., Kemish, B., Hugenholtz, P., Godwin, I. D., & Davies, J. M. (2020). Tracking seasonal changes in diversity of pollen allergen exposure: Targeted metabarcoding of a subtropical aerobiome. Science of the Total Environment, 747, 141189. https://doi.org/10.1016/j.scitotenv.2020.141189
Cecchi, L. (2013). From pollen count to pollen potency: The molecular era of aerobiology. European Respiratory Journal, 42(4), 898–900. https://doi.org/10.1183/09031936.00096413
Celenk, S. (2019). Detection of reactive allergens in long-distance transported pollen grains: Evidence from Ambrosia. Atmospheric Environment, 209, 212–219. https://doi.org/10.1016/j.atmosenv.2019.04.040
Damialis, A., Traidl-Hoffmann, C., & Treudler, R. (2019). Climate change and pollen allergies (pp. 47–66). Springer International Publishing.
De Linares, C., Alcázar, P., Valle, A. M., Díaz De La Guardia, C., & Galán, C. (2019). Parietaria major allergens versus pollen in the air we breathe. Environmental Research, 176, 108514. https://doi.org/10.1016/j.envres.2019.05.045
De Linares, C., Diaz de la Guardia, C., Nieto Lugilde, D., & Alba, F. (2010). Airborne study of grass allergen (Lol p 1) in different-sized particles. International Archives of Allergy and Immunology, 152(1), 49–57. https://doi.org/10.1159/000260083
De Linares, C., Nieto-Lugilde, D., Alba, F., Díaz De La Guardia, C., Galán, C., & Trigo, M. M. (2007). Detection of airborne allergen (Ole e 1) in relation to Olea europaea pollen in S Spain. Clinical and Experimental Allergy, 37(1), 125–132. https://doi.org/10.1111/j.1365-2222.2006.02620.x
de Weger, L. A., Bergmann, K. C., Rantio-Lehtimäki, A., Dahl, Å., Buters, J., Déchamp, C., Belmonte, J., Thibaudon, M., Cecchi, L., Besancenot, J. P., & Galán, C. (2013). Impact of pollen. In M. Sofiev & K.-C. Bergmann (Eds.), Allergenic pollen: A review of the production, release, distribution and health impacts (pp. 161–215). Springer Netherlands.
Fernández-González, D., González-Parrado, Z., Vega-Maray, A. M., Valencia-Barrera, R. M., Camazon-Izquierdo, B., De Nuntiis, P., & Mandrioli, P. (2010). Platanus pollen allergen, Pla a 1: Quantification in the atmosphere and influence on a sensitizing population. Clinical and Experimental Allergy, 40(11), 1701–1708. https://doi.org/10.1111/j.1365-2222.2010.03595.x
Fernández-González, M., Alvarez-Lopez, S., Gonzalez-Fernandez, E., Jesus Aira, M., & Rodriguez-Rajo, F. J. (2020). Cross-reactivity between the Betulaceae family and fallout in the real atmospheric aeroallergen load. Science of the Total Environment, 715, 136861. https://doi.org/10.1016/j.scitotenv.2020.136861
Fernández-González, M., Guedes, A., Abreu, I., & Rodríguez-Rajo, F. J. (2013). Pla a_1 aeroallergen immunodetection related to the airborne Platanus pollen content. Science of the Total Environment, 463–464, 855–860. https://doi.org/10.1016/j.scitotenv.2013.06.062
Fernández-González, M., Ribeiro, H., Pereira, J. R. S., Rodriguez-Rajo, F. J., & Abreu, I. (2019a). Assessment of the potential real pollen related allergenic load on the atmosphere of Porto city. Science of the Total Environment, 668, 333–341. https://doi.org/10.1016/j.scitotenv.2019.02.345
Fernández-González, M., Ribeiro, H., Pereira, J. R. S., Rodríguez-Rajo, F. J., & Abreu, I. (2019b). Assessment between Platanus pollen and Pla a 1 allergen in two cities of North-western Iberian Peninsula. Aerobiologia, 35(3), 463–475. https://doi.org/10.1007/s10453-019-09576-0
Galan, C., Antunes, C., Brandao, R., Torres, C., Garcia-Mozo, H., Caeiro, E., Ferro, R., Prank, M., Sofiev, M., Albertini, R., & Berger, U. (2013). Airborne olive pollen counts are not representative of exposure to the major olive allergen Ole e 1. Allergy, 68(6), 809–812. https://doi.org/10.1111/all.12144
Gilles, S., Behrendt, H., Ring, J., & Traidl-Hoffmann, C. (2012). The pollen enigma: Modulation of the allergic immune response by non-allergenic, pollen-derived compounds. Current Pharmaceutical Design, 18, 2314–2319. https://doi.org/10.2174/138161212800166040
Gilles, S., Mariani, V., Bryce, M., Mueller, M. J., Ring, J., Jakob, T., Pastore, S., & BehrendtTraidl-Hoffmann, H. C. (2009). Pollen-derived E1-phytoprostanes signal via PPAR-γ and NF-κB-dependent mechanisms. The Journal of Immunology, 182(11), 6653–6658. https://doi.org/10.4049/jimmunol.0802613
Global Asthma Network. (2018). The Global Asthma report 2018. Global Asthma Network.
Gong, X. M., Wang, Q. Y., Lu, S. L., Suzuki, M., Nakajima, D., Sekiguchi, K., & Miwa, M. (2017). Size distribution of allergenic Cry j 2 released from airborne Cryptomeria japonica pollen grains during the pollen scattering seasons. Aerobiologia, 33(1), 59–69. https://doi.org/10.1007/s10453-016-9450-6
Haberle, S. G., Bowman, D. M. J. S., Newnham, R. M., Johnston, F. H., Beggs, P. J., Buters, J., Campbell, B., Erbas, B., Godwin, I., Green, B. J., & Huete, A. (2014). The macroecology of airborne pollen in Australian and New Zealand urban areas. PLoS ONE, 9(5), e97925. https://doi.org/10.1371/journal.pone.0097925
Hirst, J. M. (1952). An automatic volumetric spore trap. Annals of Applied Biology, 39(2), 257–265. https://doi.org/10.1111/j.1744-7348.1952.tb00904.x
Jochner, S., Lupke, M., Laube, J., Weichenmeier, I., Pusch, G., Traidl-Hoffmann, C., Schmidt-Weber, C., Buters, J. T., & Menzel, A. (2015). Seasonal variation of birch and grass pollen loads and allergen release at two sites in the German Alps. Atmospheric Environment, 122, 83–93. https://doi.org/10.1016/j.atmosenv.2015.08.031
Johnston, F., Wheeler, A., Williamson, G., Jones, P., & Koolhof, I. S. (2018). AirRater Tasmania: Using smartphone technology to understand local environmental drivers of symptoms in people with asthma and allergic rhinitis. Journal of Allergy and Clinical Immunology, 141(2), AB84. https://doi.org/10.1016/j.jaci.2017.12.272
Jones, P. J., Koolhof, I. S., Wheeler, A. J., Williamson, G. J., Lucani, C., Campbell, S. L., Bowman, D. M., & Johnston, F. H. (2020). Can smartphone data identify the local environmental drivers of respiratory disease? Environmental Research, 182, 109118. https://doi.org/10.1016/j.envres.2020.109118
Katelaris, C. H., Lee, B. W., Potter, P. C., Maspero, J. F., Cingi, C., Lopatin, A., Saffer, M., Xu, G., & Walters, R. D. (2012). Prevalence and diversity of allergic rhinitis in regions of the world beyond Europe and North America. Clinical & Experimental Allergy, 42(2), 186–207. https://doi.org/10.1111/j.1365-2222.2011.03891.x
Kernerman, S., McCullough, J., & Ownby, D. (1992). Increased concentrations of airborne grass allergen during lawn mowing. Journal of Allergy and Clinical Immunology, 90(1), 131–133. https://doi.org/10.1016/s0091-6749(06)80022-9
Kim, K. R., Oh, J. W., Woo, S. Y., Seo, Y. A., Choi, Y. J., Kim, H. S., Lee, W. Y., & Kim, B. J. (2018). Does the increase in ambient CO2 concentration elevate allergy risks posed by oak pollen? International Journal of Biometeorology, 62(9), 1587–1594. https://doi.org/10.1007/s00484-018-1558-7
Kulthanan, K., Chusakul, S., Recto, M. T., Gabriel, M. T., Aw, D.C.-W., Prepageran, N., Wong, A., Leong, J. L., Foong, H., Quang, V. T., & Zuberbier, T. (2018). Economic burden of the inadequate management of allergic rhinitis and urticaria in Asian countries based on the GA2LEN model. Allergy, Asthma & Immunology Research, 10(4), 370. https://doi.org/10.4168/aair.2018.10.4.370
Lommen, S. T. E., Ciappetta, S., Ghiani, A., Asero, R., Gentili, R., Muller-Scharer, H., & Citterio, S. (2017). Defoliation of common ragweed by Ophraella communa beetle does not affect pollen allergenicity in controlled conditions. Plant Biosystems, 151(6), 1094–1100. https://doi.org/10.1080/11263504.2016.1244122
Matricardi, P. M., Kleine-Tebbe, J., Hoffmann, H. J., Valenta, R., Hilger, C., Hofmaier, S., Aalberse, R. C., Agache, I., Asero, R., Ballmer-Weber, B., & Barber, D. (2016). EAACI molecular allergology user’s guide. Pediatric Allergy and Immunology, 27(Suppl 23), 1–250. https://doi.org/10.1111/pai.12563
Munn, Z., Peters, M. D. J., Stern, C., Tufanaru, C., McArthur, A., & Aromataris, E. (2018). Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Medical Research Methodology, 18(1), 1–7. https://doi.org/10.1186/s12874-018-0611-x
Obersteiner, A., Gilles, S., Frank, U., Beck, I., Haring, F., Ernst, D., Rothballer, M., Hartmann, A., Traidl-Hoffmann, C., & Schmid, M. (2016). Pollen-associated microbiome correlates with pollution parameters and the allergenicity of pollen. PLoS ONE, 11(2), 16. https://doi.org/10.1371/journal.pone.0149545
Oeder, S., Alessandrini, F., Wirz, O. F., Braun, A., Wimmer, M., Frank, U., Hauser, M., Durner, J., Ferreira, F., Ernst, D., & Mempel, M. (2015). Pollen-derived nonallergenic substances enhance Th2-induced IgE production in B cells. Allergy, 70(11), 1450–1460. https://doi.org/10.1111/all.12707
Oteros, J., Bartusel, E., Alessandrini, F., Núñez, A., Moreno, D. A., Behrendt, H., Schmidt-Weber, C., Traidl-Hoffmann, C., & Buters, J. (2019). Artemisia pollen is the main vector for airborne endotoxin. Journal of Allergy and Clinical Immunology, 143(1), 369–377.e365. https://doi.org/10.1016/j.jaci.2018.05.040
Pawankar, R., Holgate, S., Walter Canonica, G., Lockey, R., & Blaiss, M. (2013). WAO white book on allergy—2013 update. World Allergy Organisation.
Plaza, M. P., Alcazar, P., & Galan, C. (2016). Correlation between airborne Olea europaea pollen concentrations and levels of the major allergen Ole e 1 in Crdoba, Spain, 2012–2014. International Journal of Biometeorology, 60(12), 1841–1847. https://doi.org/10.1007/s00484-016-1171-6
Porcel Carreño, S., Gómez Nieves, E., Fernández-Caldas, E., Abel Fernández, E., Cases, B., Tudela, J., Maghfour Martin, Y., Domínguez Domínguez, E., Alvarado Arenas, M., Jiménez Timón, S., Ahmida, T., García Ponce, J. F., Jiménez Gallardo, P., Alvarado Izquierdo, M. I., & Hernández Arbeiza, J. (2020). Immunochemical and physical quantitation of grass and olive pollen allergens: Correlation with asthma admissions in Cáceres, Spain. Journal of Investigational Allergology and Clinical Immunology, 30(5), 334–339. https://doi.org/10.18176/jiaci.0434
Riediker, M., Koller, T., & Monn, C. (2000). Determination of birch pollen allergens in different aerosol sizes. Aerobiologia, 16(2), 251–254. https://doi.org/10.1023/A:1007651106231
Rodríguez-Rajo, F. J., Jato, V., González-Parrado, Z., Elvira-Rendueles, B., Moreno-Grau, S., Vega-Maray, A., Fernández-González, D., Asturias, J. A., & Suárez-Cervera, M. (2011). The combination of airborne pollen and allergen quantification to reliably assess the real pollinosis risk in different bioclimatic areas. Aerobiologia, 27(1), 1–12. https://doi.org/10.1007/s10453-010-9170-2
Ščevková, J., Vašková, Z., Sepšiová, R., Dušička, J., & Kováč, J. (2020). Relationship between Poaceae pollen and Phl p 5 allergen concentrations and the impact of weather variables and air pollutants on their levels in the atmosphere. Heliyon, 6(7), e04421. https://doi.org/10.1016/j.heliyon.2020.e04421
Schäppi, G. F., Suphioglu, C., Taylor, P. E., & Knox, R. B. (1997a). Concentrations of the major birch tree allergen Bet v 1 in pollen and respirable fine particles in the atmosphere. Journal of Allergy and Clinical Immunology, 100(5), 656–661. https://doi.org/10.1016/S0091-6749(97)70170-2
Schäppi, G. F., Taylor, P. E., Pain, M. C., Cameron, P. A., Dent, A. W., Staff, I. A., & Suphioglu, C. (1999). Concentrations of major grass group 5 allergens in pollen grains and atmospheric particles: Implications for hay fever and allergic asthma sufferers sensitized to grass pollen allergens. Clinical and Experimental Allergy: Journal of the British Society for Allergy and Clinical Immunology, 29(5), 633–641. https://doi.org/10.1046/j.1365-2222.1999.00567.x
Schäppi, G. F., Taylor, P. E., Staff, I. A., Suphioglu, C., & Knox, R. B. (1997b). Source of Bet v 1 loaded inhalable particles from birch revealed. Sexual Plant Reproduction, 10(6), 315–323. https://doi.org/10.1007/s004970050105
Senechal, H., Visez, N., Charpin, D., Shahali, Y., Peltre, G., Biolley, J. P., Lhuissier, F., Couderc, R., Yamada, O., Malrat-Domenge, A., & Pham-Thi, N. (2015). Review of the effects of major atmospheric pollutants on pollen grains, pollen content, and allergenicity. The Scientific World Journal, 2015, 940243. https://doi.org/10.1155/2015/940243
Sercombe, J. K., Green, B. J., Rimmer, J., Burton, P. K., Katelaris, C. H., & Tovey, E. R. (2011). London plane tree bioaerosol exposure and allergic sensitization in Sydney, Australia. Annals of Allergy, Asthma & Immunology, 107(6), 493–500. https://doi.org/10.1016/j.anai.2011.08.011
Singer, B. D., Ziska, L. H., Frenz, D. A., Gebhard, D. E., & Straka, J. G. (2005). Increasing Amb a 1 content in common ragweed (Ambrosia artemisiifolia) pollen as a function of rising atmospheric CO2 concentration. Functional Plant Biology, 32(7), 667–670. https://doi.org/10.1071/FP05039
Süring, K., Bach, S., Höflich, C., & Straff, W. (2016). Flow cytometric analysis of particle-bound bet v 1 allergen in PM10. Journal of Visualized Experiments, 2016(117), e54721. https://doi.org/10.3791/54721
Tashpulatov, A. S., Clement, P., Akimcheva, S. A., Belogradova, K. A., Barinova, I., Rakhmawaty, F. D., Heberle-Bors, E., & Touraev, A. (2004). A model system to study the environment-dependent expression of the Bet v 1a gene encoding the major birch pollen allergen. International Archives of Allergy and Immunology, 134(1), 1–9. https://doi.org/10.1159/000077527
Taylor, P. E., Flagan, R. C., Valenta, R., & Glovsky, M. M. (2002). Release of allergens as respirable aerosols: A link between grass pollen and asthma. Journal of Allergy and Clinical Immunology, 109(1), 51–56. https://doi.org/10.1067/mai.2002.120759
Tham, R., Vicendese, D., Dharmage, S. C., Hyndman, R. J., Newbigin, E., Lewis, E., Osullivan, M., Lowe, A. J., Taylor, P., Bardin, P., & Tang, M. L. (2017). Associations between outdoor fungal spores and childhood and adolescent asthma hospitalizations. Journal of Allergy and Clinical Immunology, 139(4), 1140–1147.e1144. https://doi.org/10.1016/j.jaci.2016.06.046
Thien, F., Beggs, P. J., Csutoros, D., Darvall, J., Hew, M., Davies, J. M., Bardin, P. G., Bannister, T., Barnes, S., Bellomo, R., & Byrne, T. (2018). The Melbourne epidemic thunderstorm asthma event 2016: An investigation of environmental triggers, effect on health services, and patient risk factors. The Lancet Planetary Health, 2(6), e255–e263. https://doi.org/10.1016/s2542-5196(18)30120-7
Vara, A., Fernandez-Gonzalez, M., Aira, M. J., & Rodriguez-Rajo, F. J. (2016). Oleaceae cross-reactions as potential pollinosis cause in urban areas. Science of the Total Environment, 542, 435–440. https://doi.org/10.1016/j.scitotenv.2015.10.107
Wang, Q., Gong, X., Suzuki, M., Lu, S., Sekiguchi, K., Nakajima, D., & Miwa, M. (2013a). Size-segregated allergenic particles released from airborne Cryptomeria japonica pollen grains during the Yellow Sand events within the pollen scattering seasons. Asian Journal of Atmospheric Environment, 7(4), 191–198. https://doi.org/10.5572/ajae.2013.7.4.191
Wang, Q., Nakamura, S., Lu, S., Nakajima, D., Suzuki, M., Sekiguchi, K., & Miwa, M. (2013b). Diurnal and nocturnal behaviour of airborne Cryptomeria japonica pollen grains and the allergenic species in urban atmosphere of Saitama, Japan. Asian Journal of Atmospheric Environment, 7(2), 65–71. https://doi.org/10.5572/ajae.2013.7.2.065
Wang, Q., Nakamura, S., Lu, S., Xiu, G., Nakajima, D., Suzuki, M., Sakamoto, K., & Miwa, M. (2012). Release behavior of small sized daughter allergens from Cryptomeria japonica pollen grains during urban rainfall event. Aerobiologia, 28(1), 71–81. https://doi.org/10.1007/s10453-011-9212-4
Wimmer, M., Alessandrini, F., Gilles, S., Frank, U., Oeder, S., Hauser, M., Ring, J., Ferreira, F., Ernst, D., Winkler, J. B., & Schmitt-Kopplin, P. (2015). Pollen-derived adenosine is a necessary cofactor for ragweed allergy. Allergy, 70(8), 944–954. https://doi.org/10.1111/all.12642
Ziska, L. H. (2020). An overview of rising CO2 and climatic change on aeroallergens and allergic diseases. Allergy, Asthma & Immunology Research, 12(5), 771. https://doi.org/10.4168/aair.2020.12.5.771
Ziska, L. H., Makra, L., Harry, S. K., Bruffaerts, N., Hendrickx, M., Coates, F., Saarto, A., Thibaudon, M., Oliver, G., Damialis, A., & Charalampopoulos, A. (2019). Temperature-related changes in airborne allergenic pollen abundance and seasonality across the northern hemisphere: A retrospective data analysis. The Lancet Planetary Health, 3(3), e124–e131. https://doi.org/10.1016/s2542-5196(19)30015-4
Zuberbier, T., Lotvall, J., Simoens, S., Subramanian, S. V., & Church, M. K. (2014). Economic burden of inadequate management of allergic diseases in the European Union: A GA(2) LEN review. Allergy, 69(10), 1275–1279. https://doi.org/10.1111/all.12470
Acknowledgements
The authors would like to acknowledge the muwinina people, the traditional and original owners of the land that this research was undertaken, and to pay respect to those that have passed before us. The authors would like to acknowledge Michaela Venn, the University of Tasmania Clinical School librarian, for guidance with using search databases and Covidence. The authors would also like to acknowledge Professor Ray Lowenthal (Menzies Institute for Medical Research) for guidance and advice with academic writing and structuring. The findings and the conclusions in this report are those of the authors and do not necessarily represent the official position of the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention.
Funding
LT is a PhD Candidate who is funded by the MIMR Tasmania Graduate Research Scholarship. JLD is funded by a Select Foundation Principal Research Fellowship. FJ is supported by a Select Foundation Fellowship. NBA is a PhD Candidate who is supported by a MIMR Tasmania Graduate Research Scholarship, by Asthma Australia through a Top-up Scholarship and by the New South Wales Bushfire Risk Management Research Hub through a Top-up Scholarship. PJ receives funding from Public Health Services, Tasmanian Department of Health.
Author information
Authors and Affiliations
Contributions
LT, PJ and FJ conceptualised the manuscript; LT and NBA screened publications by title and abstract; LT screened full texts; LT, PJ, NBA, JD, BG, AW and FJ all contributed to writing and editing the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tegart, L.J., Johnston, F.H., Borchers Arriagada, N. et al. ‘Pollen potency’: the relationship between atmospheric pollen counts and allergen exposure. Aerobiologia 37, 825–841 (2021). https://doi.org/10.1007/s10453-021-09726-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10453-021-09726-3