Abstract
Desmids are usually abundant in shallow peatland pools. In these localities, water temperature is closely linked to seasonal fluctuations in air temperature, so with increasing temperature extremes in temperate ecosystems, these microalgae are exposed to conditions of high-temperature stress. We investigated whether the shape, size, and growth rates of Micrasterias thomasiana, a frequently occurring species, are associated with varying temperatures in cultures and natural populations. The research was based on parallel analysis of clonal populations in temperature levels from 13 to 33 °C as well as cells from natural populations collected during the season. The effects of high temperature on morphological plasticity and fluctuating asymmetry in the shape of cellular parts were investigated by the landmark-based geometric morphometrics. The results showed that variation among individuals and fluctuating asymmetry between the lateral lobes of Micrasterias cells increased at 29 °C and in natural samples taken in July and October. In parallel, the size of semicells growing at temperatures above 25 °C decreased compared to those grown at lower temperatures. However, the temperature effects on shape and size were not directly related to the growth rates. The overall bilateral asymmetry between semicell halves did not change in relation to varying temperatures. In general, the results showed that morphological variation in natural populations of M. thomasiana reflected seasonal cycles and corresponded to plasticity associated with temperature changes in clonal cultures. It might therefore be possible to use these phenotypic markers as indicators of thermal stress in natural populations inhabiting shallow pools in peatlands.
Similar content being viewed by others
Introduction
Desmid microalgae (Desmidiales, Zygnematophyceae) are an important component of the peatland phytobenthos. The vast majority of peatlands are found in the boreal biome of the Northern Hemisphere, but smaller areas of these habitats also exist in temperate zones, especially in mountain ecosystems and regions with relatively high annual precipitation (Tanneberger et al. 2017). In these acidic wetlands, a substantial part of the aquatic habitat consists of shallow pools where water temperature is closely correlated with air temperature (Jacobs et al. 1997). Consequently, the organisms living in the phytobenthos of these sites face considerable temperature differences throughout the season, ranging from freezing conditions in the winter months to summer extremes exceeding 30 °C. Thus, it is possible that part of the seasonal cycle of ectothermic organisms, such as desmidiacean green algae, in peatland pools includes periods with stress conditions at the margins of their temperature niche. Currently, temperate mountain peatlands in central Europe are seeing an increase in the frequency of extreme high-temperature events in the summer season, which are related to ongoing climate change (Neustupa et al. 2023). It is therefore possible that the amounts of temperature stress in natural desmid populations could reflect these processes.
Previous studies have shown that high-temperature stress can be markedly reflected in the morphogenesis of desmid cells, such as in the phylogenetic lineage of the genus Micrasterias, which are typical for the most complex cellular forms in the plant kingdom (Brook 1981; Škaloud et al. 2011; Lütz-Meindl 2016). Their cells are characteristically composed of two symmetrical halves (semicells), which themselves are also bilaterally symmetrical. A younger semicell develops after mitotic division in the process of cytomorphogenesis (Lütz-Meindl 2016). Consequently, each cell is composed of two modular halves, symmetric to each other but different in age (Brook 1981; Neustupa 2017). It has been shown that the bilateral symmetry of cellular parts within semicells and their species-specific patterns are severely disrupted in cells grown at temperatures above 33 °C in alpine strains of Micrasterias denticulata (Meindl 1990). Likewise, the cellular morphogenesis of this species was considerably distorted when the cells were exposed to short-term 40 °C heat shocks (Weiss et al. 1999). In addition, Neustupa et al. (2008) showed that the clonal strain of Micrasterias rotata produced cells with deviant morphology typical of so-called vegetative diploids when cultured under high-temperature conditions at 30 °C. These conditions were also associated with a reduction in vegetative cells and associated simplification of their morphology. The observed changes in cell size were largely consistent with the assumptions of the temperature–size rule for protist species, which assumes a gradual decrease in cell size related to temperature increase within a range that is normally encountered by the organism in natural habitats (Atkinson et al. 2003). However, these observations regarding the effect of temperature on the growth of Micrasterias cells were not accompanied by data on the growth rates of the populations in relation to ambient temperature. Such data might be crucial for assessing whether the observed size decrease can be attributed to earlier completion of the life cycle in rapidly growing high-temperature populations or to the adaptive responses of cells increasing their surface-to-volume ratios to cope with the lower nutrient and oxygen availability in a warmer aquatic environment (Neustupa et al. 2008; Tabi et al. 2020; Zohary et al. 2021). In addition, it is unknown whether similar dynamics of temperature-related size decreases might also take place in natural populations of desmid species inhabiting the phytobenthos of shallow pools.
It has also been repeatedly shown that stressful high-temperature conditions can lead to an increase in different types of fluctuating asymmetry (FA) in the morphology of symmetric structures in different aquatic ectotherms (Mpho et al. 2002; Nishizaki et al. 2015). This essentially means that morphogenetic processes taking place under conditions close to the margin of the thermal niche may lead to decreased developmental control, which is associated with an increase in random asymmetric fluctuations in the morphology of symmetric body parts in different individuals (Klingenberg 2015). However, no such analyses of shape FA have yet been performed for the effects of temperature on unicellular organisms.
Therefore, in this study, we focused on analysing temperature effects on different aspects of cell morphology and plasticity in the desmid species Micrasterias thomasiana, one of the frequently occurring members of the phytobenthos in minerotrophic peatlands across temperate and boreal biomes (Coesel and Meesters 2007). For the biological shape analysis, we used geometric morphometric techniques (Bookstein 2018). In addition to the analysis of shape and size variation among individuals within cultured populations reared at different temperatures and natural samples from different seasons, we also focused on quantification of shape asymmetry between different parts of Micrasterias semicells.
Thus, the following research questions were asked in this study: (1) Are there any effects of temperature on the shape, size, and growth rates of clonal populations of Micrasterias thomasiana? (2) Can the potential stress effects of high temperatures be detected in increased plasticity or FA levels among different cellular parts? (3) Are there any corresponding trends in the shape, size and cellular symmetry in the natural samples of this species from a temperate peatland pool within a season?
Material and Methods
Field sampling
Cells of Micrasterias thomasiana were sampled from the phytobenthos of a minerotrophic pool (50.509861N, 13.196075E) in a peatland headwater area of a levelled mountainous plateau in the Ore Mts. (Czech Republic) at an altitude of 848 m a.s.l. Samples of this natural population were taken four times in 2022, together with the basic physicochemical characteristics of the site. The sampling dates and their respective abiotic characteristics were January 3 (depth = 10 cm, pH = 5.35, water temperature = 1.5 °C), April 26 (8 cm, 5.30, 6.8 °C), July 25 (5 cm, 5.78, 26.9 °C), and October 21 (7 cm, 5.35, 12.5 °C). In addition, the average air temperature at 2 m for the last 8 weeks before sampling was acquired from publicly available data from the nearby meteorological station in Měděnec (CHMI 2023). These values were as follows: January—0.6 °C, April—2.8 °C, July—15.7 °C, and October—9.4 °C.
Cultivation and specific growth rates (SGR)
A clonal strain of the same species was obtained as a single-cell isolate from the studied population in April 2022. The populations were cultured in 25 ml culture wells at six temperatures (13, 17, 21, 25, 29, and 33 °C) in Heratherm IMC18 (Fisher Scientific, Ltd.) incubators, and they were illuminated at 1775 lx with LED PDGROW 35 W E27 Plant Lamp Bulbs (Sansi Electronic Engineering, Ltd.) under a light:dark (L:D) regime of 12:12 h. Four parallel populations were cultured at each of the six temperatures. The strains were cultured in DY V liquid medium (Andersen 2005) buffered by 2-(N-morpholino)ethanesulfonic acid (MES) at pH 5.5.
Up to 70 cells were inoculated into each of the cultivation chambers. After 7 days of acclimation, the specific growth rates were calculated from the cellular counts on days no. 7 and 10 of the cultivation period when the cells were in the phase of fastest growth. Vegetative reproduction of desmidial cells solely proceeds by binary division. Thus, binary logarithms were taken as the base for the formula evaluating the SGR = log2(xn)-log2(xm)/tn-tm, where xm and xn are the numbers of individuals on the mth and nth days of cultivation. Likewise, tm and tn represent days within the cultivation period at the start and the end of the SGR calculation. The
Data acquisition
A total of 50 mature semicells from each temperature and seasonal sample were photographed at 100 × magnification under a Leica DM2500 light microscope (Leica Microsystems, Wetzlar, Germany) with a Bresser MikroCam SP 5.0 digital camera (Bresser, Rhede, Germany). In total, 73 structurally homologous landmarks were positioned at the tips of the semicell lobes and lobules, as well as at the bases of individual incisions (Fig. 1a). To account for the digitisation error, the landmarks on each semicell were registered twice. In the first digitisation, they were positioned clockwise from the left margin of the semicells. The second digitisation proceeded counterclockwise from the right semicell margin. Then, they were relabelled to match the order of the first digitisation. The landmarks were registered using TpsDig, ver. 2.22 (Rohlf 2015). All these primary data are available online from Zenodo (Neustupa and Woodard 2023).
Symmetry analysis
The effects of temperature on cellular shape, size and plasticity were addressed by several parallel analyses. First, shape differences among the semicells raised at different temperatures were studied by a geometric morphometric analysis of their symmetrised configurations that did not involve any effects of bilateral symmetry within the specimens. Second, the shape asymmetry was quantified by evaluating differences between the two semicell halves of each specimen. Third, FA evaluating the random individual fluctuations in the shape of symmetric lateral lobes of semicells was used to assess possible differences among the temperatures and seasonal samples. Finally, size differentiation was quantified by the centroid size (CS) of the semicell configurations acquired from the morphometric data.
The frontal views of Micrasterias semicells are bilaterally symmetrical. In addition, their anterior and posterior sides do not differ (Savriama et al. 2010; Neustupa 2017). Thus, to compare semicell shapes among different individuals, the landmark configurations have to be symmetrised; i.e., their halves need to be averaged. This procedure was implemented by the standard formula introduced by Klingenberg et al. (2002), which involved reflection of the coordinates across the axis of bilateral symmetry (Fig. 1b), their re-labelling to match the order of landmarks in the original configuration, and averaging the mirrored (reflected and re-labelled) and original configurations of each specimen in the generalised Procrustes analysis (GPA).
Symmetric configurations of semicells aligned by GPA were then used to compare their shape and variability among the temperatures and seasonal samples. Principal component analysis (PCA) of the semicell configurations from six temperatures was carried out to illustrate the structure of the data and the shape changes associated with the main principal components (PCs). Scores of individual semicells along the first 16 PCs (spanning 93.8% of the total variation) were used in the canonical variate analysis (CVA), which aimed to visualise the differences among the temperatures. Differences in shape among groups were tested using permutational multivariate analysis of variance (MANOVA) based on Euclidean distances among the specimens (Zelditch et al. 2012). The significance of the post hoc pairwise F-values was evaluated by sequential Bonferroni p-values based on 9999 random permutations. The permutational MANOVA with a balanced design used in this analysis is very robust to heterogeneity of multivariate dispersions. In addition, resampling statistics (permutations) used for the significance assessment of shape differences among the cells raised in varying temperature levels does not assume normality and it is not affected by high dimensionality of the shape data (Cardini et al. 2015; Anderson 2017). The mean shapes of individual temperatures and seasonal samples were subjected to an additional PCA that illustrated mutual shape relationships among these populations. Differences in the amount of shape variation among groups were analysed using Procrustes distances of individual specimens to the mean configurations of their respective groups.
In order to assess whether the sample sizes of individual groups were sufficient for the analyses comparing their mean shapes, the sensitivity analysis on sample means, which has been recommended for geometric morphometric datasets, was carried out (Cardini et al 2015). Random subsamples consisting of 45, 40, 35, and 30 specimens for each group were created and their mean shapes were compared with the mean shapes original datasets by the joint non-metric multidimensional scaling (NMDS) with the Euclidean distance measure. Then, positions of the original and reduced shapes in this ordination space were compared to evaluate the changes caused by the progressive sample size reduction.
Bilateral asymmetry and FA
The overall bilateral asymmetry in shape of the opposite quadrants composing each semicell was quantified by Procrustes distances between the corresponding landmark configurations (Fig. 1b). Due to the lack of side differentiation within semicells, this measure of shape asymmetry cannot separate the side-directed, average asymmetry from individual asymmetric deviations (FA). However, such separation is possible in an analysis of shape symmetry between the lower and upper lateral sublobes because their positions can be unambiguously determined with respect to their positions within the entire lateral lobe (Fig. 1c) (Neustupa and Stastny 2018). Thus, the standard methodology of the geometric morphometric analysis of bilateral symmetry, as widely used in evolutionary biology and ecology studies (Klingenberg et al. 2002; Klingenberg 2015), can be applied here. Consequently, the GPA-aligned configurations of these lateral lobes were subjected to the two-way mixed model Procrustes analysis of variance (Procrustes ANOVA) quantifying the differences among individuals, the average (positional) asymmetry and FA (Klingenberg 2015). Note that the term "positional asymmetry" corresponds to the definition of directional asymmetry as used in studies focused on symmetry analysis of laterally oriented bilaterally symmetric structures, such as animal body parts or leaves of vascular plants (Neustupa and Stastny 2018).
The FA scores denoting the Procrustes distances of individual asymmetric deviations from the mean configuration were used for the comparison among populations. Size differences among the specimens were evaluated using CS, which is the square root of the sum of squared distances of all landmarks composing the configuration from their centroid (Zelditch et al. 2012). The 95% confidence intervals for the mean values of the univariate measures (symmetric variation, bilateral asymmetry, FA, and CS) were constructed using 9999 bootstrap repetitions of the original datasets.
Generalised Procrustes analysis (GPA) was implemented by the function procGPA in the shapes package, ver. 1.2.6, of R, ver. 4.0.5 (R Core Team 2021). The PCAs of the aligned coordinates and subsequent CVA were conducted in PAST, ver. 4.10 (Hammer et al. 2001). The shapes typical for marginal positions on individual ordination axes were visualised in TpsRegr, ver. 1.50 (Rohlf 2015), by regressing the aligned coordinates to the object scores yielded by the multivariate analyses. The analyses of cellular symmetry and asymmetry were implemented by utility scripts written in R, ver. 4.0.5 (R Core Team 2021), which used the functions in the packages geomorph, ver. 4.0.0 (Adams and Otárola-Castillo 2013),
Results
Growth rates
The mean SGRs of cultures raised at different temperatures in cultivation chambers varied from 0.283 days−1 at 25 °C to 0.006 days−1 at 33 °C (Fig. 2). The SGR rates were positive, indicating cellular growth in cultures in all chambers at temperatures from 13 to 29 °C. On the other hand, the populations exposed to 33 °C essentially showed stagnation or a slight decrease in the number of cells. Thus, these populations were not included in subsequent analyses. At the other temperature levels, the SGR values were lowest at the marginal temperatures of 13 and 29 °C. Conversely, the highest levels were found at temperatures of 21 and 25 °C,
Symmetric variation among individuals
PCA of the symmetrised configurations of semicells from five different temperatures showed that 47.3% of the total shape variation in the dataset was described by a single axis (PC1). This shape trend differentiated semicells with deep incisions and protruding polar lobes from those with typical relatively shallow incisions and less prominent polar lobes (Fig. 3).
Ordination plot showing the first two principal components based on the shape variation among the semicells of Micrasterias thomasiana raised at different temperatures. The configurations illustrate shapes typical for the opposite most marginal occupied positions on the ordination axes within the shape space. Small symbols depict the positions of individual specimens, and large symbols illustrate the mean positions of five temperatures. The mean values of adjacent temperature groups are connected by lines
In addition, a total of 16.0% of the shape variation was spanned by PC2, which mostly highlighted the shape changes involving the width of the polar lobe of the semicells. The mean positions of the populations raised at 13, 17, and 21 °C were very similar within this ordination space. However, the populations from 25 and 29 °C were clearly shifted along PC1 towards the shapes with shallow incisions of cellular lobes and lobules (Fig. 3). Notably, the right parts of the morphospace, typical for these morphological characteristics, were almost exclusively occupied by semicells raised at these two higher temperatures. The two typical semicells from temperatures 13 °C and 29 °C also clearly illustrate the shape trend described within the PCA ordination space (Fig. 4).
The sensitivity analysis for mean shapes of individual temperature groups that evaluated their stability with decreasing sample size showed that the mutual positions of the mean shapes in reduced datasets were closely similar to those of the original samples of 50 semicells. Namely, the changes in the positions of the sample means of the same temperature level in the original and progressively reduced datasets only accounted for 1.95%, 3.92%, 4.63%, and 5.13% of the shape differences among the original sample means of different temperature groups in the NMDS ordination space. Thus, we concluded that the sample sizes of 50 semicells were sufficient for subsequent analyses.
The CVA yielded the ordination space dominated by the first canonical variate (CV1) that described 51.6% of the variation. Clearly, this axis illustrated separation of two high-temperature groups (25 and 29 °C) in the left part of the ordination space, typical for shapes with shallow incisions and a concave polar lobe, from those raised at lower temperatures (Fig. 5). The overall permutational MANOVA yielded highly significant differentiation among groups (total SS = 0.3011, within-group SS = 0.2484, F = 11.69, p < 0.0001). Likewise, the post hoc pairwise analyses showed that the mean shape features of all the temperature groups, as well as the seasonal samples, differed from each other (Table 1). However, the weakest separation, indicated by relatively low F values, was typical for the cultured populations raised at 13, 17, and 21 °C. In addition, the natural samples were also relatively similar to each other, with the July, October, and January populations yielding the least divergent semicell shapes.
Ordination plot showing the first two axes of the canonical variate analysis differentiating five populations of Micrasterias thomasiana cultured at different temperatures. The configurations illustrate shapes typical for the opposite most marginal occupied positions on the ordination axes within the ordination space. The colours and symbols correspond to those in Fig. 3
The PCA of the mean shapes, combining the cultured strains and the natural samples, graphically illustrated these patterns, such as the close similarity of low-temperature populations raised at 13, 17, and 21 °C, showing typical semicells with deep incisions and protruding polar lobes (Fig. 6). Once again, the mean shapes of the populations raised at 25 and 29 °C were distinctly different, with shallower incisions and less pronounced polar lobe. Among the natural samples, these shape characteristics, characterised by their position in the left part of the ordination space along PC1 (Fig. 6), were most pronounced in populations sampled in July and partly also in October. Conversely, the April sample was the only one that was slightly skewed towards hemispherical shapes typical of lower ambient temperatures in the opposite part of this morphological trend spanned by PC1.
Ordination plot of the principal component analysis based on the mean shapes combining the cultured strains and the natural samples. The configurations illustrate shapes typical for the opposite most marginal occupied positions on the ordination axes within the shape space. Colours of the temperatures correspond to those in Fig. 3
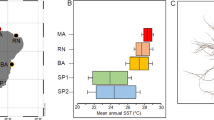
There was a slight unimodal pattern in the degree of variability among individuals in populations cultured at different temperatures (Fig. 7a). The cells grown at 13 °C were somewhat more variable, but the 95% confidence interval of the mean value overlapped slightly with those of the 17 and 21 °C populations. In contrast, the plasticity levels of the 25 and 29 °C populations were higher, and in particular, that of the 29 °C population was significantly different from that of all the populations growing at lower temperatures (Table 2). However, all the natural populations were also more variable than the cultured populations, with the exception of the one raised at 29 °C. However, the shape variability of the summer and autumn samples (July and October) was also significantly higher than that of cells from the January sample (Table 2).
a Symmetric shape variability within clonal populations and natural samples expressed by Procrustes distances (PDs) of specimens to the mean configurations of individual groups. b Size variation in clonal populations and natural samples. The violin plots show the probability density of the observed data smoothed by a kernel density estimator at different values of the variables under study. Colours of the temperatures correspond to those in Fig. 3
The size of individual semicells was significantly different for the three populations from temperatures of 13, 17, and 21 °C and the two populations from temperatures of 25 and 29 °C, and their 95% confidence intervals did not overlap with each other (Fig. 7b, Table 2). Likewise, the semicells of the January and especially the April natural populations were significantly larger than the semicells sampled in July and October.
Bilateral asymmetry and FA
The bilateral asymmetry of semicells was relatively constant across the cultured populations and seasonal samples, with no clearly detectable trends that could be attributed to the varying temperature of the environment (Fig. 8a, Table 2). On the other hand, the FA levels that reflected random asymmetric fluctuations in the shape of the lateral lobules once again followed a slight unimodal pattern across the temperature levels (Fig. 8b). Thus, the mean FA of the cells growing at marginal levels of 13 and 29 °C was slightly but significantly elevated in comparison to that of the populations from 17 and 21 °C. All of the natural samples were typical in terms of their higher FA levels. Among them, the April, July and October
a Bilateral asymmetry in shape of the semicells of the clonal populations and natural samples expressed by Procrustes distances (PDs) between the original and mirrored configurations. b Fluctuating asymmetry in the shape of the lateral lobes in the clonal populations and natural samples. The violin plots show the probability density of the observed data smoothed by a kernel density estimator at different values of the variables under study. Colours of the temperature levels correspond to those in Fig. 3
Discussion
Size and symmetric shape variation in cultures
The present analyses showed that in M. thomasiana, there are significant changes in semicell shape, size, plasticity, and symmetry among different environmental temperatures. At temperatures between 13 to 21 °C, the shape and size of mature semicells were relatively homogeneous, and these values were close to those in the taxonomic description of this species (Coesel and Meesters 2007). In contrast, at temperatures above 25 °C, substantial shifts in semicell morphology were observed, with a reduction in width of approximately 20% and associated changes in symmetric shape features, manifested mainly as comparatively shallow cellular incisions among individual lobes and lobules, and submerged polar lobes. This trend was most pronounced in cells grown at 29 °C, the highest temperature studied, at which a positive SGR of populations was still observed. However, it is interesting that these changes in semicell shape and size did not seem to be directly related to the actual SGRs of the populations. At 25 °C, where the cells already often produced the high-temperature morphotypes, the highest ever SGR was recorded, exceeding values at other temperature levels. This relatively high temperature optimum for population growth is consistent with the generally high-temperature affinities of most desmids that were described in previous studies focused on different genera this taxonomic order (Stamenković and Hanelt 2011; 2017; Santos et al. 2022). In parallel, the decrease in size of semicells with increasing temperature closely corresponded to the classical temperature–size rule for protist aquatic organisms (Atkinson et al. 2003). The associated change is apparently a manifestation of the allometric shape-to-size relationship that has previously been described in several unrelated species of the genus Micrasterias (Neustupa 2016), and thus, it is probably a deeply conserved phenotypic pattern.
The temperature–size rule has typically been explained using two underlying frameworks. The first of these assumes earlier binary division of cells in a higher-temperature environment that is nevertheless still within the temperature niche of a particular species. In this concept, high temperature leads to thermodynamically faster metabolic processes and thus morphogenetically accelerated mitotic division (Tabi et al. 2020). However, this would not explain the similar shifts in shape and plasticity in populations growing at 25 and 29 °C, which differed significantly in their SGRs. In contrast, the marginal temperature of 29 °C should, under this concept, produce large and possibly deviant cells (Atkinson et al. 2003). However, this was not the case, and the observed pattern with decreased SGRs of populations raised at high temperature levels, coupled with their smaller cell sizes and shallower cell incisions fits better with another framework for the interpretation of the temperature-related size diminution in unicellular organisms, namely, the need to regulate the uptake of substances from the environment, such as oxygen and carbon dioxide, which are less available at higher temperatures due to their reduced solubility in water (Tabi et al. 2020; Zohary et al. 2021). This is reflected by the tendency to increase the S:V ratio by decreasing the cell size at higher temperatures.
Size and symmetric shape variation in natural populations
Changes in shape and size that were very similar to those observed under experimental conditions in clonal strains also occurred in natural seasonal samples. In these populations, however, it is impossible to determine exactly how old the individual analysed semicells were. It has been recognised that cells of the genus Micrasterias are able to divide at a maximum rate of approximately once every three days under optimal conditions (Meindl 1993). This was basically consistent with the values of SGRs determined in this study in the cultured populations. In natural populations, however, a significant proportion of semicells may be considerably older, and their morphogenesis took place in the preceding weeks or perhaps even months (Brook 1981). Consistent with this idea, in our samples, the populations from July and October were most similar in terms of shape and size to the high-temperature morphotypes. Conversely, the April sample contained the most cells with typical low-temperature characteristics. Thus, the natural populations seem to have reflected the meteorological conditions of the period preceding the individual sampling dates. The period with the highest average temperature, approximately from May to September, is reflected in the morphological patterns of the summer and autumn samples. These cells are then apparently represented only to a relatively small extent in the winter population, which already contains a substantial proportion of semicells developing at the lower temperatures of the autumn months. Similarly, the spring population is largely composed of semicells that developed in the relatively cool water of the previous weeks.
Interestingly, seasonal morphological variation or plasticity of individual species of microalgae has rarely been explicitly tested in relation to temperature (Naselli-Flores and Padisák 2023). However, there are more data available for different members of the phytoplankton, and it has been shown, for example, that seasonal fluctuations in temperature are among the main drivers of observed morphological variability (Morabito et al. 2007). Likewise, Zohary et al. (2017) showed that the size of most phytoplankton species in a eutrophic subtropical lake reflected the seasonal temperature patterns, with the largest cells occurring in winter and vice versa. However, the dominant group of the phytobenthos, diatoms, were shown to be an exception to the temperature–size rule due to the considerable size ranges of individual taxa related to their size diminution series during asexual reproduction (Adams et al. 2013). For the other major groups, data at the species level are generally not available. This study provides evidence that natural populations of desmids occurring in temperate ecosystems may undergo distinct shape and size changes during the season that correlate with their plastic response to varying temperatures in clonal populations. It is therefore likely that these seasonal dynamics reflect the plastic cycles by which the populations respond to environmental temperature variability within their reaction norms.
Bilateral asymmetry and FA
In contrast to the overall shape changes and their symmetric variability, the bilateral asymmetry within semicells was essentially stable across the temperature levels and seasonal samples. Although the populations contained outlying semicells with greatly increased asymmetry, there was no significant trend correlated with temperature. An earlier study showed that corresponding parts of each semicell in their two symmetric halves are relatively strongly integrated (Neustupa 2017). Thus, looking at the data of the present study, it appears that this integration is probably not dependent on the ambient temperature. A somewhat different pattern was found for the FA levels between the adjacent lateral sublobes. Here, it was evident that high temperature conditions (29 °C or the summer samples) led to a significant increase in these random asymmetric deviations, which have typically been considered to indicate morphogenetic stress (Klingenberg 2015; Benítez et al. 2020).
This was generally in line with several previous studies, which have shown that environmental temperatures of 30 °C or more usually lead to an increasing number of cellular malformations in various strains of the genus Micrasterias, manifested either as simplification of the overall semicell pattern, or in formation of asymmetric cell shapes (Meindl 1990; Weiss et al. 1999; Neustupa et al. 2008; Lütz-Meindl 2016). Considerably increased production of heat shock proteins hsp70 and BiP (binding protein) was detected in Micrasterias denticulata as result of continuous or repeated experimental heat exposure in high temperature levels, indicating increased thermal stress (Weiss and Lütz-Meindl 1999). Local influx of Ca2+ ions has been linked to cell pattern formation in the clonal strain of M. denticulata (Lütz-Meindl 2016). Thus, deviations in cell morphology may be linked to shifts in the distribution of Ca2+ ions at the plasma membrane that were observed in the semicells developing in elevated temperature (Meindl 1990). This can then lead to abnormal growth of the primary cell wall even in parts of a developing semicell where cell incisions would normally be formed due to early stiffening of the wall material (Lütz-Meindl 2016).
Interestingly, both symmetric variation and the FA levels appeared to be significantly more variable in the natural populations than in the cultured strains. However, it is important to note that in the case of clonal populations grown at different temperature levels, the observed variation has to be considered pure phenotypic plasticity within the reaction norm of a single genotype (West-Eberhard 1989; Klingenberg 2019). In the case of the natural samples, on the other hand, the influence of genetic variation within populations, which itself may have led to increased shape variation, should be assumed. In addition, it should also be taken into account that natural populations are exposed to additional environmental influences that may manifest as an increase in stress phenotypic markers compared to those in strains under controlled experimental conditions.
Conclusions
The complex cellular shapes of desmids, characterised by their emphasis on maintaining the complex symmetry of individual lobes and lobules, have long attracted the attention of biologists (Ralfs 1848; Brook 1981). There is no indication that their evolution and development are directly related to biotic interactions, such as herbivory-induced morphological changes in scenedesmacean green algae (Lürling and Van Donk 1999). Thus, these complex cells can possibly be regarded as so-called unaddressed phenomena in the classical Portmannian framework classifying the phenotypic expressions of living organisms (Portmann 1960; 2021). This involves the phenotypic aspects of organisms that have no obvious functional significance and do not appear to be registered or stimulated by other organisms (Jaroš and Klouda 2021). However, the variation and ecological dynamics of such complex phenotypic structures are typical of intrinsic underlying patterns that can be detected by a detailed phenotypic analysis. The shapes of desmid cells represent the basic interface through which these organisms interact with the environment. Thus, the increased amounts of variation among individuals and fluctuating asymmetry between the upper and lower lateral lobes of Micrasterias cells in cultures raised at 29 °C, as well as in the natural samples taken in July and October, can be considered a direct result of high temperature during cell morphogenesis. Likewise, the decrease in size of cultured semicells grown at temperatures above 25 °C, and in the natural samples from the summer season, was consistent with the direct effects of high temperature on free-living unicellular organisms.
Thus, the observed phenotypic changes can be used for evaluating the effects of adverse environmental conditions on these important species of peatland microphytobenthos. In the context of ongoing climate change, which is increasing the frequency of extreme-temperature events, the relationship between temperature and shape dynamics of Micrasterias cells provides an intriguing system for investigating the effects of these stressful conditions on unicellular organisms.
Data availability
The datasets generated and analysed during the current study are available from the Zenodo public repository at https://doi.org/https://doi.org/10.5281/zenodo.8189080.
References
Adams DC, Otárola-Castillo E (2013) Geomorph: an R package for the collection and analysis of geometric morphometric shape data. Meth Ecol Evol 4:393–399. https://doi.org/10.1111/2041-210X.12035
Adams GL, Pichler DE, Cox EJ, O’Gorman EJ, Seeney A, Woodward G, Reuman DC (2013) Diatoms can be an important exception to temperature–size rules at species and community levels of organization. Glob Change Biol 19:3540–3552. https://doi.org/10.1111/gcb.12285
Andersen RA (2005) Algal culturing techniques. Elsevier Acad Press, London
Anderson MJ (2017) Permutational multivariate analysis of variance (PERMANOVA). In: Balakrishnan N, Colton T, Everitt B, Piegorsch W, Ruggeri F, Teugels JL (eds) Wiley StatsRef: statistics reference online. Wiley, Oxford, pp 1–15. https://doi.org/10.1002/9781118445112.stat07841
Atkinson D, Ciotti BJ, Montagnes DJS (2003) Protists decrease in size linearly with temperature: ca. 2.5% °C–1. Proc R Soc Lond B Biol Sci 270:2605–2611. https://doi.org/10.1098/rspb.2003.2538
Benítez HA, Lemic D, Villalobos-Leiva A, Bažok R, Órdenes-Claveria R, Pajač Živkovič I, Mikac KM (2020) Breaking symmetry: Fluctuating asymmetry and geometric morphometrics as tools for evaluating developmental instability under diverse agroecosystems. Symmetry 12:1789. https://doi.org/10.3390/sym12111789
Bookstein FL (2018) A course in morphometrics for biologists. Cambridge Univ Press, Cambridge. https://doi.org/10.1017/9781108120418
Brook AJ (1981) The biology of desmids. Blackwell, Oxford
Cardini A, Seetah K, Barker G (2015) How many specimens do I need? Sampling error in geometric morphometrics: testing the sensitivity of means and variances in simple randomized selection experiments. Zoomorphology 134:149–163. https://doi.org/10.1007/s00435-015-0253-z
CHMI (2023) Daily meteorological data. Czech Hydrometeorological Institute. https://www.chmi.cz/historicka-data/pocasi/denni-data/Denni-data-dle-z.-123-1998-Sb. Accessed 24 July 2023
Coesel PFM, Meesters J (2007) Desmids of the lowlands. KNNV Publ, Zeist
Dryden IL (2021) Shapes package. R Foundation for Statistical Computing. https://www.R-project.org. Accessed 24 July 2023
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeont Electr 4:1–9
Jacobs AFG, Jetten TH, Lucassen DC, Heusinkveld BG, Nieveen JP (1997) Diurnal temperature fluctuations in a natural shallow water body. Agric for Meteorol 88:269–277. https://doi.org/10.1016/S0168-1923(97)00039-7
Jaroš F, Klouda J (2021) Introduction. In: Jaroš F, Klouda J (eds) Adolf Portmann—a thinker of self-expressive life. Springer, Cham, pp 1–9
Klingenberg CP (2015) Analyzing fluctuating asymmetry with geometric morphometrics: concepts, methods, and applications. Symmetry 7:843–934. https://doi.org/10.3390/sym7020843
Klingenberg CP (2019) Phenotypic plasticity, developmental instability, and robustness: the concepts and how they are connected. Front Ecol Evol 7:56. https://doi.org/10.3389/fevo.2019.00056
Klingenberg CP, Barluenga M, Meyer A (2002) Shape analysis of symmetric structures: quantifying variation among individuals and asymmetry. Evolution 56:1909–1920. https://doi.org/10.1111/j.0014-3820.2002.tb00117.x
Lürling M, Van Donk E (1999) Grazer-induced colony formation in Scenedesmus acutus (Chlorophyceae): ecomorph expression at different temperatures. J Phycol 35:1120–1126. https://doi.org/10.1046/j.1529-8817.1999.3561120.x
Lütz-Meindl U (2016) Micrasterias as a model system in plant cell biology. Front Pl Sci 7:999. https://doi.org/10.3389/fpls.2016.00999
Meindl U (1990) Effects of temperature on cytomorphogenesis and ultrastructure ofMicrasterias denticulata Bréb. Protoplasma 157:3–18. https://doi.org/10.1007/BF01322635
Meindl U (1993) Micrasterias cells as a model system for research on morphogenesis. Microbiol Rev 57:415–433. https://doi.org/10.1128/mr.57.2.415-433.1993
Morabito G, Oggioni A, Caravati E, Panzani P (2007) Seasonal morphological plasticity of phytoplankton in Lago Maggiore (N. Italy). Hydrobiologia 578:47–57. https://doi.org/10.1007/s10750-006-0432-5
Mpho M, Callaghan A, Holloway GJ (2002) Temperature and genotypic effects on life history and fluctuating asymmetry in a field strain of Culex pipiens. Heredity 88:307–312. https://doi.org/10.1038/sj.hdy.6800045
Naselli-Flores L, Padisák J (2023) Analysis of morphological traits as a tool to identify the realized niche of phytoplankton populations: what do the shape of planktic microalgae, Anna Karenina and Vincent van Gogh have in common? Hydrobiologia. https://doi.org/10.1007/s10750-023-05195-6
Neustupa J (2016) Static allometry of unicellular green algae: scaling of cellular surface area and volume in the genus Micrasterias (Desmidiales). J Evol Biol 29:292–305. https://doi.org/10.1111/jeb.12781
Neustupa J (2017) Asymmetry and integration of cellular morphology in Micrasterias compereana. BMC Evol Biol 17:1. https://doi.org/10.1186/s12862-016-0855-1
Neustupa J, Stastny J (2018) Symmetry breaking of the cellular lobes closely relates to phylogenetic structure within green microalgae of the Micrasterias lineage (Zygnematophyceae). PeerJ 6:e6098. https://doi.org/10.7717/peerj.6098
Neustupa J, Stastny J, Hodac L (2008) Temperature-related phenotypic plasticity in the green microalga Micrasterias rotata. Aquat Microb Ecol 51:77–86. https://doi.org/10.3354/ame01184
Neustupa J, Stastny J, Woodard K (2023) Ecological monitoring of disturbed mountain peatlands: an analysis based on desmids. Biodiv Conserv 32:2671–2691. https://doi.org/10.1007/s10531-023-02624-9
Neustupa J, Woodard K (2023) Supplementary data to "The effects of temperature on plasticity, shape symmetry and seasonal variation in freshwater benthic green microalga Micrasterias thomasiana" Zenodo. https://doi.org/10.5281/zenodo.8189080
Nishizaki MT, Barron S, Carew E (2015) Thermal stress increases fluctuating asymmetry in marine mussels: environmental variation and developmental instability. Ecosphere 6:85. https://doi.org/10.1890/ES14-00399.1
Portmann A (1960) Neue Wege der Biologie. R. Piper & Co Verl, München
Portmann A (2021) New fronts of biological work. In: Jaroš F, Klouda J (eds) Adolf Portmann—a thinker of self-expressive life. Springer, Cham, pp 13–21
Ralfs J (1848) British desmidieae. Reeve, Benham, and Reeve, Wheldon & Wesleyss London
R Core Team (2021) A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.r-project.org. Accessed 24 July 2023
Rohlf FJ (2015) The tps series of software. Hystrix Ital J Mammal 26:9–12. https://doi.org/10.4404/hystrix-26.1-11264
Santos MAD, Ferragut C, Simoes NR, Da Silva DML, Moura CWN (2022) What are the main environmental predictors of diferences in the community structure of periphytic desmids in a semi-arid foodplain lake? Aquat Ecol 56:1037–1053. https://doi.org/10.1007/s10452-022-09957-7
Savriama Y, Neustupa J, Klingenberg CP (2010) Geometric morphometrics of symmetry and allometry in Micrasterias rotata (Zygnematophyceae, Viridiplantae). Nova Hedw Beih 136:43–54. https://doi.org/10.1127/1438-9134/2010/0136-0043
Stamenković M, Hanelt D (2011) Growth and photosynthetic characteristics of several Cosmarium strains (Zygnematophyceae, Streptophyta) isolated from various geographic regions under a constant light-temperature regime. Aquat Ecol 45:455–472. https://doi.org/10.1007/s10452-011-9367-7
Stamenković M, Hanelt D (2017) Geographic distribution and ecophysiological adaptations of desmids (Zygnematophyceae, Streptophyta) in relation to PAR, UV radiation and temperature: a review. Hydrobiologia 787:1–26. https://doi.org/10.1007/s10750-016-2958-5
Škaloud P, Nemjová K, Veselá J, Černá K, Neustupa J (2011) A multilocus phylogeny of the desmid genus Micrasterias (Streptophyta): evidence for the accelerated rate of morphological evolution in protists. Mol Phylog Evol 61:933–943. https://doi.org/10.1016/j.ympev.2011.08.018
Tabi A, Garnier A, Pennekamp F (2020) Testing multiple drivers of the temperature–size rule with nonlinear temperature increase. Funct Ecol 34:2503–2512. https://doi.org/10.1111/1365-2435.13676
Tanneberger F, Tegetmeyer C, Busse S et al (2017) The peatland map of Europe. Mires Peat 19(22):1–17. https://doi.org/10.19189/MaP.2016.OMB.264
Weiss D, Lütz C, Lütz-Meindl U (1999) Photosynthesis and heat response of the green alga Micrasterias denticulata (Desmidiaceae). Z Naturforsch 54:508–516. https://doi.org/10.1515/znc-1999-7-809
Weiss D, Lütz-Meindl U (1999) Heat response in the green alga Micrasterias denticulata (Desmidiaceae): immunodetection and localization of BiP and heat shock protein 70. Nova Hedw 69:217–228. https://doi.org/10.1127/nova.hedwigia/69/1999/217
West-Eberhard MJ (1989) Phenotypic plasticity and the origins of diversity. Ann Rev Ecol Syst 20:249–278. https://doi.org/10.1146/annurev.es.20.110189.001341
Zelditch ML, Swiderski DL, Sheets DH (2012) Geometric morphometrics for biologists: a primer. Elsevier Acad Press, London
Zohary T, Fishbein T, Shlichter M, Naselli-Flores L (2017) Larger cell or colony size in winter, smaller in summer—a pattern shared by many species of Lake Kinneret phytoplankton. Inland Wat 7:200–209. https://doi.org/10.1080/20442041.2017.1320505
Zohary T, Flaim G, Sommer U (2021) Temperature and the size of freshwater phytoplankton. Hydrobiologia 848:143–155. https://doi.org/10.1007/s10750-020-04246-6
Acknowledgements
The authors thank Yvonne Němcová and Táňa Neustupová for their kind assistance in field sampling and Springer Nature Author Services for English language editing and style corrections.
Funding
The study was funded by the Czech Science Foundation (Project No. 22-20989S).
Author information
Authors and Affiliations
Contributions
Both authors contributed to the study conception and design. The strains were cultured and counted by KW. The natural populations were sampled by JN. Microphotography, data acquisition, geometric morphometrics and statistical analyses were performed by JN. The first draft of the manuscript was written by JN, and both authors commented on the first version of the text. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Consent for publication
Permission for the field sampling was granted by the Regional Authority of Ustecky Region (No. KUUK/073894/21).
Additional information
Handling Editor: Man Xiao.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Neustupa, J., Woodard, K. The effects of temperature on plasticity, shape symmetry and seasonal variation in the freshwater benthic green microalga Micrasterias thomasiana. Aquat Ecol (2024). https://doi.org/10.1007/s10452-024-10093-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10452-024-10093-7