Abstract
A major threat to biodiversity is illegal trade, with many unwanted wildlife pets released into exotic environments outside their native distribution. Therefore, many potential invasive species have established in new ecosystems. Ecological niche modeling (ENM) has been used to predict and compare the environmental conditions of natural and exotic population in many groups. We used ENM to compare the climatic niche between natural and exotic areas of Trachemys venusta callirostris, one of the most traded turtles in Colombia. We generated a niche model using the MaxEnt algorithm through the R package kuenm to test several parametrizations and four sets of fresh water environmental predictors. Models were calibrated in the native distribution and projected to non-native zones in Colombia to identify suitable areas for the species. Further, we use a niche similarity test to compare native and exotic environmental space. We found few suitable areas within the projected zone even when using extrapolation; there was a greater suitability in the Magdalena River basin than in the Cauca River basin. Low similarity was detected between the niche comparison of native and exotic areas, suggesting that exotic populations have reached different environmental conditions than the native zone through ilegal trade. Although there was low extrapolation in the exotic area, the models projected ideal conditions in localities with new records for this turtle. The generalist strategies for feeding, thermoregulation, and reproduction in changing conditions may help this T. v. callirostris establish in new ecosystems, and with no current knowledge on dynamics between this exotic species and local fauna, its effects on aquatic communities are unpredictable.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Illegal wildlife trade is a major threat to biodiversity (Zhang et al. 2020; Morton et al. 2021) and is a profitable activity for some countries, especially when the final goal is to use the animals as pets (Yi-Ming et al. 2000; Zimmerman 2003). Although national and international regulations have been established to protect biodiversity, measures are insufficient to control trading (Lee et al. 2005; Spangenberg 2007). In many cases, wildlife buyers cannot keep the acquired animals and end up releasing them in places outside their native range. As a result, many potential exotic and invasive species have successfully established in new locations around the world (Johnson and Padilla 1996; Sakai et al. 2001; Rödder et al. 2009; Banha et al. 2017). The presence of a new species in an ecosystem influences the direct and indirect competition for resources with native species (Sakai et al. 2001; Brown et al. 2002; Braks et al. 2004). Therefore, detecting potential areas out of a species´ known native distribution is a main objective for ecosystems conservation (Sakai et al. 2001; Allendorf and Lundquist 2003; Mehta et al. 2007).
Ecological niche modeling (ENM) has been widely used to estimate the potential ranges of invasive or exotic species in new localities (Rödder et al. 2009; Rodrigues et al. 2016; Khosravi et al. 2019; Liu et al. 2019). Further, niche analysis allows us to understand potential species response to new environments by comparing native and invasive populations (Rödder et al. 2009; Broennimann et al. 2012; Fernández and Hamilton 2015; Di Cola et al. 2017). These tools provide valuable information to promote ecological and social strategies to mitigate or control the invasion of alien species (Peterson and Vieglais 2001; Clout and Williams 2009; Rodrigues et al. 2016).
Trachemys venusta callirostris (sensu Fritz et al. 2012 and Vargas‐Ramírez et al. 2017; Fig. 1) is a Colombian endemic turtle, currently classified as vulnerable (VU) due to hunting, habitat loss, and trafficking (Páez et al. 2022). This turtle occurs in diverse slow water bodies, mostly under 500 masl (Rueda-Almonacid et al. 2007; Bock et al. 2012; Restrepo et al. 2014). The natural distribution of T. v. callirostris in Colombia is known in the Caribbean and Magdalena basins (Bock et al. 2010, 2012; Fig. 2). However, new localities have been recorded in the interior of the country (i.e., Caldas and Quindío departments; Jaramillo and Cortés 2016; Adames-Jiménez et al. 2018). In addition to this distribution, established populations have been found (reproductive adults and neonates) in western Antioquia (Fig. 4), specifically in the municipalities of Sopetrán (6°29′17.55'' N, 75°47′15.47" W) and Santa Fe de Antioquia (6°30′39.43" N, 75°49′18.21" W). Populations were found in rivers, ponds, and artificial lakes in the Cauca River Basin, at least 230 km from the closest known record (Bock et al. 2012; Fig. 2).
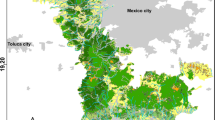
Results of potential distribution areas (blue pixels) of T. v. callirostris in the accessibility area or M (blue polygon) corresponding to its natural distribution zone, and projection area (red polygon). Zoomed in maps of a Bolivar-Atlántico, b Cesar-Magdalena, c Sucre, and d Eastern Antioquia populations. Black points represent species occurrences in its natural area used for niche modeling
It is suspected that new locations of this slider turtle in Colombia are due to the release of unwanted pets in natural areas (Adames-Jiménez et al. 2018). Further, T. v. callirostris is the most traded turtle in the country, accounting for more than 50% of the seizures by Regional Environmental Corporations RECs (Bonilla et al. 2012; Arroyave et al. 2014; Bock et al. 2015; Suárez Giorgi 2017). Therefore, this turtle occurs in new suitable regions where its population may increase competition for resources with other species (Sakai et al. 2001; Ficetola et al. 2009; Díaz-Paniagua et al. 2011). This species could shift from exotic to a potential invasive species in some regions as has occurred with other traded slider turtles (Ficetola et al. 2009; Rödder et al. 2009; Banha et al. 2017). In this study we tested if the species illegal trade allows to reach new environment al conditions in exotic areas using one of the most traded turtles in Colombia, Trachemys venusta callirostris, as a study model. We expected that the climatic conditions of introduced populations of T. v callirostris would differ statistically from the native niche using an ENM approach and we identified potential suitable regions for establishment of the turtle.
Methods
Occurrence and environmental data
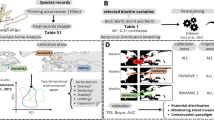
Occurrences of T. v. callirostris were obtained through the Global Biodiversity Information Facility database (GBIF 2021) and personal field observations. We split the records of the species into native and exotic areas based on previous systematic, taxonomic, and distribution studies (see Fritz et al. 2012; Vargas-Ramírez et al. 2017; Páez et al. 2022). Then, we cleaned the data based on Cobos et al. (2018), removing duplicated occurrences, then applying a spatial thin of one kilometer to have only one record in each raster pixel, and deleted potential identification results based on previous taxonomic revisions (e.g., Páez et al. 2022). Finally, we move those occurrences out the pixels calculating the Euclidean distance of each record to the surrounding pixels, then moving manually to the closest pixel, using Arcgis (ESRI 2022).
Because T. v. callirostris is restricted to freshwater habitats we obtained environmental variables from Domisch database (Domisch et al. 2015) with a spatial resolution of 30 s. These variables are more related to the ecology of freshwater species and present relevant implications within model construction and niche comparison in fresh water ecosystems (see Nori and Rojas-Soto 2019). For selection of climatic variables, we first created the accessibility area (M; Soberón and Peterson 2005) for the native distribution and a projection zone that represents the introduced populations. Both areas were estimated using level 6 HydroShed basins (Lehner et al. 2021). Then, we performed a Pearson correlation test using the variables from the native distribution area and removed those with correlation coefficients below −0.8 and above 0.8 (Echeverry-Cárdenas et al. 2021). However, because in some cases multiple highly correlated variables can be important for the species, we created four environmental sets to test during the niche modeling process (Table 2 in Supplementary material).
Ecological niche modeling
We used native occurrences (80% for training and 20% for model evaluation of each one) to model the ecological niche using the Maxent algorithm (Elith et al. 2011) in the kuenm R package (Cobos et al. 2019) and projected each model in geography for the native and exotic area using all extrapolation types to compare the results. The parametrization of the model did not include “hinge” and “threshold” features because these can lead to a complex function response (Elith et al. 2011). Further, we tested different levels of complexity for model construction as Warren and Seifert (2011) suggested, shifting the regularization multiplier values from 0.1 to 1 at 0.1 intervals and then 2, 3, 4, and 5. We selected the best model for both native and exotic based on the evaluation metrics performed in Kuenm (AUC ratio, Omission rate, and AIC) and used the ten-percentile threshold to transform the cloglog Maxent’s output into presence/absence maps. If several models fulfilled the evaluation criteria, we estimated a final model from each climatic set using the median of the replicates obtained for the native and exotic zones for all extrapolation methods. Finally, we applied the Mobility-Oriented Parity test (MOP; Owens et al. 2013) for the models with clamping extrapolation, which represent areas of potential extrapolation risk.
Niche similarity test
To test if T. v. callirostris could be reaching new environments in the exotic area, we performed a niche similarity test using the ecospat R package (Broennimann et al. 2012; Di cola et al. 2017). This test summarizes the environmental predictors in two principal components and perform a kernel density function for native and exotic occurrences. Then estimates the overlap or similarity between both (native and exotic population) when the native niche is randomly introduced in the exotic area, then a high environmental similarity value trend to one while low similarity to zero (Broennimann et al. 2012). If the observed value (obtained by comparing climates from populations from both zones) falls within the null models, then the differences are just explained by chance, because environments of the exotic zone are too different from the native one (Di cola et al. 2017). We performed niche similarity test between both areas for each environmental data set selected by kuenm evaluation metrics to obtain the best models and their corresponding potential distribution.
Results
We obtained 60 occurrences of the species for the native distribution and 32 for the exotic localities (Table 1 in Supplementary material), of which four presented evidence of reproduction (Fig. 4). We obtained four ecological niche models with different parametrization that fulfilled the evaluation criteria from kuenm (Table 3 in Supplementary material). From these four models, two used the set three and two the set four of environmental predictors. Both consensus models (using the two environmental dataset predictors) showed slight differences in the number of predicted pixels (as presence) and set four had more pixels in native and exotic zones even when using different extrapolation methods (Fig. 4). Predictions of potential distribution in the native area showed the expected presence in the geographical basins of Caribe and Magdalena where most of the occurrences have been recorded (Bock et al. 2010, 2012; Fig. 2).
The projection of the model in the exotic area showed few potential presence zones even using free extrapolation and clamping. We found a similar number of predicted pixels in the exotic zone using both predictors’ data sets when no extrapolation was performed. However, by applying clamping or free extrapolation, we found that set 4 predicted more pixels than set 3 (Fig. 3). Our models predicted suitable areas in the nearest zones of the Magdalena Basin, near urban centers in Caldas and Tolima. However, we identified few suitable areas in the Cauca basin where two new localities have recently been found with evidence of reproduction. Further, important locations such as Bogotá (Cundinamarca) and Cali (Valle del Cauca) where adults have been detected, but no juveniles, presented few potential zones (Fig. 4).
Results of the suitable areas (color lines) for establishment of the turtle in the exotic area according to A = Set 3 and B = Set 4. Gray dots = no reproduction records; green star = reproduction records. Predicted pixels are represented by CE: only from clamping extrapolation; E: only from free extrapolation; NE + EC: common pixels between No Extrapolation and Clamping; E + NE: common pixels between No Extrapolation and free extrapolation; E + CE: common pixels from clamping and free extrapolation; E + NE + CE: common pixels predicted using all extrapolation types. Shaded areas with light purple = Cauca basin, with light brown = Magdalena basin
MOP results showed a greater similarity (~1) among areas of occurrence in localities of Caldas, Cundinamarca, Quindío, and Valle del Cauca, but a low similarity of extrapolation areas occurred in localities of western Antioquia (Fig. 1 in supplementary material).
The niche similarity tests (Fig. 5) using both sets of environmental predictors suggest a great variability in the environmental space inhabited by native and exotic populations (D = 0.01; I = 0.03 for both environmental predictor sets). Further, both results were not statistically significant (p-value > 0.05). However, niches similarity is low, and the difference can be explained by random chance (null models) following the environments situated between native and invasive areas.
Niche similarity test between natural and exotic population of T. v. callirostris for set 3 (a and c) and set 4 (b and d). A and B represents the native and exotic environmental space is represented in gray and red, respectively, while the environmental overlap is represented in blue. Dashed and solid lines correspond to the 50 and 100%, respectively, of the environmental space within native and exotic areas. C and D, shows the significance test for similarity test for both set predictors
Discussion
The reduced projected suitable conditions in the exotic area can be explained by the low similarity between native and exotic distribution niches (Fig. 4a–c), showing a potential niche shifting process between T. v. callirostris native and exotic populations. Niche differences may be highly associated with the establishment of populations at upper elevations (1300–2500 masl) in Quindío and Bogotá (Adames-Jiménez et al. 2018) where lower mean temperature zones occur than those found in the native distribution below ~ 500 masl (Rueda-Almonacid et al. 2007; Bock et al. 2012). This suggest that T. v. callirostris presents a wider fundamental niche (physiological tolerances) than the environmental space occupied in its native distribution area, allowing the species to reach different climatic conditions in the exotic area. These findings have also been reported for other invasive species like Trachemys scripta (e.g. Espindola et al. 2019). Then, it is possible that some behaviors in T. v. callirostris individuals, e.g., longer time performing aerial basking for thermoregulation, are used for establishment in relatively higher latitudes and altitudes, as has been well documented in the invasions of the Emydid turtle Trachemys scripta (Polo-Cavia et al. 2012, 2014; Lambert et al. 2013; Zhang et al. 2020).
Between the years 2005 and 2009 T. v. callirostris was the most traded turtle (Bermudez et al. 2014) and is one of the most confiscated reptiles by regional environmental corporations RECs (Suárez 2017; Castro et al. 2022). With a higher trade between different Colombian regions, there is a greater chance to inhabit new localities as found in the central area of the country (exotic area), which occurs by intentional release as unwanted pets in natural areas (Adames-Jiménez et al. 2018). However, for the establishment of populations in new areas as recorded in this study in the departments of Valle del Cauca and mainly in colder environments such as Bogota (Cundinamarca department), ecological and physiological requirements must be considered (Allendorf, and Lundquist 2003; Spear 2018). For example, several studies with the invasive Emydid turtle Trachemys scripta elegans have identified that the generalist strategies (e.g., a wide diet and high tolerance to organic accumulation process in water bodies) of this species allow it an advantage in a new environment among other turtle species (Mauermann 1995; Ficetola et al. 2009; Díaz-Paniagua et al. 2011; Polo-Cavia et al. 2014; Spear 2018). Similar traits have been reported for T. v. callirostris like varied diet (consuming algae, seeds, flowers, fishes, mollusks, and arthropods) and the capability of reproducing and nesting in highly degraded habitats (De Vivero and De La Ossa 2018; Restrepo et al. 2006; Leguízamo-Pardo and Bonilla Gomez 2014). These generalist habits represent the potential of the species to respond to changing environmental conditions.
Our results showed the impact of illegal trade on the distribution of T. v. callirostris populations, showing that even in the same country an adjacent exotic area environmental differences can occur in short distances or scales. With no knowledge about the ecological dynamics of T. v. callirostris in the exotic area, e.g., interspecific competition, rates of survival, and population density, it is unclear if the turtle is behaving as an invasive species. Indeed, our results show the potential of the reptile to establish in exotic environments, and there is a need to determine whether its presence affects biotic and abiotic conditions of waterbodies. Further, future analysis from a niche modeling perspective and conservation management would help to understand which environmental conditions allows the establishment success of exotic populations.
We emphasize the need of relating the information of confiscations and relocations of individuals with all the RECs of Colombia, with special efforts in monitoring the new recorded localities from this study in the departments of Valle del Cauca and Cundinamarca. These departments are areas where adults have been inhabiting natural areas but no reproduction has been identified yet. There is also a need to improve management strategies and determine routes of traffic and potential new areas for establishment of the turtle. Further, monitoring plans to estimate the viability of the exotic populations is needed, as well as new research adding climate change scenarios to predict suitable areas in future.
References
Adames-Jiménez TA, Rivera-Robles DL, Arango-Lozano J, Botero-Botero Á (2018) New records of the turtle Trachemys venusta callirostris in the Central Andes of Colombia. Herpetol Bull 144:28–29
Allendorf FW, Lundquist LL (2003) Introduction: population biology, evolution, and control of invasive species. Conserv Biol, pp 24–30.
Arroyave FJ, Goyeneche OYR, Gómez MAB, Heredia RGH (2014) Tráfico ilegal de tortugas continentales (Testudinata) en Colombia: Una aproximación desde el análisis de redes. Acta Biol Colomb 19:381–392
Banha F, Gama M, Anastácio PM (2017) The effect of reproductive occurrences and human descriptors on invasive pet distribution modeling: Trachemys scripta elegans in the Iberian Peninsula. Ecol Modell 360:45–52
Bermudez A, Jair F, Romero Goyeneche OY, Bonilla Gómez MA, Hurtado Heredia RG (2014) Tráfico ilegal de tortugas continentales (Testudinata) en Colombia: una aproximación desde el análisis de redes. Acta Biol Colomb 19:381–391
Bock BC, Páez VP, Daza JM (2010) Trachemys callirostris (Gray, 1856) Colombian Slider, Jicotea, Hicotea, Galápago, Morrocoy de agua. In: Chelon Res Monog 5: 042.1–042.9.
Bock BC, Páez VP, Daza JM (2012) Trachemys callirostris. In: Bock VP, Morales-Betancourt MA, Lasso CA, Castaño-Mora OV, Bock BC (eds) Biología y Conservación de las Tortugas Continentales de Colombia. Serie Editorial Recursos Hidrobiológicos y Pesqueros Continentales de Colombia. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá, pp 283–291
Bock BC, Páez VP, Cortés-Duque J (2015) Trachemys callirostris Gray 18568. In: Morales-Betancourt MA, Lasso CA, Páez VP, Bock BC (eds) Libro rojo de reptiles de Colombia (2015) Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH), Universidad de Antioquia. Bogotá, D. C, Colombia, pp. 166–171.
Bonilla MA, Luque N, Cuervo MA, Pinzón M, Vásquez EA (2012) Tortugas Terrestres y de Agua Dulce de Colombia y Manejo de Decomisos. Colombia Universidad Nacional de Colombia, Bogotá
Braks MAH, Honório NA, Lounibos LP, Lourenço-de-Oliveira R, Juliano SA (2004) Interspecific competition between two invasive species of container mosquitoes, Aedes aegypti and Aedes albopictus (Diptera: Culicidae), in Brazil. Ann Entomol Soc Am 97:130–139
Broennimann O, Fitzpatrick MC, Pearman PB, Petitpierre B, Pellissier L, Yoccoz NG, Guisan A (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Glob Ecol and Biogeogr 21:481–497
Brown BJ, Mitchell RJ, Graham SA (2002) Competition for pollination between an invasive species (purple loosestrife) and a native congener. Ecology 83:2328–2336
Castro CA, Brieva C, Witte C (2022) Implications of wildlife trafficking on the health and conservation efforts of an endangered turtle species in Colombia. Conserv Sci Pract 4:e595
Clout MN, Williams PA (2009) Invasive species management: a handbook of principles and techniques. Oxford University Press, UK
Cobos ME, Jiménez L, Nuñez-Penichet C, Romero-Alvarez D, Simões M (2018) Sample data and training modules for cleaning biodiversity information. Biodivers Inform 13:49–50
Cobos ME, Peterson AT, Barve N, Osorio-Olvera L (2019) kuenm: an R package for detailed development of ecological niche models using Maxent. Peer J 7:e6281
De Vivero MM, De La Ossa J (2018) Feeding Habits of Trachemys callirostris (Gray, 1856) (Testudines: Emydidae), San Benito Abad, Sucre. Colombia Indian J Sci Technol 11:46
Díaz-Paniagua C, Pérez-Santigosa N, Hidalgo-Vila J, Florencio M (2011) Does the exotic invader turtle, Trachemys scripta elegans, compete for food with coexisting native turtles? Amphib-Reptil 32:167–175
Di Cola V, Broennimann O, Petitpierre B, Breiner FT, d’Amen M, Randin C, Guisan A (2017) ecospat: an R package to support spatial analyses and modeling of species niches and distributions. Ecography 40:774–787
Domisch S, Amatulli G, Jetz W (2015) Near-global freshwater-specific environmental variables for biodiversity analyses in 1 km resolution. Sci Data 2:150073
Echeverry-Cárdenas E, López-Castañeda C, Carvajal-Castro JD, Aguirre-Obando OA (2021) Potential geographic distribution of the tiger mosquito Aedes albopictus (Skuse, 1894) (Diptera: Culicidae) in current and future conditions for Colombia. PLOS Negl Trop Dis 15:e0008212
Elith J, Phillips SJ, Dudík HTM, Chee YE, Yates CJ (2011) A statistical explanation of MaxEnt for ecologists. Divers Dist 17:43–57
Espindola S, Parra JL, Vázquez-Domínguez E (2019) Fundamental niche unfilling and potential invasion risk of the slider turtle Trachemys scripta. PeerJ 7:e7923. https://doi.org/10.7717/peerj.7923
ESRI. Environmental Systems Research Institute (2022) ArcGIS [software GIS]. Version 10.8. Redlands, California
Fernández M, Hamilton H (2015) Ecological niche transferability using invasive species as a case study. PLoS ONE 10:e0119891
Ficetola GF, Thuiller W, Padoa-Schioppa E (2009) From introduction to the establishment of alien species: bioclimatic differences between presence and reproduction localities in the slider turtle. Diver Distrib 15:108–116
Fritz U, Stuckas H, Vargas-Ramírez M, Hundsdörfer AK, Maran J, Päckert M (2012) Molecular phylogeny of Central and South American slider turtles: implications for biogeography and systematics (Testudines: Emydidae: Trachemys). J Zoolog Syst Evol 50:125–136
GBIF (20210 Occurrences of Trachemys callirostris Gray, 1855. GBIF. 2021 https://doi.org/10.15468/dl.nhsjvk. Accessed 22 may 2022.
Jaramillo LÁF, Cortés EB (2016) Diversity of reptiles asociated with three contrasting areas in a Tropical Dry Forest (La Dorada and Victoria, Caldas). Revista De Ciencias 20:109–123
Johnson LE, Padilla DK (1996) Geographic spread of exotic species: ecological lessons and opportunities from the invasion of the zebra mussel Dreissena polymorpha. Biol Conserv 78:23–33
Khosravi R, Hemami MR, Cushman SA (2019) Multi-scale niche modeling of three sympatric felids of conservation importance in central Iran. Landsc Ecol 34:2451–2467
Lambert MR, Nielsen SN, Wright AN, Thomson RC, Shaffer HB (2013) Habitat features determine the basking distribution of introduced red-eared sliders and native western pond turtles. Chelonian Conserv Biol 12:192–199
Lee RJ, Gorog AJ, Dwiyahreni A, Siwu S, Riley J, Alexander H, Ramono W (2005) Wildlife trade and implications for law enforcement in Indonesia: a case study from North Sulawesi. Biol Conserv 123:477–488
Lehner B, Roth A, Huber M, Anand M, Grill G, Osterkamp N, Thieme M (2021) HydroSHEDS v2. 0-Refined global river network and catchment delineations from TanDEM-X elevation data. In EGU General Assembly Conference Abstracts, pp EGU21–9277.
Leguízamo-Pardo C, Bonilla Gomez MA (2014) Reproduction of Trachemys callirostris callirostris (Emydidae) in environments created by mining in La Guajira, Colombia. Acta Biol Colomb 19:363–380
Liu B, Gao X, Ma J, Jiao Z, Xiao J, Hayat MA, Wang H (2019) Modeling the present and future distribution of arbovirus vectors Aedes aegypti and Aedes albopictus under climate change scenarios in Mainland China. Sci Total Environ 664:203–214
Mauermann KJ (1995) Feeding and habitat preferences of the red-eared slider, Trachemys scripta elegans Wied. Doctoral dissertation, Texas Woman's University.
Mehta SV, Haight RG, Homans FR, Polasky S, Venette RC (2007) Optimal detection and control strategies for invasive species management. Ecol Econ 61:237–245
Morton O, Scheffers BR, Haugaasen T, Edwards DP (2021) Impacts of wildlife trade on terrestrial biodiversity. Nat Ecol Evol 5:540–548
Nori J, Rojas-Soto O (2019) On the environmental background of aquatic organisms for ecological niche modeling: a call for caution. Aquat Ecol 53:595–605
Owens HL, Campbell LP, Dornak LL, Saupe EE, Barve N, Soberón J, Peterson AT (2013) Constraints on interpretation of ecological niche models by limited environmental ranges on calibration areas. Ecol Modell 263:10–18
Páez VP, Bock BC, Alzate-Estrada DA, Barrientos-Muñoz KG, Cartagena-Otálvaro VM, Echeverry-Alcendra A, Vallejo-Betancur MM (2022) Turtles of Colombia: an annotated analysis of their diversity, distribution, and conservation status. Amphib Reptile Conserv 16:106–135
Peterson AT, Vieglais DA (2001) Predicting species invasions using ecological niche modeling: new approaches from bioinformatics attack a pressing problem: a new approach to ecological niche modeling, based on new tools drawn from biodiversity informatics, is applied to the challenge of predicting potential species’ invasions. Bioscience 51:363–371
Polo-Cavia N, López P, Martín J (2012) Feeding status and basking requirements of freshwater turtles in an invasion context. Physiol Behav 105:1208–1213
Polo-Cavia N, López P, Martín J (2014) Interference competition between native Iberian turtles and the exotic Trachemys scripta. Basic Appl Herpetol 28:5–20
Restrepo A, Piñeros VJ, Páez VP (2006) Nest site selection by Colombian slider turtles, Trachemys callirostris callirostris (Testudines: Emydidae), in the Mompos Depression, Colombia. Chelonian Conserv Biol 5:249–254
Restrepo A, Páez V, Bock BC, Daza JM, Díaz- Granados I (2014) Trachemys callirostris (Gray, 1856). Catálogo De Anfibios y Reptiles De Colombia 2:7–12
Rödder D, Schmidtlein S, Veith M, Lötters S (2009) Alien invasive slider turtle in unpredicted habitat: a matter of niche shift or of predictors studied? PLoS ONE 4:e7843
Rodrigues JF, Coelho MT, Varela S, Diniz-Filho JA (2016) Invasion risk of the pond slider turtle is underestimated when niche expansion occurs. Freshw Biol 61:1119–1127
Rueda-Almonacid JV, Carr JL, Mittermeier RA, Rodríguez-Mahecha JV, Mast RB, Vogt RC, Mittermeier CG (2007) Las tortugas y los cocodrilianos de los países andinos del trópico. Serie de guías tropicales de campo. Editorial Panamericana Bogota, Colombia.
Sakai AK, Allendorf FW, Holt JS, Lodge DM, Molofsky J, With KA, Weller SG (2001) The population biology of invasive species. Annu Rev Ecol Evol Syst 32:305–332
Soberón J, Peterson AT (2005) Interpretation of models of fundamental ecological niches and species’ distributional areas. Biodiversity Informatics 2:1–10
Spangenberg JH (2007) Biodiversity pressure and the driving forces behind. Ecol Econom 61:146–158
Spear MJ (2018) Current and projected distribution of the Red-eared Slider Turtle, Trachemys scripta elegans, in the Great Lakes Basin. Am Midl Nat 179:191–221
Suárez Giorgi CA (2017) Diagnóstico del Tráfico Ilegal de Fauna Silvestre en Jurisdicción de la Corporación Autónoma Regional de Cundinamarca–CAR, Departamentos de Cundinamarca y Boyacá. Tesis de grado, Universidad Militar Nueva Granada, Bogotá, Colombia, Colombia
Vargas-Ramírez M, del Valle C, Ceballos CP, Fritz U (2017) Trachemys medemi n. sp. from northwestern Colombia turns the biogeography of South American slider turtles upside down. J Zool Syst Evol 55:326–339
Warren DL, Seifert SN (2011) Ecological niche modeling in Maxent: the importance of model complexity and the performance of model selection criteria. Ecol Appl 21:335–342
Yi-Ming L, Zenxiang G, Xinhai L, Sung W, Niemelä J (2000) Illegal wildlife trade in the Himalayan region of China. Biodiver Conserv 9:901–918
Zhang Y, Song T, Jin Q, Huang Y, Tang X, Sun X, Bao W (2020) Status of an alien turtle in city park waters and its potential threats to local biodiversity: the red-eared slider in Beijing. Urban Ecosyst 23:147–157
Zimmerman ME (2003) The black market for wildlife: combating transnational organized crime in the illegal wildlife trade. Vand J Transnat’l L 36:1657
Acknowledgments
We thank Michael Roger Piratova and Karime Angarita Corzo for their help with methodology and formatting changes of previous versions of this manuscript.
Funding
Open Access funding provided by Colombia Consortium.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design in equivalent efforts as data collection, modeling and writing.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest are presented in this manuscript. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Data availability
All data generated or analyzed during this study are included in this article [and its supplementary information files].
Additional information
Handling Editor: Télesphore Sime-Ngando.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Julián, AL., Dahian, PS. & Felipe, TC. Reaching new environments through illegal trade: evidence of a widely traded turtle in Colombia. Aquat Ecol 57, 471–480 (2023). https://doi.org/10.1007/s10452-023-10023-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-023-10023-z