Abstract
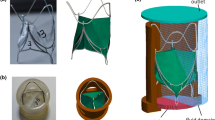
Finite element (FE) modelling can be a very resourceful tool in the field of cardiovascular devices. To ensure result reliability, FE models must be validated experimentally against physical data. Their clinical application (e.g., patients’ suitability, morphological evaluation) also requires fast simulation process and access to results, while engineering applications need highly accurate results. This study shows how FE models with different mesh discretisations can suit clinical and engineering requirements for studying a novel device designed for percutaneous valve implantation. Following sensitivity analysis and experimental characterisation of the materials, the stent-graft was first studied in a simplified geometry (i.e., compliant cylinder) and validated against in vitro data, and then in a patient-specific implantation site (i.e., distensible right ventricular outflow tract). Different meshing strategies using solid, beam and shell elements were tested. Results showed excellent agreement between computational and experimental data in the simplified implantation site. Beam elements were found to be convenient for clinical applications, providing reliable results in less than one hour in a patient-specific anatomical model. Solid elements remain the FE choice for engineering applications, albeit more computationally expensive (>100 times). This work also showed how information on device mechanical behaviour differs when acquired in a simplified model as opposed to a patient-specific model.






Similar content being viewed by others
References
Auricchio, F., and R. L. Taylor. Shape-memory alloys: modelling and numerical simulations of the finite-strain superelastic behaviour. Comput. Method. Appl. Mech. 143(1–2):175–194, 1997.
Babuska, I., and J. T. Oden. Verification and validation in computational engineering and science: basic concepts. Comput. Method. Appl. Methods 193:4057–4066, 2004.
Biglino, G., S. Schievano, V. Muthurangu, and A. M. Taylor. Cardiovascular modelling. In: Clinical Cardiac MRI-II, edited by J. Bogaart, S. Dymarowski, V. Muthurangu, and A. M. Taylor. Berlin: Springer-Verlag, 2012, pp. 669–694.
Biglino, G., C. Capelli, A. Binazzi, R. Reggiani, D. Cosentino, F. Migliavacca, P. Bonhoeffer, A. M. Taylor, and S. Schievano. Virtual and real bench testing: case study of a new percutaneous valve device. Eurointervention 8:120–128, 2012.
Biglino, G., P. Verschueren, R. Zegels, A. M. Taylor, and S. Schievano. Quantification of TangoPlus FullCure® 930 compliance for printing patient-specific vascular models. ASAIO J. 57:74, 2011.
Bonhoeffer, P. Are animal experiments the crux for decision making in whether new heart valves can be brought to clinical practice? EuroIntervention. 5:643–645, 2010.
Bonhoeffer, P., Y. Boudjemline, Z. Saliba, J. Merckx, Y. Aggoun, D. Bonnet, P. Acar, J. Le Bidois, D. Sidi, and J. Kachaner. Percutaneous replacement of pulmonary valve in a right-ventricle to pulmonary-artery prosthetic conduit with valve dysfunction. Lancet 356:1403–1405, 2000.
Bonhoeffer, P., R. Huynh, M. House, N. Douk, M. Kopcak, A. Hill, and N. Rafiee. Transcatheter pulmonic valve replacement in sheep using a grafted self-expanding stent with tissue valve. Circulation 118:S812, 2008.
Capelli, C., A. M. Taylor, F. Migliavacca, P. Bonhoeffer, and S. Schievano. Patient-specific reconstructed anatomies and computer simulations are fundamental for selecting medical device treatment: application to a new percutaneous pulmonary valve. Philos. Transact. A Math. Phys. Eng. Sci. 368:3027–3038, 2010.
Castelain, V., P. Hervé, Y. Lecarpentier, P. Duroux, G. Simonneau, and D. Chemla. Pulmonary artery pulse pressure and wave reflection in chronic pulmonary thromboembolism and primary pulmonary hypertension. J. Am. Coll. Cardiol. 37(4):1085–1092, 2001.
Cebral, J. R., M. A. Castro, J. E. Burgess, R. S. Pergolizzi, M. J. Sheridan, and C. M. Putman. Characterization of cerebral aneurysms for assessing risk of rupture by using patient-specific computational hemodynamics models. Am. J. Neuroradiol. 26(10):2550–2559, 2005.
Cebral, J. R., P. J. Yim, R. Löhner, O. Soto, and P. L. Choyke. Blood flow modeling in carotid arteries with computational fluid dynamics and MR imaging. Acad. Radiol. 9(11):1286–1299, 2002.
Condado, J. A., and M. Vélez-Gimón. Catheter-based approach to mitral regurgitation. J. Interv. Cardiol. 16(6):523–534, 2003.
Cribier, A., H. Eltchaninoff, A. Bash, N. Borenstein, C. Tron, F. Bauer, G. Derumeaux, F. Anselme, F. Laborde, and M. B. Leon. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis—first human case description. Circulation 106:3006–3008, 2002.
EN ISO 5840-03. Cardiovascular implants. Cardiac valve prostheses. Part 3: Heart valve substitutes implanted by minimally invasive techniques, 2010.
Gan, C. T. J., J. W. Lankhaar, N. Westerhof, J. T. Marcus, A. Becker, J. W. R. Twisk, A. Boonstra, P. E. Postmus, and A. Vonk-Noordergaaf. Noninvasively assessed pulmonary artery stiffness predicts mortality in pulmonary arterial hypertension. Chest. 132:1906–1912, 2007.
Grujicic, M., B. Pandurangan, A. Arakere, and J. S. Snipes. Fatigue-life computational analysis for the self-expanding endovascular nitinol stents. J. Mater. Eng. Perform. 21(1):1–13, 2012.
Guidance for industry and FDA staff. Non-clinical engineering tests and recommended labeling for intravascular stents and associated delivery systems. Document issued on April 18, 2010.
Hall, G. J., and E. P. Kasper. Comparison of element technologies for modeling stent expansion. J. Biomech. Eng-T. ASME. 128:751–756, 2006.
Koo, B. K., A. Erglis, J. H. Doh, D. V. Daniels, S. Jegere, H. S. Kim, A. Dunning, T. Defrance, A. Lansky, J. Leipsic, and J. K. Min. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms results from the prospective multicenter discover-flow (diagnosis of ischemia-causing stenoses obtained via noninvasive fractional flow reserve) study. J. Am. Coll. Cardiol. 58(19):1989–1997, 2011.
Lauten, A., M. Ferrari, K. Hekmat, R. Pfeifer, G. Dannberg, A. Ragoschke-Schumm, and H. R. Figulla. Heterotopic transcatheter tricuspid valve implantation: first-in-man application of a novel approach to tricuspid regurgitation. Eur. Heart. J. 32(10):1207–1213, 2011.
Migwi, C. M., M. I. Darby, G. H. Wostenholm, B. Yates, R. Duffy, and M. Moss. A method of determining the shear modulus and Poisson’s ratio of polymer materials. J. Mater. Sci. 29:3430–3432, 1994.
Morlacchi, S., C. Chiastra, D. Gastaldi, G. Pennati, G. Dubini, and F. Migliavacca. Sequential structural and fluid dynamic numerical simulations of a stented bifurcated coronary artery. J. Biomech. Eng. 133(12):121010, 2011.
Morlacchi, S., B. Keller, P. Arcangeli, M. Balzan, F. Migliavacca, G. Dubini, J. Gunn, N. Arnold, A. Narracott, D. Evans, and P. Lawford. Hemodynamics and in-stent restenosis: micro-CT images, histology, and computer simulations. Ann. Biomed. Eng. 39(10):2615–2626, 2011.
Oberkampf, W. L., T. G. Trucano, and C. Hirsch. Verification validation and predictive capability in computational engineering and physics. Appl. Mech. Rev. 57:345–384, 2004.
Pelton, A. R., V. Schroeder, M. R. Mitchell, X. Y. Gong, M. Barney, and S. W. Robertson. Fatigue and durability of nitinol stents. J Mech. Behav. Biomed. Mater. 1(2):153–164, 2008.
Rebelo, N., X. Y. Gong, A. Hall, A. R. Pelton, and W. Duerig. Finite element analysis on the cyclic properties of superelastic nitinol. Proceedings of the International Conference on Shape Memory and Superelastic Technologies, pp. 157–163, 2006.
Schievano, S., F. Migliavacca, L. Coats, S. Khambadkone, M. Carminati, N. Wilson, J. E. Deanfield, P. Bonhoeffer, and A. M. Taylor. Percutaneous pulmonary valve implantation based on rapid prototyping of right ventricular outflow tract and pulmonary trunk from MR data. Radiology 242:490–497, 2007.
Schievano, S., A. M. Taylor, C. Capelli, L. Coats, F. Walker, P. Lurz, J. Nordmeyer, S. Wright, S. Khambadkone, V. Tsang, M. Carminati, and P. Bonhoeffer. First-in-man implantation of a novel percutaneous valve: A new approach to medical device development. Eurointervention. 5:745–750, 2010.
Schievano, S., A. M. Taylor, C. Capelli, P. Lurz, J. Nordmeyer, F. Migliavacca, and P. Bonhoeffer. Patient specific finite element analysis results in more accurate prediction of stent fractures: application to percutaneous pulmonary valve implantation. J. Biomech. 43(4):687–693, 2010.
Schoenhagen, P., A. Hill, T. Kelley, Z. Popovic, and S. S. Halliburton. In vivo imaging and computational analysis of the aortic root. Application in clinical research and design of transcatheter aortic valve systems. J. Cardiovasc. Transl. Res. 4(4):459–469, 2011.
Simulia Abaqus analysis user’s manual, v. 6.10-EF Dassault Systèmes, Vélizy-Villacoublay, France, vol. V:31.4 (2010).
Smith, N., A. de Vecchi, M. McCormick, D. Nordsletten, O. Camara, A. F. Frangi, H. Delingette, M. Sermesant, J. Relan, N. Ayache, N. W. Krueger, W. H. W. Schulze, R. Hose, I. Valverde, P. Beerbaum, C. Staicu, M. Siebes, J. Spaan, P. Hunter, J. Weese, H. Lehmann, D. Chapelle, and R. Rezavi. EuHeart: personalized and integrated cardiac care using patient-specific cardiovascular modelling. Interface Focus. 1:349–364, 2011.
Steinman, D., and C. A. Taylor. Flow imaging and computing: large artery hemodynamics. Ann. Biomed. Eng. 33(12):1704–1709, 2005.
Taylor, C. A., and C. A. Figueroa. Patient-specific modelling of cardiovascular mechanics. Annu. Rev. Biomed. Eng. 11:109–134, 2009.
Viceconti, M., G. Clapworthy, and S. Van Sint Jan. The virtual physiological human—a European initiative for in silico human modelling. J. Physiol. Sci. 58(7):441–446, 2008.
Acknowledgments
We gratefully acknowledge the support of the following funding bodies: Rosetrees Trust, Fondation Leducq, British Heart Foundation, Royal Academy of Engineering/EPSRC and the UK National Institute of Health Research. We also thank the scientists from Medtronic Cardiovascular for their technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Joel D. Stitzel oversaw the review of this article.
Claudio Capelli and Giovanni Biglino contributed equally to this publication.
Rights and permissions
About this article
Cite this article
Capelli, C., Biglino, G., Petrini, L. et al. Finite Element Strategies to Satisfy Clinical and Engineering Requirements in the Field of Percutaneous Valves. Ann Biomed Eng 40, 2663–2673 (2012). https://doi.org/10.1007/s10439-012-0617-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-012-0617-1