Abstract
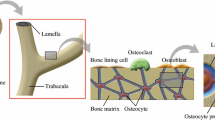
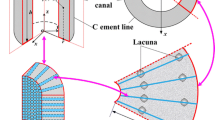
A poroelastic lacunocanalicular model was developed for the quantification of physiologically relevant parameters related to bone fluid flow. The canalicular and lacunar microstructures were explicitly represented by a dual-continuum poroelastic model. Effective material properties were calculated using the theory of composite materials. Porosity and permeability values were determined using capillaric and spherical-shell models for the canalicular and lacunar microstructures, respectively. Pore fluid pressure and fluid shear stress were calculated in response to simulated mechanical loading applied over a range of frequencies. Species transport was simulated with convective and diffusive flow, and osteocyte consumption of nutrients was incorporated. With the calculated parameter values, realistic pore fluid pressure and fluid shear stress responses were predicted and shown to be consistent with previous experimental and theoretical studies. Stress-induced fluid flow was highlighted as a potent means of species transport, and the importance of high-magnitude low-frequency loading on osteocyte nutrition was demonstrated. This new model can serve as the foundation for future hierarchical modeling efforts that may provide insight into the underlying mechanisms of mechanotransduction and functional adaptation of bone.







Similar content being viewed by others
Abbreviations
- B :
- D :
-
Species diffusion coefficient; D g = 1.0 × 10−10 m2 s−1
- E d :
- E s :
-
Young’s modulus of solid phase; E s = 17.51 GPa
- G d :
-
Drained shear modulus; Eq. (4)
- G s :
-
Shear modulus of solid phase; G s = E s/2(1 + ν s) = 6.56 GPa
- J con :
-
Convective species flux; Eq. (24)
- J diff :
-
Diffusive species flux; Eq. (25)
- K d :
-
Drained bulk modulus; Eq. (3)
- K f :
-
Bulk modulus of the pore fluid; K f = 2.3 GPa
- K s :
-
Bulk modulus of solid phase; K s = E s/3(1 − 2ν s) = 17.66 GPa
- L :
-
Distance from the osteonal canal wall to the cement line; L = r o − r i = 100 μm
- R i :
-
Radius of the osteocyte; R i = 4 μm
- R o :
-
Radius of the lacuna; R o = 5 μm
- a :
-
Radius of the osteocytic process; a = 0.1 μm
- b :
-
Radius of the canaliculus; b = 0.23 μm
- c :
-
Pressure diffusion coefficient (consolidation coefficient)
- d :
-
Cross-sectional width and height of bone section; d = 21 μm
- k c :
-
Bulk permeability of the canalicular material; Eq. (12); see Table 1
- k l :
-
Bulk permeability of the lacunar material; Eq. (13); see Table 1
- l :
-
Lacuna-to-lacuna distance; l = 30 μm
- n c :
-
Canalicular density on the osteonal canal surface; n c = 5.5/100 μm2
- p :
-
Pore fluid pressure
- q :
-
Darcy fluid velocity; Eq. (19)
- r i :
-
Inner radius of the osteon (radius of osteonal canal); r i = 25 μm
- r o :
-
Outer radius of the osteon; r o = 125 μm
- α :
- μ :
-
Dynamic viscosity of the pore fluid; μ = 0.001 Pa s
- ν s :
-
Poisson’s ratio of solid phase; ν s = 0.335
- ν d :
- τ :
-
Shear stress; Eq. (20)
- ϕc :
-
Porosity of the canalicular section with osteocyte process; Eq. (1); ϕc = 0.0074
- ϕl :
-
Porosity of the lacunar section with osteocyte body; Eq. (2); ϕl = 0.119
References
Anderson, E. J., S. Kaliyamoorthy, J. Iwan, D. Alexander, and M. L. Knothe Tate. Nano-microscale models of periosteocytic flow show differences in stresses imparted to cell body and processes. Ann Biomed Eng. 33: 52-62, 2005. doi:10.1007/s10439-005-8962-y
Bacabac, R. G., T. H. Smit, M. G. Mullender, J. J. Van Loon, and J. Klein-Nulend. Initial stress-kick is required for fluid shear stress-induced rate dependent activation of bone cells. Ann Biomed Eng. 33: 104-110, 2005. doi:10.1007/s10439-005-8968-5
Batra, N. N., Y. J. Li, C. E. Yellowley, L. You, A. M. Malone, C. H. Kim, and C. R. Jacobs. Effects of short-term recovery periods on fluid-induced signaling in osteoblastic cells. J Biomech. 38: 1909-1917, 2005. doi:10.1016/j.jbiomech.2004.08.009
Bird, R. B., W. E. Stewart, and E. N. Lightfoot. Transport Phenomena. New York: John Wiley & Sons, 1960.
Brookes, M. The Blood Supply of Bone. Butterworths, London, 1971.
Buechner, P. M., R. S. Lakes, C. Swan, and R. A. Brand. A broadband viscoelastic spectroscopic study of bovine bone: implications for fluid flow. Ann Biomed Eng. 29: 719-728, 2001. doi:10.1114/1.1385813
Chakkalakal, D. A. Mechanoelectric transduction in bone. Journal of Materials Research. 4: 1034-1046, 1989. doi:10.1557/JMR.1989.1034
Christensen, R. M. Mechanics of Composite Materials. New York: Wiley, 1979.
Civan, F., and V. Nguyen. Particle migration and deposition in porous media. In: Handbook of Porous Media, edited by K. Vafai. CRC, 2005, p. 465.
Cooper, R. R., J. W. Milgram, and R. A. Robinson. Morphology of the osteon. An electron microscopic study. J Bone Joint Surg. 48: 1239-1271, 1966.
Cowin, S. C. (1999) Bone poroelasticity. J Biomech. 32: 217-238, 1999. doi:10.1016/S0021-9290(98)00161-4
Detournay, E., and A. H.-D. Cheng. Fundamentals of poroelasticity. In: Comprehensive Rock Engineering: Principles, Practice and Projects, Vol. II, Analysis and Design Method, edited by C. Fairhust. Pergamon Press, 1993, pp. 113–171.
Dullien, F. A. L. Porous Media: Fluid Transport and Pore Structure. New York: Academic Press, 1979.
Fritton, S. P., K. J. McLeod, and C. T. Rubin. Quantifying the strain history of bone: spatial uniformity and self-similarity of low-magnitude strains. J Biomech. 33: 317-325, 2000. doi:10.1016/S0021-9290(99)00210-9
Goulet, G. C., D. M. Cooper, D. Coombe, and R. F. Zernicke. Influence of cortical canal architecture on lacunocanalicular pore pressure and fluid flow. Comput Methods Biomech Biomed Engin. 11: 379-387, 2008. doi:10.1080/10255840701814105
Goulet, G. C., N. Hamilton, D. Cooper, D. Coombe, D. Tran, R. Martinuzzi, and R. F. Zernicke. Influence of vascular porosity on fluid flow and nutrient transport in loaded cortical bone. J Biomech. 41: 2169-2175, 2008. doi:10.1016/j.jbiomech.2008.04.022
Gururaja, S., H. J. Kim, C. C. Swan, R. A. Brand, and R. S. Lakes. Modeling deformation-induced fluid flow in cortical bone’s canalicular-lacunar system. Ann Biomed Eng. 33: 7-25, 2005. doi:10.1007/s10439-005-8959-6
Harding, R. M. Survival in Space: Medical Problems of Manned Spaceflight. London: Routledge, 1989.
Iannacone, W., E. Korostoff, and S. R. Pollack. Microelectrode study of stress-generated potentials obtained from uniform and nonuniform compression of human bone. J Biomed Mater Res. 13: 753-763, 1979. doi:10.1002/jbm.820130507
Klein-Nulend, J., C. M. Semeins, N. E. Ajubi, P. J. Nijweide, and E. H. Burger. Pulsating fluid flow increases nitric oxide (NO) synthesis by osteocytes but not periosteal fibroblasts–correlation with prostaglandin upregulation. Biochem Biophys Res Commun. 217: 640-648, 1995. doi:10.1006/bbrc.1995.2822
Knothe Tate, M. L. 2001 Mixing mechanisms and net solute transport in bone. Ann Biomed Eng. 29: 810-811; author reply 812-816, 2001.
Knothe Tate, M. L., and U. Knothe. An ex vivo model to study transport processes and fluid flow in loaded bone. J Biomech. 33: 247-254, 2000. doi:10.1016/S0021-9290(99)00143-8
Knothe Tate, M. L., U. Knothe, and P. Niederer. Experimental elucidation of mechanical load-induced fluid flow and its potential role in bone metabolism and functional adaptation. Am J Med Sci. 316: 189-195, 1998. doi:10.1097/00000441-199809000-00007
Knothe Tate, M. L., R. Steck, M. R. Forwood, and P. Niederer. In vivo demonstration of load-induced fluid flow in the rat tibia and its potential implications for processes associated with functional adaptation. J Exp Biol. 203: 2737-2745, 2000.
Kufahl, R. H., and S. Saha. A theoretical model for stress-generated fluid flow in the canaliculi-lacunae network in bone tissue. J Biomech. 23: 171-180, 1990. doi:10.1016/0021-9290(90)90350-C
Li, G. P., J. T. Bronk, K. N. An, and P. J. Kelly. Permeability of cortical bone of canine tibiae. Microvasc Res. 34: 302-310, 1987. doi:10.1016/0026-2862(87)90063-X
Marotti, G., M. Ferretti, F. Remaggi, and C. Palumbo. Quantitative evaluation on osteocyte canalicular density in human secondary osteons. Bone. 16: 125-128, 1995. doi:10.1016/S8756-3282(94)00019-0
Marotti, G., M. A. Muglia, and D. Zaffe. A SEM study of osteocyte orientation in alternately structured osteons. Bone. 6: 331-334, 1985. doi:10.1016/8756-3282(85)90324-2
Maroudas, A., R. A. Stockwell, A. Nachemson, and J. Urban. Factors involved in the nutrition of the human lumbar intervertebral disc: cellularity and diffusion of glucose in vitro. J Anat. 120: 113-130, 1975.
Neuman, W. F., and M. W. Neuman. The chemical dynamics of bone mineral. Chicago: University of Chicago Press, 1958.
Oballa, V., D. A. Coombe, and W. L. Buchanan. Factors affecting the thermal response of naturally fractured reservoirs. J Can Petrol Tech. 32: 31-42, 1993.
Petrov, N., and S. R. Pollack. Comparative analysis of diffusive and stress induced nutrient transport efficiency in the lacunar-canalicular system of osteons. Biorheology. 40: 347-353, 2003.
Piekarski, K., and M. Munro. Transport mechanism operating between blood supply and osteocytes in long bones. Nature. 269: 80-82, 1977. doi:10.1038/269080a0
Rubin, C. T., and L. E. Lanyon. Regulation of bone formation by applied dynamic loads. J Bone Joint Surg Am. 66: 397-402, 1984.
Rubin, C. T., and K. J. McLeod. Promotion of bony ingrowth by frequency-specific, low-amplitude mechanical strain. Clin. Orthop. Relat. Res. 298:165–174, 1994.
Salzstein, R. A., and S. R. Pollack. Electromechanical potentials in cortical bone–II. Experimental analysis. J Biomech. 20: 271-280, 1987. doi:10.1016/0021-9290(87)90294-6
Scheidegger, A. E. The Physics of Flow Through Porous Media. Toronto: University of Toronto Press, 1974.
Smit, T. H., J. M. Huyghe, and S. C. Cowin. Estimation of the poroelastic parameters of cortical bone. J Biomech. 35: 829-835, 2002. doi:10.1016/S0021-9290(02)00021-0
Tran, D., L. Nghiem, and W. L. Buchanan. Improved iterative coupling of geomechanics with reservoir simulation. Society of Petroleum Engineers. 9: 362-369, 2004.
Wang, L., S. C. Cowin, S. Weinbaum, and S. P. Fritton. Modeling tracer transport in an osteon under cyclic loading. Ann Biomed Eng. 28: 1200-1209, 2000. doi:10.1114/1.1317531
Wang, L., S. P. Fritton, S. C. Cowin, and S. Weinbaum. Fluid pressure relaxation depends upon osteonal microstructure: modeling an oscillatory bending experiment. J Biomech. 32: 663-672, 1999. doi:10.1016/S0021-9290(99)00059-7
Weinbaum, S., S. C. Cowin, and Y. Zeng. A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses. J Biomech. 27: 339-360, 1994. doi:10.1016/0021-9290(94)90010-8
Wilkes, C. H., and M. B. Visscher. Some physiological aspects of bone marrow pressure. J Bone Joint Surg Am. 57: 49-57, 1975.
Zhang, D. Oscillatory pressurization of an animal cell as a poroelastic spherical body. Ann Biomed Eng. 33: 1249-1269, 2005. doi:10.1007/s10439-005-5688-9
Zhang, D., S. Weinbaum, and S. C. Cowin. Estimates of the peak pressures in bone pore water. J Biomech Eng. 120: 697-703, 1998. doi:10.1115/1.2834881
Zhang, D., S. Weinbaum, and S. C. Cowin. On the calculation of bone pore water pressure due to mechanical loading. Int J Solids Structures. 35: 4981-4997, 1998. doi:10.1016/S0020-7683(98)00105-X
Acknowledgments
The authors extend thanks to the Natural Sciences and Engineering Research Council of Canada, Alberta Heritage Foundation for Medical Research, and Wood Professorship in Joint Injury Research for partial funding support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goulet, G.C., Coombe, D., Martinuzzi, R.J. et al. Poroelastic Evaluation of Fluid Movement Through the Lacunocanalicular System. Ann Biomed Eng 37, 1390–1402 (2009). https://doi.org/10.1007/s10439-009-9706-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-009-9706-1