Abstract
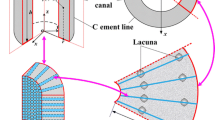
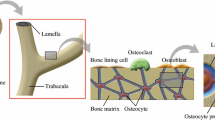
To explore the potential role that load-induced fluid flow plays as a mechano–transduction mechanism in bone adaptation, a lacunar–canalicular scale bone poroelasticity model is developed and implemented. The model uses micromechanics to homogenize the pericanalicular bone matrix, a system of straight circular cylinders in the bone matrix through which bone fluids can flow, as a locally anisotropic poroelastic medium. In this work, a simplified two-dimensional model of a periodic array of lacunae and their surrounding systems of canaliculi is used to quantify local fluid flow characteristics in the vicinity of a single lacuna. When the cortical bone model is loaded, microscale stress, and strain concentrations occur in the vicinity of individual lacunae and give rise to microscale spatial variations in the pore fluid pressure field. Furthermore, loading of the bone matrix containing canaliculi generates fluid pressures in the contained fluids. Consequently, loading of cortical bone induces fluid flow in the canaliculi and exchange of fluid between canaliculi and lacunae. For realistic bone morphology parameters, and a range of loading frequencies, fluid pressures and fluid–solid drag forces in the canalicular bone are computed and the associated energy dissipation in the models compared to that measured in physical in vitro experiments on human cortical bone. The proposed model indicates that deformation-induced fluid pressures in the lacunar–canalicular system have relaxation times on the order of milliseconds as opposed to the much shorter times (hundredths of milliseconds) associated with deformation-induced pressures in the Haversian system.
Similar content being viewed by others
References
Biot, M. A. General theory of three-dimensional consolidation. J. Appl. Phys. 12:155–164, 1941.
Biot, M. A. Theory of propagation of elastic waves in a fluid-saturated porous solid. II. Higher frequency range. J. Acoust. Soc. Amer. 28:2179–191, 1956.
Biot, M. A., and D. G. Willis. The elastic coefficients of the theory of consolidation. J. Appl. Mech. 24:594–601, 1957.
Brankov, G., and R. Blagoeva. An anatomical model for streaming potentials in osteons. J. Biomech. 17:8627–636, 1984.
Buechner, P. M., R. S. Lakes, C. Swan, and R. A. Brand. A broadband viscoelastic spectroscopic study of bovine bone: Implications for fluid flow. Ann. Biomed. Eng. 29:719–728, 2001.
Burger, E. H., J. Klein-Nulend, A. van der Plas, and P. J. Nijweide . Function of osteocytes in bone—their role in mechanotransduction. J. Nutr. 125:2020S--2023S, 1995.
Cooper, R. R., J. W. Milgram, and R. A. Robinson. Morphology of the osteons. An electron microscopic study. J. Bone Jt. Surg. Am. 48:1239–1271, 1966.
Cowin, S. C. Bone stress adaptation models. J. Biomech. Eng. 115:528–533, 1993.
Cowin, S. C. Survey article: Bone poroelasticity. J. Biomech. 32:217–238, 1999.
Cowin, S. C., L. Moss-Salentijn, and M. L. Moss. Candidates for the mechanosensory system in bone. J. Biomech. Eng. 113:191–197, 1991.
Cowin, S. C., S. Weinbaum, and Y. Zeng. A case for bone canaliculi as the anatomical site of strain generated potentials. J. Biomech. 28:1281–1297, 1995.
Currey, J. The Mechanical Adaptations of Bones. Princeton: Princeton University Press, 1984.
Djerad, S. E., F. du Burck, S. Naili, and C. Oddou. Analyse du comportement rhéologique instationnaire d’un échantillon de muscle cardiaque. C. R. Acad. Sci. Paris, série II 315:1615–1621, 1992.
Ferry, J. D. Viscoelastic Properties of Polymers. New York: Wiley, 1980.
Garner, E., R. Lakes, T. Lee, C. Swan, and R. Brand. Viscoelastic dissipation in compact bone: Implications for stress-induced fluid flow in bone. J Biomech. Eng. 122:166–172, 2000.
Glücksman, A. Studies on bone mechanics in vitro: II. The role of tension and pressure in chondrogenesis. J. Anat. 73:39–55, 1939.
Hancox, N. M. Biology of Bone. Cambridge University Press, 1972.
Iannacone, W., E. Korostoff, and S. R. Pollack. Microelectrode study of stress-generated potentials obtained from uniform and nonuniform compression of human bone. J. Biomed. Mater. Res. 13:753–763, 1979.
Jacobs, C. R., C. E. Yellowley, B. R. Davis, Z. Zhou, J. M. Cimbala, and H. J. Donahue. Differential effect of steady versus oscillating flow on bone cells. J. Biomech. 31:969–976, 1998.
Katz, J. L. Anisotropy of Young’s modulus of bone. Nature 283:106–107, 1980.
Katz, J. L. Hard tissue as a composite material. I. Bounds on the elastic behavior. J. Biomech. 4:455–473, 1971.
Knothe Tate, M. L. Whither flows the fluid in bone? An osteocyte’s perspective. J. Biomech. 36:1409–1424, 2003.
Lakes, R. S. Materials with structural hierarchy. Nature 361:511–515, 1993.
Lakes, R. S. Viscoelastic properties of cortical bone. In: Bone Mechanics Handbook, 2nd ed., edited by S. C. Cowin. Boca Raton, FL: CRC Press, 2001.
Lakes, R. S. Viscoelastic Solids. Boca Raton, FL: CRC Press, 1998.
Martin, R. B., and D. B. Burr. Structure,Function and Adaptation of Compact Bone. Raven Press, 1989.
McCreadie, B. R., and S. J. Hollister. Strain concentrations surrounding an ellipsoid model of lacunae and osteocytes. CMBBE 1:61–68, 1997.
McLean, F. C., and M. R. Urist. Bone: Fundamentals of the Physiology of Skeletal Tissue, 3rd ed. Chicago, IL: The University of Chicago Press, 1968.
Murdoch, P., S. Benzley, T. Blacker, and S. A. Mitchell. The spatial twist continuum: A connectivity based method for representing all-hexahedral finite element meshes. Finite Elem Anal. Des. 28:137–149, 1997.
Petrov, N., S. Pollack, and R. Blagoeva. A discrete model for streaming potentials in a single osteon. J. Biomech. 22(6–7):517–521, 1989.
Piekarski, K., and M. Munro. Transport mechanism operating between blood supply and osteocytes in long bones. Nature 269:80–82, 1977.
Qin, Y. X., K. J. McLeod, F. Guilak, F. P. Chiang, and C. T. Rubin. Correlation of bony ingrowth to the distribution of stress and strain parameters surrounding a porous-coated implant. J. Orthop. Res. 14:862–870, 1996.
Rubin, C. T., and K. J. McLeod. Promotion of bony ingrowth by frequency-specific, low-amplitude mechanical strain. Clin. Orthop. 298:165–174, 1994.
Rubin, C. T., and L. E. Lanyon. Limb mechanics as a function of speed and gait: A study of functional strains in the radius and tibia of horse and dog. J. Exp. Biol. 110:187–211, 1982.
Rubin, C. T., and L. E. Lanyon. Regulation of bone formation by applied dynamic loads. J. Bone Jt. Surg. Am. 66:397–402, 1984.
Salzstein, R. A., and S. R. Pollack. Electromechanical potentials in cortical bone. II. Experimental analysis. J. Biomech. 20:1271–280, 1987.
Salzstein, R. A., S. R. Pollack, A. F. T. Mak, and N. Petrov. Electromechanical potentials in cortical bone. I. A continuum approach. J. Biomech. 20:1261–270, 1987.
Scheidegger, A. E. The Physics of Flow Through Porous Media. New York: MacMillan, 1957.
Starkebaum, W., S. R. Pollack, and E. Korostoff. Microelectrode studies of stress-generated potentials in four-point bending of bone. J. Biomed. Mater. Res. 13:729–751, 1979.
Swan, C. C. http://www.engineering.uiowa.edu/∼swan/software/ fendac.pdf. FENDAC User’s Manual. 2003.
Swan, C. C. Techniques for stress- and strain-controlled homogenization of inelastic periodic composites. Comput. Meth. Appl. Mfech. Eng. 117:249–267, 1994.
Swan, C. C., R. S. Lakes, R. A. Brand, and K. J. Stewart. Micromechanically based poroelastic modeling of fluid flow in Haversian bone. J. Biomech. Eng. 125:25–37, 2003.
Tsay, R.-Y., and S. Weinbaum. Viscous flow in a channel with periodic crossbridging fibers: Exact solutions and Brinkman approximation. J. Fluid Mech. 226:125–148, 1991.
Wang, L., S. C. Cowin, S. Weinbaum, and S. P. Fritton. Modeling tracer transport in an osteons under cyclic loading. Ann. Biomed. Eng. 28:1200–1208, 2000.
Weinbaum, S., S. C. Cowin, and Y. Zeng. A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses. J. Biomech. 27:339–360, 1994.
You, L., S. C. Cowin, M. B. Schaffler, and S. Weinbaum. A model for strain amplification in the actin cytoskeleton of osteocytes due to fluid drag on the pericellular matrix. J. Biomech. 34:1375–1386, 2001.
Zhang, D., S. Weinbaum, and S. C. Cowin. Estimates of the peak pressures in bone pore water. J. Biomech. Eng. 120:697–703, 1998.
Zhang, D., S. Weinbaum, and S. C. Cowin. On the calculation of bone pore water pressure due to mechanical loading. Int. J. Solids Struct. 35:4981–4997, 1998.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gururaja, S., Kim, H.J., Swan, C.C. et al. Modeling Deformation-Induced Fluid Flow in Cortical Bone’s Canalicular–Lacunar System. Ann Biomed Eng 33, 7–25 (2005). https://doi.org/10.1007/s10439-005-8959-6
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-8959-6