Abstract
Aim
Long-term therapy with opioid analgesics bears the risk of complications, such as misuse, abuse, and dependence. An evidence-based guideline addresses the long-term use of opioid analgesics for non-cancer pain. Recent studies observed an increase in opioid prescriptions over the past two decades in Germany. However, the exact circumstances of this increase and if long-term therapy adheres to these guidelines in practice remains unknown. Therefore, this study aimed to evaluate the provision of opioid analgesics for adults with non-cancer pain from patients’ and physicians’ perspectives. Based on the results, recommendations for care strategies for the patient groups at risk of misuse, abuse, and dependence were developed.
Methods
Built on a mixed methods approach, this evaluation combines (i) interviews with primary care physicians and specialists, (ii) surveys of patients and physicians, (iii) analyses based on administrative claims data from a German statutory health insurance provider. Proceeding from the analysis of the evaluation results, the development of care strategies included (iv) semi-structured interviews with stakeholders and (v) expert workshops.
Conclusion
To our knowledge, this is the first attempt to analyze opioid prescriptions from the combined perspectives of patients and practitioners in Germany. The results are intended to facilitate the development of target group-specific care strategies and recommendations to establish a general framework for the implementation of care strategies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Opioid analgesics are utilized in the treatment of different indications, including acute severe pain, e.g., following trauma, burns or surgery, and for patients with painful terminal diseases. However, long-term use of opioid analgesics for chronic non-cancer pain (CNCP) is controversial with respect to efficacy, adverse physical effects, and aberrant behaviors such as opioid misuse (Turk et al. 2011). According to the International Association for the Study of Pain (IASP), CNCP is defined as pain lasting more than 3 months or beyond the expected period of healing of a pathology (Merskey 1986). Germany’s evidence-based S3 guideline from 2020 addresses the long-term use (≥ 3 months) of opioid analgesics for non-cancer pain (LONTS) (Häuser et al. 2020). It contains information on indications and contraindications as well as practice-related advice on therapy implementation and termination. According to the guideline the application of opioid analgesics in cases of CNCP may be advisable, e.g., for the treatment of chronic back pain, arthrosis pain, diabetic polyneuropathy, or phantom pain.
In Germany, pharmaceuticals with high potential for dependence, abuse, and health risks – such as most opioid analgesics – are covered by the German Narcotic Drugs Act (Betäubungsmittelgesetz, BtMG) and the Narcotic Drugs Prescription Ordinance (Betäubungsmittelverschreibungsverordnung, BtMVV) (BtMG 1994, s 1; BtMG 1994, annex III; BtMVV 1998, s 1). The treatment with these substances requires particular prescription forms for controlled substances (BtMVV 1998, s 8), inventory reports to the authority by all members of the supply chain (BtMG 1994, s 18), and is only permitted with reasonable causes, such as no adequate therapeutic alternatives (BtMG 1994, s 13). In the USA there are no particular prescription forms for controlled substances. Controlled substances are categorized into five schedules and regulated based upon their potential for abuse and dependence by the Controlled Substances Act (CSA). For instance, highly potent opioids – such as Oxycodone or Fentanyl – are available on written prescriptions only and for one-time use. Prescription forms for opioids with lower dependence potential (e.g. Tramadol, Buprenorphine) may be refilled up to five times if authorized by the physician (DEA 2020).
In terms of defined daily doses for statistical purposes (S-DDD), Germany ranks in second place after the United States among the countries reporting consumption of opioids for pain management in the period 2016–2018 (INCB 2019). Furthermore, according to data from a German statutory health insurance (SHI) provider from 2009, patients with CNCP received about three-quarters of all opioid-related DDDs. In some cases, they received it despite existing contraindications, such as mental comorbidities (Marschall and L'hoest 2011).
Several German analyses based on administrative claims data indicate rising numbers of opioid prescriptions with numbers varying by database and time period. Between 2000 and 2010 the percentage of individuals with at least one opioid prescription per year increased from 3.3% to 4.5% among persons insured through a regional German SHI provider (Schubert et al. 2013). Other studies based on data of other German SHI providers identified a less steep increase in patients with opioid prescriptions from 5.7% to 5.9% from 2006 to 2009 (Werber et al. 2015) and in Northern Germany from 4.4% to 4.8% from 2011 to 2015 (Verthein et al. 2020).
Despite several studies, knowledge on opioid prescriptions and related health care processes in Germany remains limited. As far as we know, no in-depth analysis covering the patient and health care provider perspectives has been conducted in Germany to date. It is essential to scrutinize current opioid analgesic provision in Germany focusing on guideline-based indication criteria. Therefore, this study aims to answer the following research questions regarding long-term opioid use for non-cancer pain:
-
Is there evidence for oversupply or inappropriate supply in the provision of opioid analgesics among persons insured by a large German SHI provider? Furthermore, is there evidence for substance dependence among patients taking opioid analgesics on long-term prescriptions? Are there certain patient groups who are affected disproportionately by oversupply, inappropriate supply, and/or dependence in the provision of opioid analgesics?
-
Are there aspects in the treatment process of patients with non-cancer pain contributing to the risk of misuse and dependence?
-
Which conditions impede guideline based-care?
The study aims to determine characteristics in which the group of insured with and without signs of dependence differ significantly. Literature on health economics and health services research gives several definitions for oversupply and inappropriate supply. For example, Chassin and Galvin (1998) define “overuse” as the provision of health care service under circumstances in which its potential for harm exceeds the possible benefit. However, the operationalization of this concise definition is challenging (Brownlee et al. 2017), and this applies to the Chassin and Galvin (1998) definition of “misuse” (misuse in terms of an inappropriate supply) as well. According to Chassin and Galvin (1998) there is an inappropriate supply when an appropriate service has been selected but a preventable complication occurs so the patient does not receive the full potential benefit of the service.
Methods/design
Health care processes of adult patients receiving long-term opioid prescriptions for non-tumor pain in Germany are studied utilizing a mixed methods approach:
-
an analysis of prescriptions and processes based on administrative claims data regardless of the indication (see Care epidemiology) and
-
a patient survey in patients with back pain and/or arthrosis (see Care analysis) on their health care situation, additionally the primary data were merged with the administrative claims of the SHI.
-
The physician perspective is evaluated by semi-structured guided interviews and a survey of primary care physicians and specialists (see Physician survey).
Based on these results and additional interviews with stakeholders, target group-specific strategies and reform approaches were developed. The study follows the LONTS guideline’s specification of long-term opioid therapy, which is defined as a 3-month use (Häuser et al. 2020).
The study was ethically approved by the University of Duisburg-Essen (ref. no. 21-9964-BO).
In addition, the operationalization of oversupply and inappropriate supply is based on the LONTS guideline. According to its recommendations, exceeding a 120 mg oral morphine equivalent dosage or prescriptions despite existing contraindications (primary headache, pain in functional/somatoform orders, chronic pancreatitis, chronic inflammatory bowel disease, harmful use of opioids, mental and behavioral disorders caused by opioids, severe mood disorder, suicidality) should be avoided due to increased risk of harming patients (Häuser et al. 2020). Therefore among others, the occurrence of high doses or contraindications are used as indicators for oversupply and/or inappropriate supply and therefore contributes to the bigger picture of long-term opioid therapy in Germany.
Dependence is defined as ICD-10 diagnosis F11.- (care epidemiology) or accords with diagnosis criteria of the American Psychiatric Association (care analysis). For subgroup analyses, age, gender, ethnicity, relevant indications, or postal code were considered. In addition, framework conditions or aspects of the treatment process – such as therapy duration, treating specialist group, or utilization of multimodal therapy – that contribute to oversupply or inappropriate supply or impede guideline-based care were identified. Based on these findings, recommendations for care strategies were developed with regard to correspondingly adapted framework conditions that promote an appropriate care situation.
Care epidemiology
The care epidemiology is intended to facilitate an overview on the provision of opioid analgesics in patients with CNCP. Demographic and indication-specific characteristics were identified and the health care services were analyzed. The care epidemiology aims at the first research questions (evidence for oversupply or inappropriate supply in the provision of opioid analgesics and in whether certain patient groups are affected disproportionately). Furthermore, indicators of multimodal therapy in which remedies (such as psychotherapy, physiotherapy, or occupational therapy) were prescribed in addition to opioid analgesics were analyzed (Fig. 1).
The analysis is based on administrative claims data of the DAK-Gesundheit, including master data (e.g., age, gender), inpatient and outpatient care data, prescription data regarding medication, aids and remedies, and data regarding rehabilitation, incapacity to work or sick pays (see Fig. 2).
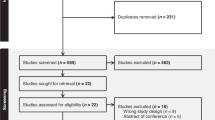
The selection period of the study population starts in 2018 and ends in quarter 1 of 2019. Each individual was selected by the date of the first prescription of opioid analgesics during the selection period (see Fig. 3). After selection, participants were observed for a two-year period or until death. For individuals selected in quarter 1 of 2018, information is included on whether and in how many quarters prescriptions of opioid analgesics occurred in 2017. For patients included in selection 2 to 5, this information is not provided. However, only patients with a long-term opioid therapy (≥ 3 months) were selected for care epidemiology as prescriptions have to occur in at least two consecutive quarters.
Inclusion and exclusion criteria for care epidemiology
All insured persons who meet the following criteria were subject to analysis:
-
In the period from 1 January 2017 until 31 March 2021, continuously insured by DAK-Gesundheit. Decedents during the period who meet the preceding criteria are included.
-
At least one prescription of opioid analgesics per quarter in two successive quarters. In case there are only prescriptions in two successive quarters, there must be at least 90 days between the first and the last prescription. The first prescription during the selection period has to take place between the first quarter of 2018 and the first quarter of 2019.
-
Minimum age of 18 years by the time of the first prescription of an opioid analgesic in one of the observed quarters.
Exclusion criteria were:
-
Diagnosis of a malignant growth in the two quarters of selection (ICD-10-GM: C00-C97 ending with G (reliable) and Z (condition after diagnosis) for outpatient care or ICD-10-GM: C00-C97 for inpatient care).
-
Documented palliative care in the two quarters of selection (ICD-10-GM: Z51.5; EBM: 01425-01426, 03370-03373, 04370-04373, 37300–37320; DRG: ZE60, ZE145, ZE2020-133, ZE2020-134; OPS: 8-982, 8-98e, 8-98h, 1-265.b, 1-773, 1-774).
Anatomical Therapeutic Chemical Classification System (ATC) codes constitute the basis for identifying the prescription of opioid analgesics. Table 1 shows the analgesics and the respective ATC-codes intended for all routine data analyses that were conducted. This selection of opioids represents the vast majority of prescribed opioid DDDs in the German Statutory Health Insurance market (Ludwig et al. 2021). Codeine, Methadone, Levomethadone, and their combinations were left out due to their minor role in therapy of non-cancer pain.
Data analysis of the care epidemiology
All analyses of administrative claims data were performed in Stata/SE version 12.1. A descriptive analysis of the medical care situation was performed, focusing on patient characteristics such as gender, age, comorbidities, region, and population density of residency. Further, health care services such as physician contacts separated according to specialist group, remedy prescriptions, and incapacity for work were examined. Subgroup analyses regarding individuals concerning oversupply or inappropriate supply provided information about whether certain groups are affected disproportionately (e.g., by age, gender, diagnoses, etc.). In addition, absolute and relative frequencies of misuse and its different types were calculated.
Oversupply and inappropriate supply were measured by different indicators based on the LONTS guideline’s recommendations regarding oral morphine equivalent dose, co-medications, contraindications, or possible signs of doctor shopping. This helped to examine patterns of care or opioid prescriptions and identify characteristics of the patient groups concerned.
Frequency distributions, location parameters, and correlations were analyzed by appropriate statistical measures (e.g., mean, median, frequency), depending on scale level. Standard deviations were calculated wherever appropriate. Moreover, cross tables and correlation analyses were used. The statistical tests for significant differences between the subgroups include the Chi-square test/Fisher’s exact test, t-test, Mann–Whitney U test, and Wilcoxon test for paired differences. Possible predictive factors for oversupply and/or inappropriate supply indicated by claims data were determined by logistic regression. Significance levels are fixed at .05 and the statistical power at 80% for all analyses.
Patient survey and care analysis
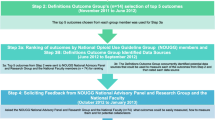
As a complement to the care epidemiology, the care analysis and the patient survey capture the treatment procedures from the patient perspective, including SHI administrative claims data and survey data. The care analysis addresses research questions 2 (aspects in the treatment process contributing to the risk of abuse, misuse or dependence) and 3 (conditions impeding guideline based-care) from the patient perspective. A cross-sectional patient survey was conducted on a sample of long-term opioid recipients drawn from 2020 SHI data. Since back and arthrosis pain represent widespread conditions and are conditions with strong guideline recommendations for opioid use, these patients were included in the care analysis (see Fig. 4) (Häuser et al. 2020). The survey started in November 2021 and gathered information mainly on the patients’ perspectives regarding their current state of pain and medical care. In addition, it contained selected retrospective questions on their conditions in 2020.
Patients with a valid declaration of consent and a completed questionnaire were included as study population. Administrative claims data for 2020 were included for the study population and linked to the survey data.
Inclusion and exclusion criteria for patient survey and care analysis
The inclusion criteria for the cross-sectional patient survey:
-
In the period from 1 January 2019 until 31 March 2020, no change of insurance provider (decedents included).
-
At least one prescription of opioid analgesics per quarter in quarter 1 and quarter 2 of 2020. In case there were prescriptions in quarter 1 and quarter 2 of 2020 solely, they had to be at least 90 days apart. The selected opioid prescriptions were identified via ATC-codes.
-
Diagnosis of chronic back pain or arthrosis pain in one of the quarters in 2020 (Chronic back pain: ICD-10-GM M42.16-19, M42.90, M42.96-99, M43.0-1, M47.26-27, M47.29, M47.86-88, M47.99, M48.06, M48.2, M54.16, M54.5, M55.3, M.99.33; M99.43, M99.53; Arthrosis pain: ICD-10-GM M15-M19).
-
Minimum age of 18 in quarter 1 of 2020 (born 1 January 2002 and before)
The following criteria for exclusion apply:
-
Diagnosis of a malignant growth in one of the observed quarters in 2020 (ICD-10-GM: C00-C97 ending with G and Z (outpatient) or no ending (inpatient))
-
Utilization of palliative care in one of the observed quarters in 2020 (ICD-10-GM: Z51.5; EBM: 01425-01426, 03370-03373, 04370-04373, 37300-37320; DRG: ZE60, ZE145, ZE2020-133, ZE2020-134; OPS: 8-982, 8-98e, 8-98h, 1-265.b, 1-773, 1-774.)
Questionnaire from patient survey
A standardized questionnaire was developed based on a literature search and S3 guideline recommendations. The patient questionnaire (see Online Resource 1) collected data with regard to pain situation, quality of care, health-related quality of life, indicators of substance use disorder, care situation (guideline compliance), and socio-demographic information.
Initially, the patients were asked to describe when and for how long pain problems occurred. In order to comprehend the patients’ pain situation, excerpts of the standardized German pain questionnaire (anamnesis, version 2019) commonly utilized among German pain relief specialists (Nagel et al. 2015) were used. The questionnaire was adjusted and shortened for the purpose of primary data collection in scientific projects and contained questions about the causes of pain and the care situation. Additional instruments to cover pain-related items were the
-
Brief Pain Inventory (BPI) to detect the pain intensity and effects in the past 24 hours (Radbruch et al. 1999),
-
Graded Chronic Pain Scale Items and Scoring (GCPS) to acquire data on pain effects in the past 3 months (Von Korff et al. 1992),
-
Self-Administered Comorbidity Questionnaire to collect data on comorbidities (Streibelt et al. 2012),
-
Patient Health Questionnaire-4 (PHQ-4) to record anxiety and depression symptoms (Löwe 2015).
To evaluate the quality of care regarding chronically sick persons, the German version of the Patient Assessment of Chronic Illness Care (PACIC 5A) was used. Its purpose is to assess to what extent the quality of care coincides with the Chronic Care Model (Rosemann et al. 2007). Further, potential substance dependence problems in the context of opioid prescriptions were taken from the German epidemiological survey of substance abuse 2015 (IFT 2015) to cover the DSM-5 criteria for substance use disorders. The questions on guideline compliant treatment were developed based on the current S3 guideline LONTS (Häuser et al. 2020). This includes the scope of guideline compliance of healthcare provision and opposing barriers from the patient’s point of view. In particular, the active involvement of patients regarding therapeutic options, objectives, decision-making, as well as treatment adherence and the related problems were interrogated. Furthermore, socio-demographic questions were asked, including school education and vocational training.
The selected individuals were sent a postal questionnaire by DAK-Gesundheit with a prepaid reply envelope directed to an independent department at the University of Duisburg-Essen not involved in further analyses to assure data privacy. This department examined the validity of informed consent and pseudonymized the questionnaires. Pseudonyms and patient information were reported to DAK-Gesundheit so administrative claims data could be selected.
Afterwards, the results of the questionnaire were merged with administrative claims data of the respective individuals via pseudonym. Additionally, an indicator variable specified if and in how many quarters of 2019 opioid analgesics were prescribed (see Fig. 4).
Sample size for the patient survey
From the entire DAK-Gesundheit collective meeting the above-mentioned inclusion and exclusion criteria, a random sample of approximately 2300 insured adults with long-term use of opioid analgesics was drawn. Accordingly, the sample consisted of patients with back pain and/or arthrosis pain, which were subdivided in one indication group, respectively. At least 290 participants per indication group are required to receive statistically significant mean differences at a power of 80% and a significance level of .05 with medium effect size. Based on an expected response rate of 30%, at least 967 persons per indication group must be contacted. This response rate is based on the findings of a German study on patients with back pain insured by another statutory health insurance (Bitzer et al. 2015). The study showed an overall response rate of 37%, though this varied with treatment from 31% to 42%. To additionally perform a logistic regression at a power of 80% and an alpha error of .05 for medium effect size, a minimum of 348 participants were needed.
Data analysis of the patient survey and care analysis
The data gathered by questionnaire of patients with valid declaration of consent were included into the analysis and linked to administrative claims data of 2020. Assessment tools were processed according to the developer’s recommendations. Psychometric properties such as internal constancy were assessed.
Univariate descriptive analyses were performed for variables such as pain situation, side effects, guideline based treatment, and dependence. Subgroups were analyzed to provide more detailed knowledge about the inappropriate supply with regard to various age groups and pain situations, among other aspects. Possible predictive factors for dependence and/or misuse indicated by the German epidemiological survey of substance abuse were determined by logistic regression.
First, descriptive analyses regarding missing values, frequency distributions, location parameters, and correlation were performed. The overall collective and individual subgroups were described by appropriate statistical measures (e.g., mean, median, frequency), depending on measuring scale and distribution. Standard deviations were calculated wherever appropriate. Moreover, cross tables and correlation analyses were used. The statistical tests for significant differences between the subgroups include the Chi-square test/Fisher’s exact test, t-test, Mann–Whitney-U test, and Wilcoxon test for paired differences. A logistic regression model tested for predictive factors regarding long-term use of opioid analgesics dependence, oversupply, and inappropriate supply. These findings are intended to help identify individual subgroups whose treatment could be improved by innovative health care strategies.
Physician interview/survey
The physician survey is a cross-sectional evaluation of the situation of opioid prescriptions in long-term treatment of non-cancer pain from the physician perspective. The physician survey addresses research questions 2 (aspects in the treatment process contributing to the risk of addiction or misuse) and 3 (conditions impeding guideline based-care) from the physician perspective.
Semi-structured interviews with primary care physicians and medical specialists
Semi-structured interviews with primary care physicians and medical specialists were conducted to cover their view on impediments and obstacles for guideline-compliant care. The findings will contribute to the conducting of a standardized questionnaire and will also be utilized to develop target group-specific strategies and alternative care strategies.
The semi-structured interviews and their analysis are based on Meuser and Nagel (1991, 2009) (Meuser and Nagel 1991, 2009) and Mayring (1991, 2015) (Mayring 1991; Mayring 2015). The interview-questions were developed based on a literature search and addressed aspects of guideline-compliant care of patients suffering from CNCP and treated with opioid analgesics. Furthermore, they contained questions on risks for and identification of misuse, abuse, and dependence.
Four groups of medical specialists were included: SHI-authorized primary care physicians, orthopedists and/or trauma surgeons, neurologists, and pain therapists. Interviewees were recruited with the help of the Professional Association of German Doctors and Psychotherapists practicing in Pain Medicine and Palliative Care (Berufsverband der Ärzte und Psychologischen Psychotherapeuten in der Schmerz- und Palliativmedizin in Deutschland e.V., BVSD).
Physician survey
The aim of the cross-sectional survey of general practitioners and specialists using a standardized questionnaire (see Online Resource 2) was to quantify the impressions gained from the interviews. In addition, it was taken into account to develop target group-specific care strategies and reform approaches to create suitable framework conditions for the implementation of the care strategies.
The study population included orthopedists, trauma surgeons, and neurologists as per the indications listed in the LONTS guideline and considered for opioid prescriptions. Further, primary care physicians and members of the BVSD, physicians practicing in pain medicine, were included.
The development of the questionnaire was based on the results of the semi-structured interviews, the evidence-based (S3) German guideline LONTS, and the evidence-based (S3) German guideline for drug related disorders. The questionnaire aimed to gather information in three different fields. First, it aimed to collect information about physicians’ attitudes toward opioid analgesics and their attitudes toward (long-term) opioid prescribing. Second, it focused on guideline-based treatment of patients with CNCP and implementation of care standards. In this context, barriers to guideline-based care for patients with CNCP treated with opioid analgesics remain in focus. Third, the problem of opioid-related abuse, misuse, and dependence was brought into focus.
Sample size for the physician survey
At least 290 participants are required to receive medium effect sizes for statistically significant mean differences at a power of 80% and a significance level of .05. According to an expected response rate of 25%, a minimum of 1160 physicians must be considered in total. Therefore, a sample of approximately 1300 physicians was drawn based on addresses purchased from a commercial address provider. The shares of specialist groups were fitted to the distribution of medical specialties on a national level according to the National Association of Statutory Health Insurance Physicians (KBV 2020). In addition, approximately 550 members of the BVSD practicing in pain medicine were contacted. The expected response rate was based on experiences of the researchers on previous physician surveys.
Data analysis of the physician survey
To evaluate the data gathered from the standardized surveys with primary care physicians and medical specialists, descriptive and analytical statistical measures, such as the absolute and relative frequencies of the reported impediments to guideline-compliant care provision, were used. In addition, subgroup analyses were performed, including specialist groups, additional qualifications, perception of and knowledge about the guideline, as well as addiction-related, structural, and regional aspects (e.g., differences between urban and rural).
First, descriptive analyses regarding missing values, frequency distributions, location parameters, and correlation were performed. The overall collective and individual subgroups were described by appropriate measures (e.g., mean, median, frequency), depending on measuring scale and distribution. Standard deviations were calculated wherever appropriate. Moreover, cross tables and correlation analyses were used. The statistical tests for significant differences between the subgroups include the Chi-square test/Fisher’s exact test, t-test, Mann–Whitney-U -test and Wilcoxon test for paired differences. To test for significant differences regarding regional aspects for example, the community size was tested against variables describing the physicians’ perception of the care situation. Possible barriers to guideline-compliant care provision (e.g., guideline knowledge and utilization) were determined by means of logistic regression.
Additionally, the psychometric quality of the survey instruments used in the physician survey were assessed. Furthermore, descriptive analyses regarding missing values, frequency distributions, location parameters, and correlation were performed. Significance levels are fixed at .05 and the statistical power at 80% for all analyses.
Development of target group-specific care strategies and reform approaches
Development of target group-specific alternative care strategies
The basis for the development of target group-specific care strategies were the results of the care epidemiology as well as the patient and physician survey.
The strategies mainly addressed two patient groups:
-
1.
Patients who are already affected by oversupply and/or inappropriate supply of opioid analgesics, and
-
2.
Patients who are at increased risk of oversupply and/or inappropriate supply of opioid analgesics due to specific framework conditions.
Depending on the conducted analyses, other identified problems as well as demographic and socio-economic criteria might be considered additionally.
The aim was to develop differentiated instruments that facilitate guideline-compliant care for patients with CNCP treated with opioid analgesics or via alternative treatment methods.
Development of reform approaches
To develop suitable framework conditions for the implementation of the compiled target group-specific alternative care strategies the role of, e.g., capacity planning, quality management and quality indicators, reimbursement, and the scope of service may be discussed.
Considering the perspective of physicians, patients and other stakeholders, health policy scenarios will be created to refine the framework conditions within an expert workshop with representatives of the joint self-government of Germany’s healthcare system, of physicians and patients, and of other stakeholders if necessary. Aspects such as practicability and incentive effects will be discussed. The results of the expert workshop will be used to elaborate concrete policy recommendations.
Discussion
To the authors’ knowledge, the approach to analyze the provision of opioid analgesics for non-cancer pain from the provider perspective on the one hand and the patient perspective on the other hand in one study has not been conducted to date in Germany.
In general, taking opioids in higher than the recommended doses (Häuser et al. 2020) or longer than 3 months may promote substance abuse (Verthein et al. 2020). Opioid abuse is a substantial problem in the United States. Over the past 20 years, it has become increasingly common, reaching epidemic levels (Rudd et al. 2016). There may be multiple reasons for the spread; Helmerhorst et al. mainly name aggressive marketing by pharmaceutical companies, easy access to illicitly or falsely prescribed opioids, as well as improvidence regarding opioid prescriptions suggested by scientific reports from the past century (Helmerhorst et al. 2017). These circumstances seem not to apply for Germany.
Nevertheless, opioid prescriptions have also become more common in Germany (Buth et al. 2019; Kraus et al. 2019; Schubert et al. 2013; Werber et al. 2015). However, researchers suggest that the number of opioid addicts and deaths remained mostly unchanged over the past 20 years and that there is no indication of epidemic proportions (Rosner et al. 2019; Seitz et al. 2019). They also state that findings based on self-reported information (Seitz et al. 2019) as well as those based on administrative data (Rosner et al. 2019) reveal gaps of knowledge about the German situation. Therefore, this study intended to combine both aspects, and the authors aimed to examine whether oversupply and/or inappropriate supply currently exist among persons with German SHI coverage as well as their socio-demographic attributes. Further, its purpose was to detect potentially restrictive conditions which hamper guideline-compliant care provision.
The survey of general practitioners and medical specialists using a standardized questionnaire will allow for quantifying statements and contribute to the development of care strategies and corresponding implementation strategies.
Data availability
Not applicable.
Code availability
Not applicable.
Abbreviations
- ATC:
-
Anatomical Therapeutic Chemical Classification System
- BPI:
-
Brief Pain Inventory
- BtMG:
-
Betäubungsmittel Gesetz; German Narcotic Drugs Act
- BVSD:
-
Berufsverband der Ärzte und Psychologischen Psychotherapeuten in der Schmerz- und Palliativmedizin in Deutschland e.V.
- CNCP:
-
Chronic Non-Cancer Pain
- GCPS:
-
Graded Chronic Pain Scale Items and Scoring
- IASP:
-
International Association for the Study of Pain
- PHQ-4:
-
Patient Health Questionnaire-4
- PACIC:
-
Patient Assessment of Chronic Illness Care
- S-DDD:
-
Defined Daily Doses for Statistical Purposes
- DRG:
-
Diagnosis Related Groups
- EBM:
-
Einheitlicher Bewertungsmaßstab; Uniform Value Scale
- ICD-10-GM:
-
International Statistical Classification of Diseases and Related Health Problems, 10th revision, German Modification
- LONTS:
-
Long-term Use of Opioids for Chronic non-cancer Pain
- OPS:
-
Operationen- und Prozedurenschlüssel; procedure classification
- PACIC 5A:
-
Patient Assessment of Chronic Illness Care
- Q:
-
Quarter
- SHI:
-
Statutory Health Insurance
- WHO:
-
World Health Organization
References
Bitzer E, Lehmann B, Bohm S, Priess HW (2015) BARMER GEK Report Krankenhaus 2015. Schwerpunkt: Lumbale Rückenschmerzen. BARMER GEK. https://www.barmer.de/resource/blob/1026504/788aab584c80c6cba0e5eebe1d5a6b79/barmer-gek-report-krankenhaus-2015-data.pdf. Accessed 17 Aug 2022
Brownlee S, Chalkidou K, Doust J, Elshaug AG, Glasziou P, Heath I, Nagpal S, Saini V, Srivastava D, Chalmers K, Korenstein D (2017) Evidence for overuse of medical services around the world. Lancet 390(10090):156–168. https://doi.org/10.1016/S0140-6736(16)32585-5 Erratum in: Lancet. 2022 Mar 5;399(10328):908
Buth S, Holzbach R, Martens M-S, Neumann-Runde E, Meiners O, Verthein U (2019) Problematic medication with benzodiazepines, “Z-drugs”, and opioid analgesics. Dtsch Arztebl Int 116(37):607–614
Chassin MR, Galvin RW (1998) The urgent need to improve health care quality. Institute of Medicine National Roundtable on Health Care Quality. JAMA 280(11):1000–1005. https://doi.org/10.1001/jama.280.11.1000
DEA (2020) Drugs of Abuse: A DEA Resource Guide. Drug Enforcement Department, U.S. Department of Justice. https://www.dea.gov/sites/default/files/2020-04/Drugs%20of%20Abuse%202020-Web%20Version-508%20compliant-4-24-20_0.pdf. Accessed 20 Aug 2021
Gesetz über den Verkehr mit Betäubungsmitteln - (Betäubungsmittelgesetz - BtMG) (1994) BGBl I no. 13 p. 358
Häuser W, Bock F, Hüppe M et al (2020) Empfehlungen der zweiten Aktualisierung der Leitlinie LONTS. Schmerz 34(3):204–244. https://doi.org/10.1007/s00482-020-00472-y
Helmerhorst GT, Teunis T, Janssen SJ, Ring D (2017) An epidemic of the use, misuse and overdose of opioids and deaths due to overdose, in the United States and Canada: is Europe next? Bone Joint J 99-b(7):856–864. https://doi.org/10.1302/0301-620x.99b7.Bjj-2016-1350.R1
INCB (2019) Report of the International Narcotics Control Board for 2019. International Narcotics Control Board (INCB). https://www.incb.org/documents/Publications/AnnualReports/AR2019/Annual_Report_Chapters/English_ebook_AR2019.pdf. Accessed 20 Aug 2021
KBV - Kassenärztliche Bundesvereinigung (2020) Statistische Informationen aus dem Bundesarztregister. https://www.kbv.de/media/sp/2020-12-31_BAR_Statistik.pdf. Accessed 15 June 2021
Kraus L, Seitz NN, Schulte B, Cremer-Schaeffer P, Braun B, Verthein U (2019) Estimation of the number of people with opioid addiction in Germany. Dtsch Arztebl Int 116(9):137–143. https://doi.org/10.3238/arztebl.2019.0137
Löwe B (2015) Gesundheitsfragebogen für Patienten (PHQ-4). Universitätsklinikum Hamburg-Eppendorf. https://www.uke.de/kliniken-institute/kliniken/psychosomatische-medizin-und-psychotherapie/forschung/downloads/index.html. Accessed 30 Mar 2021
Ludwig WD, Mühlbauer B, Seifert L (2021) Arzneiverordnungs-Report 2021. Springer, Berlin Heidelberg. https://doi.org/10.1007/978-3-662-63825-5
Marschall U, L'hoest H (2011) Opioidtherapie in der Versorgungsrealität. Ein Beitrag zur Diskussion um ein weitverbreitetes Arzneimittel. BARMER GEK. https://www.barmer.de/blob/71128/5bed89a4369e9ffa1b47d48a592ed571/data/opioidtherapie-in-der-versorgungsrealitaet.pdf. Accessed 14 Apr 2021
Mayring P (1991) Qualitative Inhaltsanalyse. In: Handbuch qualitative Forschung: Grundlagen, Konzepte. Methoden und Anwendungen. Beltz, München, pp 209–213
Mayring P (2015) Qualitative Inhaltsanalyse: Grundlagen und Techniken. Beltz, Weinheim Basel
Merskey H (Ed.) (1986) Classification of chronic pain: Descriptions of chronic pain syndromes and definitions of pain terms. 3:226
Meuser M, Nagel U (1991) ExpertInneninterviews - vielfach erprobt, wenig bedacht: ein Beitrag zur qualitativen Methodendiskussion. In: Qualitativ-empirische Sozialforschung: Konzepte, Methoden, Analysen. Westdt. Verl., Opladen, pp 441-471
Meuser M, Nagel U (2009) Das Experteninterview — konzeptionelle Grundlagen und methodische Anlage. In: Methoden der vergleichenden Politik- und Sozialwissenschaft: Neue Entwicklungen und Anwendungen. VS Verlag für Sozialwissenschaften, Wiesbaden, pp 465–479
Nagel B, Pfingsten M, Lindena G, Kohlmann T (2015) Deutscher Schmerz-Fragebogen. Handbuch. Deutsche Schmerzgesellschaft e.V. https://www.schmerzgesellschaft.de/fileadmin/user_upload/DSF-Handbuch_2015.pdf. Accessed 01 Feb 2021
Radbruch L, Loick G, Kiencke P et al (1999) Validation of the German version of the brief pain inventory. J Pain Symptom Manag 18(3):180–187. https://doi.org/10.1016/S0885-3924(99)00064-0
Rosemann T, Laux G, Droesemeyer S, Gensichen J, Szecsenyi J (2007) Evaluation of a culturally adapted German version of the Patient Assessment of Chronic Illness Care (PACIC 5A) questionnaire in a sample of osteoarthritis patients. J Eval Clin Pract 13(5):806–813. https://doi.org/10.1111/j.1365-2753.2007.00786.x
Rosner B, Neicun J, Yang JC, Roman-Urrestarazu A (2019) Opioid prescription patterns in Germany and the global opioid epidemic: Systematic review of available evidence. PLoS One 14(8):e0221153. https://doi.org/10.1371/journal.pone.0221153
Rudd RA, Aleshire N, Zibbell JE, Gladden RM (2016) Increases in drug and opioid overdose deaths — United States, 2000–2014. Morb Mortal Wkly Rep 64(50 & 51):1378–1382
Schubert I, Ihle P, Sabatowski R (2013) Increase in opiate prescription in Germany between 2000 and 2010: a study based on insurance data. Dtsch Arztebl Int 110(4):45–51. https://doi.org/10.3238/arztebl.2013.0045
Seitz NN, Lochbühler K, Atzendorf J, Rauschert C, Pfeiffer-Gerschel T, Kraus L (2019) Trends in substance use and related disorders. Dtsch Arztebl Int 116(35-36):585–591
Streibelt M, Schmidt C, Brünger M, Spyra K (2012) Comorbidity from the patient perspective - does it work? Validity of a questionnaire on self-estimation of comorbidity (SCQ-D). Orthopade 41(4):303–310. https://doi.org/10.1007/s00132-012-1901-3
Turk DC, Wilson HD, Cahana A (2011) Treatment of chronic non-cancer pain. Lancet 377(9784):2226–2235. https://doi.org/10.1016/s0140-6736(11)60402-9
Verthein U, Buth S, Daubmann A, Martens M-S, Schulte B (2020) Trends in risky prescriptions of opioid analgesics from 2011 to 2015 in Northern Germany. J Psychopharmacol 34(11):1210–1217. https://doi.org/10.1177/0269881120936544
Verordnung über das Verschreiben, die Abgabe und den Nachweis des Verbleibs von Betäubungsmitteln (Betäubungsmittel-Verschreibungsverordnung - BtMVV) (1998) BGBl. I p. 74, 80
Von Korff M, Ormel J, Keefe FJ, Dworkin SF (1992) Grading the severity of chronic pain. Pain 50(2):133–149. https://doi.org/10.1016/0304-3959(92)90154-4
Werber A, Marschall U, L'Hoest H, Hauser W, Moradi B, Schiltenwolf M (2015) Opioid therapy in the treatment of chronic pain conditions in Germany. Pain Physician 18(3):E323–E331
WIdO - Wissenschaftliches Institut der AOK (2022) Anatomisch-therapeutisch-chemische Klassifikation mit Tagesdosen für den deutschen Arzneimittelmarkt. https://www.wido.de/fileadmin/Dateien/Bilder/Publikationen_Produkte/Arzneimittel-Klassifikation/ATC_2022/wido_arz_amtliche_fassung_des_atc-index_2022.zip
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was entirely funded by the innovation fund of the German Federal Joint Committee (G-BA; funding-no. 01VSF19059). The funding body has in no way influenced the design of the study, the collection, analysis, and interpretation of data or the writing process of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study organization and conception of the surveys. Nils Frederik Schrader, Anja Niemann, Christian Speckemeier, Wolfgang Straßmeir, and Silke Neusser developed the concepts for data collection and analysis, whereas Carina Abels and Nikola Blase provided the concepts behind the interviews and workshops. Nils Frederik Schrader and Silke Neusser were the major contributors in writing this manuscript. All authors contributed to the manuscript in different stages and also read and approved the final version. Silke Neusser is the project leader and responsible for the study conception.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the ethics committee of the medical faculty of the University Duisburg-Essen (ref. no. 21-9964-BO).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Nils Frederik Schrader and Anja Niemann shared first-authorship.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schrader, N.F., Niemann, A., Speckemeier, C. et al. Prescription of opioid analgesics for non-cancer pain in Germany: study protocol for a mixed methods analysis. J Public Health (Berl.) 31, 1909–1919 (2023). https://doi.org/10.1007/s10389-022-01748-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10389-022-01748-6