Abstract
Knowing the ecology of game species is important to define sustainable hunting pressure and to plan management actions aimed to maintain viable populations. Common pheasant (Phasianus colchicus) is one of the main gamebird species in Europe and North America, despite its native range extending from the Caucasus to Eastern China. This research aimed to define the environmental variables shaping the spatial distribution of male pheasants and to estimate their breeding density in an agroecosystem of northern Italy. During the breeding season, 2015, we carried out 372 point counts with unlimited distances, randomly placed following a stratified sampling survey design. The habitat requirements of the pheasant were evaluated following a presence vs. availability approach, using environmental variables related to land use cover and landscape configuration. We built generalized linear models with a binary distribution, selecting variables following an information-theoretic approach. Densities were estimated through both conventional and multiple-covariate distance sampling. We estimated a density of 1.45 males/km2, with 4.26 males/km2 in suitable areas and 0.91 males/km2 in unsuitable ones. We found pheasants in areas with meadows and tree plantations, which were used to find food and refuges from predators and bad weather conditions. Similarly, woodlands have a positive effect on species occurrence, whereas arable lands were avoided, specifically maize and paddy fields. We found little evidence that landscape configuration affects pheasant occurrence. We found pheasants to be negatively affected by the length of edges between woodlands and arable lands, whereas edges between woodlands and grasslands seem to be beneficial for the species. These findings could help landscape and wildlife managers to plan habitat improvement actions useful to maintain self-sustaining populations of this species, by increasing cover of woodlands, meadows, and tree plantations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Common pheasant (Phasianus colchicus Linnaeus, 1758) (hereafter, pheasant) is one of the main gamebird species in Europe, with 10–57 million birds released annually in the UK (Mason et al. 2020; Madden 2021). The introduction of pheasants into hunting grounds is a widespread practice also in other countries of Western Europe, such as in France (with 10–15 million pheasants released annually), though remains rare in Fennoscandia (for instance, in Finland, 15,000 pheasants were released each year) (Arroyo and Beja 2002; Mustin et al. 2012). Its native range extends from Russian Far East, Korea, Eastern China, and Northern Vietnam, across Central Asia, west to the Caucasus, and western shores of the Caspian Sea. European native populations are found in Russia, and notably in Georgia, with small populations also present in Turkey, Greece and Armenia (BirdLife International 2021). However, it was introduced all over the world for hunting purposes, with viable and widespread populations mainly in Europe and North America (del Hoyo et al. 1994). In its native range, it is found mainly in the overgrown edges of rivers, hilly areas close to cultivations having small thickets, and flat arable lands. Introduced populations breed mostly in arable lands, including mosaics of cereal crops, grasslands, fallows, small woodlands, and hedgerows (Johnsgard 1986; del Hoyo et al. 1994). The European native population is estimated at 9,700–16,300 mature individuals with a stable trend, whereas the overall European population, including introduced populations, is estimated at 4,140,000–5,370,000 males, with an increasing trend (BirdLife International 2017, 2021). For this reason, the species was classified as least concern by IUCN (International Union for Conservation of the Nature) (BirdLife International 2021). Nonetheless, some populations are declining locally owing to habitat loss and over-hunting (BirdLife International 2016). In Italy, it is impossible to define its current distribution and to estimate the size and trend of the breeding population owing to the massive releases of birds for hunting because most stocks survive just a few months between shooting seasons. However, self-sustaining populations thrive in protected areas or in well-managed estates, where a few thousand territorial males can be estimated (Brichetti and Fracasso 2018). Furthermore, the conservation status of the Italian population is not evaluated by IUCN because it is considered an introduced species (Gustin et al. 2019). The aims of this research were (i) to define the environmental variables shaping the spatial distribution of male pheasants and (ii) to estimate its breeding density in an agroecosystem of northern Italy. We expected the species to be positively affected by meadows, hedgerows, woodlands, and tree plantations while avoiding a large amount of maize and paddy fields (Meriggi 1983, 1992; del Hoyo et al. 1994; Genovesi et al. 1999; Leif 2005; Nelli et al. 2012). This research is relevant because despite the pheasant being an important game species, there are few studies about its ecology in northern Italy (Meriggi 1983; Meriggi et al. 1996; Genovesi et al. 1999; Nelli et al. 2012). Moreover, knowing the environmental factors affecting the distribution and density of game species is very important to define sustainable hunting pressure and to plan management actions aimed to maintain viable populations (Sinclair et al. 2006; Sands et al. 2012; Robertson et al. 2017). Furthermore, increasing knowledge about gamebirds’ ecology could be helpful to better understand the ecological effects occurring in areas where they are released and managed (Madden 2021). Additionally, few studies are aimed to investigate the ecology of common species, such as the pheasant (EBCC 2019); therefore, if the preservation of overall species richness patterns is considered a valid conservation goal, then attention must be given to common species as well as rare ones (Lennon et al. 2003).
Materials and methods
Study area
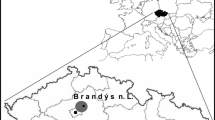
This study was carried out in the western Po Plain (north-western Italy), on a surface of about 2900 km2 (45° 11′ N 9° 05′ E) (Fig. 1). The Ticino River crosses the study area from north to south, flowing into the Po River that runs from west to east. The Sesia River and the Lambro River flow along the western and the eastern boundaries of the study area, respectively. The landscape is characterized by cultivated areas, especially paddy fields (39.4%), other annual crops (mainly maize, soybean, oil-seed rape, winter wheat, and alfalfa) (29.1%), and tree plantations (6.8%). Broad-leaved forests and urban areas represent 4.9% and 10.3% of the total area, respectively (ERSAF 2014). Continuous forests (composed mainly of Quercus robur, Carpinus betulus, Salix sp., Poplar sp., and the invasive Robinia pseudoacacia) are located along the Ticino River and in the southern part of the study area, near the Apennine slopes, whereas remnants of broad-leaved forest fragments are scattered in the cultivated area (95% of which are smaller than 10 ha) (ERSAF 2012). Tree plantations are mainly constituted by monospecific plantations of hybrid poplar species, but also of reforestations and short rotation coppices (SRCs). Due to its particular geographical location, this area is difficult to relate to other European lowlands. Even though the Po Plain presents a subcontinental climate, it is separated from the rest of continental Europe by the Alps, which could act as a zoogeographical barrier (Bianco 1990; Hermansen et al. 2011). Furthermore, even if it is attributable to the Mediterranean Basin, the climate, the land use, and the vegetation in the Po Plain differ from the rest of the Mediterranean area (Mikusiński and Angelstam 1997; Capotorti et al. 2012).
Survey design and data collection
To obtain a representative sample in this large and heterogeneous study area, a stratified sampling design was planned (Krebs 1999; Sutherland 2006). According to this approach, the area was firstly divided into landscape units (LUs), i.e., homogeneous portions of the area in terms of habitat or ecological characteristics likely to induce systematic variations in population density (Sutherland 2006). To this aim, a 2 × 2 km grid of sampling cells was superimposed on the study area and each cell was assigned to a LU, based on its characteristics. More details on LU definition are reported in Appendix S1 (Supplementary Materials, SM). Then, data collection was conducted in 62 sampling cells randomly selected (approximatively 10% of the study area). The number of sampling cells selected for each LU was proportional to the LU extent, in such a way that all the landscape characteristics of the study area were represented and each stratum was investigated with an effort proportional to its surface (Krebs 1999). Within each selected sampling cell, six point counts were carried out (Bibby et al. 2000; Sutherland 2006) during the breeding season 2015, between May and early June, because in this period, there is the peak of the call and the breeding activity (Cramp and Simmons 1980; Brichetti and Fracasso 2004). Data on species occurrence were therefore collected in 372 point counts. The selection of the six point counts inside the 2-km cell was carried out by a multi-level sampling (Sutherland 2006), subdividing the 2 km cells into cells of 250 × 250 m and randomly selecting six of them (Fig. 1). By using this second grid, it was possible to obtain more accurate estimates. Each point, placed randomly inside the cells, was surveyed once from dawn to 10:30, and the count lasted for 10 min (Chamberlain and Rolando 2014); overall, data collection took place for 32 effective days. During the fieldwork, we measured the exact distance from the observer to the birds with a laser rangefinder (Leica Rangemaster 900; Leica, Solms, Germany). When we did not see a calling bird (23% of cases), we mapped its approximate position on aerial photographs (1:5000 scale) based on the likely attenuation and direction of its vocalization. We then measured the distance from observer to the position of each calling bird using the software QGIS v.3.14.16 “Pi.”
Environmental variables
The habitat requirements of the pheasant were evaluated following a presence vs. availability approach (Manly et al. 2002; Boyce et al. 2002). The environmental variables (Table 1) were measured at the home range scale (Manly et al. 2002; Morrison et al. 2006) at presence sites and subsequently compared with those of an equal number of availability sites which were randomly selected in the study area without any constraints (Manly et al. 2002; Boyce et al. 2002). We measured variables related both to land use cover (N = 14) and to landscape configuration (N = 13); formulas and descriptions of landscape metrics used (McGarigal and Marks 1994) are provided in SM Table S2. In addition to these metrics, we measured also the edges (in meters) between woodlands and both arable lands and grasslands, as well as between shrublands and both arable lands and grasslands. To take into account the spatial ecology of the species (Brennan et al. 2002; Manly et al. 2002), presence/availability sites corresponded to cells the extent of which was defined based on the home range size of the species during the breeding season, as suggested by the available literature. Specifically, the home range of the species is equal to 2.1–52.7 ha (for details and references, see SM Table S2); hence, considering an average of about 18.4 ha, a grid was generated with square cells of 359 m on side.
Habitat suitability
We used generalized linear models (GLM) with a binary distribution (link function = logit) (Rushton et al. 2004; Keating and Cherry 2004) to relate the presence of male pheasants to the environmental variables. The presence of the species was 1 when at least one male was detected in the 359 m grid cells, or 0 otherwise. To model the spatial distribution of pheasant, first, we selected only the variables with a remarkable effect on its occurrence (therefore with some evidence of importance), with a pairwise comparison of the second-order Akaike information criterion (AICc; Akaike 1973) of two simple GLM: one with the intercept only and the other with each variable (Burnham et al. 2011; Chiatante et al. 2021). When the AICc value of the GLM with the variable was lesser than the one with the intercept only (the null model), with a difference of at least two (Δ AICc ≤ 2), that variable was retained (Burnham and Anderson 2002). Once the number of variables was reduced, we ran a priori sets of GLM built with all combinations of the retained environmental variables; these models were built using not correlated variables (|r|< 0.70). Then, for each model, the AICc was calculated and the models with the lowest AICc (Δ AICc ≤ 2) were selected as the best (Burnham and Anderson 2002). These models were averaged and the importance of each variable (Σwi) in the set was calculated (Burnham and Anderson 2002). For this analysis, all the variables considered were standardized by normalization; that is, each variable had a mean of zero and a standard deviation of one (Quinn and Keough 2002; Zuur et al. 2007). The model’s ability to distinguish between occupied and available sites was tested through the area under the curve (AUC) of the receiver operating characteristic plot (ROC curve) (Pearce and Ferrier 2000; Fawcett 2006). Moreover, we tested the residuals’ spatial autocorrelation by the Moran I test (Zuur et al. 2007; Bivand et al. 2008). We used the variance inflation factor (VIF) with a threshold of 3 to exclude variable collinearity (Fox and Monette 1992; Zuur et al. 2010) and the explained deviance D2 was used as a measure of the variance explained by the model (Zuur et al. 2007). Finally, we predicted the probability of occurrence of the pheasant in the study area. Precisely, using a grid covering the whole area and with a spatial resolution of 359 m, first, we measured for each square of the grid the environmental variables included in the model. Then, after their standardization (as we did to build the models), we reclassified these values using the coefficients estimated by the average model. In this way, we obtained the probability of occurrence of the species for each square of the grid, allowing us to calculate the average (± SD) probability of occurrence of the pheasant in the study area. Data used to measure the environmental variables were obtained from the regional land use map DUSAF 5.0 (ERSAF 2017) and the regional forest map “Carta dei tipi forestali reali della Lombardia” (ERSAF 2012) and processed by the software Quantum GIS v.3.14.16 “Pi.” All the analyses were performed using the statistical software R v.3.3.2 (R Core Team 2019) and the packages MuMIn (Tillé and Matei 2016), car (Fox and Weisberg 2011), landscapemetrics (Hesselbarth 2021), spdep (Bivand et al. 2015), and ROCR (Sing et al. 2007).
Density estimation
The density of male pheasants was estimated through the distance sampling method, particularly the conventional distance sampling (CDS) (Buckland et al. 1993). After a visual inspection of distances distribution, we truncated the 10% of the greatest distances as suggested by Buckland et al. (1993) and transformed the distance data into equal intervals of 60 m. As suggested by Buckland et al. (1993) and Thomas et al. (2010), we tested the following combinations of key functions and series adjustments: (1) uniform key with cosine adjustments, (2) half-normal key with cosine adjustments, (3) half-normal key with Hermite polynomial adjustments, and (4) hazard-rate key with simple polynomial adjustments. Anyway, the probability of detecting a bird depends not only on distance but also on many other factors, such as habitat, weather, observer, and bird behavior (Buckland et al. 1993), a circumstance that could exist, at least in part, in this research due to the variability of our data. Therefore, ignoring all these other factors, besides distances, could cause some bias in the estimate (Beavers and Ramsey 1998; Bas et al. 2008; Anderson et al. 2015). For this reason, besides CDS, we used also multiple-covariate distance sampling (MCDS) (Marques et al. 2007), an extension of CDS that allow modelling the detection probability as a function of variables other than distance. Therefore, a graphical exploratory analysis was run to assess if the habitat around points might bias the estimate of the density. We considered the cover of open areas (arable lands, meadows, and fallows), shrublands, and arboreal vegetation (woodlands, poplar plantations, other tree plantations) in a 300-m circular plot around the sampling points (Chiatante et al. 2020, 2021). The results of this analysis (ESM Figure S2) showed that both the cover of open areas (rs = 0.223, P = 0.003) and arboreal vegetation (rs = − 0.255, P < 0.001) could bias our estimate because the detection distance changed with them; the correlation with shrublands was not significant (rs = − 0.072, P = 0.337). Therefore, we used the cover of open areas and arboreal vegetation as covariates. To estimate density with MCDS, only half-normal and hazard-rate keys are allowed (Marques et al. 2007). We built the detection functions using the habitat suitability as strata, i.e., suitable vs. unsuitable areas. To this purpose, we reclassified the map of probability of occurrence (see section “Habitat suitability models”) into a binary map of suitable/unsuitable areas using as threshold the average probability in presence sites (equal to 0.803) (Liu et al. 2005). Once the models were computed, ran both with CDS and MCDS, we used Akaike information criterion (AIC) to select the best model (Buckland et al. 1993; Thomas et al. 2010). Considering that data were binned, the goodness-of-fit of the models was assessed by χ2 tests (Buckland et al. 1993; Thomas et al. 2010). Finally, the average probability of detection was estimated and the effective detection radius (EDR) was defined. For each estimate, both the coefficient of variation (CV) and the 95% confidence intervals (CI) were calculated. The analyses were performed using the statistical software R v.3.6.1 (R Core Team 2019) and the package Distance (Miller et al. 2019).
Results
Habitat suitability
We detected 183 male pheasants in 100 sampling points, corresponding to 139 cells (42.2%). The pairwise comparison suggested that 10 environmental variables affected in a remarkable way the species occurrence (SM Tables S3 and S4). These almost encompassed exclusively land use variables, but also two variables of landscape configuration. Indeed, we found pheasants in areas with meadows, tree plantations (including poplar plantations), and woodlands, in the proximity of water bodies and with long edges between woodlands and grasslands (SM Figure S3). Contrarily, they avoided urban areas, arable lands (especially maize and paddy fields), and areas with long edges between woodlands and arable lands (SM Figure S3).
Nine models best explained the occurrence of the pheasant (SM Table S5). The average GLM showed that four variables affected in a marked way the pheasant occurrence (Table 2, Fig. 2). In particular, meadows and poplar plantations increased its probability of occurrence, while urban areas and edges between woodlands and arable lands decreased it. Contrarily, the length of edges between woodlands and grasslands had a positive effect on the species distribution. The VIF revealed no collinearity among variables (Table 2) and the model residuals did not show any spatial correlation (Moran test, I = 1.051, P = 0.668). ROC analysis showed an excellent ability of the model to distinguish between occupied and random sites (AUC = 0.945, P < 0.001) and the explained deviance D2 was equal to 55.8%. The average probability of occurrence estimated in the study area by the model was equal to 0.482 ± 0.273 (SD) (min. 0.00, max. 1.00) (SM Figure S4).
Density estimation
We collected 179 observations of 183 pheasants. On average, 1.02 pheasants per point were detected (SE = 0.018, min = 1, max = 4). The best detection probability function was the hazard-rate with simple polynomial adjustments with open areas and arboreal vegetation as covariate (Table 3; Fig. 3). The goodness-of-fit of the model was good (χ2 = 0.622, df = 2, P = 0.733). This model gave an EDR of 273 m and the average probability of detection was estimated to be 0.422 ± 0.050 SE. The estimated density in unsuitable areas was 0.91 males/km2 (SE = 0.204, LCI 95% = 0.60, UCI 95% = 1.42, CV = 22.2%), whereas in suitable areas was 4.26 males/km2 (SE = 0.854, LCI 95% = 2.88, UCI 95% = 6.30, CV = 20.1%). In general, the density of pheasant estimated in the study area was 1.45 males/km2 (SE = 0.240, LCI 95% = 1.06, UCI 95% = 2.01, CV = 16.5%).
Discussion
This research was aimed to explore the relationships between the occurrence of male pheasants during the breeding season and environmental variables, particularly land use cover and landscape configuration. As expected, we found them in areas with meadows and poplar plantations. Pheasants typically select meadows, grasslands, and other herbaceous areas (Wasilewski 1986; Leptich 1992; Clark et al. 1999; Smith et al. 1999; Holá et al. 2015). Nonetheless, in some cases, pheasants avoid grasslands during the breeding season (Genovesi et al. 1999; Smith et al. 1999) likely to avoid predators (Smith et al. 1999). This happens especially with tall dense vegetation cover, having significantly more predated individuals than the short or sparse cover (Smith et al. 1999). Regarding poplar plantations, they are commonly used during the breeding season in northern Italy (Meriggi et al. 1996). Likewise, other tree plantations, such as reforestations and short rotation coppices, have a positive effect on the species occurrence. This is not surprising, because the selection for this land use type is well known, both in the native range (Ashoori et al. 2018) and in Europe (Baxter et al. 1996; Nelli et al. 2012; Chiatante et al. 2019). Particularly, Baxter et al. (1996) found the species more often in willow coppices than in poplar coppices. Still, Nelli et al. (2012) found a negative relationship between crowing males and distance from reforestations; similarly, densities increased with a higher cover of reforestations. Indeed, these kinds of tree plantations, when mature, show characteristics that make them similar to natural woodlands, having a high density of stems near ground level, a characteristic that seems to enhance pheasant survival (Leif 2005). Therefore, pheasants can use tree plantations to find food and refuges from predators and bad weather conditions (Baxter et al. 1996; Leif 2005; Nelli et al. 2012). Similarly, woodlands have a positive effect on the species occurrence, although this relationship was found only for univariate analyses. However, this result is in accordance with previous research. In general, this species avoids large forests but a small amount of woody vegetation in the landscape, as in our study area, is beneficial for this species (Hill and Robertson 1988; del Hoyo et al. 1994; Ronnenberg et al. 2016; Keller et al. 2020). Indeed, the cover provided by woody habitats reduces home range sizes and enhances home range suitability for breeding male pheasants (Leif 2005). In addition, woody habitats provide shelter and refuge from hunters and predators throughout the year (Hill and Robertson 1988; Leif 2005; Holá et al. 2015). Nonetheless, in North America, generally, woody vegetation does not affect the occurrence and abundance of pheasants (Riley et al. 1998; Schmitz and Clark 1999). Besides, we did not find any relation with hedgerow density or shrubland cover. This result is a little unexpected because hedgerows and shelterbelts are usually essential for the species occurrence (Hill and Robertson 1988; Gabbert et al. 1999; Leif 2005). Similarly, also Nelli et al. (2012) did not find any relation with hedgerows, probably due to the low presence of these landscape features in their study area, which could also be true in our case, considering that on average, there are only 13 m of hedgerows per hectare. Univariate analyses showed that in the study area, arable lands are avoided, specifically maize and paddy fields. This is in accordance with the fact that maize and paddy fields are not suitable habitats during the breeding season (Meriggi et al. 1996). Indeed, in North America, conversion of barley to paddy fields has been the strongest driver of habitat loss for the species likely because they are flooded for much of the growing season (Coates et al. 2017). On the other hand, in Germany, with approximately 15–35% of maize cover, the effect is moderately positive, whereas, at the highest percentages, maize has a negative effect on the species densities (Ronnenberg et al. 2016). In general, cultivated areas were positively selected by pheasants (del Hoyo et al. 1994; Keller et al. 2020). In North America, pheasants are largely limited to irrigated croplands (Johnsgard 1986), selecting wheat and alfalfa during the breeding season (Whiteside and Guthery 1983) but avoiding maize and soybean (Clark et al. 1999; Leif 2005). Selection for irrigated crops might be due to the higher availability of arthropods in moist soils with respect to dry ones (Hart et al. 2009). In addition, irrigated croplands require ditches and canals that could provide both arthropods and safe places, especially if grassed. Nonetheless, intensive agriculture and changes in agricultural practices are detrimental for pheasants (Ronnenberg et al. 2016; Coates et al. 2017). For instance, monocultures and the consequent use of herbicides and insecticides reduce both the availability of refugia and food resources, increasing the effects of predation and mortality (Rands 1986; Meriggi et al. 1996; Riley and Schulz 2001; Coates et al. 2017). The analyses showed also a positive effect of rivers and water bodies, which is common for the species both in native and introduced ranges (Cramp and Simmons 1980; Johnsgard 1986; Holá et al. 2015). Indeed, in Russia, it occurs in shrublands and thickets in river valleys and desert-adapted subspecies are largely limited to riverine areas or other areas of available fresh water (Johnsgard 1986). Similarly, in the western Palearctic, the pheasants occur in broad river valleys. Likewise, wetlands are very important habitat throughout the year (Gatti et al. 1989; Leptich 1992; Smith et al. 1999; Ronnenberg et al. 2016). Avoidance of urban areas is also in accordance with general habits of the species. For instance, road density has a strong negative effect on the species, probably because the increase mortality rate of individuals is caused by intensive road traffic (Holá et al. 2015; Madden and Perkins 2017).
We found little evidence that landscape configuration affects pheasant occurrence, which was shown to be important in other studies. For instance, Clark et al. (1999) found that areas with small patches of grasslands clustered have a moderate probability to be selected. On the other hand, it was shown that patches with smaller sizes and characterized by long and irregular edges are more suitable for pheasants, compared to bigger and more regular ones (Baxter et al. 1996; Nelli et al. 2012). Moreover, habitat heterogeneity was shown to be important, as in Germany, where municipalities with a low crop diversity host fewer pheasants than more diverse areas (Ronnenberg et al. 2016). The only effect we found was related to edges, which are generally beneficial for the pheasant (Clark et al. 1999; Genovesi et al. 1999). Particularly, we found pheasants to be negatively affected by the length of edges between woodlands and arable lands, which agrees with the general avoidance of arable lands we found. Generally, in croplands, pheasants spent most of their time in spontaneous vegetation growing along drainage ditches, field edges, roadsides, and shelterbelts (Warner and Joselyn 1986; Wasilewski 1986; Meriggi et al. 1996; Genovesi et al. 1999). Contrarily, we found a positive effect of edges between woodlands and grasslands. This could be due to the higher suitability of meadows with respect to arable lands we observed, related also to lesser disturbances common to cultivated areas (e.g., due to spraying, weed control). Anyhow, along edges, vulnerability to predation is high (Schmitz and Clark 1999; Kuehl and Clark 2002); therefore, one may ask why pheasants do not avoid edges that increase mortality risk, perhaps because breeding activity around edges is a stronger factor in habitat selection that compete with predator avoidance (Schmitz and Clark 1999) and the selection of edges between woodlands and grasslands is a better compromise with respect to use edges between woodlands and arable lands because of their higher suitability.
The second aim of this study was to estimate the pheasant density. Our results showed a density of 1.45 males/km2, with 4.26 males/km2 in suitable areas and 0.91 males/km2 in unsuitable ones. These values are below other densities estimated in northern Italy. Nelli et al. (2012) estimated densities of 6.6–27.0 males/km2 in four protected areas near Milan. However, in these areas, habitat improvements (e.g., game crops, stubble maintenance, and maintenance of linear grassy vegetation) were carried out to increase the density and productivity of wild pheasants. Other Italian studies estimated densities between 2.8 and 255 pheasants/km2, but these values are likely biased by releases of birds for hunting (Brichetti and Fracasso 2018). Our results differed from other estimates likely because of the method of data analysis in as much these estimates usually were calculated in relation to fixed-radius plots, which assume the perfect detectability. Indeed, many studies have found differences between indices of relative abundances calculated within fixed-radius plots and distance sampling estimates (Norvell et al. 2003; Buckland et al. 2008; Gottschalk and Huettmann 2011). All of them stressed the more convincing estimates of distance sampling, which gives precise unbiased results with respect to a study design based on a relative abundance approach, in as much it takes into account the variable detectability of individuals (Buckland et al. 1993). However, relative abundances reflect species responses to ecological gradients and can be easily employed to investigate trends, therefore are very useful in some circumstances (Hutto and Young 2003; Johnson 2008).
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on a reasonable request.
Change history
27 July 2022
Missing Open Access funding information has been added in the Funding Note.
References
Akaike H (1973) Information theory as an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) Second International Symposium on Information Theory. Akademiai Kiado, Budapest 267–281
Anderson AS, Marques TA, Shoo LP, Williams SE (2015) Detectability in audio-visual surveys of tropical rainforest birds: the influence of species, weather and habitat characteristics. PLoS One 10:e0128464. https://doi.org/10.1371/journal.pone.0128464
Arroyo B, Beja P (2002) Impact of hunting management practices on biodiversity. Report WP2 to REGHAB project. European Commission, Brussels
Ashoori A, Kafash A, Varasteh Moradi H et al (2018) Habitat modeling of the common pheasant Phasianus colchicus (Galliformes: Phasianidae) in a highly modified landscape: application of species distribution models in the study of a poorly documented bird in Iran. Eur Zool J 85:372–380. https://doi.org/10.1080/24750263.2018.1510994
Bas Y, Devictor V, Moussus JP, Jiguet F (2008) Accounting for weather and time-of-day parameters when analysing count data from monitoring programs. Biodivers Conserv 17:3403–3416. https://doi.org/10.1007/s10531-008-9420-6
Baxter DA, Sage RB, Hall DO (1996) A methodology for assessing gamebird use of short rotation coppice. Biomass Bioen 10:301–306
Beavers SC, Ramsey FL (1998) Detectability analysis in transect surveys. J Wildlife Manage 62:948–957
Bianco PG (1990) Potential role of the palaeohistory of the Mediterranean and Paratethys basins on the early dispersal of Euro-Mediterranean freshwater fishes. Ichthyol Explor Fres 1:167–184
Bibby CJ, Burgess ND, Hill DA, Mustoe SH (2000) Bird census techniques, 2nd edn. Academic Press, London, UK
BirdLife International (2021) European Red List of Birds. Publications Office of the European Union, Luxembourg
BirdLife International (2017) European birds of conservation concern: populations, trends and national responsabilities. BirdLife International, Cambridge, UK
BirdLife International (2016) Phasianus colchicus: The IUCN Red List of Threatened Species 2016: e.T45100023A85926819. https://doi.org/10.2305/IUCN.UK.2016-3.RLTS.T45100023A85926819.en. Accessed 16 Sep 2021
Bivand R, Altman M, Anselin L et al (2015) Package spdep: Spatial Dependence - weighting schemes, statistics and models. www.cran.r-project.org, Wien. Accessed 16 Sep 2021
Bivand R, Pebesma EJ, Gómez-Rubio V (2008) Applied spatial data analysis with R. Springer, New York
Boyce MS, Vernier PR, Nielsen SE, Schmiegelow FKA (2002) Evaluating resource selection functions. Ecol Model 157:281–300. https://doi.org/10.1016/S0304-3800(02)00200-4
Brennan JM, Bender DJ, Contreras TA, Fahrig L (2002) Focal patch landscape studies for wildlife management: optimizing sampling effort across scales. Integrating landscape ecology into natural resource management. Cambridge University Press, Cambridge, pp 68–91
Brichetti P, Fracasso G (2018) The birds of Italy. Volume 1. Anatidae-Alcidae. Edizioni Belvedere, Latina, IT
Brichetti P, Fracasso G (2004) Ornitologia Italiana. Vol 2 - Tetraonidae-Scolopacidae. Alberto Perdisa editore, Bologna, IT
Buckland ST, Anderson DR, Burnham KP, Laake JL (1993) Distance sampling: estimating abundance of biological populations. Chapman & Hall, London
Buckland ST, Marsden SJ, Green RE (2008) Estimating bird abundance: making methods work. Bird Conserv Intern 18:91–108. https://doi.org/10.1017/S0959270908000294
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Burnham KP, Anderson DR, Huyvaert KP (2011) AIC model selection and multimodel inference in behavioral ecology: some background, observations, and comparisons. Behav Ecol Sociobiol 65:23–35. https://doi.org/10.1007/s00265-010-1029-6
Capotorti G, Guida D, Siervo V et al (2012) Ecological classification of land and conservation of biodiversity at the national level: the case of Italy. Biol Conserv 147:174–183. https://doi.org/10.1016/j.biocon.2011.12.028
Chamberlain D, Rolando A (2014) The effects of a settling-down period on estimates of bird species richness and occurrence from point counts in the Alps. Bird Study 61:121–124. https://doi.org/10.1080/00063657.2013.870527
Chiatante G, Giordano M, Meriggi A (2019) Bird diversity in short rotation coppice in Northern Italy. Ardea 107:5–17. https://doi.org/10.5253/arde.v107i1.a10
Chiatante G, Giordano M, Vidus Rosin A et al (2021) Spatial distribution of the Barbary Partridge (Alectoris barbara) in Sardinia explained by land use and climate. Eur J Wildl Res 67:78. https://doi.org/10.1007/s10344-021-01519-w
Chiatante G, Vidus Rosin A, Cinerari CE et al (2020) Habitat selection and density of the Barbary partridge in Sardinia. Mediterranean Sea Eur J Wildlife Res 66:22. https://doi.org/10.1007/s10344-020-1360-9
Clark WR, Schmitz RA, Bogenschutz TR (1999) Site selection and nest success of ring-necked pheasants as a function of location in Iowa landscapes. J Wildlife Manage 63:976–989
Coates PS, Brussee BE, Howe KB et al (2017) Long-term and widespread changes in agricultural practices influence ring-necked pheasant abundance in California. Ecol Evol 7:2546–2559. https://doi.org/10.1002/ece3.2675
Cramp S, Simmons KEL (1980) Handbook of the birds of Europe, the Middle East and North Africa, vol II. Oxford University Press, New York, Hawks to Bustards
del Hoyo J, Elliot A, Sargatal J (eds) (1994) Handbook of the Birds of the World. Vol. 2 New World Vultures to Guineafowl. Lynx Edicions, Barcelona
EBCC (2019) Final species classification for 2019 update in Europe and its biogeographical regions. European Bird Census Council, Beek-Ubbergen (NL)
ERSAF (2014) DUSAF 4.0 Destinazione d’Uso dei Suoli Agricoli e Forestali. Regione Lombardia, Milan
ERSAF (2012) Carta dei tipi forestali reali della Lombardia. Regione Lombardia, Milan
ERSAF (2017) DUSAF 5.0 Destinazione d’Uso dei Suoli Agricoli e Forestali. Regione Lombardia, Milan
Fawcett T (2006) An introduction to ROC analysis. Pattern Recogn Lett 27:861–874. https://doi.org/10.1016/j.patrec.2005.10.010
Fox J, Monette G (1992) Generalized collinearity diagnostics. J Am Stat Assoc 87:178–183. https://doi.org/10.2307/2290467
Fox J, Weisberg S (2011) Package car: an R Companion to Applied Regression. www.cran.r-project.org, Wien. Accessed 16 Sep 2021
Gabbert AE, Leif AP, Purvis JR, Flake LD (1999) Survival and habitat use by ring-necked pheasants during two disparate winters in South Dakota. J Wildlife Manage 63:711–722
Gatti RC, Dumke RT, Pils CM (1989) Habitat use and movements of female ring-necked pheasants during fall and winter. J Wildlife Manage 53:462–475
Genovesi P, Besa M, Toso S (1999) Habitat selection by breeding pheasants Phasianus colchicus in an agricultural area of northern Italy. Wildlife Biol 5:193–201
Gottschalk TK, Huettmann F (2011) Comparison of distance sampling and territory mapping methods for birds in four different habitats. J Ornithol 152:421–429. https://doi.org/10.1007/s10336-010-0601-1
Gustin M, Nardelli R, Brichetti P et al (2019) Lista Rossa IUCN degli uccelli nidificanti in Italia 2019. Comitato Italiano IUCN e Ministero dell’Ambiente e della Tutela del Territorio e del Mare, Roma (IT)
Hart CM, Brueggemann SJ, Fien CA (2009) A new perspective and methods for pheasant management. Calif Fish Game 95:1–37
Hermansen JS, Saether SA, Elgvin TO et al (2011) Hybrid speciation in sparrows I: phenotypic intermediacy, genetic admixture and barriers to gene flow. Mol Ecol 20:3812–3822. https://doi.org/10.1111/j.1365-294X.2011.05183.x
Hesselbarth MHK (2021) Package landscapemetrics: Landscape metrics for categorical map patterns. www.cran.r-project.org, Wien. Accessed 16 Sep 2021
Hill D, Robertson P (1988) The Pheasant. Ecology, Management and Conservation. BSP Professional Books, Oxford (UK)
Holá M, Zíka T, Šálek M et al (2015) Effect of habitat and game management practices on ring-necked pheasant harvest in the Czech Republic. Eur J Wildl Res 61:73–80. https://doi.org/10.1007/s10344-014-0875-3
Hutto RL, Young JS (2003) On the design of monitoring programs and the use of population indices: a reply to Ellingson and Lukacs. Wildlife Soc B 31:903–910
Johnsgard PA (1986) The pheasants of the world. Oxford University Press, Oxford (UK)
Johnson DH (2008) In defense of indices: the case of bird surveys. J Wildlife Manage 72:857–868. https://doi.org/10.2193/2007-294
Keating KA, Cherry S (2004) Use and interpretation of logistic regression in habitat-selection studies. J Wildlife Manage 68:774–789. https://doi.org/10.2193/0022-541X(2004)068[0774:UAIOLR]2.0.CO;2
Keller V, Herrando S, Voříšek P et al (2020) European Breeding Bird Atlas 2: distribution, abundance and change. European Bird Census Council & Lynx Edicions, Barcelona
Krebs CJ (1999) Ecological methodology, 2nd ed. Benjamin/Cummings, Menlo Park
Kuehl AA, Clark WR (2002) Predator activity related to landscape features in Northern Iowa. J Wildlife Manage 66:1224–1234
Leif AP (2005) Spatial ecology and habitat selection of breeding male pheasants. Wildlife Soc B 33:130–141
Lennon JJ, Koleff P, Greenwood JJD, Gaston KJ (2003) Contribution of rarity and commonness to patterns of species richness: Richness patterns and rarity/commonness. Ecol Lett 7:81–87. https://doi.org/10.1046/j.1461-0248.2004.00548.x
Leptich DJ (1992) Winter habitat use by hen pheasants in Southern Idaho. J Wildlife Manage 56:376–380
Liu C, Berry PM, Dawson TP, Pearson RG (2005) Selecting thresholds of occurrence in the prediction of species distributions. Ecography 28:385–393. https://doi.org/10.1111/j.0906-7590.2005.03957.x
Madden JR (2021) How many gamebirds are released in the UK each year? Eur J Wildl Res 67:72. https://doi.org/10.1007/s10344-021-01508-z
Madden JR, Perkins SE (2017) Why did the pheasant cross the road? Long-term road mortality patterns in relation to management changes. R Soc Open Sci 4:170617. https://doi.org/10.1098/rsos.170617
Manly BFJ, McDonald LL, Thomas DL et al (2002) Resource selection by animals: statistical design and analysis for field studies, 2nd edn. Kluwer Academic Publishers, Dordrecht
Marques TA, Thomas L, Fancy SG, Buckland ST (2007) Improving estimates of bird density using multiple-covariate distance sampling. Auk 124:1229–1243
Mason LR, Bicknell JE, Smart J, Peach WJ (2020) The impacts of non-naitve gamebird release in the UK: an updated evidence review. RSPB Centre Conserv Sci, Sandy (UK)
McGarigal K, Marks BJ (1994) Fragstat: spatial pattern analysis program for quantifying landscape structure. Oregon State University, Corvallis, OR, USA, Forest Science Department
Meriggi A (1983) Territorialismo, preferenze ambientali e produttività di una popolazione di fagiano. Avocetta 7:1–12
Meriggi A (1992) Fagiano comune. In: Brichetti P, de Franceschi P, Baccetti N (eds) Fauna d’Italia, vol XXIX. Aves 1. Gaviidae-Phasianidae. Calderini Editore, Bologna (IT), pp 824–840
Meriggi A, Pandini W, Cesaris C (1996) Demography of the pheasant in relation to habitat characteristics in northern Italy. J Wildl Res 1:15–23
Mikusiński G, Angelstam P (1997) European woodpeckers and anthropogenic habitat change: a review. Vogelwelt 118:277–284
Miller DL, Rexstad E, Thomas L et al (2019) Distance Sampling in R. J Stat Soft 89:1–28. https://doi.org/10.18637/jss.v089.i01
Morrison ML, Marcot BG, Mannan RW (2006) Wildlife-habitat relationships: concepts and applications, 3rd edn. Island Press, Washington
Mustin K, Newey S, Irvine J et al (2012) Biodiversity impacts of game bird hunting and associated management practices in Europe and North America. The James Hutton Institute, Aberdeen
Nelli L, Meriggi A, Vidus-Rosin A (2012) Effects of habitat improvement actions (HIAs) and reforestations on pheasants Phasianus colchicus in northern Italy. Wildlife Biol 18:121–130. https://doi.org/10.2981/11-022
Norvell RE, Howe FP, Parrish JR (2003) A seven-year comparison of relative-abundance and distance-sampling methods. Auk 120:1013–1028. https://doi.org/10.1093/auk/120.4.1013
Pearce J, Ferrier S (2000) Evaluating the predictive performance of habitat models developed using logistic regression. Ecol Model 133:225–245. https://doi.org/10.1016/S0304-3800(00)00322-7
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Wien
Rands MRW (1986) The survival of gamebird (Galliformes) chicks in relation to pesticide use on cereals. Ibis 128:57–64
Riley TZ, Clark WR, Ewing E, Vohs PA (1998) Survival of ring-necked pheasant chicks during brood rearing. J Wildlife Manage 62:36–44
Riley TZ, Schulz JH (2001) Predation and ring-necked pheasant population dynamics. Wildlife Soc B 29:33–38
Robertson PA, Mill AC, Rushton SP et al (2017) Pheasant release in Great Britain: long-term and large-scale changes in the survival of a managed bird. Eur J Wildl Res 63:100. https://doi.org/10.1007/s10344-017-1157-7
Ronnenberg K, Strauß E, Siebert U (2016) Crop diversity loss as primary cause of grey partridge and common pheasant decline in Lower Saxony. Germany BMC Ecol 16:39. https://doi.org/10.1186/s12898-016-0093-9
Rushton SP, Ormerod SJ, Kerby G (2004) New paradigms for modelling species distributions? J Appl Ecol 41:193–200. https://doi.org/10.1111/j.0021-8901.2004.00903.x
Sands JP, DeMaso SJ, Schnupp MJ, Brennan LA (2012) Wildlife science: connecting research with management. CRC Press, Boca Raton, FL
Schmitz RA, Clark WR (1999) Survival of Ring-necked Pheasant hens during spring in relation to landscape features. J Wildlife Manage 63:147–154
Sinclair ARE, Fryxell JM, Caughley G, Caughley G (2006) Wildlife ecology, conservation, and management, 2nd edn. Blackwell Publishing, Malden, MA, Oxford
Sing T, Sander O, Beerenwinkel N, Lengauer T (2007) Package ROCR: Visualizing the performance of scoring classifiers. www.cran.r-project.org, Wien. Accessed 16 Sep 2021
Smith SA, Stewart NJ, Gates JE (1999) Home ranges, habitat selection and mortality of Ring-necked Pheasants (Phasianus colchicus) in North-Central Maryland. Am Midl Nat 141:185–197
Sutherland WJ (2006) Ecological census techniques: a handbook, 2nd edn. Cambridge University Press, Cambridge
Thomas L, Buckland ST, Rexstad EA et al (2010) Distance software: design and analysis of distance sampling surveys for estimating population size. J Appl Ecol 47:5–14
Tillé Y, Matei A (2016) Package sampling: Survey Sampling. www.cran.r-project.org, Wien. Accessed 16 Sep 2021
Warner RE, Joselyn GB (1986) Responses of Illinois ring-necked pheasant populations to block roadside management. J Wildlife Manage 50:525–532
Wasilewski M (1986) Territories of pheasant cocks in breeding season. Ekol Pol 34:681–688
Whiteside RW, Guthery FS (1983) Ring-necked Pheasant movements, home ranges, and habitat use in West Texas. J Wildlife Manage 47:1097–1104
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems: Data exploration. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Zuur AF, Ieno EN, Smith GM (2007) Analysing Ecological Data. Springer Science + Business Media, LLC, New York
Acknowledgements
We would like to thank Arianna Bazzocchi, Kati Sara Bovo, Martina Lucchelli, Arianna Musacchio, and Zeno Porro for their help during the fieldwork.
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Gianpasquale Chiatante. The first draft of the manuscript was written by Gianpasquale Chiatante and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This research was conducted with ethical approval from the University of Pavia (Department of Earth and Environmental Sciences). Bird surveys were conducted with permission from local landowners where necessary. Data collection did not involve sampling procedure, and experimental manipulation of birds and the fieldwork was conducted under the Law of the Republic of Italy on the Protection of Wildlife (February 25, 1992).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chiatante, G., Meriggi, A. Habitat selection and density of common pheasant (Phasianus colchicus) in Northern Italy: effects of land use cover and landscape configuration. Eur J Wildl Res 68, 26 (2022). https://doi.org/10.1007/s10344-022-01575-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-022-01575-w