Abstract
The use of intensive silviculture to increase forest productivity in commercial plantations will undoubtedly persist into the future. This review synthesizes and analyses studies that have investigated the influence of fertilization on density, microfibril angle (MFA), and modulus of elasticity (MOE) of different Pinus species under intensive management. It was found that in general, ring width or growth rate demonstrated a negative correlation with density, and a positive relationship with MFA. Also, the relationship between growth rate and density and MFA were age specific and often non-significant at the whole stem level. Earlywood and latewood were influenced differently by fertilization: Earlywood width and density increased while in latewood, width stayed constant but density decreased. This resulted in a decrease in within-ring heterogeneity of density and density-related properties. Since latewood generally contributed more to whole-ring density, the net effect was that whole-ring wood density of pines mostly decreased with fertilization. Given that fertilization often resulted in a decrease in wood density and an increase in MFA, MOE decreased with fertilization. Most fertilization-wood-property studies on Pinus involved nitrogen and phosphorus fertilizers with differing quantity and ratios. Results suggest that nitrogen may cause the greatest change in wood growth and properties. An important question that was often unanswered in research, was whether the effect of specific fertilization treatments on wood properties were purely based on the influence of increased growth or whether there were direct influences not explained by growth. Also, despite potassium being an important elemental component of many fertilizers, relatively little work has been done to determine its effect on wood properties.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, silviculture in commercial plantation forests has developed from the conventional extensive tending of trees into intensive forest management (Schimleck et al. 2018; Carter et al. 2021). Intensive management involves the manipulation of forest lands with productive potential to achieve the highest possible yields (Irland 2011; Shmulsky and Jones 2019; Ameray et al. 2021). Indeed, literature report thriving intensively managed forests around the world such as in Southeastern United States (Samuelson et al. 2004; Love-Myers et al. 2009; Jokela et al. 2010; Munsell and Fox 2010; Campoe et al. 2013; Albaugh et al. 2021); New Zealand (Davis et al. 2015; Moore and Clinton 2015; Smaill and Clinton 2016; Burdon and Moore 2018; Clinton 2018); South America (Will et al. 2015; Barros Filho et al. 2017; Topanotti et al. 2021; Pereira et al. 2022); Australia (Davis et al. 2015) Nordic countries (Hedwall et al. 2014; Ameray et al. 2021; Smolander et al. 2022); and South Africa (Campion 2008; du Toit et al. 2010; Kotze and du Toit 2012). These studies demonstrate that the increased growth rates achieved through intensive silviculture has led to a reduction in rotation age.
Intensive silviculture adopts both direct and indirect practices to influence the growth rate of trees (Irland 2011; Mäkinen and Hynynen 2012; Burdon and Moore 2018). Indirect practices include those that manage stocking such as site preparation, initial spacing and thinning. On the other hand, direct practices include those that alter the soil nutrition and uptake such as fertilization and irrigation. Unlike the other practices, fertilization has gathered significant attention of both forest researchers and managers for two reasons. First for the influence it has on productivity (Campion 2008; du Toit et al. 2010; Trazzi et al. 2019). It moved from being a good practice at establishment to being prescribed as a pre-requisite in plantations (Fox et al. 2007a, b; Burdon and Moore 2018; Loewe-Muñoz et al. 2020). In many silvicultural regimes, fertilization is implemented not just to ameliorate deficiencies but to maximize growth of trees too (Smaill and Clinton 2016; Zhao et al. 2016; Clinton 2018). Secondly, fertilizer application is known to increase growth rate with often negative effects on the mechanical properties of wood (Kennedy 1995; Barrette et al. 2023).
The shift to increased productivity has been to ensure that forestry competes favorably with alternative land uses in meeting wood demands of a rapidly increasing global population (du Toit et al. 2010; Moore et al. 2012; Moore and Clinton 2015; Clinton 2018). Recent projections postulate that the global construction-driven demand for roundwood will more than triple by 2030 (FAO 2022). This implies that the use of intensive silviculture to achieve faster growth will most certainly persist into the future and so will the use of fertilizers (Barrette et al. 2023). It is, however, important for forest nutritionists to understand responses to different but also increasing nutrient supply scenarios (Kotze and du Toit 2012; Moore and Cown 2015). The research in fertilizer application and its effect on wood properties has evolved and forest managers are required to adapt to the changing environment.
This paper is a review that focuses on synthesis and analysis of studies that have investigated the influence of fertilization on properties of different Pinus species under intensive management. The deliberate focus has been on Pinus species because they are usually managed to provide timber for structural end-uses. Additionally, silvicultural practices such as fertilization tend to negatively impact properties that determine quality of structural products. The core objective is to provide pointers for research and highlight the possibility of adjusting fertilizer practices to achieve high-quality wood and growth rate in coordination with other silvicultural practices. Specific search terms relating to the species, properties of interest and silvicultural treatment were used in different databases to obtain the first set of publications. After that, trickle-up and trickle-down approaches were adopted as executed by (Dickinson et al. 2019). Many of the studies reviewed have been conducted on loblolly and slash pine in Southeastern United States; and radiata pine in New Zealand and Australia but effort has been made to find relevant studies in other intensively managed pine plantations around the world.
The paper starts by contextualizing the wood properties that form the basis of the discussions and how they are impacted by growth rate. It then discusses fertilization practices with a particular focus on the growth trends, elemental composition and formulations, rates, and frequency of application. This is followed by the effects of application on density, microfibril angle and Young’s modulus or modulus of elasticity (MOE, also referred to as stiffness). Before the concluding section, a brief review is conducted on how fertilization has been synchronized with other silvicultural practices and how this has impacted wood properties. In the conclusion, a synthesis is provided with an emphasis on how fertilizer application practices can be adapted to ensure that the targeted properties are either preserved or improved while productivity is boosted.
Growth rate and wood properties
The discussions on growth-rate and wood properties have focused on three key concepts. Firstly, increase in the proportion of juvenile wood in structural products. This has been attributed to an interaction of accelerated growth and reduction in rotation age (Erasmus et al. 2020; Kennedy 1995; Long et al. 2004; Clark III et al. 2008; Antony et al. 2012; Carson et al. 2014; Butler et al. 2015; Auty et al. 2016; Dahlen et al. 2020). Where fertilization occurred soon after planting (as is most often the case), juvenile wood proportion will be maximized, especially at the base of the tree stem. Juvenile wood is undesirable because it has inferior structural properties (Amarasekara and Denne 2002; Moore and Cown 2017). Secondly, increased growth rate directly alters type, number, size, shape, physical structure, and chemical composition of the wood elements (Makinen and Hynynen 2014; Rocha et al. 2019; Shmulsky and Jones 2019). These changes occur in patterns that are often detrimental to important properties, including density (specific gravity) and microfibril angles (MFA), which are key determinants of strength and stiffness (mechanical properties). Thirdly, increased growth rate increases heterogeneity of some within and between annual growth ring components of earlywood and latewood (Peltola et al. 2007; Shmulsky and Jones 2019) and high heterogeneity has long been identified as one of the greatest quality problems facing wood utilization industries (Zobel 1992). However, literature was not consistent, and some workers reported that in some cases there are certain positive externalities that can be harnessed with fertilizer application besides increased productivity. For instance, it has been reported that fertilization can lead to greater within-ring uniformity of density by increasing cell wall thickness in earlywood while consequently thinning latewood cell walls (Zobel 1992). Also, adjusting fertilizer formulations (Wessels et al. 2015; Shmulsky and Jones 2019) and soil moisture could achieve quality wood with appreciable growth rates (du Toit 2006; Kotze and du Toit 2012; Hans 2013; Fischer and du Toit 2019).
Variation of properties
A considerable amount of literature has investigated wood properties. These studies have established that important determinants for ultimate use in structures include Young’s modulus or modulus of elasticity and modulus of rupture (MOR, bending strength). The most useful of these is stiffness, which defines the amount of deformation that wood undergoes when subjected to a load (Antony et al. 2011a, b, c, d, 2012; Wessels et al. 2014). In fact, it has been suggested that for several Pinus species such as Pinus radiata and Pinus patula, stiffness is a common predictor of timber suitability for construction and can be measured non-destructively (Antony et al. 2009a; b; Wessels et al. 2014; Davis et al. 2015; Wessels and Petersen 2015). Also, there is broad consensus among researchers that the variation in stiffness is mainly due to the combined influence of density (specific gravity) and MFA (Downes et al. 2002a, b; Xu and Walker 2004; Donaldson 2008; Ivković et al. 2009; Lasserre et al. 2009; Antony et al. 2011a, b, c, d, 2012; Auty et al. 2013; Wagner et al. 2013; Moore et al. 2015; Salmén 2018).
Wood properties vary between and within trees under the influence of factors related (but not limited) to genetic, environmental, or silvicultural practices. In addition, proliferation of knowledge on juvenile and mature wood has highlighted the significance of relating patterns of variation in properties to cambial age (where the cambial age is effectively a surrogate for prevailing nutritional, hormonal and mechanical factors affecting wood formation at a specific point in the tree stem). For most softwoods, properties change rapidly across the juvenile wood zone, followed by a levelling off in the mature wood zone (Amarasekara and Denne 2002; Auty et al. 2013; Moore and Cown 2017; Kidombo and Dean 2018). The radial pattern for density of a majority of Pinus spp. is demonstrated by either an increase with cambial age or a decrease for the first few years before increasing with cambial age (Antony et al. 2011a, b, c, d; Mäkinen and Hynynen 2012; Wagner et al. 2013; Auty et al. 2014; Barrios et al. 2017; Schimleck et al. 2022).
On the other hand, radial variation in MFA is characterized by a decrease from pith to bark at all heights along the stem (Donaldson 2008; Antony et al. 2011a, b, c, d; Auty et al. 2013; Moore et al. 2015; Barrios et al. 2017). Some studies have noted that these changes are more pronounced in the juvenile wood zone (Evans et al. 2001; Xu et al. 2004). Along the tree axis there is an exponential decrease in MFA from pith to bark in the lower trunk, typically up to 7 m, beyond which the rate decreases to more stable values (Donaldson 2008; Moore et al. 2015). There is low radial variation for most part of the stem before increasing near the top of the stem. Simply put, as the tree height increases, variability in MFA closer to the pith is less than in the lower sections of the stem. According to a study on P. radiata, MFA at breast height stabilized around the 20th annual ring, but at 8 m, stable values were obtained at the 10th annual ring (Donaldson 2008).
Stiffness increases from pith to bark (Xu and Walker 2004; Ivković et al. 2009; Antony et al. 2011a, b, c, d; Wagner et al. 2013; Barrios et al. 2017). Along the tree axis, however, stiffness increases towards the crown in the juvenile wood zone, while a decrease has been observed in the mature wood zone (Antony et al. 2011a, b, c, d). Several studies that have investigated variation in stiffness have used density and MFA as proxies. When considered individually, MFA has a higher influence on bending stiffness than density especially in the juvenile wood zone (Alteyrac et al. 2006; Auty et al. 2013; Wessels et al. 2015). As such, the properties of interest in subsequent sections of this paper will be wood density, MFA and MOE.
Influence of growth rate on wood properties
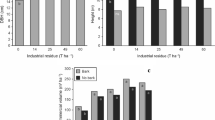
Despite the existing complexities in the relationship between wood properties and growth rate (Zobel and Buijtenen 1989; Zobel 1992; Love-Myers et al. 2009), several attempts have been made to explain the influence of growth rate using ring width as a proxy (Mäkinen and Hynynen 2012; Auty et al. 2013, 2014; Moore et al. 2015; Dahlen et al. 2018; Rodriguez and Tomazello-Filho 2019). Some studies have attempted to explain how different properties interrelate (Downes et al. 2002a, b; Ivković et al. 2009) and a path analysis conducted by Ivkovic et al. (2009) demonstrated how MOE relates to density, MFA, spiral grain and ring width (Fig. 1).
An illustration of the use of path analysis to explain direct effects (straight lines- solid represents significant effect) and indirect (curves—solid represents significant correlation) paths between different wood properties including ring width (RW), density (DEN), microfibril angle (MfA), spiral grain (SLG) and modulus of elasticity (MoE) modified from (Ivković et al. 2009)
Generally, density demonstrates a negative correlation with ring width (Saarsalmi and Mälkönen 2001; Mäkinen and Hynynen 2012; Auty et al. 2014; Kimberley et al. 2015; Rodriguez and Tomazello-Filho 2019). For MFA, a positive correlation has been reported (Auty et al. 2013; Moore et al. 2015; Barrios et al. 2017). It is important to note that cambial age has an influence on the variation of wood properties (Schneider et al. 2008; Ivković et al. 2013; Kuprevicius et al. 2013; Barrios et al. 2017). A study on jack pine in Canada reported that ring width did not affect the density of wood from trees less than 5 years old (Schneider et al. 2008). Similarly, (Barrios et al. 2017) found no correlation between ring width and properties in juvenile wood of P. radiata; yet, significant correlations were present in mature wood. For MOE, however, significant negative correlations with ring width were reported for young P. radiata (Lasserre et al. 2009) and P. patula (Wessels et al. 2014) trees.
Whereas several studies report non-significant impacts of growth rate (ring width) on wood properties at the whole stem cross-section (Auty et al. 2013, 2014; Makinen and Hynynen 2014; Kimberley et al. 2015; Moore et al. 2015; Barrios et al. 2017; Kohler et al. 2019), noticeable changes are reported at the ring level. The changes at ring level are closely related to the difference in properties in earlywood and latewood (Schneider et al. 2008). Thus, to gain clearer understanding of how growth rate affects wood properties, it is better to focus on how changes in ring width are translated in annual growth rings with careful consideration and knowledge of the causal factor. To this end, the reader is referred to a detailed schematic provided by Downes et al. (2002a, b:45).
Fertilization practices
In most softwood plantations, fertilizers are applied at establishment and or in synchronisation with other practices during the rotation period (Campion 2008; Antony et al. 2009a, b; Albaugh et al. 2017, 2021; du Toit and Scheepers 2020; Carter et al. 2021). At establishment, seedlings are most vulnerable to external factors and the demand for nutrients uptake from the soil is relatively high (Miller 1984). However, due to the decomposition of litter from the previous rotation, it is not mandatory to plant with fertilizer unless that specific site requires it (Albaugh et al. 2015; Zhao et al. 2016). Thus, balancing nutrient needs with soil supply potential is a key component of good establishment practices that is implemented when required (Kotze and du Toit 2012; Albaugh et al. 2015). After establishment, many studies report that the decision to fertilize is based on foliar nutrient concentrations or some other indicator of soil nutrient supply potential (Louw and Scholes 2003; Campion 2008; Kotze and du Toit 2012; Hedwall et al. 2014; Ramírez Alzate et al. 2016; du Toit and Scheepers 2020; Albaugh et al. 2021; Shephard et al. 2021). After all, growth to canopy closure usually outweighs the soil’s ability to meet nutrient requirements and this causes semi-mature stands to exhibit reduced growth rates (Miller 1984; du Toit and Scheepers 2020). For this reason, mid-rotation fertilization has been widely practiced to supplement the soil with the necessary nutrients (Antony et al. 2009a; b; Love-Myers et al. 2010; du Toit and Scheepers 2020; Carter et al. 2021). As foliar content analyses are prone to sampling and interpretation errors, Rubilar et al. (2018) suggested that it is important to complement them with leaf area analysis to ensure more accurate fertilization recommendations at mid-rotation.
However, no clear definitions and qualifications of plantations as mid-rotation in terms of age have been established (Campion 2008). Some studies considered all plantations between 5 and 14 years as mid-rotation (Jokela and Long 2018; Shephard et al. 2021; Hackman et al. 2022). This ambiguity in mid-rotation ages is understandable because plantations are established and managed for different objectives. In their detailed review of 50-years of fertilization practices in Southeastern United States, all plantations between 3 and 25 years old were considered mid-rotation (Albaugh et al. 2018). This was on the premise that all plantations within this age bracket respond similarly to fertilization (Albaugh et al. 2015).
Elemental composition
The quantity and elemental composition of fertilizers applied depends, among others, on existing site deficiencies and species requirements. As such, different localities may have different requirements of growth nutrients. Studies from intensively managed plantations have established that nitrogen (N), phosphorus (P) and potassium (K), individually or in combination, are the nutrients that most commonly limit growth in forest soils (Barrette et al. 2023). In the southeastern United States, which has the most intensively managed loblolly and slash pine plantations in the world, mostly combinations of N and P have been applied (Fox et al. 2007a, b; Zhao et al. 2016; Albaugh et al. 2018; Carter et al. 2021). Similarly, deficiencies of N and P have been reported to limit growth of radiata pine in Australia and New Zealand (Nyakuengama et al. 2002; McGrath et al. 2003; Davis et al. 2015; Burdon and Moore 2018; Clinton 2018). In the Northern Scots pine plantations, however, N is the major limiting factor and N-rich fertilizers are common (Saarsalmi and Mälkönen 2001; Hedwall et al. 2014; Smolander et al. 2022). Conversely, in South Africa, the growth of pine plantations have been mainly limited by P and K (Campion 2008; Crous et al. 2009; du Toit et al. 2010; Kotze and du Toit 2012) although N is also applied in some regions (Campion 2008; Chikumbu 2011; du Toit and Scheepers 2020). It is critical to note that the addition of macro-nutrients often accentuates the deficiencies of other nutrients. This has been reported for magnesium and boron in New Zealand (Clinton 2018) and the northern forests (Smolander et al. 2022), zinc in Australia (Davis et al. 2015) and magnesium in South Africa (Kotze and du Toit 2012).
The majority of forest fertilization research has been conducted with combined treatments of N and P. Plant responses to N have been mostly classified as short-term (Long et al. 2004; Ameray et al. 2021). P, on the other hand, can improve site qualities over a longer period (Snowdon 2002; Long et al. 2004; Fox et al. 2007a, b; Antony et al. 2011a, b, c, d). The application of N and P, thus, meets both short- and long-term needs of the forest plantation. Additionally, when N and P are applied together, higher growth was obtained than each individually, on specific sites (Fox et al. 2007a, b; Ramírez Alzate et al. 2016; du Toit and Scheepers 2020). This is explained by the idea that balancing the two nutrients promotes their synergistic relationship where abundance of P in soil increases N uptake by plants (McGrath et al. 2003; Campion 2008). The synergy is corroborated in a recent study which reported that the application of N and P together quadrupled the growth in comparison to N applied individually at the same rate (Albaugh 2021). Additionally, in Southern Africa, fertilization with only P is common because of the highly weathered, and acid soils (Payn et al. 1988; Zwolinski et al. 2002; Kotze and du Toit 2012). In these regions, N mineralisation rate together with pH and carbon contents of soils have been used to predict the additional responses to fertilization with N in the presence of P (Donald 1987; du Toit and Scheepers 2020).
Given that the decision to fertilize is contextualized based on sites and stand requirements, as well as soil water availability, it is impractical to make generalisations based on quantities. This review, thus, adopted ratios to represent the proportions of the elements in fertilizer formulations. Optimum ratios of N and P fertilizers have been suggested over the years. A 10:1 ratio was suggested for conifers by (Ingestad 1979) and was popularized by (Linder 1995). More recently, it has also been adopted by Moilanen et al. (2015). Additionally, (Fox et al. 2007a, b) introduced an 8:1 ratio that has been adopted as a global reference in most intensively managed pine plantations. However, considering that nutrient deficiencies are localized, several N:P ratios have been examined (Liechty and Fristoe 2013; Gyawali and Burkhart 2015; Haveraaen and Frivold 2015). Recent research, however, recommends that the quantity of P applied should be equivalent to 10% of total N applied—a N:P ratio of 10:1 (Albaugh et al. 2018).
Despite being an essential element in forest nutrition, little research has been done on K and on its effect on growth and properties beyond amelioration (Davis et al. 2015; Hackman et al. 2022). In fact, according to some studies, K is usually added as a base element with other micronutrients such as boron, depending on site-specific requirements (Carlson et al. 2014; Ramírez Alzate et al. 2016). This is substantiated by Albaugh et al. (2021) who report K to have been added only when foliar concentrations showed insufficiency. This potentially explains the lack of information on the specialized effects of K or its proportions in relation to N and P. This notwithstanding, the existing studies on K point to the fact that it plays a significant role in drought resistance and ensuring that plants survive environmental stresses (Ingestad 1979; Zörb et al. 2014; Salazar-Tortosa et al. 2018). A study by Fromm (2010) reported that potassium was found to accumulate in the latewood cells of Norway spruce suggesting K as a player in formation of latewood.
Furthermore, Wessels et al. (2015) also suggested that the N:K ratio may play a key role in determining cell wall thickness and transitions from earlywood to latewood, thus pointing to stiffer and stronger solid wood. To ascertain the recommended proportions of K, reference is made to studies that applied NPK fertilizers or declared recommended quantities of K in case deficiencies were detected. NPK is usually applied at a ratio of 8:1:1.5–2.6 for Slash and Loblolly Pine in Southeastern US (Hackman et al. 2022); at 2.7:1:1.3 for radiata pine in New Zealand (Davis et al. 2015); and a 7.5:1:2.3 in boreal coniferous stands (Jacobson and Pettersson 2010). In South Africa, K is applied in equal proportion with P in both early and mid-rotation plantations on specific site types that are targeted for sawlogs (Campion 2008; Crous et al. 2009; du Toit et al. 2010; Kotze and du Toit 2012).
Fertilization, growth, and wood properties
Duration of response
The impacts of fertilization on growth, and consequently, wood properties are reported to only last a certain number of years after application (Haveraaen and Frivold 2015; Albaugh et al. 2021). Research points to N causing the greatest change in wood growth and properties and these changes are proportional to the quantities of N supplied per unit area (Zobel 1992; Fox et al. 2007a, b; Munsell and Fox 2010; Antony et al. 2011a, b, c, d; Shmulsky and Jones 2019). For instance, research conducted on Loblolly pine with three N treatments (112, 224 and 336 kg N/ha) reported that the 224 N and 336 N treatments had longer response durations than the 112 kg N/ha treatment (Antony et al. 2009a, 2013; b; Albaugh et al. 2021). Also, because N is associated with short-term responses, the effective duration is usually capped at a decade (Long et al. 2004; Fox et al. 2007a, b; Albaugh et al. 2021). An earlier study asserted that the effect of mid-rotation fertilization is usually limited to maximum periods between 8 and 10 years, and this is corroborated by studies that reported a response duration for loblolly pine of 8 years (Fox et al. 2007a, b; Albaugh et al. 2021). Furthermore, periods between 2 and 3 years have been reported for slash pine that received 224 kg N/ha (Love-Myers et al. 2009), 5 years for radiata with 200 kg N/ha (Nyakuengama et al. 2002), and 7–11 years for Scots pine that received 150 kg N/ha (Saarsalmi and Mälkönen 2001; Jacobson and Pettersson 2010; Hedwall et al. 2014; Smolander et al. 2022). However, a recent study reported a response period of 15 years after the last application of N (Smolander et al. 2022).
Responses to P application are usually longer lived than N and K because it is effectively retained in forest ecosystems (Zwolinski et al. 2002; Long et al. 2004; Fox et al. 2007a, b; Crous et al. 2009; Antony et al. 2011a, b, c, d; Kotze and du Toit 2012). Studies show that P produces responses for over 20 years (Everett and Palm-Leis 2009; Davis et al. 2015; Hackman et al. 2022) with potential to affect growth even in subsequent rotations (Everett and Palm-Leis 2009). On poor soils, however, this response period can be shorter and more applications may be required (Davis et al. 2015).
The type of fertilizer and method of application also influence the duration of response (Gyawali and Burkhart 2015; Carter et al. 2021). Usually, broadcasting leads to longer response periods than direct applications to the soil (Davis et al. 2015). Further, slow and controlled release fertilizers may produce responses over longer periods than conventional fertilizers. This was demonstrated in a study which reported that 150 kg N/ha of conventional fertilizer led to sharp increase in growth that lasted 7 years while an equal amount of controlled release fertilizer caused a much slower growth increment that lasted 10 years longer (Smolander et al. 2022). Owing to the fact that the response durations of fertilizer vary greatly, researchers and forest plantation managers rely on routine nutrient assessments (Campion 2008; Albaugh et al. 2021). After the duration of response, tree growth returns to pre-treatment levels, however, impact on properties of the wood formed is permanent (Long et al. 2004; Rodriguez and Tomazello-Filho 2019).
Although frequency of fertilization was considered an important factor by earlier researchers, interest in recent years is shifting to the cumulative amount applied within the rotation (Rubilar et al. 2018; Carter et al. 2021). Moreover, it has been established that similar growth responses of fertilization can be achieved by either applying multiple small doses or one large dose. This happens if the cumulative application rates are comparable and the response time sufficient for full utilization of the applied fertilizer (Albaugh et al. 2015, 2018). Not only is this finding valuable for adapting forest operations, but it can also potentially be used to influence properties of the wood produced (Saarsalmi and Mälkönen 2001; Trazzi et al. 2019).
Fertilization influences on wood properties.
In eucalypts, an increase in wood density and pulpability factor has often been observed with fertilization of short-rotation crops (DeBell et al. 2001; du Toit et al. 2001). In contrast to this, wood properties in pines are generally negatively affected by fertilizers within the response period (Downes et al. 2002a, b; Antony et al. 2009a, b; 2013; Makinen and Hynynen 2014; Moore et al. 2021). The magnitude of the effect is usually dependent on the quantity and frequency of fertilizer applied—especially N (Zobel 1992; Beets et al. 2001; Fox et al. 2007a, b; Crous et al. 2009; Munsell and Fox 2010; Antony et al. 2011a, b, c, d; Shmulsky and Jones 2019). The change in properties is often only practically meaningful if large doses of fertilizer are applied and are proportional to increase in ring width. (Saarsalmi and Mälkönen 2001; Rodriguez and Tomazello-Filho 2019).
Fertilization of pine stands has been reported to generally reduce wood density (Campion 2008; Love-Myers et al. 2009, 2010; Pereira et al. 2022). A long-term fertilization study on mature Scots pine reported a 4% reduction in wood density (Makinen and Hynynen 2014). In this study 150: 75: 75 kg/ha of NPK was applied at five-year intervals for 30 years. Another study on the same species maintained a 1.3% foliar nitrogen concentration for 8 years and analyzed juvenile and mature sections of the core separately (Love-Myers et al. 2010). They reported a 5% reduction in juvenile wood density versus a 9% reduction in mature wood. Under similar nutrient conditions and species, another study reported an average reduction of 7.5% in density of an 8-year plantation monitored for 9 years (Albaugh et al. 2004). Application of 58:283:113 kg/ha of NPK to a 12 years loblolly pine reduced density by up to 10% (Clark III et al. 2004). Similarly, a 10% decrease in density was reported in the 6th year after fertilization of 17-year loblolly pine with compost pulp mill sludge (Rodriguez and Tomazello-Filho 2019). In another investigation, 13-year-old loblolly pine was fertilized with maximum of 336 kg/ha of N and four annual rings post treatment was analyzed (Antony et al. 2009a; b). The authors reported that the average density in these rings was significantly lower than the unfertilized trees by 8% and lasted a maximum of 3 years after fertilization before returning to pre-treatment values. Another study that also applied 336 kg/ha of N to thinned and un-thinned mid-rotation loblolly pine observed a 0.062 g/cm3 decrease in latewood density and consequently ring density (Antony et al. 2011a, b, c, d). Furthermore, application of 80:80 kg/ha of P:K per ha of P. patula across a 15-year-rotation lead to a 3% reduction in wood density (Crous et al. 2009).
The changes in density due to fertilization are driven by the differences in effects on earlywood and latewood but is more pronounced in the latter (Antony et al. 2011a, b, c, d; Makinen and Hynynen 2014; Barrios et al. 2017; Dahlen et al. 2018; Shmulsky and Jones 2019). Some studies revealed that fertilization increased the width of earlywood and the thickness of cell walls therein (Antony et al. 2009a, b; Ulvcrona and Ahnlund Ulvcrona 2011; Shmulsky and Jones 2019). If considered individually, this change will usually be perceived as positive as it increases the earlywood density. This, however, is countered by the subsequent changes in the latewood: the thinning of cell walls without necessarily affecting latewood width (Clark III et al. 2004; Love-Myers et al. 2009, 2010; Makinen and Hynynen 2014; Shmulsky and Jones 2019; Eberhardt et al. 2022). This means that it is the reduction in latewood density caused by fertilization which in turn influences the whole ring density. Thus, the decrease in annual ring density in fertilized trees is driven by the decrease in latewood wood density rather than a lesser amount of higher density latewood (Clark III et al. 2004; Antony et al. 2009a, b; 2011a, b, c, d; Davis et al. 2015). On the flip side, this opposite effect of fertilization on cell-walls in earlywood and latewood has a positive effect of increasing uniformity in ring density (Zobel 1992; Shmulsky and Jones 2019). A study on Pinus maximinoi grown in South Africa demonstrated that uniformity in density not only enhances machining and finishing properties of boards but also improves the predictability of their behavior boards while in use (Malan 2006).
Latewood is one of the most important factors determining wood quality, and differences in MOE and density are usually driven by the characteristics of latewood (Barrios et al. 2017). However, this effect is said to be less pronounced in the juvenile wood zone and a study found that latewood percentage determined ring density largely for rings that were over 7 years old in jack pine (Schneider et al. 2008). This was supported by another study which reported that the effects of fertilization and irrigation on latewood was more significant when the trees were producing mature wood in loblolly pine trees (Love-Myers et al. 2010). For some species of pine such as P. radiata and P. elliottii, trees start producing mature wood by the 10th annual growth ring (Moore and Cown 2017).
The reduction in the latewood wood density is caused by the decrease in thickening of the cell walls after fertilization, which is attributed to the interaction between foliage-phytohormones-carbohydrate levels (Clark III et al. 2004; Antony et al. 2009a, b; Luo and Li 2022). Although auxins, cytokinins and brassinosteroids are listed as the main phytohormones controlling cambial activity, the differences exhibited in morphology of earlywood and latewood cells are attributed largely to the concentration of auxin (Traversari et al. 2022). Auxins control secondary cambial growth in plants based on a concentration gradient that varies with distance from the live crown (Uggla et al. 1996; Kijidani et al. 2014) and cell differentiation stages (Fajstavr et al. 2018). Auxin levels have been found to be higher in the upper trunk and in zones of active cell division and lower in the lower trunk and in mature xylems. This distinguishes the crown as the source of control of growth and wood development—high auxin levels induce formation of cells with large lumen and thin cell walls. Especially in juvenile trees that have more variability in crown dimensions, quality of wood produced can sometimes be predicted using crown metrics (Kuprevicius et al. 2013). This confirms why silvicultural practices that increase crown dimensions such as fertilization potentially affect wood properties negatively.
Some studies, however, have reported no net effect of fertilization on wood properties especially when effects are either averaged beyond the reaction period or analysed at the whole stem cross-section (Auty et al. 2013, 2014; Makinen and Hynynen 2014; Kimberley et al. 2015; Moore et al. 2015; Barrios et al. 2017; Kohler et al. 2019). For instance, Makinen and Hynynen (2014) reported that fertilization enhanced growth without major negative changes to wood properties of Scots pine. This was supported by a study which reported no significant change in density of a 15-year loblolly pine years after application of 227:28 kg/ha of N:P (Schulte et al. 2020).
Few investigations have focussed on the effect of fertilization on MFA, however, in those where MFA was considered, fertilization generally resulted in an increased average MFA (Downes et al. 2002a, b; Antony et al. 2009a, b). A 2.3° increase in annual average MFA was reported during the 5 years after fertilization of a 14 years old P. radiata with 200:100:100 kg/ha of NPK (Downes et al. 2002a, b). An increase of 3° was reported in 33-yr loblolly pines that received 336 kg/ha of N (Antony et al. 2009a; b). Although the negative correlation with ring width can be used as a proxy (Auty et al. 2013; Moore et al. 2015; Barrios et al. 2017), more research is needed to explore MFA variation with different prescriptions of fertilizers. This is even more relevant considering that MFA in some species has a stronger influence on MOE than density.
Wood produced immediately after fertilization has been reported to have lower stiffness values (Moore et al. 2021). A 12% reduction was reported for 5 years after fertilization of a 14-year-old P. radiata with 200:100:100 kg/ha of N:P:K (Downes et al. 2002a, b). A similar percentage was reported for a 33-year-old loblolly pine treated with an 336:28 kg/ha of N:P (Antony et al. 2009a, 2013; b). Some studies suggest that effects of fertilizer are more pronounced on the formation of juvenile wood (Love-Myers et al. 2010) (Clark III et al. 2004). However, reductions in stiffness have also been reported in the mature wood zone although not to levels that significantly affect the quality of the wood for structural use. (Downes et al. 2002a, b; Clark III et al. 2004; Antony et al. 2009a, b). For instance, a small but significant reduction in the MOE was reported for 14-year-old radiata pine in Australia (Downes et al. 2002a, b) while 17 years old radiata pine in New Zealand did not have any alterations in MOE (Watt et al. 2008). Although this could be attributed to site effects, it can be safely concluded that in the juvenile wood zone, changes in density and MFA gives a clear representation of variation in MOE (Donaldson 2008; Ivković et al. 2009; Lasserre et al. 2009; Antony et al. 2011a, b, c, d, 2012; Auty et al. 2013; Wagner et al. 2013; Moore et al. 2015; Salmén 2018). Also, Ulvcrona and Ahnlund Ulvcrona (2011) argued that the effect of fertilization in juvenile wood is linked strongly to ring width.
Combined effect of fertilization and other silvicultural practices on wood properties
Thinning
Applying fertilizer after thinning is a common practice in commercial pine plantations. Thinning reduces both the basal and leaf area of a plantation which decreases competition and enhances the responsiveness of the remaining trees to fertilization (Long et al. 2004; Fox et al. 2007a, b). Additionally, fertilization is reported to increase growth of the larger diameter trees more than those of smaller diameters (Campoe et al. 2013; Liechty and Fristoe 2013). Fertilization serves to overcome nutrient limitations with the overall effect of reducing the time it takes for a stand to regain maximum wood production. This is achieved by providing the required nutrients that result in accelerating post-thinning diameter growth (Campion 2008; Antony et al. 2009a, b; Carter et al. 2021). Thinning alone is reported to increase radial growth without significantly altering properties such as density, but, the interaction between fertilization and thinning reduces density (Clark III et al. 2008; Antony et al. 2009a, b; Ulvcrona and Ahnlund Ulvcrona 2011; Li et al. 2021). Fertilization and thinning reduced density by up to 8% and this reduction was mainly associated with fertilization (Zobel 1992; Zhang 1995; Makinen and Hynynen 2014). It can be concluded that the effects of fertilization and thinning treatments on radial growth rate and on wood and tracheid properties were often additive.
Weed control
Fertilization and vegetation control are highly synchronized in plantation culture (Albaugh et al. 2021; Carter et al. 2021). The addition of nutrients usually encourages the growth of other unwanted vegetation as well, necessitating vegetation control measures, whether chemical or manual. This ensures that growth components are only available for the target crop. There is no doubt that performing fertilization in combination with vegetation control improves growth significantly and better financial returns than each of the practices individually (Clark III et al. 2004; Antony et al. 2011a, b, c, d; Liechty and Fristoe 2013; Li et al. 2021). Combating weed alone does not affect the properties of the wood, but changes have been observed when synchronized with activities such as thinning and/or fertilizer application. The following order has generally been reported regarding impact on both growth and wood properties in pine stands: fertilizer plus vegetation control > fertilizer > vegetation control (Carter et al. 2021).
Irrigation
Irrigation of plantation trees is usually not applicable on a commercial scale but research using irrigation give us an insight into the effect of water availability on growth and wood properties. Water availability is one of the site factors that strongly determines the response of trees to fertilization (Albaugh et al. 1998; Love-Myers et al. 2010; Hans 2013; Will et al. 2015). In other words, water is the ‘vehicle’ for the nutrients. However, it is recommended that fertilization is implemented when soils are moist, not saturated, to ensure that nutrient retention is enhanced (Albaugh et al. 2014; Davis et al. 2015). Irrigation alone has some effect on biomass accumulation, however, much higher growth is usually observed when implemented in combination with fertilization. (Coyle and Coleman 2005; Campoe et al. 2013). For example, a study with P. radiata reported a 60% increase in growth with irrigation and a 200–300% increase when both water and nitrogen were available (Benson et al. 1992).
Also, an increase in total biomass production of 120% from loblolly pines that were fertilized and irrigated versus 91% with fertilization alone and 29% with irrigation was reported in another study (Albaugh et al. 1998). These growth gains observed from fertilization and irrigation together are due to improvements in light capture (due to the increase in leaf area index) and use efficiency (which may include changes in carbon allocation patterns) (Samuelson et al. 2004; Campoe et al. 2013).
In addition to the increase in growth, it has also been found that the combined effect of fertilization and irrigation affects wood properties more than either treatment individually (Love-Myers et al. 2010; Shmulsky and Jones 2019). Most notable effect is when irrigation increases latewood density of fertilized trees and bring overall ring density closer to levels achieved in unfertilized and unirrigated trees (Love-Myers et al. 2010). This change in ring density is attributed to increases in the growing season, which in turn increase the proportion of latewood, resulting in an overall increase in ring density (Moilanen et al. 2015). This is corroborated by a recent study on 17 years old loblolly pine which showed that the latewood percentage of fertilized and irrigated trees were closer to those that received none of the treatment (Eberhardt et al. 2022). However, this effect is said to be more significant in mature wood than juvenile wood because of different proportions of earlywood in the annual rings (Love-Myers et al. 2010; Moilanen et al. 2015).
Discussion
The changes in wood elements and tree ring components caused by fertilization are mainly characterized by an increase in MFA and a decrease in density and stiffness (Downes et al. 2002a, b; Antony et al. 2009a, b, 2013; Makinen and Hynynen 2014; Moore et al. 2021). It is argued that fertilization does not affect wood properties to an extent that makes it unusable, and that diameter increases more than compensates for possible negative effects (Albaugh et al. 2004). Especially when the effect it has on proportion of juvenile wood is not considered. This has been demonstrated by studies that show that the effect of fertilization on wood properties averaged over the whole stem cross-section is often not significant (Auty et al. 2013, 2014; Makinen and Hynynen 2014; Kimberley et al. 2015; Moore et al. 2015; Barrios et al. 2017; Kohler et al. 2019). At the annual ring level, the effects of specific fertilizer application (N) are undeniable and are reported to be within specific response periods of 3–8 years depending on the amount and frequency of N applied (Long et al. 2004; Fox et al. 2007a, b; Albaugh et al. 2021). The challenge is that the effect of fertilizers is permanent for the rings affected, regardless of the reaction time (Long et al. 2004), and the properties of a piece of wood is an average of the properties of the annual rings that make it up. If not implemented appropriately, there is a risk that trees will be produced with multiple bands of weaker wood within the juvenile zone of the logs. (Nyakuengama et al. 2002; Antony et al. 2011a, b, c, d). For some countries such as South Africa, where structural grade wood often comes from the juvenile zone, this will have an important impact (Wessels et al. 2014; Wessels and Petersen 2015).
As important as it is to consider the properties at whole stem cross-section, it is also valuable to examine ring-by-ring variations, especially regarding effects of fertilizer application. It might not be beneficial in some cases to have intermittent 3–4 years of fast growth with lower mechanical properties since it is possible that this reduction in the properties can affect the commercial value of the boards. For instance, a band of lower strength or stiffness wood closer to the pith might have more profound commercial consequences than a similar band in the mature wood section close to the bark. This means that the fertilization operations must be planned to consider the effect on wood properties. The effect of fertilizers is proportional to both the amount and concentration of nitrogen, and large amounts of N may produce much higher growth, but also produce significant changes in wood properties (Zobel 1992; Fox et al. 2007a, b; Munsell and Fox 2010; Antony et al. 2011a, b, c, d; Shmulsky and Jones 2019). It has been demonstrated that it is possible to apply multiple small doses of fertilizer as opposed to a single large dose and still get the same growth gains (Albaugh et al. 2018).
Furthermore, consistently small amounts of N lead to significant growth gains without altering wood properties (Antony et al. 2009a; b; Trazzi et al. 2019). This is also true for slow or controlled release fertilizers that are argued to produce more uniform wood (Smolander et al. 2022). Although one-off applications may be more justifiable in terms of running operational costs, it is better for wood quality to distribute them at carefully chosen intervals over the rotation to produce more consistent and uniform wood. This is best achieved when forest operations are designed not only to maximize productivity, but also to optimize product quality. This operational adjustment applies whether the wood is juvenile or mature. (Downes et al. 2002a, b; Mora 2003; Schimleck et al. 2018). This review considers that regardless of where in the stem cross-section, growth rate (ring width) has an impact on wood properties, particularly those induced by fertilization.
Most studies have focused on N and P applications, probably because these elements have been described as the most constraining in forest soils. K, on the other hand, has received little attention. Given that silviculture has evolved from mere growth limitation to maximum productivity per unit area (Schimleck et al. 2018; Carter et al. 2021), fertilizer ratios that not only provide the best growth rates, but also result in trees of required quality must be investigated. It has been proposed that high N: K can increase the proportions of latewood (Wessels et al. 2015), but limited investigations have been conducted to this end. Future studies should also quantify the effects of the different fertilizer combinations especially with K since it is suggested to maintain growth even during environmental stress.
The rapid growth of young trees, whatever the cause, increases the proportion of juvenile wood and, consequently, changes wood properties within the ring (Erasmus et al. 2020; Long et al. 2004; Clark III et al. 2008; Antony et al. 2012; Carson et al. 2014; Butler et al. 2015; Auty et al. 2016; Dahlen et al. 2020). Increased proportions of juvenile wood due to the reduction in the rotation age is one of the greatest challenges for the timber industry today and in the foreseeable future. This is because the use of intensive silviculture has facilitated the shift of commercial plantations into agroecosystems to maximize growth rate and profit (Munsell and Fox 2010). Scientists and arborists are quite aware that the percentage of juvenile wood can be genetically controlled, and steps have been taken, through breeding practices, to reduce the juvenile core (Moore and Cown 2017). The growth of a tree, once planted, is also shaped by how silvicultural practices influence the proportions of juvenile and mature wood (Clark III et al. 2006, 2008; Lasserre et al. 2009). As a note, better definitions in terms of the differentiation of wood as juvenile or mature is required, especially when using variations in density or MFA (Clark III et al. 2006; Mora et al. 2007).
Conclusion
This review shows that the effect of fertilization on wood properties is clearly a complex phenomenon where there are direct and indirect effects as well as interactions between different factors that influence wood properties. Results of Pinus fertilizer-wood property studies are likely species, site, and management specific and were therefore not always consistent. However, based on current literature available, the following general conclusions can be drawn:
-
The primary aim of fertilization is increased growth rate. Where fertilization occurred at or soon after planting (as is most often the case), early growth and subsequently juvenile wood proportion will be increased with a resultant negative effect on wood properties such as density and MFA.
-
In general, ring width (i.e. growth rate) demonstrated a negative correlation with density, and a positive relationship with MFA. However, results were often age specific, sometimes having an effect on mature wood but not on juvenile wood. Also, the relationship between growth rate and density and MFA were often non-significant at the whole stem level.
-
Fertilization, in some cases, seems to also have an effect on wood properties independent of the growth rate effect. However, this aspect was mostly not analysed in studies.
-
Earlywood and latewood were often influenced differently by fertilization: Earlywood width and density was increased while latewood width stayed constant and latewood density decreased. This resulted in a decrease in within-ring heterogeneity of density and density-related properties. Since latewood generally contributed more to whole-ring density, the net effect was that whole-ring wood density of pines mostly decreased with fertilization due to a decrease in cell wall thickness.
-
Although only a few studies considered the effect of fertilization on MFA, the general response was an increase in MFA after fertilization. Similarly, stiffness or MOE was only considered in a few studies and generally decreased after fertilization. Given that fertilization often resulted in a decrease in wood density and an increase in MFA, it was unsurprising that wood stiffness (or MOE) usually decreased with fertilization.
-
Most studies found that the combined effect of fertilization and silvicultural practices reducing competition such as thinning and weed control, resulted in significant changes to some wood properties. The magnitude of changes was additive, but fertilization often had a larger effect than silvicultural practices.
-
Increased water availability (through irrigation) combined with fertilization, affected wood properties more than either treatment individually. However, unlike thinning and weeding, irrigation has been shown to interact with fertilizer application in a way that counteracts the negative effects of fertilizer on wood properties by increasing the proportion of latewood. It was suggested that the beneficial effects of combined fertilization and irrigation is more likely in mature wood than juvenile wood.
-
Most fertilization wood property studies on Pinus involved nitrogen and phosphorus fertilizers with the quantity and ratios determined by site requirements. Nitrogen provides short term responses with phosphorus responding over a longer period. Results suggest that nitrogen may cause the greatest change in wood growth and properties.
-
Despite potassium being an essential element in forest nutrition and one often included in fertilizers, there has been relatively little research on the effect of potassium on wood properties.
The literature review showed that a particularly important knowledge gap exists in understanding the interrelationship between the various effects of fertilizing pine trees. A majority of the studies on fertilization thus far have focused on wood density, however, more focused studies need to be conducted on MFA and MOE. Studies designed to use methods such as path analysis (see Fig. 1) might be well suited to understand this complicated cause-and-effect scenario. An important question that was often unanswered in research, was whether the effect of specific fertilization treatments on wood properties were purely based on the influence of increased growth or whether there were direct influences not explained by growth. Future research should emphasize trial designs where these influences can be untangled in the statistical analyses process. Finally, despite potassium being an important elemental component of many fertilizers, relatively little work has been done to determine the effect of this element on wood properties. Work on this aspect will be especially beneficial in countries such as South Africa where potassium is often used for pine plantation fertilization.
References
Albaugh T, Allen L, Dougherty P, Kress L, King J (1998) Leaf area and above and belowground growth responses of loblolly pine to nutrient and water additions. For Sci 44(2):317–328
Albaugh T, Allen L, Dougherty P, Johnsen K (2004) Long term growth responses of loblolly pine to optimal nutrient and water resource availability. For Ecol Manag 192(1):3–19. https://doi.org/10.1016/j.foreco.2004.01.002
Albaugh T, Kiser LC, Fox T, Allen HL, Rubilar R, Stape JL (2014) Ecosystem nutrient retention after fertilization of Pinus taeda. For Sci 60(6):1131–1139. https://doi.org/10.5849/forsci.13-159
Albaugh T, Fox T, Allen HL, Rubilar R (2015) Juvenile southern pine response to fertilization is influenced by soil drainage and texture. Forests 6(8):2799–2819. https://doi.org/10.3390/f6082799
Albaugh T, Fox T, Rubilar R, Cook RL, Amateis RL, Burkhart H (2017) Post-thinning density and fertilization affect Pinus taeda stand and individual tree growth. For Ecol Manag 396(2017):207–216. https://doi.org/10.1016/j.foreco.2017.04.030
Albaugh T, Fox T, Cook R, Raymond J, Rubilar R, Campoe O (2018) Forest fertilizer applications in the southeastern United States from 1969 to 2016. For Sci 65(3):355–362. https://doi.org/10.1093/forsci/fxy058
Albaugh T, Albaugh J, Carter D, Cook R, Cohrs C, Rubilar R, Campoe O (2021) Duration of response to nitrogen and phosphorus applications in mid-rotation Pinus taeda. For Ecol Manag. https://doi.org/10.1016/j.foreco.2021.119578
Alteyrac J, Cloutier A, Ung CH, Zhang SY (2006) Mechanical properties in relation to selected wood characteristics of black spruce. Wood Fiber Sci 38(2):229–237
Amarasekara H, Denne MP (2002) Effects of crown size on wood characteristics of Corsican pine in relation to definitions of juvenile wood, crown formed wood and core wood. Forestry 75(1):51–61. https://doi.org/10.1093/forestry/75.1.51
Ameray A, Bergeron Y, Valeria O, Montoro Girona M, Cavard X (2021) Forest carbon management: a review of silvicultural practices and management strategies across boreal, temperate and tropical forests. Curr for Rep 7(4):245–266. https://doi.org/10.1007/s40725-021-00151-w
Antony F, Jordan L, Schimleck L, Daniels R, Clark A III (2009a) The effect of mid-rotation fertilization on the wood properties of loblolly pine (Pinus taeda). IAWA J 30(1):49–58. https://doi.org/10.1163/22941932-90000202
Antony F, Jordan L, Schimleck L, Daniels R, Clark A III, Hall D (2009b) Effect of midrotation fertilization on growth and specific gravity of loblolly pine. Can J for Res 39(2009):928–935. https://doi.org/10.1139/X09-021
Antony F, Jordan L, Schimleck L, Clark A III, Souter R, Daniels R (2011a) Regional variation in wood modulus of elasticity (stiffness) and modulus of rupture (strength) of planted loblolly pine in the United States. Can J for Res 41(2011):1522–1533. https://doi.org/10.1139/X11-064
Antony F, Schimleck L, Jordan L, Clark A, Daniels R (2011b) Effect of early age woody and herbaceous competition control on wood properties of loblolly pine. For Ecol Manag 262(8):1639–1647. https://doi.org/10.1016/j.foreco.2011.07.015
Antony F, Schimleck L, Hall D, Clark A III, Daniels R (2011c) Modeling the effect of midrotation fertilization on specific gravity of loblolly pine (Pinus taeda L.). For Sci 57(2):145–152
Antony F, Schimleck L, Daniels R, Clark A III (2011d) Effect of fertilization on growth and wood properties of thinned and unthinned midrotation loblolly pine (Pinus taeda L.) stands. South J Appl for 35(3):142–147
Antony F, Schimleck L, Jordan L, Daniels R, Clark A III (2012) Modeling the effect of initial planting density on within tree variation of stiffness in loblolly pine. Ann for Sci 69(5):641–650. https://doi.org/10.1007/s13595-011-0180-1
Antony F, Schimleck L, Daniels R, Clark A III (2013) Effect of mid-rotation fertilization on stiffness and strength of loblolly pine wood. IAWA J 34(2):127–134. https://doi.org/10.1163/22941932-00000011
Auty D, Gardiner B, Achim A, Moore J, Cameron A (2013) Models for predicting microfibril angle variation in Scots pine. Ann for Sci 70(2):209–218. https://doi.org/10.1007/s13595-012-0248-6
Auty D, Achim A, Macdonald E, Cameron AD, Gardiner BA (2014) Models for predicting wood density variation in Scots pine. Forestry 87(3):449–458. https://doi.org/10.1093/forestry/cpu005
Auty D, Achim A, Macdonald E, Cameron A, Gardiner B (2016) Models for predicting clearwood mechanical properties of Scots Pine. For Sci 62(August):403–413
Barrette J, Achim A, Auty D (2023) Impact of intensive forest management practices on wood quality from conifers: literature review and reflection on future challenges. Curr for Rep 9(2):101–130. https://doi.org/10.1007/s40725-023-00181-6
Barrios A, Trincada G, Watt M (2017) Wood properties of juvenile and mature wood of Pinus radiata D. Don trees growing on contrasting sites in Chile. For Sci 63(2):184–191. https://doi.org/10.5849/forsci.2016-060
Barros Filho NF, Schumacher MV, Neves JCL, de Barros NF, Valadares SV (2017) Tree growth and nutrient dynamics in pine plantations in Southern Brazil. Revista Brasileira de Ciencia do Solo 41:1–12. https://doi.org/10.1590/18069657rbcs20160400
Beets P, Gilchrist K, Jeffreys M (2001) Wood density of radiata pine: effect of nitrogen supply. For Ecol Manag 145(3):173–180. https://doi.org/10.1016/S0378-1127(00)00405-9
Benson ML, Myers BJ, Raison RJ (1992) Dynamics of stem growth of Pinus radiata as affected by water and nitrogen supply. For Ecol Manag 52:117–137
Burdon R, Moore J (2018) Adverse genetic correlations and impacts of silviculture involving wood properties: analysis of issues for radiata pine. Forests 9(6):1–17. https://doi.org/10.3390/f9060308
Butler M, Dahlen J, Daniels R, Eberhardt T, Antony F (2015) Bending strength and stiffness of loblolly pine lumber from intensively managed stands located on the Georgia Lower Coastal Plain. Eur J Wood Wood Prod 74(1):91–100. https://doi.org/10.1007/s00107-015-0956-3
Campion J (2008) The effects of mid- and late-rotation fertiliser application on tree growth and wood quality in softwood saw-timber stands: a critical review. South for 70(1):7–17. https://doi.org/10.2989/SOUTH.FOR.2008.70.1.2.512
Campoe O, Stape JL, Albaugh T, Lee Allen H, Fox T, Rubilar R, Binkley D (2013) Fertilization and irrigation effects on tree level aboveground net primary production, light interception and light use efficiency in a loblolly pine plantation. For Ecol Manag 288(2013):43–48. https://doi.org/10.1016/j.foreco.2012.05.026
Carlson C, Fox T, Allen HL, Albaugh T, Rubilar R, Stape J (2014) Growth responses of loblolly pine in the southeast united states to midrotation applications of nitrogen, phosphorus, potassium, and micronutrients. For Sci 60(1):157–169. https://doi.org/10.5849/forsci.12-158
Carson SD, Cown DJ, McKinley RB, Moore J (2014) Effects of site, silviculture and seedlot on wood density and estimated wood stiffness in radiata pine at mid-rotation. N Z J for Sci 44(1):1–12. https://doi.org/10.1186/s40490-014-0026-3
Carter D, Allen HL, Fox T, Albaugh T, Rubilar R, Campoe O, Cook R (2021) A 50-year retrospective of the forest productivity cooperative in the Southeastern United States: regionwide trials. J for 119(1):73–85. https://doi.org/10.1093/jofore/fvaa046
Chikumbu V (2011) Growth responses to fertilizer application of thinned , mid-rotation Pinus radiata stands across a soil water availability gradient in the Boland area of the Western Cape. Stellenbosch University
Clark A III, Borders B, Daniels R (2004) Impact of vegetation control and annual fertilization on properties of loblolly pine wood at age 12. For Prod J 54(12):90–96
Clark A III, Daniels R, Jordan L (2006) Juvenile/mature wood transition in loblolly pine as defined by annual ring specific gravity, proportion of latewood, and microfibril angle. Wood Fiber Sci 38(2):292–299
Clark A III, Jordan L, Schimleck L, Daniels R (2008) Effect of initial planting spacing on wood properties of unthinned loblolly pine at age 21. For Prod J 58(10):78–83
Clinton P (2018) Future expectations of forest soils: increasing productivity within environmental limits using new knowledge. N Z J Agric Res 61(3):389–401. https://doi.org/10.1080/00288233.2018.1446992
Coyle D, Coleman M (2005) Forest production responses to irrigation and fertilization are not explained by shifts in allocation. For Ecol Manag 208(1–3):137–152. https://doi.org/10.1016/j.foreco.2004.11.022
Crous JW, Morris AR, Scholes MC (2009) Effect of phosphorus and potassium fertiliser on tree growth and dry timber production of Pinus patula on gabbro-derived soils in Swaziland. South for 71(3):235–243. https://doi.org/10.2989/SF.2009.71.3.8.920
Dahlen J, Auty D, Eberhardt T (2018) Models for predicting specific gravity and ring width for loblolly pine from intensively managed plantations, and implications for wood utilization. Forests 9(6):1–20. https://doi.org/10.3390/f9060292
Dahlen J, Nabavi M, Auty D, Schimleck L, Eberhardt T (2020) Models for predicting the within-tree and regional variation of tracheid length and width for plantation loblolly pine. Forestry 94(1):127–140. https://doi.org/10.1093/forestry/cpaa018
Davis M, Xue J, Clinton P (2015) Planted forest Nutrition. New Zealand Forest Research Institute Limited. https://doi.org/10.13140/RG.2.1.1773.9604
DeBell D, Keyes C, Gartner B (2001) Wood density of Eucalyptus saligna grown in Hawaiian plantations: effects of silvicultural practices and relation to growth rate. Aust for 64(2):106–110. https://doi.org/10.1080/00049158.2001.10676173
Dickinson Y, Wang X, Wiedenbeck J, Ross R (2019) Effects of silvicultural practices on engineering properties of northern hardwood species of the great lakes region a literature review. United States Department of Agriculture, Madison
Donald DGM (1987) The application of fertiliser to pines following second thinning. S Afr for J 142(1):13–16. https://doi.org/10.1080/00382167.1987.9630278
Donaldson LA (2008) Microfibril angle: measurement, variation and relationships—a review. IAWA J 29(4):345–386. https://doi.org/10.1163/22941932-90000192
Downes GM, Nyakuengama G, Evans R, Northway R, Blakemore P, Dickson RL, Lausberg M (2002a) Relationship between wood density, microfibril angle and stiffness in thinned and fertilized Pinus radiata. IAWA J 23(3):253–265. https://doi.org/10.1163/22941932-90000302
Downes GM, Wimmer R, Evans R (2002b) Understanding wood formation: gains to commercial forestry through tree-ring research. Dendrochronologia 20(1–2):37–51. https://doi.org/10.1078/1125-7865-00006
du Toit B (2006) Information requirements to fertilize plantations with greater precision in a dry country. In: Ackerman P, Langin D, Antonides M (eds) Precision forestry in plantations, semi-natural and natural forests. Stellenbosch University, South Africa, Cape Town, pp 245–260
du Toit B, Scheepers GP (2020) Modelling soil nitrogen mineralisation in semi-mature pine stands of South Africa to identify nutritional limitations and to predict potential responses to fertilisation. Ann for Sci 77(2):24. https://doi.org/10.1007/s13595-020-0923-y
du Toit B, Arbuthnot A, Oscroft D, Job A (2001) The effects of remedial fertilizer treatments on growth and pulp properties of Eucalyptus grandis stands established on infertile soils of the Zululand coastal plain. South Afr for J 192:9–18. https://doi.org/10.1080/20702620.2001.10434129
du Toit B, Smith CW, Little KM, Boreham G, Pallett RN (2010) Intensive, site-specific silviculture: manipulating resource availability at establishment for improved stand productivity. A review of South African research. For Ecol Manag 259(9):1836–1845. https://doi.org/10.1016/j.foreco.2009.07.015
Eberhardt T, Leduc D, Samuelson L (2022) Wood properties of loblolly pine grown under intensive management in the Upper Coastal Plain of southwest Georgia. Can J for Res 52(7):1110–1118. https://doi.org/10.1139/cjfr-2022-0016
Erasmus J, Drew DM, Wessels CB (2020) The flexural lumber properties of Pinus patula Schiede ex Schltdl. & Cham. improve with decreasing initial tree spacing. Ann for Sci 77:1–15. https://doi.org/10.1007/s13595-020-00975-9
Evans R, Booker R, Kibblewhite PR (2001) Variation of microfibril angle, density and stiffness in fifty radiata pine trees. In 55th appita annual conference. Proceedings. Appita Inc., Hobart, Australia, pp 9–13
Everett C, Palm-Leis H (2009) Availability of residual phosphorus fertilizer for loblolly pine. For Ecol Manag 258(10):2207–2213. https://doi.org/10.1016/j.foreco.2008.11.029
Fajstavr M, Paschová Z, Giagli K, Vavrčík H, Gryc V, Urban J (2018) Auxin (IAA) and soluble carbohydrate seasonal dynamics monitored during xylogenesis and phloemogenesis in scots pine. Iforest 11(5):553–562. https://doi.org/10.3832/ifor2734-011
FAO (2022) The State of the World’s Forests 2022. Forest pathways for green recovery and building inclusive, resilient and sustainable economies. Rome. https://doi.org/10.4060/cb9360en
Fischer P, du Toit B (2019) Use of δ 13 c as water stress indicator and potential silvicultural decision support tool in Pinus radiata stand management in South Africa. Iforest 12(1):51–60. https://doi.org/10.3832/ifor2628-011
Fox T, Allen HL, Albaugh T, Rubilar R, Carlson C (2007a) Tree nutrition and forest fertilization of pine plantations in the Southern United States. South J Appl for 31(1):5–11. https://doi.org/10.1093/sjaf/31.1.5
Fox T, Jokela E, Allen HL (2007b) The development of pine plantation silviculture in the Southern United States. J for 105(7):337–347
Fromm J (2010) Wood formation of trees in relation to potassium and calcium nutrition. Tree Physiol 30(9):1140–1147. https://doi.org/10.1093/treephys/tpq024
Gyawali N, Burkhart H (2015) General response functions to silvicultural treatments in loblolly pine plantations. Can J for Res 45(3):252–265. https://doi.org/10.1139/cjfr-2014-0172
Hackman JJ, Rose BD, Frank HER, Vilgalys R, Cook RL, Garcia K (2022) NPK fertilizer use in loblolly pine plantations: Who are we really feeding ? For Ecol Manag 520(July):120393. https://doi.org/10.1016/j.foreco.2022.120393
Hans R (2013) Initial growth responses to controlled release fertilizer application at establishment of commercial forestry species in South Africa. Dessertation, Stellenbosch University
Haveraaen O, Frivold LH (2015) Effect of repeated fertilization on stem growth in old stands of Pinus sylvestris in South East Norway. J for Sci 61(2):72–79. https://doi.org/10.17221/110/2014-JFS
Hedwall PO, Gong P, Ingerslev M, Bergh J (2014) Fertilization in northern forests—biological, economic and environmental constraints and possibilities. Scand J for Res 29(4):301–311. https://doi.org/10.1080/02827581.2014.926096
Ingestad T (1979) Mineral nutrient requirements of Pinus silvestris and Picea abies seedlings. Physiol Plant 45(4):373–380. https://doi.org/10.1111/j.1399-3054.1979.tb02599.x
Irland L (2011) Timber productivity research gaps for extensive forest management. Small-Scale for 10(4):389–400. https://doi.org/10.1007/s11842-011-9155-1
Ivković M, Gapare W, Abarquez A, Ilic J, Powell MB, Wu HX (2009) Prediction of wood stiffness, strength, and shrinkage in juvenile wood of radiata pine. Wood Sci Technol 43(3–4):237–257. https://doi.org/10.1007/s00226-008-0232-3
Ivković M, Gapare W, Wu H, Espinoza S, Rozenberg P (2013) Influence of cambial age and climate on ring width and wood density in Pinus radiata families. Ann for Sci 70(5):525–534. https://doi.org/10.1007/s13595-013-0290-z
Jacobson S, Pettersson F (2010) An assessment of different fertilization regimes in three boreal coniferous stands. Silva Fenn 44(5):815–827. https://doi.org/10.14214/sf.123
Jokela E, Long A (2018) Using soils to guide fertilizer recommendations for Southern Pines. Institute of Food and Agricultural Science, University of Florida, Florida
Jokela E, Martin T, Vogel J (2010) Twenty-five years of intensive forest management with Southern Pines: important lessons learned. J for 108(7):338–347
Kennedy RW (1995) Coniferous wood quality in the future: concerns and strategies. Wood Sci Technol 29(5):321–338. https://doi.org/10.1007/BF00202581
Kidombo SD, Dean TJ (2018) Growth of tree diameter and stem taper as affected by reduced leaf area on selected branch whorls. Can J for Res 48(4):317–323. https://doi.org/10.1139/cjfr-2017-0279
Kijidani Y, Ohshiro N, Matsumura J, Koga S (2014) Effects of crown length on indole acetic acid (IAA) amounts in cambial region tissues in lower and upper trunks of sugi cultivars (Cryptomeria japonica) in September. J Wood Sci 60(4):235–242. https://doi.org/10.1007/s10086-014-1400-8
Kimberley MO, Cown DJ, McKinley RB, Moore J, Dowling LJ (2015) Modelling variation in wood density within and among trees in stands of New Zealand-grown radiata pine. N Z J for Sci 45(1):1–13. https://doi.org/10.1186/s40490-015-0053-8
Kohler M, Niederberger J, Wichser A, Bierbaß P, Rötzer T, Spiecker H, Bauhus J (2019) Using tree rings to reconstruct changes in soil P availability—results from forest fertilization trials. Dendrochronologia 54:11–19. https://doi.org/10.1016/j.dendro.2019.01.001
Kotze H, du Toit B (2012) Silviculture of industrial pine plantations in South Africa. In: Bredenkamp B, Upfold S (eds) South African forestry handbook, 5th edn. Southern African Institute for Forestry, Scottsville, South Africa, pp 123–140
Kuprevicius A, Auty D, Achim A, Caspersen J (2013) Quantifying the influence of live crown ratio on the mechanical properties of clear wood. Forestry 86(3):361–369. https://doi.org/10.1093/forestry/cpt006
Lasserre JP, Mason EG, Watt M, Moore J (2009) Influence of initial planting spacing and genotype on microfibril angle, wood density, fibre properties and modulus of elasticity in Pinus radiata D. Don Corewood. For Ecol Manag 258(9):1924–1931. https://doi.org/10.1016/j.foreco.2009.07.028
Li Y, Dickens ED, Sault M, Siry JP, Izlar RL, Clabo DC, Tyson WG (2021) Does it pay to conduct midrotation competition control and/or fertilization? Results from two thinned loblolly pine studies in the coastal plain of Georgia. J for 119(4):337–350. https://doi.org/10.1093/jofore/fvab008
Liechty HO, Fristoe C (2013) Response of midrotation pine stands to fertilizer and herbicide application in the western gulf coastal plain. South J Appl for 37(2):69–74. https://doi.org/10.5849/sjaf.11-044
Linder S (1995) Foliar analysis for detecting and correcting nutrient imbalances in Norway spruce. Ecol Bull 44(44):178–190. https://doi.org/10.2307/20113161
Loewe-Muñoz V, Delard C, Del Río R, Balzarini M (2020) Long-term effect of fertilization on stone pine growth and cone production. Ann for Sci 77(3):1–9. https://doi.org/10.1007/s13595-020-00978-6
Long JN, Dean TJ, Roberts SD (2004) Linkages between silviculture and ecology: examination of several important conceptual models. For Ecol Manag 200(1–3):249–261. https://doi.org/10.1016/j.foreco.2004.07.005
Louw JR, Scholes MC (2003) Foliar nutrient levels as indicators of site quality for Pinus patula in the Mpumalanga escarpment area. South Afr for J 197(1):21–30. https://doi.org/10.1080/20702620.2003.10431718
Love-Myers K, Clark A III, Schimleck L, Jokela E, Daniels R (2009) Specific gravity responses of slash and loblolly pine following mid-rotation fertilization. For Ecol Manag 257(12):2342–2349. https://doi.org/10.1016/j.foreco.2009.03.031
Love-Myers K, Clark A III, Schimleck L, Dougherty P, Daniels R, Jokela E, Daniels R (2010) The effects of irrigation and fertilization on specific gravity of loblolly pine. For Sci 56(5):484–493. https://doi.org/10.1093/forestscience/56.5.484
Luo L, Li L (2022) Molecular understanding of wood formation in trees. For Res 2(1):5. https://doi.org/10.48130/fr-2022-0005
Mäkinen H, Hynynen J (2012) Predicting wood and tracheid properties of Scots pine. For Ecol Manag 279(2012):11–20. https://doi.org/10.1016/j.foreco.2012.05.024
Makinen H, Hynynen J (2014) Wood density and tracheid properties of Scots pine: responses to repeated fertilization and timing of the first commercial thinning. Forestry 87(3):437–447. https://doi.org/10.1093/forestry/cpu004
Malan FS (2006) The wood properties and sawn-board quality of South African-grown Pinus maximinoi (HE Moore). South Afr for J 208(1):39–47. https://doi.org/10.2989/10295920609505260
McGrath JF, Copeland B, Dumbrell IC (2003) Magnitude and duration of growth and wood quality responses to phosphorus and nitrogen in thinned Pinus radiata in southern Western Australia. Aust for 66(3):223–230. https://doi.org/10.1080/00049158.2003.10674916
Miller H (1984) Dynamics of nutrition cycling in plantation ecosystems. In: Bowen G, Nambiar EKS (eds) Nutrition of plantation forests. Academic Press Inc., London, p 516
Moilanen M, Hytönen J, Hökkä H, Ahtikoski A (2015) Fertilization increased growth of Scots pine and financial performance of forest management in a drained peatland in Finland. Silva Fen- Nica 49(3):1–18
Moore J, Clinton P (2015) Enhancing the productivity of radiata pine forestry within environmental limits. NZ J for 60(3):35–41
Moore J, Cown DJ (2015) Wood quality variability—what is it, what are the consequences and what we can do about it. N Z J for 59(4):3–9
Moore J, Cown DJ (2017) Corewood (juvenile wood) and its impact on wood utilisation. Curr for Rep 3(2):107–118. https://doi.org/10.1007/s40725-017-0055-2
Moore J, Lyon AJ, Lehneke S (2012) Effects of rotation length on the grade recovery and wood properties of Sitka spruce structural timber grown in Great Britain. Ann for Sci 69(3):353–362
Moore J, Cown DJ, McKinley RB, Sabatia CO (2015) Effects of stand density and seedlot on three wood properties of young radiata pine grown at a dry-land site in New Zealand. N Z J for Sci 45(1):4. https://doi.org/10.1186/s40490-015-0035-x
Moore J, Nanayakkara B, McKinley RB, Garrett LG (2021) Effects of nutrient removal by harvesting practices and fertiliser addition on end-of-rotation radiata pine wood quality. For Ecol Manag 494:119269. https://doi.org/10.1016/j.foreco.2021.119269
Mora C (2003) Effects of early intensive silviculture on wood properties of loblolly pine. North Carolina State University
Mora C, Allen HL, Daniels R, Clark A (2007) Modeling corewood–outerwood transition in loblolly pine using wood specific gravity. Can J for Res 37(6):999–1011. https://doi.org/10.1139/X06-250
Munsell JF, Fox T (2010) An analysis of the feasibility for increasing woody biomass production from pine plantations in the southern United States. Biomass Bioenerg 34(12):1631–1642. https://doi.org/10.1016/j.biombioe.2010.05.009
Nyakuengama G, Downes GM, Ng J (2002) Growth and wood density responses to later-age fertilizer application in Pinus radiata. IAWA J 23(4):431–448. https://doi.org/10.1163/22941932-90000313
Payn T, De Ronde C, Grey DC (1988) Phosphate fertilisation of mature Pinus radiata stands. S Afr for J 147(1):26–31. https://doi.org/10.1080/00382167.1988.9628967
Peltola H, Kilpeläinen A, Sauvala K, Räisänen T, Ikonen VP (2007) Effects of early thinning regime and tree status on the radial growth and wood density of scots pine. Silva Fenn 41(3):489–505. https://doi.org/10.14214/sf.285
Pereira M, Bassaco MVM, Motta ACV, Maeda S, Prior SA, Marques R, Magri E, Bognola IA et al (2022) Influence of industrial forest residue applications on Pinus taeda: soil, litter, growth, nutrition, and wood quality characteristics. New for. https://doi.org/10.1007/s11056-021-09902-w
Ramírez Alzate MV, Rubilar R, Montes C, Allen HL, Fox T, Sanfuentes E (2016) Mid-rotation response to fertilizer by Pinus radiata D. Don at three contrasting sites. J for Sci 62(4):153–162. https://doi.org/10.17221/106/2015-JFS
Rocha MFV, Veiga TRLA, Soares BCD, de Araújo ACC, Carvalho AMM, Hein PRG (2019) Do the growing conditions of trees influence the wood properties? Floresta e Ambient 26(3):1–16. https://doi.org/10.1590/2179-8087.035318
Rodriguez D, Tomazello-Filho M (2019) Clues to wood quality and production from analyzing ring width and density variabilities of fertilized Pinus taeda trees. New for 50(5):821–843. https://doi.org/10.1007/s11056-018-09702-9
Rubilar R, Allen HL, Fox T, Cook R, Albaugh T, Campoe O (2018) Advances in silviculture of intensively managed plantations. Curr for Rep 4(1):23–34. https://doi.org/10.1007/s40725-018-0072-9
Saarsalmi A, Mälkönen E (2001) Forest fertilization research in Finland: a literature review. Scand J for Res 16(6):514–535. https://doi.org/10.1080/02827580152699358
Salazar-Tortosa D, Castro J, Villar-Salvador P, Viñegla B, Matías L, Michelsen A, Rubio de Casas R, Querejeta JI (2018) The “isohydric trap”: a proposed feedback between water shortage, stomatal regulation, and nutrient acquisition drives differential growth and survival of European pines under climatic dryness. Glob Change Biol 24(9):4069–4083. https://doi.org/10.1111/gcb.14311
Salmén L (2018) Wood cell wall structure and organisation in relation to mechanics. In: Geitmann A, Gril J (eds) Plant biomechanics: from structure to function at multiple scales. Springer Nature, Switzerland, pp 3–18
Samuelson L, Johnsen K, Stokes T (2004) Production, allocation, and stemwood growth efficiency of Pinus taeda L. stands in response to 6 years of intensive management. For Ecol Manag 192(1):59–70. https://doi.org/10.1016/j.foreco.2004.01.005
Schimleck L, Antony F, Dahlen J, Moore J (2018) Wood and fiber quality of plantation-grown conifers: a summary of research with an emphasis on loblolly and radiata pine. Forests 9(6):1–16. https://doi.org/10.3390/f9060298
Schimleck L, Dahlen J, Auty D (2022) Radial patterns of specific gravity variation in North American conifers. Can J for Res 52(6):889–900. https://doi.org/10.1139/cjfr-2021-0338
Schneider R, Zhang SY, Swift DE, Bégin J, Lussier J-MM, Zhang Y, Swift DE, Begin J et al (2008) Predicting selected wood properties of jack pine following commercial thinning. Can J for Res 38(7):2030–2043. https://doi.org/10.1139/X08-038
Schulte M, Cook R, Albaugh T, Burkhart H, Creighton J, Campoe O, Carter D, Rubilar R et al. (2020) Fertilization and thinning effects on plantation loblolly pine taper and wood quality. In: Bragg D, Koerth N, Holley G (eds) Proceedings of the 20th biennial southern silvicultural research conference. Asheville: Department of Agriculture, Forest Service, Southern Research Station, pp 273–278
Shephard NT, Joshi O, Meek CR, Will RE (2021) Long-term growth effects of simulated-drought, mid-rotation fertilization, and thinning on a loblolly pine plantation in southeastern Oklahoma, USA. For Ecol Manag 494(April):119323. https://doi.org/10.1016/j.foreco.2021.119323
Shmulsky R, Jones PD (2019) Silvicultural practices and wood quality. Forest products and wood science, 7th edn. Wiley, West Sussex, UK, pp 257–292. https://doi.org/10.1002/9781119426400.ch11
Smaill S, Clinton P (2016) Overview of the issues affecting fertiliser use in New Zealand’s radiata pine forests. N Z J for 61(2):11–15
Smolander A, Henttonen HM, Nöjd P, Soronen P, Mäkinen H (2022) Long-term response of soil and stem wood properties to repeated nitrogen fertilization in a N-limited Scots pine stand. Eur J for Res 141(3):421–431. https://doi.org/10.1007/s10342-022-01448-6
Snowdon P (2002) Modeling type 1 and type 2 growth responses in plantations after application of fertilizer or other silvicultural treatments. For Ecol Manag 163(1–3):229–244. https://doi.org/10.1016/S0378-1127(01)00582-5
Topanotti LR, Vaz DR, Carvalho SPC, Rios PD, Tomazello-Filho M, Dobner M, Nicoletti MF (2021) Growth and wood density of Pinus taeda L. as affected by shelterwood harvest in a two-aged stand in Southern Brazil. Eur J for Res 140(4):869–881. https://doi.org/10.1007/s10342-021-01372-1
Traversari S, Giovannelli A, Emiliani G (2022) Wood formation under changing environment: omics approaches to elucidate the mechanisms driving the early-to-latewood transition in conifers. Forests 13(4):1–17. https://doi.org/10.3390/f13040608
Trazzi PA, dos Santos JA, Caldeira MVW, Roters DF, Carvalho D, Dobner Júnior M (2019) Initial growth of Pinus taeda by fertilization response at planting. Floresta e Ambient 26(Spe1):1–9. https://doi.org/10.1590/2179-8087.037018
Uggla C, Moritz T, Sandberg G, Sundberg B (1996) Auxin as a positional signal in pattern formation in plants. Proc Natl Acad Sci USA 93(17):9282–9286. https://doi.org/10.1073/pnas.93.17.9282
Ulvcrona T, Ahnlund Ulvcrona K (2011) The effects of pre-commercial thinning and fertilization on characteristics of juvenile clearwood of Scots pine (Pinus sylvestris L.). Forestry 84(3):207–219. https://doi.org/10.1093/forestry/cpr007
Wagner L, Bader T, Auty D, de Borst K (2013) Key parameters controlling stiffness variability within trees: a multiscale experimental–numerical approach. Trees-Struct Funct 27(1):321–336. https://doi.org/10.1007/s00468-012-0801-9
Watt M, Clinton P, Coker G, Davis M, Simcock R, Parfitt R, Dando J (2008) Modelling the influence of environment and stand characteristics on basic density and modulus of elasticity for young Pinus radiata and Cupressus lusitanica. For Ecol Manag 255(3–4):1023–1033. https://doi.org/10.1016/j.foreco.2007.09.086
Wessels CB, Petersen NO (2015) Strength and stiffness capacity utilisation of timber members in roof truss structures. South for 77(4):305–307. https://doi.org/10.2989/20702620.2015.1063033
Wessels CB, Malan FS, Nel DG, Rypstra T, Wessels B, Malan FS, Nel DG, Rypstra T (2014) Variation in strength, stiffness and related wood properties in young South African-grown Pinus patula. South for 76(1):37–46. https://doi.org/10.2989/20702620.2013.870406
Wessels CB, Malan FS, Seifert T, Louw JH, Rypstra T (2015) The prediction of the flexural lumber properties from standing South African-grown Pinus patula trees. Eur J Forest Res 134(1):1–18. https://doi.org/10.1007/s10342-014-0829-z
Will RE, Fox T, Akers M, Domec JC, González-Benecke C, Jokela E, Kane M, Laviner MA et al (2015) A range-wide experiment to investigate nutrient and soil moisture interactions in loblolly pine plantations. Forests 6(6):2014–2028. https://doi.org/10.3390/f6062014
Xu P, Walker JCF (2004) Stiffness gradients in radiata pine trees. Wood Sci Technol 38(1):1–9. https://doi.org/10.1007/s00226-003-0188-2
Xu P, Donaldson L, Walker J, Evans R, Downes GM (2004) Effects of density and microfibril orientation on the vertical variation of low-stiffness wood in radiata pine butt logs. Holzforschung 58(6):673–677. https://doi.org/10.1515/HF.2004.122
Zhang SY (1995) Effect of growth rate on wood specific gravity and selected mechanical properties in individual species from distinct wood categories. Wood Sci Technol 29(6):451–465. https://doi.org/10.1007/BF00194204
Zhao D, Kane M, Teskey R, Fox T, Albaugh T, Allen HL, Rubilar R (2016) Maximum response of loblolly pine plantations to silvicultural management in the southern United States. For Ecol Manag 375(2016):105–111. https://doi.org/10.1016/j.foreco.2016.05.035
Zobel B (1992) Silvicultural effects on wood properties. Wood Sci Technol 29(6):451–465. https://doi.org/10.1007/BF00194204
Zobel B, Buijtenen J (1989) Wood variation: its causes and control. Springer-Verlag, Berlin Heildelberg
Zörb C, Senbayram M, Peiter E (2014) Potassium in agriculture—status and perspectives. J Plant Physiol 171(9):656–669. https://doi.org/10.1016/j.jplph.2013.08.008
Zwolinski J, Johnston M, Kotze H (2002) Soil treatments for improved timber production in Pinus radiata plantations in South Africa. For Ecol Manag 171(1–2):199–207. https://doi.org/10.1016/S0378-1127(02)00473-5
Funding
Open access funding provided by Stellenbosch University.
Author information
Authors and Affiliations
Contributions
SA wrote the paper with contributions of BMB, BdT and CBW. All authors reviewed the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Peter Biber.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Akello, S., du Toit, B., Balboni, B.M. et al. A review of the effect of fertilization on the wood properties of Pinus trees. Eur J Forest Res (2024). https://doi.org/10.1007/s10342-024-01709-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10342-024-01709-6