Abstract
Climate changes have dramatically affected forest succession. However, how trees at different successional stages respond to climate warming is unclear in the subtropics. Here, we compared the radial growth (RW) and xylem features of the early-successional stage Castanea mollissima and late-successional stage Quercus fabri in subtropical forests and assessed their response to climate changes. All parameters, including RW, number of vessels (VN), vessel density (VD), mean (MVA) and total (TVA) vessel area, percentage of conductive area within xylem (RCTA), theoretical (Kh) and xylem-specific (Ks) hydraulic conductivity, except for MVA of C. mollissima were significantly higher than those of Q. fabri. During the drought period, a notable adverse correlation between two cluster parameters of Q. fabri, associated with hydraulic safety and efficiency, suggested a pronounced hydraulic trade-off in Q. fabri under drought conditions. Castanea mollissima was more sensitive to climate and more prone to hydraulic failure than Q. fabri. Temperatures and moisture conditions positively and negatively affected the hydraulic efficiency-related parameters of C. mollissima. Moisture conditions in the previous summer and winter were significantly negatively and positively related to the radial growth of both species. The impact of generalized warming was not evident due to variations in hydraulic strategies and species characteristics, trade-offs between non-growing and growing season climates, and specific competition. If climate warming continues, C. mollissima growth will probably significantly decline due to the increasing risk of hydraulic failure. Warming may accelerate species replacement and forest succession in the study area by changing their lifespans and competitive relationships.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forest succession is an important process in the regeneration of forest resources and plays an essential role in the global carbon cycle (Teixeira et al. 2020). It is strongly influenced by climate conditions, such as temperature and precipitation (Arroyo-Rodríguez et al. 2017; Cadotte and Tucker 2017; Lohbeck et al. 2014). Recently, warming has been shown to have a dramatic impact on the rate and pathways of forest succession (Anderson-Teixeira et al. 2013). For example, Shuman et al. (2011) found that warming will accelerate forest succession in Siberia, which may make a shift in the ability of larch (early-successional stage trees) to establish; Molina et al. (2021) pointed out that warming favors the growth of early-successional stage trees. Therefore, understanding the impact of warming on forest succession helps us to accurately predict forest dynamics and to better manage forests or reforestation (Taylor et al. 2009).
Tree-ring data, especially ring widths, are one of the most visual representations of tree growth and forest productivity (Cherubini et al. 2021; Tei et al. 2017; Zhu et al. 2018). Trees of different successional stages play different roles in forest succession (Brzeziecki and Kienast 1994). They also respond differently to climate change and take different strategies to cope with changing climate (Kitao et al. 2016). Tree ring analysis has been successfully used to reconstruct past stand history and forest succession processes (Abrams et al. 1995; Altman et al. 2018; Brandes et al. 2018). For example, Brienen et al. (2009) examined the potential of tree rings of 70 species for studying forest succession in southern Mexico and proved that tree ring analysis is a promising tool for studies on secondary forest succession in the tropics. Boulanger et al. (2017) found that warming may lead to fewer late-successional stage trees and more early-successional stage trees, which suggests that warming may favor to the growth of early-successional stage trees. Esposito et al. (2018) also found that early-successional stage trees showed better tolerance in environments affected by many disturbances than late-successional stage trees. However, previous studies on exploring the specificity growth-climate response of tree species of different successional stages are mostly based on traditional ring widths. The process explanations and mechanisms behind the differential response of early- and late-successional stage species to rapid warming are still unclear, which is crucial for understanding the impact of climate change on forest succession.
Xylem anatomical features, a new class of tree-ring parameters at cellular and subcellular scales, are directly related to the key functions and physiological processes of trees (Fonti et al. 2010; Zhu et al. 2021). Compared to ring widths, studying their plastic adjustments can provide important insights into the physiological explanations, adaptation processes, and response strategies of tree growth to climate (Carrer et al. 2015; Sperry et al. 2008). Vessels are the most important hydraulic structures for broad-leaved trees and directly determine their hydraulic conductivity (Fonti et al. 2010; Zhu et al. 2017). Trees respond to changing environments by constantly adjusting vessel features, such as size, number, and density, to balance hydraulic efficiency and safety (Guada et al. 2021; Zhu et al. 2021). In a few studies on forest succession, wood anatomy has been performed to obtain a more accurate tree age (Brienen et al. 2009; Verheyden et al. 2004). Recently, Nola et al. (2020) compared the response of xylem anatomical features of native and alien species to climate and concluded that local climate change might slow forest succession. Therefore, quantitative xylem anatomy could be a promising approach to understanding forest succession processes under climate change and their behind mechanisms (Cannone et al. 2008).
Subtropical forests in China account for ca. 1/4 of the entire national land area having an important ecological significance and a high capacity for carbon storage (Yu et al. 2014). Nevertheless, the regional climax forests, the subtropical monsoon evergreen forests, were severely damaged by anthropogenic and natural disturbances (e.g., logging, climate change, and land use change) (Zhang and Song 2006; Zhou et al. 2013), resulting in drastic changes in ecological conditions. During the last few decades, southern China has experienced extensive afforestation programs to reconstruct forest ecosystems, restore eco-environments, and mitigate climate change (Bastin et al. 2019). The subtropical forests in south China are now fragmenting and exhibiting a remarked variability along successional gradients (temporal scale) (Zhou et al. 2013). Castanea mollissima and Quercus fabri, as vital economic tree species, are early-successional and late-successional stage species, respectively, both widely used in afforestation in southern China (Wen 2018). Previous studies mainly focused on improving their yield or stress resistance from genetic and physiological aspects, and few studies were conducted on their growth responses to recent climate changes (Lin et al. 2018; Zhu et al. 2019). They are both ring-porous species belonging to the family Fagaceae with distinctive ring boundaries and big earlywood vessels, which are suitable for tree-ring analysis. Herein, based on the previous research on trees at different-successional stages, we hypothesize that: (1) the two tree species have significant different xylem anatomical features, and they have different xylem adjustment strategies in response to climate change; (2) the early-successional stage C. mollissima can better adapt to recent warming than the late-successional stage Q. fabri. In this study, we measured both species’ tree-ring width and earlywood vessel features, then compared their differences in xylem anatomical features and responses to climate changes. Finally, their different climatic responses were used to infer the effects of climate change on forest succession.
Materials and methods
Study area and climate
The study site is located in Dashanchong forest park (28°23′58′′–28°24′58′′ N and 113°17′46′′–113°19′08′′ E, 55-260 m asl), Changsha city, Hunan province, China (Fig. 1). The terrain of this study area is hilly with red soils on slate and shale rocks, covered by vegetation mainly characterized by plantations and natural secondary forests (Wen 2018). The forest coverage rate reaches 80%, and the total vegetation coverage rate could be as high as 98%. The evergreen broad-leaved forests consist mainly of Schima superba, Choerospondias axillaris, Liquidambar formosana, Cyclobalanopsis glauca, C. mollissima, and Q. fabri (Wen 2018).
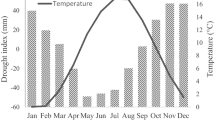
The study site has a subtropical monsoon climate with a hot and rainy summer and a mild and moist winter. Annual precipitation of the study area ranges from 932 to 1824 mm, and 70% of the total precipitation falls from March to August. Annual mean temperature ranges from 16.3 to 19.2 °C. The warmest and coldest months are July (29.2 °C) and January (4.9 °C). The temperature in this region has a significant increasing trend (0.03 °C a−1), while the relative humidity has a significant (k = − 0.165, R2 = 0.318, p < 0.001) decreasing trend (Fig. 2). From 2003 to 2015, the study area experienced a period of prolonged drought stress, characterized by high temperatures, low precipitation, low standardized precipitation-evapotranspiration index (SPEI) and low relative humidity (Fig. 2).
Trends in main climate factors during 1970–2019: a annual total precipitation, b mean temperature, c standardized precipitation evapotranspiration index (SPEI), and d relative humidity. The thin straight line represents the linear regression trend. The colored shaded area represents 95% confidence intervals. The grey dark shaded area represents the drought period during 2003–2015
Sample collection and processing
A forest stand without apparent recent disturbances in the study area was chosen as the sampling site. It is a natural secondary forest that was regenerating without any management after significant human disturbance of a subtropical evergreen broadleaved forest. The forest was dominated by C. mollissima and Q. fabri, and mixed very few Choerospondias axillaris, Schima superba, Liriodendron chinensis, and Cinnamomum camphora. For each species, we chose 30 healthy dominant trees growing in similar micro-site conditions at the study site (mean attitude: 65 m, asl). The mean diameter at breast height (1.3 m) of C. mollissima and Q. fabri were respectively 30.69 ± 4.99 cm and 26.90 ± 4.15 cm. One core per tree was collected at breast height using an increment borer with an inner diameter of 5.15 mm (Haglöf, Långsele, Sweden).
The cores were pre-processed by fixing, air drying, and polishing with a series of sandpaper of successively finer grits (up to 1200 grit) until the tree rings were clearly visible. Tree-ring widths were measured and cross-dated by the CooRecorder & CDendro 9.6 software (Cybis, Saltsjöbaden, Sweden). The COFECHA program was used to test the quality of measurement and cross-dating (Holmes 1983). Tree cores that could not be successfully cross-dated or showed weak correlations with the master sequence were removed. Based on the principle of trees with similar age and core surfaces without any obvious abnormality, a total of 10 C. mollissima and 11 Q. fabri trees were finally retained for quantitative wood anatomy.
The selected sample surfaces were further rubbed with white chalk to improve the contrast of earlywood vessels. These high-quality samples of each species were then scanned with a flatbed scanner at 3200 dpi resolution (Epson Expression 11000XL Scanner, Seiko Epson Corporation, Suwa, Japan). Subsequently, xylem anatomical features were recognized, measured, and corrected manually with ROXAS (von Arx and Carrer 2014). The ring width (RW), mean vessel area (MVA), number of vessels (VN), total vessel area (TVA), mean percentage of conductive area (RCTA), vessel density (VD), theoretical hydraulic conductivity (Kh) and theoretical xylem-specific hydraulic conductivity (Ks) of both species were obtained in this study (Zhu et al. 2021).
Because the target area used to measure the xylem anatomical features was not uniform due to different arc or angle of the growth ring boundary line. The raw data of TVA, VN, and Kh were normalized with a 5000-micron tangential width for quantitative analysis (Zhu et al. 2020). A spline with a 50% frequency cutoff at two-thirds of an individual series was used to remove the age-related growth trend in the normalized data of RW and all anatomical features using the ‘dplR’ package (Bunn 2008; Chen et al. 2021) in R software (R Core Team 2022). Finally, we got eight standard chronologies for each tree species (Fig. S1).
Meteorological data
Climate data used in this study were obtained from the China Meteorological Data Service Center (http://data.cma.cn/). The instrumental meteorological data from 1970 to 2019 at the Changsha meteorological station (Changsha, 112.55°E and 28.13°N, 68.0 m asl, 80.4 km from the study site) were used in this study, including the monthly total precipitation (P), mean relative humidity (Rh), mean temperature (T), maximum temperature (Tmax), and minimum temperature (Tmin). The SPEI was calculated from mean temperature and precipitation (Vicente-Serrano et al. 2010).
Seasonal climate variables were defined as follows: pSum = previous summer (from previous June to August), pAut = previous autumn (from previous September to November), pWin = previous winter (from previous December to current February), SPR = current spring (from current March to May), SUM = current summer (from current June to August), AUT = current autumn (from current September to November), PGS = previous growing season (from previous March to November), PNG = previous non-growing season (from previous December to current February), GS = current growing season (from current March to November).
Statistical analysis
To compare the similarities and differences between the xylem features in both species, we first calculated the mean value of each anatomical variable per tree and then compared those mean values between species using the independent sample t-test. We used Pearson correlation analysis to investigate the interrelations by standard chronologies among the vessel features and ring width. To highlight the effects of drought on xylem formation, we performed the analyses mentioned above over three periods: the whole common period of two species (1970–2019), the normal period (1990 to 2002), and the drought period (2003–2015). Here, we defined these years in the same length before the drought period (2003–2015) as the normal period. Since some anatomical features were highly correlated and responded similarly to climate, we classified anatomical features to remove redundant information. Hierarchical clustering of all the xylem anatomical features standard chronologies was carried out by Ward’s method in IBM SPSS Statistics for Windows, version 26.0 (IBMCorp., Armonk, N.Y., USA). The Z-Score normalization was used to view the similarities between each category of xylem anatomical features. To explore how trees respond to climate, we also calculated the Pearson correlations between the mean Z-Score for each category of xylem features and the main climate factors in the main seasons in three periods (the whole, normal, and drought periods). Finally, moving correlation analysis with a 20-yr window was performed to explore the temporal stability of dendroclimatic relations. The Pearson correlations between xylem features and main climate factors during the whole period were performed by the R package ‘treeclim’ (Zang and Biondi 2015) in R statistical programming environment, while the correlations during the normal and drought periods were calculated using the SPSS software. Moving correlation analyses were performed by the ‘treeclim’ package in R software. All data used for the t-test and correlation analysis were checked for the normality and homoscedasticity assumptions by Shapiro–Wilk test and Levene's test respectively. The Mann–Whitney U test was applied for very few datasets that do not follow a normal distribution.
Results
Comparisons of xylem features between species
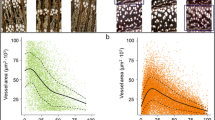
The xylem features of C. mollissima and Q. fabri were significantly different. Tree-ring width (RW) and all xylem features except for MVA of C. mollissima were significantly higher than Q. fabri (Fig. 3). The RW and VN of the two species decreased with the increase of cambium age, especially for C. mollissima. The MVA, RCTA, Kh, and Ks of Q. fabri increased with cambium age, while those of C. mollissima increased initially and then decreased after the cambium age of 30. The TVA and VD of the two species decreased and increased during the recent three decades (Fig. 4).
Differences in tree-ring width and main vessel features between Castanea mollissima (CM) and Quercus fabri (QF). a ring width (RW), b mean vessel area (MVA), c number of vessels (VN), d total vessel area (TVA), e mean percentage of conductive area within xylem (RCTA), f vessel density (VD), g tree-ring theoretical hydraulic conductivity (Kh), h xylem-specific theoretical hydraulic conductivity (Ks). ns = p > 0.05, *, = p < 0.05, ** = p < 0.01, *** = p < 0.001, **** = p < 0.0001
Trends of tree ring widths and main vessel features of Castanea mollissima (CM) and Quercus fabri (QF). a ring width (RW), b mean vessel area (MVA), c number of vessels (VN), d total vessel area (TVA), e mean percentage of conductive area within xylem (RCTA), f vessel density (VD), g tree-ring theoretical hydraulic conductivity (Kh), h xylem-specific theoretical hydraulic conductivity (Ks)
Different relationships among xylem features between species
All features were divided into three clusters (Fig. 5). The first cluster (HC1) was RW, representing the carbon fixation capacity. The second cluster (HC2) was more relevant to hydraulic safety, the RCTA, VD, MVA, and Ks for C. mollissima and the RCTA, Ks, and VD for Q. fabri. And the third cluster (HC3) was more relevant to hydraulic efficiency, for C. mollissima including VN, TVA, and Kh, and for Q. fabri including TVA, MVA, Kh, and VN (Fig. 5). Indicators within each category (HC2 or HC3) had similar inter-annual trends, except for VN in HC3 of Q. fabri (Fig. S2). Almost all indicators within the HC2 and HC3 categories of C. mollissima and Q. fabri had significantly positive correlations with each other (Fig. 6).
The result of hierarchical clustering of Castanea mollissima (CM) and Quercus fabri (QF). RW = Ring width, MVA = Mean vessel area, VN = Number of vessels, TVA = Total vessel area, RCTA = Mean percentage of conductive area within xylem, VD = Vessel density, Kh = Tree-ring theoretical hydraulic conductivity, Ks = Xylem-specific theoretical hydraulic conductivity
Correlations between RW and main vessel features of Castanea mollissima (CM) and Quercus fabri (QF) in the common period of 1970–2019. RW = Ring width, MVA = mean vessel area, VN = Number of vessels, TVA = Total vessel area, RCTA = Mean percentage of conductive area within xylem, VD = Vessel density, Kh = Tree-ring theoretical hydraulic conductivity, Ks = Xylem-specific theoretical hydraulic conductivity. In each graph, numbers that have been crossed out indicate non-significance (p > 0.05)
During the common period (1970–2019), RW (HC1) was significantly positively related to VN, TVA, and Kh for both species and significantly negatively correlated with RCTA, VD, and Ks of Q. fabri (Fig. 6). The relationship between HC1 and HC2 of the two species was significantly negative during the normal (1990–2002) and drought (2003–2015) periods (Fig. 7). The HC1 positively correlated with HC3 for the two species, especially for Q. fabri in the drought period which reached a 99% significant level. There was a significantly negative correlation relationship between HC2 and HC3 of Q. fabri during the drought period (Fig. 7). Relationships between indicators with or within each cluster during the normal and drought periods had similar correlation patterns (Fig. S3).
Different climate-growth relationships of C. mollissima and Q. fabri
Castanea mollissima was more sensitive to climate than Q. fabri (Fig. 8). The xylem features of C. mollissima were generally limited by both temperatures (T, Tmax, and Tmin) and water conditions (P, Rh, and SPEI), while these of Q. fabri were only limited by water conditions (Fig. 8). For C. mollissima, the HC1 was significantly positively correlated with the T in pSum and SPR, Tmax in pSum, and Tmin in SPR, AUT, and GS. The HC3 was significantly positively correlated with the T and Tmin in pSum and SUM, and significantly negatively correlated with Tmax in pWin/PNG. The HC1 was significantly negatively correlated with the P and SPEI in pSum and significantly positively correlated in pWin/PNG. It was also significantly negatively correlated with the Rh in pSum and PGS. The HC3 was significantly negatively correlated with the P and Rh in pSum, PGS, and SUM, and SPEI in pSum and SUM (Fig. 8). For Q. fabri, the HC1 was significantly negatively correlated with the P in pSum and AUT, and significantly positively correlated with the P and SPEI in pWin/PNG. The HC3 was significantly negatively correlated with the P, Rh, and SPEI in pSum and SUM, and P and Rh in PGS (Fig. 8).
Correlations between three clusters (HC1, HC2, and HC3) of xylem features and climate factors of Castanea mollissima (CM) and Quercus fabri (QF) during the common years of 1970–2019. See Fig. 5 for more details on HC1, HC2, and HC3 for two species. P = precipitation, Rh = relative humidity, T = mean temperature, Tmax = maximum temperature, Tmin = minimum temperature, SPEI = standardized precipitation evapotranspiration index. pSum = previous summer (previous June to August), pAut = previous autumn (previous September to November), pWin = previous winter (from previous December to current February), SPR = current spring (current March to May), SUM = current summer, AUT = current autumn, PGS = previous growing season (previous March to November), PNG = previous non-growing season (previous December to current February), GS = current growing season. * = p < 0.05
Contrasting response with normal and dry climatic conditions
The HC3 of both species was the most responsive to the climate, and it changed significantly between the normal and the dry period (Fig. 9). Compared to the normal period, the correlation during the dry period in C. mollissima was often enhanced, especially the positive correlation effect of moisture conditions in pWin/PNG, the negative correlation effect of temperatures in pWin/PNG and the positive correlation effect of temperatures in GS (Fig. 9). For Q. fabri, the correlation during the dry period was usually changed. The non-significant negative correlation of HC3 with P in pWin/PNG shifted to a significant positive correlation, and the significant positive correlation between them in SPR shifted to a non-significant negative correlation during the dry period. The significant positive correlation of T and Tmax in pWin/PNG became a non-significant negative correlation, and the non-significant negative correlation of T and Tmax in SPR became a significant positive correlation (Fig. 9).
Pearson correlations between xylem anatomical features and climate factors of Castanea mollissima (CM) and Quercus fabri (QF) during the normal (1990–2002) and drought (2003–2015) periods. See Fig. 5 for more details on HC1, HC2, and HC3 for two species. P = precipitation, Rh = relative humidity, T = mean temperature, Tmax = maximum temperature, Tmin = minimum temperature, SPEI = standardized precipitation evapotranspiration index. pSum = previous summer (from previous June to August), pAut = previous autumn ( previous September to November), pWin = previous winter (previous December to current February), SPR = current spring (current March to May), SUM = current summer, AUT = current autumn, PGS = previous growing season (previous March to November), PNG = previous non-growing season (previous December to current February), GS = current growing season. Dashed lines indicate significant levels of correlation (p < 0.05)
Compared with the normal period, the relationships between the HC1 and HC2 of both species and climate had insignificantly changed (Fig. 9). For C. mollissima, only the significant positive correlation between HC1 and P and SPEI in the pWin/PNG became insignificant during drought period. For Q. fabri, only the significant negative correlation of HC2 to P in GS changed to a weak positive correlation during the drought period (Fig. 9).
The temporal variations of dendroclimatic relationships
Moving correlation analysis showed that the impacts of GS temperatures (T, Tmin and Tmax) on the HC1 and HC3 of C. mollissima were shifted from non-significant positive to significant positive after the period of 1987–2006 (Fig. 10). The impacts of PNG temperatures (T, Tmin and Tmax) on the HC1 and HC3 of C. mollissima were shifted from non-significant positive to non-significant negative (HC1) or significant negative (HC3) around the period of 1978–1997 (Fig. 10). The impacts of P and SPEI of the GS on the HC3 of C. mollissima shifted from non-significant positive to significant negative around the period of 1981–2000, while the impacts of the Rh of the GS on the HC1 and HC3 of C. mollissima shifted from non-significant positive and negative to significant negative around the period of 1985–2004 (Fig. 10). The impacts of P and SPEI of the PNG on the HC1 and HC3 of C. mollissima shifted from non-significant positive to significant positive around the period of 1987–2006, while the impacts of Rh of the PNG on the HC1 and HC3 of C. mollissima shifted from non-significant positive to negative around the period of 1986–2005 (Fig. 10). The non-significant negative impacts of P and SPEI of the PNG on the HC2 of C. mollissima became significant around the period of 1987–2006 and then weakened around the period 1995–2014. Although the temporal variation pattern of the relationships between main climate factors and the HC1, HC2, and HC3 of Q. fabri was not as clear as or weaker than that of C. mollissima, it was generally similar to that of C. mollissima (Fig. 10).
Moving correlation analysis with a 20-year window between xylem anatomical features and climate factors of Castanea mollissima (CM) and Quercus fabri (QF) during the 1971–2019. See Fig. 5 for more details on HC1, HC2, and HC3 for two species. P = precipitation, Rh = relative humidity, T = mean temperature, Tmax = maximum temperature, Tmin = minimum temperature, SPEI = standardized precipitation evapotranspiration index. PNG = previous non-growing season (previous December to current February), GS = current growing season (March to November). * = p < 0.05
Discussion
Differences in xylem anatomical features of C. mollissima and Q. fabri
Species-specific differences in anatomical features can reflect differential growth or xylem adjustment to external environments (Fonti et al. 2010). Vessel features controlled potential water conductivity, reflecting the hydraulic safety or efficiency (Sperry et al. 2008). We found that the HC1 of both study species had a positive correlation with HC3, indicating carbon fixation capacity was positively related to hydraulic efficiency. Castanea mollissima had more but larger earlywood vessels than Q. fabri (Fig. 4), which allowed the former to have a higher TVA and Kh (reflecting water conductivity ability) (Hacke and Sperry 2001). The higher hydraulic conductivity of C. mollissima corresponded to its wider rings (RW) compared with Q. fabri, suggesting that improving water transport efficiency promotes radial growth by increasing the rate of gas exchange and photosynthetic efficiency (Mencuccini 2003; Zanne et al. 2010). This is in agreement with studies on other broadleaf tree species in tropical, temperate, and Mediterranean forests (Castagneri et al. 2020; Islam et al. 2018; Zhu et al. 2020). It seems to confirm and explain that the early-successional stage trees often grew faster than the late-successional trees (Martínez-Garza et al. 2013). A negative correlation between hydraulic efficiency and radial growth was found in Quercus canariensis (Gea-Izquierdo et al. 2012). This disagreement might be related to different relationships between xylem structure and carbon allocation among species or sites (Rita et al. 2015).
Moreover, the RW of the two species was significantly positively related to VN but had no correlations with MVA, suggesting trees improved water transport efficiency more by increasing the number of vessels than vessel size. This further confirmed the finding that radial growth of broadleaf tree species is more dependent on vessel numbers rather than size (Zhu et al. 2020). We also found a significantly negative correlation existed between HC2 and HC3 of Q. fabri during the drought period, suggesting Q. fabri experienced a strong trade-off between hydraulic efficiency and safety under drought conditions that guaranteed trees have a certain water transport capacity while reducing the occurrence of embolism and cavitation (Fonti et al. 2010; Sperry et al. 2008). With this trade-off strategy, late-successional stage Q. fabri may exhibit stronger self-regulation than early-successional stage C. mollissima during future warm and dry climates. Different hydraulic strategies of the two species, with C. mollissima focusing more on hydraulic efficiency and Q. fabri more on hydraulic safety, also corresponded well with the rapid growth of early-successional stage trees and the long lifespan of late-successional stage trees above.
The MVA of both species basically increased with cambial age (Fig. 4), which seems to be a common pattern (Rosell et al. 2017; Zhu et al. 2021). This pattern is related to tree height that increases with ontogeny: as trees grow and their height increases, vessel size is also larger (Carrer et al. 2015). The change in vessel size with cambium age is the adjustment of the pathway length resistance of water transport to tree height growth (Anfodillo et al. 2006; Carrer et al. 2015). In addition, many xylem features of C. mollissima showed a more evident downward trend than those of Q. fabri (Fig. 4). This confirmed the differences in trees at different successional stages: late-successional stage Q. fabri had a longer lifespan than early-successional stage C. mollissima (Navas et al. 2010). The relationship between the growth rate and longevity of trees at different successional stages may be an important component of biodiversity and is important for the development of forests (Black et al. 2008).
Climatic response of xylem anatomical features
As previously noted, differences existed in the xylem anatomical features of C. mollissima and Q. fabri, and likewise, differences were observed in their responses to climate. The early-successional stage C. mollissima was more sensitive to main climate factors than the late-succession stage Q. fabri (Fig. 9), which was in line with previous research (Oliveira et al. 2017). This was probably due to species-specific growth strategies. As a late-succession tree, Q. fabri might adapt to environmental changes by coordinating multiple complementary traits, so its response to climate was not significant (Gea-Izquierdo et al. 2013; Hacke et al. 2001). The late-successional trees usually had a higher shade tolerance, and a lower light compensates point and dark respiration rate than the early-successional stage trees (Kitao et al. 2016), implying the late-successional stage Q. fabri might be more adaptable and have wider ecological amplitudes than the early-successional stage C. mollissima. Besides, late-successional trees also needed longer time to fix more carbohydrates in order to achieve higher wood density (Krajnc et al. 2021). Higher wood density could bring higher security of embolism (Hacke et al. 2001), which seemed to be a compensation for lower tree growth, that is, trees produce safer xylem to compensate for their lower growth rate (Domec and Gartner 2003). It was consistent with our above conclusion that the late-succession stage Q. fabri has a slower growth rate but a safer xylem than the early-successional stage C. mollissima.
High temperatures could increase radial growth and vessel development (cell division and differentiation) by promoting effective cumulative temperatures, improving photosynthesis rate, and extend the length of growth period (Liu et al. 2012; Souto-Herrero et al. 2017). Hence, warming promoting radial growth and vessel features was found lots of studies (Bryukhanova and Fonti 2012; Tumajer and Lehejček 2019). For example, Zhu et al. (2020) pointed out that rapid warming around 1980 significantly promoted carbon assimilation and hydraulic efficiency of Fraxinus mandshurica from 19 temperate forest sites in northeast China. In our study, however, the effect of generalized warming is not evident, and we did not find the two study species showed a significant growth increase. The climate-sensitive C. mollissima even showed a slight growth decline in the last decades. The possible reasons for this difference are as follows: (1) A strong trade-off between the impacts of non-growing and growing season climates. In PNG, except for Rh, the responses of climate factors to HC1 and HC3 were opposite to that of GS. For example, a previous warm non-growing season decreases RW and HC3, but a current warm growing season produces the opposite effect. (2) The constantly changing competitive relationships among populations. Both interspecific and intraspecific competition within a forest stand has also increased over the years, which may lead to a growth decrease (Liang et al. 2019). (3) Trade-off between hydraulic efficiency and safety in xylem (Fonti et al. 2010; Zhu et al. 2017). For C. mollissima, the enhancing negative correlations between HC3 and water conditions (P, Rh, SPEI) in GS after the period of 1987–2006 (Fig. 10) indicated there was a strategy of maximization of water transport efficiency to adapt to slightly increasing water stress (Fig. 2). The C. mollissima probably had a greater risk of hydraulic failure caused by drought because of without a trade-off between hydraulic efficiency and safety in its xylem (Zhu et al. 2020). However, Q. fabri with this trade-off means it improving hydraulic safety at the cost of losing hydraulic efficiency, resulting in insignificant warming effects. (4) In addition, we speculated that this may be due to species characteristics of early-successional trees. They usually exhibited a clear growth decline after rapid growth.
This contrary impacts of climate in PNG and GS on tree growth might be related to species characteristics. The two species are deciduous trees without photosynthesize in winter. Warming winter temperature may increase the consumption of organic matter instead of accumulation (Harvey et al. 2020). For C. mollissima, moisture (P, Rh, and SPEI) showed a negative correlation with HC1 and HC3 in different seasons. In the rainy growing season, excessive moisture in pSum and SUM was not favorable for radial growth and vessel development. It was also noted by Jacob et al. (2020), who pointed out that precipitation impeded tree growth in wet conditions. This phenomenon was probably related to the soil property of the study area: the laterite had a high viscosity. In excessively humid environments, the respiration of the root system was affected by oxygen deficiency (González-González et al. 2015), and soil hypoxia increased root consumption and limited the role of carbohydrates in earlywood formation. Besides, this might be related to stomatal regulation in plants that balances plant photosynthesis and water loss (Lima Neto et al. 2015). Stomatal closure due to the pressure in the epidermal cells caused by excessive humidity inhibits transpiration, water uptake, and carbon assimilation (Nola et al. 2020). However, in the colder non-growing seasons, excessive humidity brought a greater risk of frost (Zhang et al. 2023; Zhu et al. 2021). In addition, moisture especially relative humidity was closely related to vapor pressure deficit that was closely related to gas exchange and photosynthesis (Li et al. 2019; Zhang et al. 2023).
In general, moving correlation analysis revealed that the relationship between three clusters of xylem features and main climate factors in PNG and GS changed significantly with time for C. mollissima but almost kept stable for Q. fabri (Fig. 10). This finding corresponded well with our previous suggestion that late-successional Q. fabri were less sensitive and more adaptable to climate changes and had wider ecological amplitudes than that of early-successional C. mollissima. The unstable dendroclimatic relationships with time, which could be related to species competition, warming, and the series of changes they caused.
The effects of climate warming on subtropical humid forest
Althought, we did not find a direct evidence that warming can significantly affect the radial growth of C. mollissima and Q. fabri as mentioned in previous studies in subtropics of China (Jing et al., 2022a, b; Zhang et al. 2023). However, their different hydraulic strategies, C. mollissima focused more on hydraulic efficiency and Q. fabri more on hydraulic safety, which also made C. mollissima potentially more vulnerable in the face of extreme climates. It is worth noting that C. mollissima is more sensitive to climate than Q. far and it will show more clear decline due to increasing the risk of hydraulic failure under continuous climate warming (Zhu et al. 2020). This indicates that warming is probably more favorable for the early-successional stage C. mollissima to shorten its lifespans and end its life history but no effect on that of the late-successional stage Q. fabri (Brienen et al. 2020; Loehle 1988). Hence, we inferred that warming may accelerate species replacement and forest succession. Besides, although we do not have data, warming may enhance tree establishment in the early stages of succession, which may facilitate early forest succession (Shuman et al. 2011). Our previous research in this region showed that warming-induced drought stress has led to a significant decline of some early-successional stage tree species, such as widely planted Cunninghamia lanceolata and Pinus massoniana (Jing et al., 2022a, b; Zhang et al. 2023), which further suggested that warming probably favored early forest succession.
Furthermore, it is important to note that we only studied the response of the early-successional stage C. mollissima and the late-successional stage Q. fabri to warming and were not able to determine whether climate warming would change community relationships, such as species competitive relationships that are important drivers of forest succession and often lead to species turnover (De Mesel et al. 2015; Pulsford et al. 2016). Although we didn’t measure species competition, tree-to-tree competition will increase as they grow. Considering the age of the trees and the features of different-succession stages species, and taking into account the trend of tree growth in recent years (Fig. 4), their competitive relationships did appear to have changed. The late-successional stage Q. fabri will gradually be at an advantage compared to C. mollissima, which indirectly speeds up the forest succession process. This seems to suggest that competition and species characteristics, apart from climates, may also play important roles in affecting tree growth. Also, climate warming will amplify the effects of competition due to their differential climate sensitivity.
In addition, our study only looked at two tree species in one sampling site in the subtropics humid forest. Using them as subjects may not accurately infer changes in forest succession processes. About ten trees of each species were used in the final analyses due to quantitative wood anatomy being very time-consuming (von Arx and Carrer 2014). Although many studies with similar or even lower sample sizes have been successfully published (Chen et al. 2021; Zhu et al. 2020; Zhu et al. 2021), we have to admit that the limited number of trees may bring uncertainty to this study to some extent. Nevertheless, our study at least proved that a great different xylem adjustment and growth responses to warming existed between the early- and late-succession tree species, and xylem anatomical features could provide new insights into how warming affects forest succession. In the future, xylem anatomy studies of multiple species on a large scale, combined with models or other methods, may be able to exactly infer forest succession processes under warming.
Conclusions
Our study shows that the early-successional stage C. mollissima and the late-successional stage Q. fabri adopted different growth strategies to cope with climate changes. All xylem parameters, except for MVA, of C. mollissima were significantly higher than that of Q. fabri. The radial growth (HC1) of both species was negatively and positively related to those hydraulic safety-related (HC2) and hydraulic efficiency-related (HC3) xylem features. The HC2 and HC3 of Q. fabri showed a significant negative correlation during the drought period, indicating that Q. fabri experienced stronger trade-offs between hydraulic efficiency and safety than early-successional stage C. mollissima under drought conditions. Future continuous climate warming will probably lead to a clear growth decline of C. mollissima because of the increasing risk of hydraulic failure.
Castanea mollissima was more sensitive to climate than Q. fabri. The HC3 of C. mollissima was positively and negatively correlated with temperatures (T, Tmin, and Tmax) and moisture conditions (P, Rh, and SPEI), especially in pSum and SUM. The HC1 of both species was significantly negative and positively related to moisture conditions in pSum and pWin. The relationship between the three clusters and climate generally changed significantly with time for C. mollissima but almost kept stable for Q. fabri, which might be related to warming, species competition, and the series of changes they caused. Differences in hydraulic strategies and species characteristics, trade-offs between non-growing and growing season climates, and specific competition led to no clear effects of generalized warming on tree growth in this study.
Changing species competitive relationships and lifespans of the early- and late-successional stage species caused by warming may accelerate species replacement and forest succession. Our study highlighted the significant differences in xylem adjustment and growth responses to warming between the early- and late-succession tree species and provided some new insights into quantitative wood anatomy for studying how warming affects forest succession. This study was limited to study area, number of species and sites, and sample depths, and whether our findings could be generalized to a wide range remains to be investigated.
Data availability
Data will be made available on request.
References
Abrams MD, Orwig DA, Demeo TE (1995) Dendroecological analysis of successional dynamics for a presettlement- origin white-pine-mixed-oak forest in the southern Appalachians, USA. J Ecol 83(1):123–133. https://doi.org/10.2307/2261156
Altman J, Ukhvatkina ON, Omelko AM, Macek M, Plener T, Pejcha V, Cerny T, Petrik P, Srutek M, Song JS, Zhmerenetsky AA, Vozmishcheva AS, Krestov PV, Petrenko TY, Treydte K, Dolezal J (2018) Poleward migration of the destructive effects of tropical cyclones during the 20th century. Proc Natl Acad Sci 115(45):11543–11548. https://doi.org/10.1073/pnas.1808979115
Anderson-Teixeira KJ, Miller AD, Mohan JE, Hudiburg TW, Duval BD, Delucia EH (2013) Altered dynamics of forest recovery under a changing climate. Glob Chang Biol 19(7):2001–2021. https://doi.org/10.1111/gcb.12194
Anfodillo T, Carraro V, Carrer M, Fior C, Rossi S (2006) Convergent tapering of xylem conduits in different woody species. New Phytol 169(2):279–290. https://doi.org/10.1111/j.1469-8137.2005.01587.x
Arroyo-Rodríguez V, Melo FPL, Martínez-Ramos M, Bongers F, Chazdon RL, Meave JA, Norden N, Santos BA, Tabarelli Leal IR, M, (2017) Multiple successional pathways in human-modified tropical landscapes: new insights from forest succession, forest fragmentation and landscape ecology research. Biol Rev 92(1):326–340. https://doi.org/10.1111/brv.12231
Bastin JF, Finegold Y, Garcia C, Mollicone D, Rezende M, Routh D, Zohner CM, Crowther TW (2019) The global tree restoration potential. Science 365(6448):76–79 https://www.science.org/doi/10.1126/science.aax0848
Black BA, Colbert JJ, Pederson N (2008) Relationships between radial growth rates and lifespan within North American tree species. Écoscience 15(3):349–357. https://doi.org/10.2980/15-3-3149
Boulanger Y, Taylor AR, Price DT, Cyr D, McGarrigle E, Rammer W, Sainte-Marie G, Beaudoin A, Guindon L, Mansuy N (2017) Climate change impacts on forest landscapes along the Canadian southern boreal forest transition zone. Landscape Ecol 32(7):1415–1431. https://doi.org/10.1007/s10980-016-0421-7
Brandes AFN, Sánchez-Tapia A, Sansevero JBB, Albuquerque RP, Barros CF (2018) Fire records in tree rings of Moquiniastrum polymorphum: potential for reconstructing fire history in the Brazilian Atlantic Forest. Acta Botanica Brasilica 33(1):61–66. https://doi.org/10.1590/0102-33062018abb0282
Brienen RJW, Lebrija-Trejos E, Van Breugel M, Pérez-García EA, Bongers F, Meave JA, Martínez-Ramos M (2009) The potential of tree rings for the study of forest succession in southern Mexico. Biotropica 41(2):186–195. https://doi.org/10.1111/j.1744-7429.2008.00462.x
Brienen RJW, Caldwell L, Duchesne L, Voelker S, Barichivich J, Baliva M, Ceccantini G, Di Filippo A, Helama S, Locosselli GM, Lopez L, Piovesan G, Schöngart J, Villalba R, Gloor E (2020) Forest carbon sink neutralized by pervasive growth-lifespan trade-offs. Nat Commun 11(1):4241. https://doi.org/10.1038/s41467-020-17966-z
Bryukhanova M, Fonti P (2013) Xylem plasticity allows rapid hydraulic adjustment to annual climatic variability. Trees 27(3):485–496. https://doi.org/10.1007/s00468-012-0802-8
Brzeziecki B, Kienast F (1994) Classifying the life-history strategies of trees on the basis of the Grimian model. For Ecol Manag 69(1–3):167–187. https://doi.org/10.1016/0378-1127(94)90227-5
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26(2):115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Cadotte MW, Tucker CM (2017) Should environmental filtering be abandoned? Trends Ecol Evol 32(6):429–437. https://doi.org/10.1016/j.tree.2017.03.004
Cannone N, Diolaiuti G, Guglielmin M, Smiraglia C (2008) Accelerating climate change impacts on alpine glacier forefield ecosystems in the European Alps. Ecol Appl 18(3):637–648. https://doi.org/10.1890/07-1188.1
Carrer M, von Arx G, Castagneri D, Petit G (2015) Distilling allometric and environmental information from time series of conduit size: the standardization issue and its relationship to tree hydraulic architecture. Tree Physiol 35(1):27–33. https://doi.org/10.1093/treephys/tpu108
Castagneri D, Carrer M, Regev L, Boaretto E (2020) Precipitation variability differently affects radial growth, xylem traits and ring porosity of three Mediterranean oak species at xeric and mesic sites. Sci Total Environ 699:134285. https://doi.org/10.1016/j.scitotenv.2019.134285
Chen L, Zhu L, Liu S, Lei P, Yuan D, Li Z, Wang X (2021) Xylem features detrending methods matter: a case study on earlywood vessels of Fraxinus mandshurica. Ecol Indicators 130: 108041. https://doi.org/10.1016/j.ecolind.2021.108041
Cherubini P, Battipaglia G, Innes JL (2021) Tree vitality and forest health: can tree-ring stable isotopes be used as indicators? Curr for Rep 7:69–80. https://doi.org/10.1007/s40725-021-00137-8
De Mesel I, Kerckhof F, Norro A, Rumes B, Degraer S (2015) Succession and seasonal dynamics of the epifauna community on offshore wind farm foundations and their role as stepping stones for non-indigenous species. Hydrobiologia 756(1):37–50. https://doi.org/10.1007/s10750-014-2157-1
Domec J-C, Gartner BL (2003) Relationship between growth rates and xylem hydraulic characteristics in young, mature and old-growth ponderosa pine trees. Plant, Cell Environ 26(3):471–483. https://doi.org/10.1046/j.1365-3040.2003.00978.x
Esposito MP, Nakazato RK, Pedroso ANV, Lima MEL, Figueiredo MA, Diniz AP, Kozovits AR, Domingos M (2018) Oxidant-antioxidant balance and tolerance against oxidative stress in pioneer and non-pioneer tree species from the remaining Atlantic Forest. Sci Total Environ 625:382–393. https://doi.org/10.1016/j.scitotenv.2017.12.255
Fonti P, von Arx G, García-González I, Eilmann B, Sass-Klaassen U, Gärtner H, Eckstein D (2010) Studying global change through investigation of the plastic responses of xylem anatomy in tree rings. New Phytol 185(1):42–53. https://doi.org/10.1111/j.1469-8137.2009.03030.x
Gea-Izquierdo G, Fonti P, Cherubini P, Martín-Benito D, Chaar H, Cañellas I (2012) Xylem hydraulic adjustment and growth response of Quercus canariensis Willd to climatic variability. Tree Physiol 32(4):401–413. https://doi.org/10.1093/treephys/tps026
Gea-Izquierdo G, Battipaglia G, Gärtner H, Cherubini P (2013) Xylem adjustment in Erica arborea to temperature and moisture availability in contrasting climates. IAWA J 34(2):109–126. https://doi.org/10.1163/22941932-00000010
González-González BD, Vázquez-Ruiz RA, García-González I (2015) Effects of climate on earlywood vessel formation of Quercus robur and Q. pyrenaica at a site in the northwestern Iberian Peninsula. Can J Forest Res 45(6):698–709. https://doi.org/10.1139/cjfr-2014-0436
Guada G, Sass-Klaassen U, Souto-Herrero M, García-González I (2021) Anatomical tree-ring chronologies and seasonal patterns of cambial dynamics are valuable indicators of tree performance of two oak species at the Atlantic-Mediterranean boundary. Dendrochronologia 70:125893. https://doi.org/10.1016/j.dendro.2021.125893
Hacke UG, Sperry JS (2001) Functional and ecological xylem anatomy. Perspect Plant Ecol Evolut Syst 4(2):97–115. https://doi.org/10.1078/1433-8319-00017
Hacke UG, Sperry JS, Pockman WT, Davis SD, McCulloh KA (2001) Trends in wood density and structure are linked to prevention of xylem implosion by negative pressure. Oecologia 126(4):457–461. https://doi.org/10.1007/s004420100628
Harvey JE, Smiljanić M, Scharnweber T, Buras A, Cedro A, Cruz-García R, Drobyshev I, Janecka K, Jansons Ā, Kaczka R, Klisz M, Läänelaid A, Matisons R, Muffler L, Sohar K, Spyt B, Stolz J, van der Maaten E, van der Maaten-Theunissen M, Vitas A, Weigel R, Kreyling J, Wilmking M (2020) Tree growth influenced by warming winter climate and summer moisture availability in northern temperate forests. Glob Change Biol 26(4):2505–2518. https://doi.org/10.1111/gcb.14966
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bulletin 43:69–78
Islam M, Rahman M, Bräuning A (2018) Long-term hydraulic adjustment of three tropical moist forest tree species to changing climate. Front Plant Sci 9:1761. https://doi.org/10.3389/fpls.2018.01761
Jacob M, De Ridder M, Vandenabeele M, Asfaha T, Nyssen J, Beeckman H (2020) The response of Erica arborea L. tree growth to climate variability at the Afro-alpine tropical highlands of North Ethiopia. Forests 11(3):310. https://doi.org/10.3390/f11030310
Jing M, Zhu L, Liu S, Cao Y, Zhu Y, Yan W (2022a) Warming-induced drought leads to tree growth decline in subtropics: evidence from tree rings in central China. Front Plant Sci 13: 964400. https://doi.org/10.3389/fpls.2022.964400
Jing M, Zhu L, Cherubini P, Yuan D, Li Z, Wang X, Liu S (2022b) Responses of radial growth of Pinus massoniana and Castanopsis eyrei to climate change at different elevations in south China. Ecol Indic 145: 109602. https://doi.org/10.1016/j.ecolind.2022.109602
Kitao M, Hida T, Eguchi N, Tobita H, Utsugi H, Uemura A, Kitaoka S, Koike T (2016) Light compensation points in shade-grown seedlings of deciduous broadleaf tree species with different successional traits raised under elevated CO2. Plant Biol 18(S1):22–27. https://doi.org/10.1111/plb.12400
Krajnc L, Hafner P, Gričar J (2021) The effect of bedrock and species mixture on wood density and radial wood increment in pubescent oak and black pine. For Ecol Manag 481: 118753. https://doi.org/10.1016/j.foreco.2020.118753
Li Q, Wei M, Li Y, Feng G, Wang Y, Li S, Zhang D (2019) Effects of soil moisture on water transport, photosynthetic carbon gain and water use efficiency in tomato are influenced by evaporative demand. Agric Water Manag 226:105818. https://doi.org/10.1016/j.agwat.2019.105818
Liang H, Huang J-G, Ma Q, Li J, Wang Z, Guo X, Zhu H, Jiang S, Zhou P, Yu B, Luo D (2019) Contributions of competition and climate on radial growth of Pinus massoniana in subtropics of China. Agric for Meteorol 274:7–17. https://doi.org/10.1016/j.agrformet.2019.04.014
Lima Neto MC, Martins MD, Ferreira-Silva SL, Silveira JA (2015) Jatropha curcas and Ricinus communis display contrasting photosynthetic mechanisms in response to environmental conditions. Sci Agric 72:260–269. https://doi.org/10.1590/0103-9016-2014-0325
Lin Y, Lu Y, Song Z, Huang D (2018) Characterizations of the endogenous starch hydrolase inhibitors in acorns of Quercus fabri Hance. Food Chem 258:111–117. https://doi.org/10.1016/j.foodchem.2018.03.001
Liu B, Li Y, Eckstein D, Zhu L, Dawadi B, Liang E (2012) Has an extending growing season any effect on the radial growth of Smith fir at the timberline on the southeastern Tibetan Plateau? Trees 27(2):441–446. https://doi.org/10.1007/s00468-012-0819-z
Loehle C (1988) Tree life history strategies: the role of defenses. Can J Forest Res 18(2):209–222. https://doi.org/10.1139/x88-032
Lohbeck M, Poorter L, Martinez-Ramos M, Rodriguez-Valázquez J, van Breugel M, Bongers F (2014) Changing drivers of species dominance during tropical forest succession. Funct Ecol 28(4):1052–1058. https://doi.org/10.1111/1365-2435.12240
Martínez-Garza C, Bongers F, Poorter L (2013) Are functional traits good predictors of species performance in restoration plantings in tropical abandoned pastures? For Ecol Manag 303:35–45. https://doi.org/10.1016/j.foreco.2013.03.046
Mencuccini M (2003) The ecological significance of long-distance water transport: short-term regulation, long-term acclimation and the hydraulic costs of stature across plant life forms. Plant, Cell Environ 26(1):163–182. https://doi.org/10.1046/j.1365-3040.2003.00991.x
Molina E, Valeria O, De Grandpre L, Ramirez JA, Cyr D, Boulanger Y (2021) Projecting future aboveground biomass and productivity of managed eastern Canadian mixedwood boreal forest in response to climate change. For Ecol Manag 487: https://doi.org/10.1016/j.foreco.2021.119016
Navas M-L, Roumet C, Bellmann A, Laurent G, Garnier E (2010) Suites of plant traits in species from different stages of a Mediterranean secondary succession. Plant Biol 12(1):183–196. https://doi.org/10.1111/j.1438-8677.2009.00208.x
Nola P, Bracco F, Assini S, von Arx G, Castagneri D (2020) Xylem anatomy of Robinia pseudoacacia L. and Quercus robur L. is differently affected by climate in a temperate alluvial forest. Ann Forest Sci 77(1):8. https://doi.org/10.1007/s13595-019-0906-z
Oliveira HC, da Silva LMI, de Freitas LD, Debiasi TV, Marchiori NM, Aidar MPM, Bianchini E, Pimenta JA, Stolf-Moreira R (2017) Nitrogen use strategies of seedlings from neotropical tree species of distinct successional groups. Plant Physiol Biochem 114:119–127. https://doi.org/10.1016/j.plaphy.2017.03.003
Pulsford SA, Lindenmayer DB, Driscoll DA (2016) A succession of theories: purging redundancy from disturbance theory. Biol Rev 91(1):148–167. https://doi.org/10.1111/brv.12163
R Core Team. (2022) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria
Rita A, Cherubini P, Leonardi S, Todaro L, Borghetti M (2015) Functional adjustments of xylem anatomy to climatic variability: insights from long-term Ilex aquifolium tree-ring series. Tree Physiol 35(8):817–828. https://doi.org/10.1093/treephys/tpv055
Rosell JA, Olson ME, Anfodillo T (2017) Scaling of xylem vessel diameter with plant size: causes, predictions, and outstanding questions. Curr for Rep 3(1):46–59. https://doi.org/10.1007/s40725-017-0049-0
Shuman JK, Shugart HH, Ohalloran TL (2011) Sensitivity of Siberian larch forests to climate change. Glob Change Biol 17(7):2370–2384. https://doi.org/10.1111/j.1365-2486.2011.02417.x
Souto-Herrero M, Rozas V, García-González I (2017) A 481-year chronology of oak earlywood vessels as an age-independent climatic proxy in NW Iberia. Global Planet Change 155:20–28. https://doi.org/10.1016/j.gloplacha.2017.06.003
Sperry JS, Meinzer FC, McCulloh KA (2008) Safety and efficiency conflicts in hydraulic architecture: scaling from tissues to trees. Plant, Cell Environ 31(5):632–645. https://doi.org/10.1111/j.1365-3040.2007.01765.x
Taylor AR, Chen HYH, VanDamme L (2009) A review of forest succession models and their suitability for forest management planning. Forest Sci 55(1):23–36. https://doi.org/10.1093/forestscience/55.1.23
Tei S, Sugimoto A, Yonenobu H, Matsuura Y, Osawa A, Sato H, Fujinuma J, Maximov T (2017) Tree-ring analysis and modeling approaches yield contrary response of circumboreal forest productivity to climate change. Glob Change Biol 23(12):5179–5188. https://doi.org/10.1111/gcb.13780
Teixeira HM, Cardoso IM, Bianchi FJ, da Cruz Silva A, Jamme D, Peña-Claros M (2020) Linking vegetation and soil functions during secondary forest succession in the Atlantic forest. For Ecol Manag 457: 117696. https://doi.org/10.1016/j.foreco.2019.117696
Tumajer J, Lehejček J (2019) Boreal tree-rings are influenced by temperature up to two years prior to their formation: a trade-off between growth and reproduction? Environ Res Lett 14(12): 124024. https://doi.org/10.1088/1748-9326/ab5134
Verheyden A, Kairo JG, Beeckman H, Koedam N (2004) Growth rings, growth ring formation and age determination in the mangrove Rhizophora mucronata. Ann Bot 94(1):59–66. https://doi.org/10.1093/aob/mch115
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming: the standardized precipitation evapotranspiration index. J Clim 23(7):1696–1718. https://doi.org/10.1175/2009JCLI2909.1
von Arx G, Carrer M (2014) ROXAS—a new tool to build centuries-long tracheid-lumen chronologies in conifers. Dendrochronologia 32(3):290–293. https://doi.org/10.1016/j.dendro.2013.12.001
Wen Y (2018) Reseach on optimization of spatial structure of typical broad-leaved forest stands around dongting lake area base on stand distribution pattern. PhD dissertation, Central South University of Forestry and Technology, Changsha.
Yu G, Chen Z, Piao S, Peng C, Ciais P, Wang Q, Zhu X (2014) High carbon dioxide uptake by subtropical forest ecosystems in the East Asian monsoon region. Proc Natl Acad Sci 111(13):4910–4915. https://doi.org/10.1073/pnas.1317065111
Zang C, Biondi F (2015) treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38(4):431–436. https://doi.org/10.1111/ecog.01335
Zanne AE, Westoby M, Falster DS, Ackerly DD, Loarie SR, Arnold SEJ, Coomes DA (2010) Angiosperm wood structure: Global patterns in vessel anatomy and their relation to wood density and potential conductivity. Am J Bot 97(2):207–215. https://doi.org/10.3732/ajb.0900178
Zhang Y, Song C (2006) Impacts of afforestation, deforestation, and reforestation on forest cover in China from 1949 to 2003. J Forest 104(7):383–387. https://doi.org/10.1093/jof/104.7.383
Zhang R, Hu Z, Cherubini P, Cooper DJ, Zhu L, Lei P (2023) Tree-ring data reveal trees are suffering from severe drought stress in the humid subtropical forest. For Ecol Manag 546: 121330. https://doi.org/10.1016/j.foreco.2023.121330
Zhou G, Peng C, Li Y, Liu S, Zhang Q, Tang X, Liu J, Yan J, Zhang D, Chu G (2013) A climate change-induced threat to the ecological resilience of a subtropical monsoon evergreen broad-leaved forest in Southern China. Glob Change Biol 19(4):1197–1210. https://doi.org/10.1111/gcb.12128
Zhu L, Wang X, Pederson N, Chen Z, Cooper DJ, Zhang Y, Li Z (2018) Spatial variability in growth-climate relationships of Amur cork tree (Phellodendron amurense) and their connections with PDO in Northeast China. J Geophys Res Biogeosci 123:1625–1636. https://doi.org/10.1029/2017JG004292
Zhu L, Wang X, Li Z (2017) Anatomical characteristics of xylem in tree rings and its relationship with environments. Chinese J Plant Ecol 41(2):238–251. https://doi.org/10.17521/cjpe.2016.0198
Zhu C, Shi F, Chen Y, Wang M, Zhao Y, Geng G (2019) Transcriptome analysis of Chinese chestnut (Castanea mollissima Blume) in response to Dryocosmus kuriphilus Yasumatsu infestation. Int J Mol Sci 20(4):855. https://doi.org/10.3390/ijms20040855
Zhu L, Cooper DJ, Yuan D, Li Z, Zhang Y, Liang H, Wang X (2020) Regional scale temperature rather than precipitation determines vessel features in earlywood of Manchurian Ash in temperate forests. J Geophys Res Biogeosci 125(11):e2020JG005955. https://doi.org/10.1029/2020JG005955
Zhu L, Liu S, Arzac A, Cooper DJ, Jin Y, Yuan D, Zhu Y, Zhang X, Li Z, Zhang Y, Liang H, Wang X (2021) Different response of earlywood vessel features of Fraxinus mandshurica to rapid warming in warm-dry and cold-wet areas. Agric for Meteorol 307:108523. https://doi.org/10.1016/j.agrformet.2021.108523
Acknowledgements
We would like to thank the two anonymous reviewers for their suggestions and comments. We are grateful to the Forestry Bureau staff for their assistance in the field.
Funding
Open Access funding provided by Lib4RI – Library for the Research Institutes within the ETH Domain: Eawag, Empa, PSI & WSL. The study was supported by the National Natural Science Foundation of China (42107476, 31971455), the Natural Science Foundation of Hunan Province (2021JJ41075), the China Postdoctoral Science Foundation (2020M682600), the China Postdoctoral International Exchange Fellowship Program (PC2021099), and the Science and Technology Innovation Program of Hunan Province (2020RC2058).
Author information
Authors and Affiliations
Contributions
Conceptualization: PC, SL, PL and LZ; Methodology: ZH, RZ and LZ; Formal analysis and investigation: ZH and RZ; Writing—original draft preparation: ZH; Writing—review and editing: ZH, LZ, PC, SL, PL, and RZ; Funding acquisition: PL and LZ; Supervision: LZ.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Miren del Río.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hu, Z., Zhu, L., Liu, S. et al. Xylem adjustment and growth response of early- and late-successional tree species to rapid warming. Eur J Forest Res 143, 785–801 (2024). https://doi.org/10.1007/s10342-023-01655-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-023-01655-9