Abstract
Among the vascular plants there is a lack of the typical epixylous species but they are a constant component on decaying wood. Their distribution patterns on this kind of substrate seem to be the least known among phototrophs. A total of 454 dead logs of Picea abies were analyzed with regard to cover of vascular plants and the independent morphometric features of logs and altitude. Four types of forest were compared, and the frequency and cover of the most frequent species were analyzed across the forest communities along the decomposition stage. Among the logs that were studied, 292 were colonized by vascular plants. The highest number of colonized logs was recorded in Calamagrostio villosae-Piceetum and the lowest in a deciduous beech forest of the Fagetalia order. Detrended correspondence analysis revealed that the dead logs occurring in the four forest communities differed significantly in species composition. Constrained correspondence analysis showed that six variables significantly explained the species variation, i.e., altitude, shade, moisture, decomposition stage, cover of bryophytes and status of forest (protected vs. managed). The results of the indicator value method indicated that of the 34 found species, ten could be treated as indicator species for the forest communities that were analyzed. The statistical analysis did not confirm significant role of coarse woody debris as a secondary habitat for rare and protected vascular plants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The role of dead wood in a forest ecosystem is well recognized (Holeksa 2001). Depending on its decomposition stage, decaying wood provides a habitat for numerous types of organisms including bacteria, fungi and invertebrates. Standing dead trees, i.e., snags, can be used by nesting birds and mammals. Vascular plants seem to be the least known as colonizers of dead wood among phototrophs (Stokland et al. 2012). One of the main reasons for the lack of studies is the conviction that in contrast to mosses, liverworts or lichens, only common vascular plants are to be found on dead logs. Some bryophytes and lichen species prefer coarse woody debris (CWD), which plays a significant role in the maintenance of biodiversity (Jonsson et al. 2005; Riffell et al. 2011; Dittrich et al. 2014). Little is known about the patterns of dead wood colonization by vascular plants, i.e., species composition and species diversity, their relations with the physical–chemical properties of the decaying wood or the community assembly of vascular plants that inhabit dead wood. Vascular plants inhabiting mechanisms were partially described by Harmon et al. (1986). Vascular plants can send their roots through bark to extract water and nutrients or can develop roots through the mat of decaying fine litter that accumulates during the decaying process. Plants growing on CWD influence decomposition and vice versa. During the decaying of plants on CWD, the amount of organic matter increases, which is followed by settlement of more demanding plants (Harmon et al. 1986). This phenomenon resembles the process of succession and community assembly that is driven by abiotic factors. The assembly rules for natural and close-natural forest communities have been reported to be better explained by niche partitioning and trade-off processes than by a neutral model (Nakashizuka 2001). We assume that the colonization of CWD by vascular plants is also better predicted by the niche theory in spite of the fact that CWD is of minor significance and is only one of the many types of substrates in forest ecosystems. The dead wood that is present in various forest communities should differ in the species composition of colonizing flora due to differences in the niches of plants.
This study focused on fallen logs of the Norway spruce Picea abies L. It is commonly acknowledged that CWD enhances the regeneration of this type of tree (Mayer et al. 1972; Korpel 1989; Hofgaard 1993; Kuuluvainen 1994; Zielonka 2006). Generally, cryptogams colonizing the CWD of P. abies are already known (Kruys and Jonsson 1999; Żarnowiec and Staniaszek-Kik 2009). Only a few studies have focused on the vascular flora on spruce dead wood. Zielonka and Piątek (2004) studied colonization by herbaceous and dwarf plants on decaying logs of P. abies in a subalpine spruce forest (Plagiothecio-Piceetum) in the Polish Tatra Mountains. They presented the species composition and related it to the stage of decomposition. Subsequently, Kushnevskaya et al. (2007) analyzed vegetation including vascular plants on fallen spruce trees in northwestern Russia and Dittrich et al. (2013, 2014), among others, reported which vascular plants were observed on dead lying trunks in the Harz Mts in Germany. In our study, which focused on vascular plants, we decided to select logs from among other types of CWD. Lying trunks (logs) are more frequently colonized than standing dead trees and other types of dead wood (Lee and Sturgess 2001; Dittrich et al. 2013), which may prove that not only epiphyte species on living trees are potential dead wood colonizers but also other plants that begin to appear on CWD during decomposition. This process is attributable to the more efficient water supply on lying trunks than on standing trunks (Söderström 1988; Laaka-Lindberg et al. 2005; Dittrich et al. 2013). It is believed that the process of colonization starts with epiphytic lichens which grow on bark of living trees, followed by bryophytes and then vascular plants from forest floor in the last stage (Andersson and Hytteborn 1991; Zielonka and Piątek 2004). Taking this into account, we were curious about how the neighborhood, in terms of the forest type, affected the species composition of colonized logs. The decomposition stage and other properties of the logs as well as the site characteristics were included in the study. Despite the fact that there were no known obligate epixylic vascular plant species, we believe that process of colonization by vascular plants is not random but is dependent on many intrinsic and extrinsic factors and that some species can be efficient colonizers. Among forest communities, extrinsic factors such as altitude and light conditions can have an impact on species diversity, whereas within forest communities the traits of CWD affect colonizing species. In particular, we hypothesized that: (1) colonization frequency (i.e., the proportion of colonized vs. non-colonized logs) does not depend on the type of forest community in which logs are lying; (2) logs located in different forest communities differ in species composition in terms of the biodiversity and frequency of particular plant species; (3) some environmental factors, i.e., decomposition and altitude, are the most important explanatory variables in species composition across forest communities, and (4) the status of a forest, i.e., protection versus management, also contribute to the differences in the species composition of colonized logs.
Materials and methods
Study area and design
The studies were conducted in the area of the Karkonosze Mts in the years 2003–2008. The area included the Karkonosze National Park (KNP), which lies within the Sudety Mts. This area is characterized by a severe high mountainous climate (Fig. 1). At the top of the highest peak of Sudety Mts (Śnieżka, 1602 m a.s.l.), the vegetation period lasts 131 days, the mean annual temperature is 0.1 °C, and precipitation is 1158 mm. In the lower parts, i.e., in Bystrzyca Kłodzka (at 368 m a.s.l.), these values are 221 days, 7.3 °C and 705 mm, respectively (Kosiński and Bednorz 2003). The forest ecosystems occupy ca 70 % of Karkonosze Mts., which amounts to 13,505 ha. In total, 30 % of forested areas are situated in KNP. The percentage of spruce P. abies in the tree stand is estimated at 90 %, beech Fagus sylvatica (3 %), Scots pine Pinus sylvestris (3 %), birch Betula pendula (2 %) and the remaining ones (2 %). The dominance of the spruce forests is the consequence of the forest management practice from nineteenth and twentieth century that lasted until 1914 (Danielewicz et al. 2013). Spruce was cultivated in the lower montane belt, i.e., (500–1000 m a.s.l.) by foresters with seeds that originated from the Alps on beech forest sites. The spruce forests occupy 85 % of the forest area in the lower montane belt. Their provenance is believed to be the reason for the decline in P. abies in recent decades due to its weak adaptation to Polish conditions both in terms of abiotic ones, i.e., climate and air pollution (years 1960–1980) and biotic ones, mainly fungal diseases (Fabiszewski and Wojtuń 1994; Stachurski et al. 1994; Jadczyk 2009). However, in the upper forest montane belt, i.e., (1000–1250 m a.s.l.) where the spruce forests originated, a natural decline of this tree was also observed. At present, the accumulation of pollutants, SO2 and NO x emissions and climate change are regarded as the main reasons for the defoliation and dying out of P. abies in the Polish Sudety Mts (Modrzyński 2003; Korzybski et al. 2013). The forest communities selected for this study are described in Table 1. These included a subalpine reedgrass spruce forest—Calamagrostio villosae-Piceetum, which forms the upper forest montane belt (1000–1250 m a.s.l.). This forest is considered to be the only near-natural climate-conditioned forest community in this altitude range in the mountains. It is a climax forest community (Matuszkiewicz 2008a). It occupies almost all of the available places and grows on podzolic and stony soils. The other forest types were a lower montane belt (500–1000 m a.s.l.) comprising a fir-spruce mixed coniferous forest—Abieti-Piceetum and a beech forest of the Fagetalia order (acidophilus beech forest—Luzulo luzuloidis-Fagetum, and a fertile beech forest—Dentario enneaphylli-Fagetum). In addition, a forest planted with P. abies, which is mainly distributed between 600 and 1000 m a.s.l., was also taken into account. In order to avoid pseudoreplication sensu Hurlbert (1984), random-stratified sampling was applied to cover all distinguished forest communities and protected and non-protected forests. The entire area was searched in terms of the absence/presence of vascular plants on decaying logs of P. abies. In total, 154 10 m × 10 m plots were established. Both protected (belonging to the KNP) and managed forests (outside of the KNP) were subjects of the study. In the protected forests, the management practices are aimed at the reconstruction of forests into natural forests.
According to Danielewicz et al. (2013) when the threat to the environment that is associated with air pollution by industry ceased, foresters, scientists and conservationists began numerous revitalization programs of forest ecosystems. CWD such as snags and uprooted trees is left in forests. Forests regenerate in a natural way. Although reconstruction is also conducted in managed forests, cuttings and planting trees also take place (Danielewicz et al. 2013).
Only forest areas, i.e., natural forest communities (according to the potential vegetation concept of Matuszkiewicz 2008b) and seminatural forests (partially planted), were included. The part of a destroyed stand of spruce due to very intensive forest management, in which the removal of dead wood was recorded, was excluded from the study. Within the forest areas, only logs longer than 0.5 m and with a lower diameter of more than 10 cm were included in the research. A single log was treated as an independent sample in the statistical analyses. The numbers of investigated logs in particular forest types and categories are given in Table 2. The following parameters of the dead logs were recorded (see Table 3 for a more detailed description): altitude a.s.l., light availability (shading) based on visual estimation of canopy of tree cover above log, the moisture of the log, the area of the log surface (m2), length of a log and the upper and lower diameter of lying log. Decomposition was expressed according to the eight decomposition classes that were originally used by McCullough (1948) with the modifications of Dynesius and Jonsson (1991) and Holeksa (2001), and which were further used by Zielonka and Piątek (2004). When logs were colonized, the percentage cover of particular vascular plant species was noted according to Londo’s decimal scale (1976). The mean percentage values of the respective cover classes were adopted for statistical analyses, i.e., the following values were taken into account: 1, 2, 4, 8, 10 and 10 percent intervals (10, 20 … 90 and 97.5) when cover >10. The frequency of a species (number of occurrences) was also computed. The appearance of a species on a log was treated as a single occurrence. The number of logs on which a species was present was regarded as species frequency. Species that were encountered on more than ten logs were regarded as common. The total cover of vascular plants also was recorded. Moreover, the cover of bryophytes (mosses and liverworts) and the cover of lichens were recorded. For each log, based on the cover of vascular plants, the following diversity indices were computed: S—number of species, H—Shannon–Wiener index and E—Pielou’s Evenness.
Data analysis
The differences in the frequency of colonization, i.e., the number of colonized versus the number of non-colonized of logs among particular forest communities, were compared using the Chi-square test. The selected morphometric features of the logs and conditions such as decomposition stage, shade and humidity were compared among the studied forest communities using the Kruskal–Wallis test followed by the Conover test for multiple comparisons. The same procedure was applied in the case of the total cover of vascular plants, bryophytes, lichens and diversity indices. Detrended correspondence analysis (DCA) was used to examine the direction of species diversity and to assess the ecological distance among the colonized dead logs. Rare species were not downweighted, but outliers were removed based on the initial results of this analysis (Hill and Gauch 1980), and a second DCA was carried out. As far as the species diversity is concerned, DCA enabled us to relate the plots to the distinguished plant communities in which logs were present using the vector fitting onto DCA procedure. The differences in centroids were tested using the Monte Carlo test with 999 permutations. In order to examine whether there were differences in the beta diversity of the plots among plant communities, the nonparametric MANOVA, i.e., multivariate ANOVA based on dissimilarities, was also carried out. The significance of the partitions was tested using the permutational ANOVA (Oksanen et al. 2013). These two analyses did not indicate to what extent the communities overlapped in terms of species composition, which are significantly different. Therefore, the gradients (axes length), which are expressed in the standard deviation values of species turnover and range of site scores along the first two axes, which are believed to be a measure of beta diversity, were computed (Eilertsen et al. 1990). Next, the Kruskal–Wallis test with the Conover test as post hoc was applied for pairwise comparisons. The constrained correspondence analysis (CCA) was adopted in order to examine the responses to environmental factors of particular species. The length of the gradient in the DCA analysis was higher than 4.0 SD, and therefore, CCA was selected instead of redundancy analysis (RDA) following the suggestions of Ter Braak and Šmilauer (2002). Nine explanatory variables were subjected to this analysis. The variables that exhibited a high variance inflation factor, which indicated a correlation with other variables, were excluded from the analysis. Two models were applied—conditional effect and marginal effect with 999 permutations of the Monte Carlo test. In order to show how species respond to decomposition across forest communities, the most frequent species were analyzed in terms of their frequency and cover in the different decomposition classes. To study how a forest community influences the colonization of dead logs by plant species that are derived from the forest floor, indicator species for a forest community were distinguished. The classification of indicator species into the four selected forest communities, and in a further step, the combination of two and three groups of vegetation were performed using the indicator value, i.e., the IndVal method (Dufręne and Legendre 1997) as modified by Cáceres et al. (2010). The statistical significance of this relationship was tested using a permutation test. Only those indicator species for groups of sites that had a statistically significant IndVal are presented.
Results
Differences in the characteristics of the logs and colonization frequency
A total of 454 dead logs of P. abies were analyzed. Among them, 292 were colonized by vascular plants. There was a significant difference in the frequency of colonized logs vs. non-colonized logs among the forest communities (Chi-squared = 48.17, p < 0.001). The highest percentage (75 %) of colonized logs was found in Calamagrostio villosae-Piceetum followed by a P. abies-planted forest (72 %), Abieti-Piceetum (48 %), and the lowest (31 %) was recorded in the beech forest Fagetalia (Fig. 2).
There were no differences in the decomposition stage of dead logs among the forest communities, and the light conditions were similar with the exception of the P. abies-planted forest in which there was a higher light availability. The cover of bryophytes was the highest in the Calamagrostio villosae-Piceetum and in P. abies-planted forests. No differences in the diversity indices including species richness, Shannon–Wiener and Evenness were found among the communities studied. Only the total cover of vascular plants was significantly higher in Calamagrostio villosae-Piceetum (Table 4).
Species composition of colonized logs
An indirect gradient analysis using DCA (Fig. 3) revealed that the dead logs that occur in the four forest communities differed in species composition but that both axes explained only 22.3 %. The main direction of species turnover among the forest communities correlated with the second vertical axis (Fig. 3). Both the nonparametric ANOVA (F = 0.07, p = 0.0049) and the goodness of fit of community type as a factor in the DCA (p < 0.001) revealed that the dead logs differed in species composition in terms of beta diversity expressed as the dissimilarity and centroids of communities along particular axes of the DCA, respectively (Table 4). The Kruskal–Wallis and Conover tests demonstrated that the species composition of vascular plants on logs from Calamagrostio villosae-Piceetum differed significantly from the logs lying in different forest communities. The logs of Fagetalia were characterized by the highest range of the DCA followed by Calamagrostio villosae-Piceetum, the P. abies-planted forest and Abieti-Piceetum (Table 4).
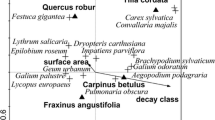
The Monte Carlo test, which was based on the results of the CCA, showed that three axes of this ordination explained the relationships between the species and the environmental variables (p = 0.005). In the conditional effect four variables, i.e., DECOMPOSITION, SHADE, ALTITUDE and BRYOPHYTES significantly explained species diversity (Table 5). Some factors such as MOISTURE, SHADE, PROTECTION, ALTITUDE and BRYOPHYTES had an impact across the forest communities to a marginal degree. The goodness of fit of the environmental variables onto the DCA partially confirmed the significance of some parameters such as DECOMPOSITION, ALTITUDE and BRYOPHYTES. Some species were associated with higher values of these environmental factors. For instance, some plants such as Homogyne alpina, Luzula sylvatica, Deschampsia flexuosa and Galium hercynicum were confined to areas in which there was a higher degree of decomposition. Digitalis purpurea and Deschampsia caespitosa were associated with a cover of bryophytes (Fig. 4).
Biplot of the species on the first two CCA axes (λ 1 = 0.43, λ 2 = 0.19) for the decaying logs of Picea abies. Explanations—frequent species are bolded >10 logs present, species that are significant for forest communities are underlined. Ath.dis—Athyrium distentifolium, Bet.pen—Betula pendula, Cal.aru—Calamagrostis arundinacea, Cal.vil—Calamagrostis villosa, Cal.vul—Calluna vulgaris, Car.sp.—Carex species, Des.cae—Deschampsia caespitosa, Des.fle—Deschampsia flexuosa, Dig.pur—Digitalis purpurea, Dry.car—Dryopteris carthusiana, Dry.dil—Dryopteris dilatata, Dry.exp—Dryopteris expensa, Dry.fil,—Dryopteris filix-mas, Dry.sp.—Dryopteris sp., Fag.syl,—Fagus sylvatica, Gal.her,—Galium hercynicum, Gym.dry—Gymnocarpium dryopteris, Hom.alp—Homogyne alpina, Imp.par—Impatiens parviflora, Lam.gal—Lamium galeobdolon, Lar.dec—Larix decidua (seedlings), Luz.luz—Luzula luzuloides, Luz.syl—Luzula sylvatica, Lyc.ann—Lycopodium annotinum, Mai.bif—Maianthemum bifolium, Oxa.ace—Oxalis acetosella, Phe.con—Phegopteris connectilis, Pic.abi—Picea abies (seedlings), Rub.ide—Rubus idaeus, Rub.sp—Rubus species, Sor.auc—Sorbus aucuparia, Tri.eur—Trientalis europaea, Vac.myr—Vaccinium myrtillus, Vac.vit—Vaccinium vitis-idaea
The results of the IndVal method indicated that of the 34 species that were present, only ten could be treated as indicator species for the forest communities that were analyzed (Table 6). These were herbaceous species except for a seedling of Larix decidua that was found in the P. abies-planted forest. Some species were characteristic for the forest floor of a particular community, i.e., Calamagrostis villosa, Deschampsia caespitosa and Trientalis europaea in the case of Calamagrostio villosae-Piceetum. Three plant species, i.e., Calamagrostis arundinacea, Oxalis acetosella and Dryopteris carthusiana, which are more common, were significant indicators for more than one community (Table 6). For the Fagetalia forests, three species were found only in this community and were weak indicators, e.g., Phegopteris connectilis. Five species were frequent or relatively frequent but without fidelity to any of communities and 20 were only encountered sporadically.
Discussion
Differences in colonization frequency among the forest communities
The first hypothesis that colonization frequency does not depend on the type of forest community was rejected. The decomposition stage of logs was similar in all of the communities, which indicates that colonization does not depend on the conditions that occur in a particular type of forest. The amount of dead wood in a particular forest community also cannot be a constraining factor; however, in this study we did not analyze the volume of dead wood or the wood of living tress as was done in the works of other authors. It partially confirms the findings of Zielonka and Piątek (2004), who stated that the distribution of decaying logs is relatively equal in a small spatial scale. They suggested that this is a consequence of constancy in the delivery of dead wood to the forest floor over the years. This process can occur independently of the forest community. Both the natural coniferous community—reedgrass spruce forest—Calamagrostio villosae-Piceetum and the P. abies-planted forest were colonized most frequently. In natural types of forest, the thinning of the tree stand and the fall of standing dead trees are more a common and stable phenomenon than they are in managed, exploited forests. In Central Europe, managed spruce forests differ from old-growth forests by the near absence of dead wood (Dittrich et al. 2013). This study also showed that the total number of fallen trees per hectare was lower in managed forest than in protected forest (Table 1). In the case of the second forest, i.e., the P. abies-planted forest, which is a previously managed forest, spruce was cultivated in the past in habitats of Abieti-Piceetum and beech phytocoenoses. However, the common feature with Calamagrostio villosae-Piceetum is the dominance of spruce in the tree stand. Due to its long persistence, the phytocoenoses of the P. abies-planted forest resembled the forest in the upper montane zone. As was already mentioned, colonization by vascular plants can be enhanced by bryophytes and lichens. As far as the former is concerned, a previous work demonstrated a positive correlation between the cover of bryophytes and vascular plants on the logs of F. sylvatica (Staniaszek-Kik et al. 2014). It is worth stressing that in this study the mean cover of colonizing bryophytes changed depending on the type of forest community. It was the lowest in the beech forests and in the Abieti-Piceetum. The phenomenon of a higher cover of bryophytes in the reedgrass spruce forest and in the managed spruce forest is interesting because in another study (Staniaszek-Kik et al. 2014), it was revealed that moss cover decreased with increasing altitude. However, in this study both the cover of bryophytes and vascular plants increased, weakly but significantly, with the altitude a.s.l. (rs = 0.13, p < 0.001, rs = 0.26, p < 0.001, respectively). This is obviously connected with the vertical distribution of spruce trees, which are more abundant in the upper parts. The previous work focused on the dead logs of F. sylvatica whose zonation in the mountains of Central Europe is well known. This is an important finding because it shows that an important factor for colonization by bryophytes and vascular plants is not only extrinsic environmental factors but also the species identity of the log. As far as the extrinsic environmental factors are concerned, light availability is higher in coniferous forests than in beechwood forests where the CWD of spruce was also present in this study. Both vascular plants and bryophytes respond positively to the amount of light on the forest floor (Proctor 2000; Glime 2007). The numbers of species, according to the values of the Shannon–Wiener index, Evenness, did not depend on the type of surrounding community, which indicates that the type of decomposition that is usual among the forest types has a major impact on the biodiversity indices.
Species composition of the flora colonizing dead logs
As we expected, the species composition varied significantly among the forest communities. The species that are present on dead logs in particular forest communities, especially those that are indicating for them (Table 4), are also typical for the ground layer of these types of forests (Matuszkiewicz 2008a). It confirms niche theory that better explain shift in species’ distributions among forest communities located along environmental gradient. This pattern is particularly visible for the beech forest in which there were three indicating species, and especially for the reedgrass spruce forest Calamagrostio villosae-Piceetum, which also had three species. Dittrich et al. (2014) showed that species, which occasionally occurred on logs, i.e., Calamagrostis villosa, Deschampsia flexuosa, Trientallis europaea, were indicators for the forest floor in the same forest community—Calamagrostio villosae-Piceetum, which was also found in this study. These species were found to be indicators for this community in our research. Other plants mentioned as indicator species by Dittrich et al. (2014) such as Vaccinium myrtillus, Oxalis acetosella, Luzula sylvatica were reported in this study, but they were not only indicating for Calamagrostio villosae-Piceetum but were generally frequent or relatively frequent. Kushnevskaya et al. (2007), who examined a Taiga forest, indicated 22 species on spruce logs of which 14 were in common with our study. Similarly, Vaccinium myrtillus, Oxalis acetosella, Vaccinium vitis-idea had the highest frequency. Taking into account climatic and latitudinal differences, the patterns in species composition were congruent. In turn, in a study by Zielonka and Piątek (2004) of the 20 species that were present on spruce logs in Plagiothecio-Piceetum in the Tatra Mts, 14 were also encountered in our study. For the spruce logs located in Fagetalia forests, Impatiens parviflora along with F. sylvatica as seedlings and Phegopteris connectilis was the most frequent inhabiting species. This plant was not present in other forest communities. It can be inferred that the colonization ability of the species is limited by its short dispersal, which results in its frequent occurrence in one community.
Role of log decay and other environmental factors in colonization by vascular plants
Some vascular species, which are shown in Fig. 5, are quite frequent and do not show a preference for any community or groups of forest communities, e.g., Oxalis acetosella. According to Zielonka and Piątek (2004), this species is mainly confined to decomposition stages 5 and 6. A similar pattern was found in this study despite the fact that a wider ecological amplitude was encompassed because four types of forests were examined rather than only one community of subalpine spruce forest as was the case in the Zielonka and Piątek (2004) study. Dryopteris carthusiana also exhibited a distribution similar to O. acetosella with stage 6 being optimum for it. However, other species, e.g., P. abies (seedlings), Vaccinium myrtillus and Deschampsia flexuosa, were most frequent in stages 4 and 6. Stage 4 seems to be the most suitable in terms of conditions for plant development for all of the species. This is contrast to the Zielonka and Piątek (2004) results, which indicated that stage 6 of log decomposition was the best.
Another important feature is the mean cover of particular plants on logs. It can be seen that there are no rapid differences in the cover of the species among the decomposition stages, although there are exceptions. Two grass plants, i.e., Calamagrostis arundinacea and Deschampsia flexuosa, exhibited a high degree of variation in both their frequency and mean cover among the decomposition stages. The latter was the most abundant in the last stage of decay when the shape of a log is flattened without any remains of the bark. Moreover, the architecture of the plant facilitates its coexistence with the higher cover of bryophytes in the final stages of decomposition. In the case of the other grass, C. villosa, a great deal of variation was observed only in its frequency but not in its mean cover, which was rather low. Like D. flexuosa, this species is indicating for both the ground layer of Calamagrostio villosae-Piceetum (Schlüter 1969; Matuszkiewicz 2008a) and dead logs (Table 6). Apart from forbs, tree seedlings were found on dead logs. These were P. abies, Larix decidua and F. sylvatica. It has been reported by many authors that the logs of dead mature trees have favorable conditions for the germination of tree seeds, and in the late decomposition stage, seedlings are even able to survive and grow to a mature developmental stage, e.g., P. abies (Holeksa 2001; Zielonka 2006). Others such as F. sylvatica were encountered because of the logs in the beech forest where F. sylvatica is the dominant species in the tree stand. Herbaceous species are thought to germinate in stage 3 in which the logs are of a round shape and have crevices that are several millimeters deep. These are the minimal conditions for germination and growth. However, sometimes the presence of a thin layer of soil or decomposed leaves is enough for the appearance of vascular plants, which can be manifested by a single record of a plant in stage 2 of decomposition (Fig. 5) since a log is round, smooth and has a bark that is partially intact at this stage (Zielonka and Piątek 2004). Semi-decomposed logs, i.e., around stage 4, are characterized by increased moisture, which independent of the species of the dead logs is one of the most crucial variables that influence the cover of bryophytes and vascular plants. In a previous study on F. sylvatica (Staniaszek-Kik et al. 2014), logs that had a positive correlation between the cover of plants and the moisture of dead wood were found. Sollins et al. (1987) and recent papers (Bütler et al. 2007) have stressed the important role of moisture. In our study, moisture was only marginally significant (Table 5). In addition to the cover of bryophytes and altitude, whose roles were already mentioned, another significant factor that can be regarded as being responsible for species distribution and cover is shade (Table 3). In this study, shade was a rather limiting factor (Fig. 4). Logs are usually situated under the canopy gaps created by fallen trees; thus, light conditions are better than in forest interiors. Species that turned out to be shade tolerant plants were Gymnocarpium dryopteris, Maianthemum bifolium and Dryopteris filix-mas. Surprisingly, moisture and shade were in almost opposite directions on the CCA diagram (Fig. 4), which seems illogical at first sight. However, larger logs retain moisture longer than smaller ones (Harmon et al. 1986), which is reflected by the close positions of moisture and area (Fig. 4). Moreover, moisture was a marginally significant variable and shade was only expressed on a three-degree scale.
Role of forest management
The presence/absence of protection or management was also important for creating the species composition of herbaceous species on dead logs (Table 3). Vaccinium myrtillus, Calamagrostis villosa and Trientallis europaea were associated with protected forests, whereas Dryopteris expansa and D. dilatata were more closely linked with managed forests (Fig. 4). The work did not confirm that type of community had an impact on the differences in the species composition among the status of the forests. All of the analyzed forest communities were found in both groups (Table 1). There is a lack of information in the literature about how forest management practices, e.g., cutting trees and the abandonment of logs, affect the colonization processes when compared to natural broken or uprooted wind-fallen trees. While the causes of the colonization process itself must be the same, the age, size (diameter of trunk) and the area of tree cutting can have an impact on the presence and abundance of epiphytes and in the next stage—after death—on epixylic flora. A previous study (Staniaszek-Kik et al. 2014) showed that there was a higher cover of vascular plants on the logs of F. sylvatica in managed forests. This special situation was connected with the ongoing forest reconstruction in the region. The reconstruction of tree stands in the Sudety Mts, aiming at changes in tree species composition, and in other parts of southern Poland is the consequence of the damage to Norway spruce stands from air pollution and a massive attack of pests that was highest in the 1980s (Stachurski et al. 1994). Besides spruce trees, foresters also removed many other species. It can be inferred that an increase in the colonization of logs of dead trees is a side effect of forest management. Nevertheless, as far as vascular plants are concerned, common forest herbs or common tree species usually benefit most in contrast to cryptogamous plants.
Conclusions
To summarize, we did not confirm significant role of CWD, irrespective of the type of forest, the altitude at which it lies or management status, as a secondary habitat for rare and protected vascular plants. None of the species found on logs in our study is protected or considered to be rare according to the national or regional red lists. Species that are frequent colonizers are usually common forest floor plants, which have been shown in other studies. There are no obligatory epixylic and epiphytic vascular plants. However, their role in the community assembly of epixylic vegetation cannot be neglected. They co-occur at the later decomposition stages of logs with lichens, liverworts and mosses, which owing to the presence of dead wood, can persist in forest ecosystems. Further studies are needed in order to recognize the relationships between vascular plants and cryptogamous species in detail. The nature of these interactions, whether positive or negative, can be important for biodiversity.
References
Andersson LI, Hytteborn H (1991) Bryophytes and decaying wood—a comparison between managed and natural forest. Ecography 14:121–130. doi:10.1111/j.1600-0587.1991.tb00642.x
Bütler R, Patty L, Le Bayon RC, Guenat C, Schlaepfer R (2007) Log decay of Picea abies in the Swiss Jura Mountains of central Europe. For Ecol Manag 242:791–799. doi:10.1016/j.foreco.2007.02.017
Cáceres MD, Legendre P, Moretti M (2010) Improving indicator species analysis by combining groups of sites. Oikos 119(10):1674–1684. doi:10.1111/j.1600-0706.2010.18334.x
Danielewicz W, Raj A, Zientarski J (2013) Ekosystemy leśne [Forests]. In: Knapik R, Raj A (eds) Przyroda Karkonoskiego Parku Narodowego [The Nature of the Karkonosze National Park]. Karkonoski Park Narodowy, Jelenia Góra, pp 279–302 (in Polish)
Dittrich S, Hauck M, Jacob M, Rommerskirchen A, Leuschner C (2013) Response of ground vegetation and epiphyte diversity to natural age dynamics in a Central European mountain spruce forest. J Veg Sci 24:675–687. doi:10.1111/j.1654-1103.2012.01490.x
Dittrich S, Jacob M, Bade C, Leuschner C, Hauck M (2014) The significance of deadwood for total bryophyte, lichen, and vascular plant diversity in an old-growth spruce forest. Plant Ecol 215(10):1123–1137. doi:10.1007/s11258-014-0371-6
Dufręne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366. doi:10.1890/0012-9615(1997)067%5B0345:SAAIST%5D2.0.CO;2
Dynesius M, Jonsson BG (1991) Dating uprooted trees: comparison and application of eight methods in a boreal forest. Can J For Res 21:655–665. doi:10.1139/x91-089
Eilertsen O, Okland RH, Okland T, Pedersen O (1990) Data manipulation and gradient length estimation in DCA ordination. J Veg Sci 1(2):261–270. doi:10.2307/3235663
Fabiszewski J, Wojtuń B (1994) Zjawiska ekologiczne towarzyszące wymieraniu lasów w Sudetach (Die begleitenden ökologischen Erscheinungen des Waldsterbenprozesses im Sudetengebrige). Prace Instytutu Badawczego Leśnictwa B 21(2):195–210 (in Polish with German summary)
Glime JM (2007) Bryophyte ecology. Ebook sponsored by Michigan Technological University and the International Association of Bryologists. http://www.bryoecol.mtu.edu. Accessed April 5 2016
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Linkaemper GW, Cromack K, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302. doi:10.1016/S0065-2504(03)34002-4
Hill MO, Gauch HG Jr (1980) Detrended correspondence analysis: an improved ordination technique. Vegetatio 42(1–3):47–58. doi:10.1007/BF00048870
Hofgaard A (1993) Structure and regeneration patterns in a virgin Picea abies forest in northern Sweden. J Veg Sci 4:601–608. doi:10.2307/3236125
Holeksa J (2001) Coarse woody debris in a Carpathian subalpine spruce forest. Forstwes Cent 120:256–270. doi:10.1007/BF02796097
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54(2):187–211. doi:10.2307/1942661
Jadczyk P (2009) Natural effects of large-area forest decline in the Western Sudeten. Environ Prot Eng 35(1):49–56
Jonsson BG, Kruys N, Ranius T (2005) Ecology of species living on dead wood—lessons for dead wood management. Silva Fenn 39(2):289–309. doi:10.14214/sf.390
Korpel S (1989) Pralesy Slovenska. Veda, Bratislava
Korzybski D, Mionskowski M, Dmyterko E, Bruchwald A (2013) Stopień uszkodzenia świerka jodły i modrzewia w Sudetach Zachodnich (Degree of damage to spruce, fir and larch stands in the Western Sudetes). Sylwan 157(2):104–112 (in Polish with English abstract and summary)
Kosiński P, Bednorz L (2003) Trees and shrubs of the Polish part of the Eastern Sudety Mts. Dendrobiology 49:31–42
Kruys N, Jonsson BG (1999) Fine woody debris is important for species richness on logs in managed boreal spruce forests of northern Sweden. Can J For Res 29(8):1295–1299. doi:10.1139/x99-106
Kushnevskaya H, Mirin D, Shorohova E (2007) Patterns of epixylic vegetation on spruce logs in late-successional boreal forests. For Ecol Manag 250(1):25–33. doi:10.1016/j.foreco.2007.03.006
Kuuluvainen T (1994) Gap disturbance, ground microtopography and the regeneration dynamics of boreal coniferous forests in Finland: a review. Ann Zool Fenn 31(1):35–51. doi:10.5735/085.050.0506
Laaka-Lindberg S, Pohjamo M, Korpelainen H (2005) Niche breadth and niche overlap in three epixylic hepatics in a boreal old-growth forest, southern Finland. J Bryol 27:119–127. doi:10.1179/037366805X53031
Lee P, Sturgess K (2001) The effects of logs, stumps, and root throws on understory communities within 28-year-old aspen-dominated boreal forests. Can J Bot 79(8):905–916. doi:10.1139/b01-072
Londo G (1976) The decimal scale for relevés of permanent quadrats. Vegetatio 33:61–64. doi:10.1007/BF00055300
Matuszkiewicz W (2008a) Przewodnik do oznaczania zbiorowisk roślinnych Polski [A key for determination of plant communities in Poland]. Wydawnictwo Naukowe PWN, Warszawa (in Polish)
Matuszkiewicz JM (2008b) Potential natural vegetation of Poland. Institute of Geography and Spatial Organization Polish Academy of Sciences, Warszawa
Mayer H, Schenker S, Zukrigl K (1972) Der Urwaldrest Neuwald bei Lahnsattel. Centerblat fur das Gesamte Forstwesen 89:147–190
McCullough HA (1948) Plant succession on fallen logs in a virgin spruce-fir forest. Ecology 29(4):508–513. doi:10.2307/1932645
Modrzyński J (2003) Defoliation of older Norway spruce (Picea abies/L./Karst.) stands in the Polish Sudety and Carpathian mountains. For Ecol Manag 181(3):289–299. doi:10.1016/S0378-1127(02)00657-6
Nakashizuka T (2001) Species coexistence in temperate, mixed deciduous forests. Trends Ecol Evol 16(4):205–210. doi:10.1016/S0169-5347(01)02117-6
Oksanen J, Guillaume Blanchet FG, Kindt K, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens HMH, Wagner H (2013) vegan: Community Ecology. Package. R package version 2.0-8. http://CRAN.R-project.org/package=vegan
Proctor MC (2000) The bryophyte paradox: tolerance of desiccation, evasion of drought. Plant Ecol 151(1):41–49. doi:10.1023/A:1026517920852
Riffell S, Verschuyl J, Miller D, Wigley TB (2011) Biofuel harvests, coarse woody debris, and biodiversity—a meta-analysis. For Ecol Manag 261(4):878–887. doi:10.1016/j.foreco.2010.12.021
Schlüter H (1969) Das Calamagrostio villosae-Piceetum des Thüringer Waldes im Vergleich zu anderen Mittelgebirgen. Vegetatio 17(1):157–164. doi:10.1007/BF01965906
Söderström L (1988) Sequences of bryophytes and lichens in relation to substrate variables of decaying coniferous wood in Northern Sweden. Nordic J Bot 8:89–97. doi:10.1111/j.1756-1051.1988.tb01709.x
Sollins P, Cline SP, Verhoeven T, Sachs D, Spycher G (1987) Patterns of log decay in old-growth Douglas-fir forests. Can J For Res 17:1585–1595. doi:10.1139/x87-243
Stachurski A, Zimka JR, Kwiecien M (1994) Forest decline in Karkonosze Poland. I. Chlorophyll, phenols, defoliation index and nutrient status of the Norway spruce (Picea abies L.). Ekol Pol 42(3):286–316
Staniaszek-Kik M, Żarnowiec J, Chmura D (2014) Colonization patterns of vascular plant species on decaying logs of Fagus sylvatica L. in a lower mountain forest belt: a case study of the Sudeten Mountains, (Southern Poland). Appl Ecol Environ Res 12(3):601–613. doi:10.15666/aeer/1203_601613
Stokland JH, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Ter Braak CJF, Šmilauer P (2002) Canoco reference and CanoDraw for Windows User’s guide: Software for Canonical Community Ordination (version 4.5). Microcomputer Power (Ithaca, NY, USA)
Żarnowiec J, Staniaszek-Kik M (2009) Lichens and plants inhabiting snags in mountains forests of the Karkonosze National Park (Sudetes Mts, SW Poland). Nowellia Bryologica, revue specialisee de briologie—numero special “Troisièmes Rencontres Bryologiques internationales de juin 2009”. pp 78–90
Zielonka T (2006) When does dead wood turn into a substrate for spruce replacement? J Veg Sci 17(6):739–746. doi:10.1658/1100-9233(2006)17%5B739:WDDWTI%5D2.0.CO;2
Zielonka T, Piątek G (2004) The herb layer and dwarf shrubs colonization of decaying logs in subalpine forest in the Polish Tatra Mountains. Plant Ecol 172:63–72. doi:10.1023/B:VEGE.0000026037.03716.fc
Acknowledgements
Authors thank two anonymous reviewers for many valuable remarks on the previous version of manuscript. Michele Simmons and Anna Beckmann helped to edit it.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Gediminas Brazaitis.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Staniaszek-Kik, M., Żarnowiec, J. & Chmura, D. The vascular plant colonization on decaying Picea abies logs in Karkonosze mountain forest belts: the effects of forest community type, cryptogam cover, log decomposition and forest management. Eur J Forest Res 135, 1145–1157 (2016). https://doi.org/10.1007/s10342-016-1001-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-016-1001-8