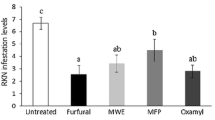
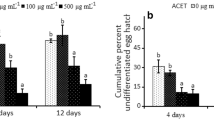
Abstract
Seeds of papaya fruit (Carica papaya) are usually unused for the industry, but they have been previously reported to be anthelmintic. However, the effects and mechanisms involved in papaya seeds against root-knot nematodes (RKN) are poorly studied in soil incorporation. Here, we investigated soil biofumigation with papaya seeds against the RKN Meloidogyne incognita and the effect of their volatile organic compounds (VOCs) separately from non-VOCs. Soil biofumigation reduced the number of galls and eggs of RKN about 100% in tomato roots, by using seeds macerates at 4.0 g 100 mL−1, which is a feasible amount for field applications. The VOCs emitted by the seeds killed 80% of M. incognita second-stage juveniles after a few hours of exposure. Furthermore, the whole volatilome of the papaya seed was identified by gas chromatography. Some of the compounds have already been studied against RKN. Therefore, we examined the efficacy of all nonstudied compounds against M. incognita. The most promising compounds were vinyl acetate, phenylacetaldehyde and benzylacetonitrile with lethal concentrations (LC50) of 61.5, 101.3 and 14.4 μg mL−1, respectively. When vinyl acetate and phenylacetaldehyde were applied to the soil at the recommended commercial nematicide dose (396 μg mL−1), the effect on M. incognita was better than the commercial nematicide (Carbofuran), reducing 70% of the number of galls and eggs. However, benzylacetonitrile reduced galls and eggs only by 25%. These results provide evidence for biofumigation with papaya seeds as a sustainable approach to control RKN and the potential use of their VOCs as nematicidal compounds.

Similar content being viewed by others
References
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry. Allured Pub Corp, Carol Stream
Aissani N, Urgeghe PP, Oplos C, Saba M, Tocco G, Petretto GL, Eloh K, Menkissoglu-Spiroudi U, Ntalli N, Caboni P (2015) Nematicidal activity of the volatilome of Eruca sativa on Meloidogyne incognita. J Agric Food Chem 63:6120–6125. https://doi.org/10.1021/acs.jafc.5b02425
Barros AF, Campos VP, da Silva JCP, Pedroso MP, Medeiros FHV, Pozza EA, Reale AL (2014) Nematicidal activity of volatile organic compounds emitted by Brassica juncea, Azadirachta indica, Canavalia ensiformis, Mucuna pruriens and Cajanus cajan against Meloidogyne incognita. Appl Soil Ecol 80:34–43. https://doi.org/10.1016/j.apsoil.2014.02.011
Boneti JIS, Ferraz S (1981) Modificação do método de Hussey & Barker para extração de ovos de Meloidogyne exigua de raízes de cafeeiro. Fitopatol Bras 6:553
Caboni P, Saba M, Oplos C, Aissani N, Maxia A, Menkissoglu-Spiroudi U, Casu L, Ntalli N (2015) Nematicidal activity of furanocoumarins from parsley against Meloidogyne spp. Pest Manag Sci 71:1099–1105. https://doi.org/10.1002/ps.3890
Cadena MB, Preston GM, Van der Hoorn RA, Townley HE, Thompson IP (2018) Species-specific antimicrobial activity of essential oils and enhancement by encapsulation in mesoporous silica nanoparticles. Ind Crops Prod 122:582–590. https://doi.org/10.1016/j.indcrop.2018.05.081
Coutinho MM, Freitas LG, Dallemole-Giaretta R, Neves WS, Lopes EA, Ferraz S (2009) Controle de Meloidogyne javanica com Pochonia chlamydosporia e farinha de sementes de mamão. Nematol Bras 33:169–175
Dicke M (2016) Plant phenotypic plasticity in the phytobiome: a volatile issue. Curr Opin Plant Biol 32:17–23
Estupiñan-López L, Campos VP, Silva AP, Barros AF, Pedroso MP, Silva JCP, Terra WC (2017) Volatile organic compounds from cottonseed meal are toxic to Meloidogyne incognita. Trop Plant Pathol 42:443–450. https://doi.org/10.1007/s40858-017-0154-4
Fagundes GR, Yamanishi OK (2002) Estudo da comercialização do mamão em Brasília-DF. Rev Bras Frutic 24:091–095. https://doi.org/10.1590/S0100-29452002000100020
Gaugler R, Bilgrami A (2004) Nematode behaviour. CABI, New York
Girardi NS, García D, Robledo SN, Passone MA, Nesci A, Etcheverry M (2016) Microencapsulation of Peumus boldus oil by complex coacervation to provide peanut seeds protection against fungal pathogens. Ind Crops Prod 15:93–101. https://doi.org/10.1016/j.indcrop.2016.07.045
Jardim IN, Oliveira DF, Silva GH, Campos VP, de Souza PE (2017) (E)-cinnamaldehyde from the essential oil of Cinnamomum cassia controls Meloidogyne incognita in soybean plants. J Pest Sci 91:479–487. https://doi.org/10.1007/s10340-017-0850-3
Jones JT, Haegeman A, Danchin EG, Gaur HS, Helder J, Jones MG, Kikuchi T, Manzanilla-López R, Palomares-Rius JE, Wesemael WM, Perry RN (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 57:946–961
Kermanshai R, McCarry BE, Rosenfeld J, Summers PS, Weretilnyk EA, Sorger GJ (2001) Benzyl isothiocyanate is the chief or sole anthelmintic in papaya seed extracts. Phytochemistry 57:427–435. https://doi.org/10.1016/S0031-9422(01)00077-2
Lal J, Chandra S, Raviprakash V, Sabir M (1976) In vitro anthelmintic action of some indigenous medicinal plants on Ascaridiagalli worms. Indian J Physiol Pharmacol 20:64–68. https://doi.org/10.7324/JAPS.2018.8224
Lima T, Rinald M, Ascheri D (2006) Característica físico-químicas, composição centesimal de cascas e semente de mamão. https://ainfo.cnptia.embrapa.br/digital/bitstream/item/75815/1/bolpd-263.pdf. Accessed 16 Apr 2019
Neves WS, Freitas LG, Coutinho MM, Dallemole-Giaretta R, Ferraz S (2008) Uso de Sementes de Mamão e Solarização do Solo no Controle de Meloidogyne javanica e Meloidogyne incognita. Nematol Bras 32:253–259
Neves WS, Freitas LG, Dallemole-Giaretta R, Coutinho MM, Ferraz S, Parreira DF (2011) Incorporação de Farinha de Semente de Mamão ao Solo para o Controle de Meloidogyne javanica. Nematol Bras 36:25–31
Nicol JM, Turner SJ, Coyne DL, Nijs L, Hockland S, Maafi ZT (2011) Current nematode threats to world agriculture. In: Jones J, Gheysen G, Fenoll C (eds) Genomics and molecular genetics of plant-nematode interactions. Springer, Dordrecht, pp 21–43. https://doi.org/10.1007/978-94-007-0434-3_2
Nist (2019) Chemistry Webook–National Institute of Standards and Technology. http://webbook.nist.gov/chemistry/. Accessed 5 Apr 2019
Ntalli N, Caboni P (2017) A review of isothiocyanates biofumigation activity on plant parasitic nematodes. Phytochem Rev 16:827–834. https://doi.org/10.1007/s11101-017-9491-7
Ntalli NG, Ferrari F, Giannakou I, Menkissoglu-Spiroudi U (2011) Synergistic and antagonistic interactions of terpenes against Meloidogyne incognita and the nematicidal activity of essential oils from seven plants indigenous to Greece. Pest Manag Sci 67:341–351. https://doi.org/10.1002/ps.2070
Ntalli N, Monokrousos N, Rumbos C, Kontea D, Zioga D, Argyropoulou MD, Menkissoglu-Spiroudi U, Tsiropoulos NG (2018) Greenhouse biofumigation with Melia azedarach controls Meloidogyne spp. and enhances soil biological activity. J Pest Sci 91:29–40. https://doi.org/10.1007/s10340-017-0909-1
Nunes AS, Campos VP, Mascarello A, Stumpf TR, Chiaradia-Delatorre LD, Machado ART, Oliveira DF (2013) Activity of chalcones derived from 2, 4, 5-trimethoxybenzaldehyde against Meloidogyne exigua and in silico interaction of one chalcone with a putative caffeic acid 3-O-methyltransferase from Meloidogyne incognita. Exp Parasitol 135:661–668. https://doi.org/10.1016/j.exppara.2013.10.003
Oka Y (2001) Nematicidal activity of essential oil components against the root-knot nematode Meloidogyne javanica. Nematology 3:159–164. https://doi.org/10.1163/156854101750236286
Pedroso LA, Campos VP, Pedroso MP, Barros AF, Freire ES, Resende FM (2019) Volatile organic compounds produced by castor bean cake incorporated into the soil exhibit toxic activity against Meloidogyne incognita. Pest Manag Sci 75:476–483. https://doi.org/10.1002/ps.5142
Ploeg A, Stapleton J (2001) Glasshouse studies on the effects of time, temperature and amendment of soil with broccoli plant residues on the infestation of melon plants by Meloidogyne incognita and M. javanica. Nematology 3:855–861. https://doi.org/10.1163/156854101753625353
Romero JJ, Zarate MA, Ogunade IM, Arriola KG, Adesogan AT (2018) Tropical plant supplementation effects on the performance and parasite burden of goats. Asian Aust J Anim Sci 31:208–217. https://doi.org/10.5713/ajas.17.0321
Silva JCP, Campos VP, Barros AF, Pedroso MP, Terra WC, Lopez LE, Souza JT (2018) Plant volatiles reduce the viability of the root-knot nematode Meloidogyne incognita either directly or when retained in water. Plant Dis 102:2170–2179. https://doi.org/10.1094/PDIS-01-18-0143-RE
Silva MF, Campos VP, Barros AF, Terra WC, Pedroso MP, Gomes VA, Ribeiro CR, Silva FF (2019) Volatile emissions of watercress (Nasturtium officinale) leaves and passion fruit (Passiflora edulis) seeds against Meloidogyne incognita. Pest Manag Sci. https://doi.org/10.1002/ps.5654
Sousa RC, Halasz MRT (2006) Recuperação e reaproveitamento de rejeitos de mamão. Rev Educ e Tecnol 1:1–12
Sousa RMOF, Rosa JS, Silva CA, Almeida MTM, Novo MT, Cunha AC, Fernandes-Ferreira M (2015) Larvicidal, molluscicidal and nematicidal activities of essential oils and compounds from Foeniculum vulgare. J Pest Sci 88:413–426. https://doi.org/10.1007/s10340-014-0628-9
Srinivasan MR (2004) Probit analysis. In: Palaniswamy S, Kuttalam S, Chandrasekaran S, Kennedy JS, Srinivasan MR (eds) Electronic manual on pesticides and environment department of agricultural entomology. TNAU, Coimbatore
Terra WC, Campos VP, Martins SJ, Costa LSAS, Silva JCP, Barros AF, Lopez LE, Santos TCN, Smant G, Oliveira DF (2018) Volatile organic molecules from Fusarium oxysporum strain 21 with nematicidal activity against Meloidogyne incognita. Crop Prot 106:125–131. https://doi.org/10.1016/j.cropro.2017.12.022
Trudgill DL, Block VC (2001) Apomictic, polyphagous root-knot nematodes: exceptionally successful and damaging biotrophic root pathogens. Annu Rev Phytopathol 39:53–77. https://doi.org/10.1146/annurev.phyto.39.1.53
Ullah I, Khan AL, Ali L, Khan AR, Waqas M, Hussain J, Lee I-J, Shin J-H (2015) Benzaldehyde as an insecticidal, antimicrobial, and antioxidant compound produced by Photorhabdus temperata M1021. J Microbiol 53:127–133. https://doi.org/10.1007/s12275-015-4632-4
Walter M, Ramaley L (1973) Purification of acetonitrile. Anal Chem 45:165–166. https://doi.org/10.1021/ac60323a041
Wang D, Rosen C, Kinkel L, Cao A, Tharayil N, Gerik J (2009) Production of methyl sulfide and dimethyl disulfide from soil-incorporated plant materials and implications for controlling soilborne pathogens. Plant Soil 324:185–197. https://doi.org/10.1007/s11104-009-9943-y
Zasada IA, Ferris H (2003) Sensitivity of Meloidogyne javanica and Tylenchulus semipenetrans to isothiocyanates in laboratory assays. Phytopathology 93:747–750. https://doi.org/10.1094/PHYTO.2003.93.6.747
Zhan J, Thrall PH, Papaix J, Xie L, Burdon JJ (2015) Playing on a pathogen’s weakness: using evolution to guide sustainable plant disease control strategies. Annu Rev Phytopathol 53:19–43. https://doi.org/10.1146/annurev-phyto-080614-120040
Acknowledgements
The authors gratefully acknowledge financial support and fellowships from: Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals (vertebrates) performed by any of the authors.
Additional information
Communicated by M.B. Isman.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.

Supplemental Fig.
1. Technique setups used to evaluate papaya seed VOCs and non-VOCs. a, Biofumigation in plastic cup described by Estupiñan-López et al. (2017). Artificial substrate and papaya seeds macerate were mixed in polythene bags resulting in mixtures with different macerate concentrations. Together, eggs of M. incognita were added to each plastic bag. The bags were closed and the contents stirred. Then, substrate + eggs + seed macerate were placed into a 100 mL plastic cup. Microtubes were introduced over the mixture. Then, the cups were sealed with plastic film. After 3 days the eggs were injected inside the microtube and 3 days later the plastic film was removed, the eggs in the microtube were also removed and a tomato seedling transplanted to the cup. b, Supelco vial techniques described by Barros et al. (2014). Papaya seeds macerate was placed into the Supelco vial over steril dry sand. In all vials, a microtube was halfway inserted in the sand. Subsequently, the Supelco vials were closed and left for 3 days to perform VOCs accumulation. Next, a suspension containing J2 of M. incognita was injected into the microtube with a syringe. The J2 were exposed to the VOCs for six different periods. After, the vials were opened and part of J2 was removed to estimate mortality. The remaining suspension of each period was inoculated in a tomato seedling to later estimation of galls and eggs. (JPEG 6998 kb)

Supplemental Fig.
2. The structural formula of the vinyl acetate, phenylacetaldehyde and benzylacetonitrile molecules. (JPEG 282 kb)
Rights and permissions
About this article
Cite this article
Gomes, V.A., Campos, V.P., da Silva, J.C.P. et al. Activity of papaya seeds (Carica papaya) against Meloidogyne incognita as a soil biofumigant. J Pest Sci 93, 783–792 (2020). https://doi.org/10.1007/s10340-020-01192-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-020-01192-z