Abstract
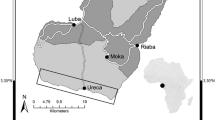
We assessed the normal values for 15 blood plasma biochemistry parameters and three indices of body condition (IBCs) in free-living Osprey (Pandion haliaetus) nestlings in Brandenburg (Germany). Values were compared with those of other raptors, and possible sexual and age-related differences were examined. In addition, we looked for possible relationships of habitat quality (measured in terms of foraging conditions and human disturbances) to nestling nutritional condition and productivity. Female nestlings showed higher mean urea levels and lower glucose values than males, which could be related to higher growth rates and nutrient demand of females at the end of the nestling period. Seven parameters also showed variation with age, probably relating to increasing body mass, metabolic rates, and physical activity during the pre-fledging stage. Conversely, the IBCs showed poor correlations with selected nutritional parameters, probably due to the homogeneously acceptable nutritional conditions of the nestlings. Finally, we found that Ospreys did not seem to adjust initial parental investment in relation to habitat quality, since productivity was not affected by habitat indicators. However, in large broods, but not in small ones, nestling nutritional condition improved as foraging conditions improved. These results suggest that females that are in better physical condition seem to produce more and better nourished nestlings in better foraging conditions.
Zusammenfassung
Einflussfaktoren auf biochemische Blutplasma-Parameter und körperliche Kondition nestjunger Fischadler ( Pandion haliaetus )
Wir beurteilten Normalwerte für 15 biochemische Blutplasma-Parameter und drei Indizes für die Körperkondition (Indices of Body Condition—IBCs) bei freilebenden nestjungen Fischadlern (Pandion haliaetus) in Brandenburg (Deutschland). Die Werte wurden mit denen anderer Greifvogelarten verglichen und mögliche geschlechts- und altersabhängige Unterschiede wurden untersucht. Zusätzlich haben wir denkbare Zusammenhänge zwischen der Habitatqualität—gemessen anhand der Lage von Gebieten der Nahrungssuche und anhand menschlicher Störung—und dem Ernährungszustand der Nestlinge sowie der Reproduktion untersucht. Weibliche Nestlinge zeigten höhere Harnstoff-Durchschnittswerte und geringere Glukose-Werte als männliche, was mit einer höheren Wachstumsrate und einem höheren Nährstoffbedarf bei den jungen Weibchen gegen Ende der Nestlingsperiode zusammenhängen kann. Sieben Parameter zeigten Veränderungen abhängig vom Alter, wahrscheinlich im Zusammenhang mit der zunehmenden Körpermasse, mit dem höheren Metabolismus und mit der steigenden körperlichen Aktivität während der Phase kurz vor dem Flüggewerden. Umgekehrt zeigten die IBCs geringe Korrelationen mit ausgewählten Parametern bezüglich der Nährstoffversorgung, wahrscheinlich aufgrund angemessener und gleichmäßiger Versorgung der Nestlinge. Schließlich fanden wir, dass Fischadler ihre ursprünglichen elterlichen Investitionen vermutlich nicht der Habitatqualität anpassten, da die Reproduktion nicht von den Habitateigenschaften beeinflusst wurde. Jedoch verbesserte sich die Nährstoffversorgung in großen Bruten mit besseren Bedingungen zur Nahrungssuche, nicht aber in kleinen Bruten. Unsere Ergebnisse legen es nahe, dass Weibchen mit besserer Körperkondition mehr und besser ernährte Jungvögel hervorbringen, insbesondere unter besseren Bedingungen für die Nahrungssuche.

Similar content being viewed by others
References
Alonso-Álvarez C (2005) Age-dependent changes in plasma biochemistry of yellow-legged gulls (Larus cachinnans). Comp Biochem Physiol A 140:512–518
Alonso-Álvarez C, Ferrer M (2001) A biochemical study of fasting, subfeeding, and recovery processes in Yellow-Legged Gulls. Physiol Biochem Zool 74:703–713
Bai M-L, Schmidt D, Gottschalk E, Mühlenberg M (2009) Distribution pattern of an expanding Osprey (Pandion haliaetus) population in a changing environment. J Ornithol 150:255–263
Balasch J, Musquera S, Palacios L, Jiménez M, Palomeque J (1976) Comparative hematology of some falconiforms. Condor 78:258–273
Balbontín J, Ferrer M (2002) Plasma chemistry reference values in free-living Bonelli’s eagle (Hieraaetus fasciatus) nestlings. J Raptor Res 36:231–235
Bell DJ, Freeman BM (1971) Physiology and biochemistry of the domestic fowl. Academic, London
BirdLife International (2012) Pandion haliaetus. In: IUCN (eds) IUCN Red List of threatened species, version 2012.1. http://www.iucnredlist.org/
Casado E, Balbontín J, Ferrer M (2002) Plasma chemistry in Booted Eagle (Hieraaetus pennatus) during breeding season. Comp Biochem Physiol A 131:233–241
Chalmers GA, Barret MW (1982) Capture myopathy. In: Hoff GL, Davis JW (eds) Noninfectious diseases of wildlife. Iowa State University Press, Ames, pp 84–94
Costa ND, MacDonald DE, Swan RA (1993) Age-related changes in plasma biochemical values of farmed Emus (Dromaius novaehollandiae). Aust Vet J 70:341–344
De le Court C, Aguilera E, Recio F (1995) Plasma chemistry values of free-living White Spoonbills (Platalea leucorodia). Comp Biochem Physiol A 112:137–141
Del Hoyo J, Elliot A, Sargatal J (1994) Handbook of the birds of the world, vol 2: New World vultures to guineafowl. Lynx Edicions, Barcelona
Dobado-Berrios PM, Ferrer M (1997) Age-related changes of plasma alkaline phosphatase and inorganic phosphorus, and late ossification of the cranial roof in the Spanish imperial eagle (Aquila adalberti C. L. Brehm, 1861). Physiol Biol 70:421–427
Dobado-Berrios PM, Tella JL, Ceballos O, Donázar JA (1998) Effects of age and captivity on plasma chemistry values of the Egyptian vulture. Condor 100:719–725
Dujowich M, Mazet JK, Zuba JR (2005) Hematologic and biochemical reference ranges for captive California condors (Gymnogyps californianus). J Zoo Wildl Med 36:590–597
Eriksson MOG (1986) Fish delivery, production of young, and nest density of Osprey (Pandion haliaetus) in southwest Sweden. Can J Zool 64:1961–1965
Ferguson-Lees J, Christie DA (2001) Raptors of the world. Houghton Mifflin Company, Boston
Ferrer M (1992a) Regulation of the period of postfledgling dependence in the Spanish Imperial eagle Aquila adalberti. Ibis 134:128–133
Ferrer M (1992b) Natal dispersal in relation to nutritional condition in Spanish imperial eagles. Ornis Scand 23:104–107
Ferrer M (1993) Blood chemistry studies in birds: some applications to ecological problems. Comp Biochem Physiol 1:1031–1044
Ferrer M (1994) Nutritional condition of Spanish Imperial Eagle nestlings Aquila adalberti. Bird Study 41:120–123
Ferrer M, Dobado-Berrios P (1998) Factors affecting plasma chemistry values of the Spanish Imperial Eagle, Aquila adalberti. Comp Biochem Physiol A 120:209–217
Ferrer M, García-Rodríguez T, Carrillo JC, Castroviejo J (1987) Hematocrit and blood chemistry values in captive raptors (Gyps fulvus, Buteo buteo, Milvus migrans, Aquila heliaca). Comp Biochem Physiol A 87:1123–1127
Fourie F, Hattingh J (1980) Variability in bird haematology. Comp Biochem Physiol A 65:147–150
García-Rodríguez T, Ferrer M, Carrillo JC, Castroviejo J (1987a) Circadian rhythms of determined blood chemistry values in buzzards and eagle-owls. Comp Biochem Physiol A 88:663–669
García-Rodríguez T, Ferrer M, Carrillo JC, Castroviejo J (1987b) Metabolic responses of Buteo buteo to long-term fasting and refeeding. Comp Biochem Physiol A 87:381–386
González JL, Hiraldo F (1991) Some hematological data from marsh harriers (Circus aeroginosus) in central Spain. Comp Biochem Physiol A 100:735–737
Green AJ (2001) Mass/length residuals: measures of body condition or generators of spurious results? Ecology 85:1473–1483
Griffiths R, Double MC, Orr K, Dawson RJG (1998) A DNA test to sex most birds. Mol Ecol 7:1071–1075
Griminger P (1976) Protein metabolism. In: Sturkie PD (ed) Avian physiology. Springer, New York, pp 233–251
Grove RA, Henny CJ, Kaiser JL (2009) Osprey: worldwide sentinel species for assessing and monitoring environmental contamination in rivers, lakes, reservoirs, and estuaries. J Toxicol Env Health B 12:25–44
Hanauska-Brown LA, Dufty AM, Roloff GJ (2003) Blood chemistry, cytology, and body condition in adult Northern Goshawks (Accipiter gentilis). J Raptor Res 37:299–306
Harmata PJ, Restani M, Harmata AR (2007) Settlement patterns, foraging behavior, and reproductive success of ospreys along a heterogeneous riverine corridor. Can J Zool 85:56–62
Hotchleithner M (1994) Biochemistries. In: Ritchie BW, Harrison GJ, Harrison LR (eds) Avian medicine: principles and application. Wingers, Lake Worth, pp 223–245
Jambu M (1991) Exploratory and multivariate data analysis. Academic, New York
Jamieson IG, Seymour NR, Bancroft RP, Sullivan R (1983) Sibling aggression in nestling ospreys in Nova Scotia. Can J Zool 61:466–469
Jerzak L, Sparks TH, Kasprzak M, Bochenski M, Kaminski P, Wisniewska E, Mroczkowski S, Tryjanowski P (2010) Blood chemistry in white stork Ciconia ciconia chicks varies by sex and age. Comp Biochem Physiol B 156:144–147
Johnson DH, Krapu GL, Reinecke KJ, Jorde DG (1985) An evaluation of condition indices for birds. J Wildlife Manage 49:569–575
Juráni M, Výboh P, Zeman M, Lamošová D, Košt’ál L, Blažíček P (2004) Post-hatching dynamics of plasma biochemistry in free living European starlings (Sturnus vulgaris). Comp Biochem Physiol A 138:89–95
Kjellén N, Hake M, Alerstam T (2001) Timing and speed of migration in male, female and juvenile Ospreys Pandion haliaetus between Sweden and Africa as revealed by field observations, radar and satellite tracking. J Avian Biol 32:57–67
Klaassen M, Abraham KF, Jefferies RL, Vrtiska M (2006) Factors affecting the site of investment, and the reliance on saving for arctic breeders: the capital-income dichotomy revisited. Ardea 94:371–383
Lener HRL, Mindell DP (2005) Phylogeny of eagles, Old World vultures, and other Accipitridae based on nuclear and mitochondrial DNA. Mol Phylogenet Evol 37:327–346
Littell RC, Milliken GA, Stroup WW, Wolfinger RD, Schabenberger O (2006) SAS for mixed models. SAS Institute Inc., Cary
Löhmus A (2001) Habitat selection in a recovering Osprey Pandion haliaetus population. Ibis 143:651–657
Mealey B, Parks GM, Pages CM, Millsap BA, Bass OL, Bossart GD (2004) Serum chemistry values for nestling bald eagles (Haliaeetus leucocephalus) in Florida Bay, Everglades National Park. J Raptor Res 38:96–100
Muriel R, Casado E, Schmidt D, Calabuig CP, Ferrer M (2010) Morphometric sex determination of young Ospreys Pandion haliaetus using discriminant analysis. Bird Study 57:336–343
Niezgoda J, Pierzchala K, Lang D (1984) Levels of calcium and phosphatase in blood plasma of Japanese Quails. In: Janowski TM, Zakl NI (eds) Wybrane. Zagadnienia Fizjol. Endokrynol. Zwierzat Uzytkowych. Ossolinskich, Wroclaw, Poland, pp 103–114
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118:1883–1891
Peig J, Green AJ (2010) The paradigm of body condition: a critical reappraisal of current methods based on mass and length. Funct Ecol 24:1323–1332
Polo FJ (1995) Estudio bioquímico y enzimático del plasma de aves en cautividad (Ph.D. dissertation). Universitat de Barcelona, Barcelona
Poole AF (1989) Ospreys. A natural and unnatural history. Cambridge University Press, Cambridge
Sarasola JH, Negro JJ, Travaini A (2004) Nutritional condition and serum biochemistry for free-living Swainson’s Hawks wintering in central Argentina. Comp Biochem Physiol A 137:697–701
Saurola PL (1997) The osprey (Pandion haliaetus) and modern forestry: a review of population trends and their causes in Europe. J Raptor Res 31:129–137
Saurola PL, Koivu J (1987) Saaksi (The Osprey)—Kanta-Hameen Lintumiehet r.y., Forssa
Schaadt CP, Bird DM (1993) Sex-specific growth in Ospreys: the role of sexual size dimorphism. Auk 110:900–910
Schmidt D (1999) Untersuchungen zur Populationsbiologie und Habitatnutzung des Fischadlers Pandion haliaetus in Deutschland. ILN-Werkstattreihe 6, Singen
Schmidt D (2001) Die Bestandsentwicklung des Fischadlers Pandion haliaetus in Deutschland im ausgehenden 20. Jahrhundert Vogelwelt 122:117–128
Schmidt D (2010) Der Brutbestand des Fischadlers Pandion haliaetus in Deutschland im frühen 21. Jahrhundert Charadrius 46:10–17
Shmueli M, Izhaki I, Zinder O, Arad Z (2000) The physiological state of captive and migrating Great White Pelicans (Pelecanus onocrotalus) revealed by their blood chemistry. Comp Biochem Physiol A 125:25–32
Smith EE, Bush M (1978) Hematologic parameters on various species of strigiformes and falconiformes. J Wildlife Dis 14:447–450
Steeger C, Ydenberg RC (1993) Clutch size and initiation date of ospreys: natural patterns and the effect of natural delay. Can J Zool 71:2141–2146
Steidl RJ, Griffin CR, Niles LJ (1991) Contaminant levels of osprey eggs and prey reflect regional differences in reproductive success. J Wildlife Manage 55:601–608
Stinson CH (1977) Growth and behaviour of young ospreys Pandion haliaetus. Oikos 28:299–303
Stinson CH (1978) The influence of environmental conditions on aspects of the time budgets of breeding ospreys. Oecologia 36:127–139
Tilgar V, Ots I, Mänd R (2004) Bone alkaline phosphatase as a sensitive indicator of skeletal development in birds: a study of the great tit nestlings. Physiol Biochem Zool 77:530–535
Tilgar V, Kilgas P, Mägi M, Mänd R (2008) Age-related changes in the activity of bone alkaline phosphatase and its application as a marker of prefledging maturity of nestlings in wild passerines. Auk 125:456–460
Toschik PC, Rattner BA, McGowan PC, Christman MC, Carter DB, Hale RC, Matson CW, Ottinger MA (2005) Effects of contaminant exposure on reproductive success of osprey (Pandion haliaetus) nesting in Delaware River and Bay, USA. Environ Toxicol Chem 24:617–628
Umminger B (1977) Relation of whole blood sugar concentrations in vertebrates to standard metabolic rate. Comp Bioschem Physiol A 56:457–460
Van Daele LJ, Van Daele HA (1982) Factors affecting the productivity of Ospreys nesting in West-Central Idaho. Condor 84:292–299
Van Wyk E, Van der Bank H, Verdoorn GH (1998) Dynamics of haematology and blood biochemistry in free-living African whitebacked vulture (Pseudogyps africanus) nestlings. Comp Biochem Physiol A 120:495–508
Vergara P, Fargallo JA, Martínez-Padilla J (2010) Reaching independence: food supply, parent quality, and offspring phenotypic characters in kestrels. Behav Ecol 21:507–512
Villegas A, Sánchez JM, Costillo E, Corbacho C (2002) Blood chemistry and haematocrit of the black vulture (Aegypius monachus). Comp Biochem Physiol A 132:489–497
Viñuela JM, Ferrer M, Recio F (1991) Age-related variations in plasma levels of alkaline phosphatase, Calcium and inorganic phosphotus in chicks of two species of raptors. Comp Biochem Physiol 99:49–54
Weber M, Schmidt D, Hädrich J (2003) Chlororganische Rückstánde in Eiern des Fischadlers (Pandion haliaetus) aus Deutschland. J Ornithol 144:45–58
Wink M, Sauer-Gürth H (2000) Advances in the molecular systematics of African raptors. In: Chancellor RD, Meyburg BU (eds) Raptors at risk: proceedings of the V World Conference on Birds of Prey and Owls, Midrand, Johannesburg, South Africa 1998. WWGBP & Hancock House, Berlin, pp 135–147
Wolf SH, Schreiber RW, Kahama L, Torres JJ (1985) Seasonal, sexual and age-related variation in the blood composition of the Brown Pelican (Pelecanus occidentalis). Comp Biochem Physiol A 82:837–846
Acknowledgments
We are grateful to the Kreig and Bennewitz families for their hospitality and help in Germany, to Mr. Weiss and Mr. Heuer for their collaboration in the field, as well as to J. Giralt and M. Ríos, who collaborated as volunteers during the field work. We thank the Director and staff of the Botanical Zoo of Jerez, who allowed us to use the laboratory, the Office of Environment, Health and Consumer Protection of Brandenburg State for providing GIS data, and the German power company for facilitating the sampling of nests on certain pylons. We are also grateful to T. Mackrill and two anonymous referees who helped to improve the manuscript. R. Muriel was an FPU fellow (Ministry of Education, Government of Spain), and his work was partially funded by a temporary stay grant from the FPU program. The work of D. Schmidt was partly funded by the Deutsche Ornithologen-Gesellschaft DO-G. This study complies with the current laws of Germany and Spain.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. G. Guglielmo.
Appendix
Rights and permissions
About this article
Cite this article
Muriel, R., Schmidt, D., Calabuig, C.P. et al. Factors affecting plasma biochemistry parameters and physical condition of Osprey (Pandion haliaetus) nestlings. J Ornithol 154, 619–632 (2013). https://doi.org/10.1007/s10336-012-0925-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-012-0925-0