Abstract
Water pollution is calling for a sustainable remediation method such as the use of metallic iron (Fe0) to reduce and filter some pollutants, yet the reactivity and hydraulic conductivity of iron filters decline over time under field conditions. Here we review iron filters with focus on metallic corrosion in porous media, flaws in designing iron filters, next-generation filters and perspectives such as safe drinking water supply, iron for anaemia control and coping with a reactive material. We argue that assumptions sustaining the design of current Fe0 filters are not valid because proposed solutions address the issues of declining iron reactivity and hydraulic conductivity separately. Alternatively, a recent approach suggest that each individual Fe0 atom corroding within a filter contributes to both reactivity and permeability loss. This approach applies well to alternative iron materials such as bimetallics, composites, hybrid aggregates, e.g. Fe0/sand, and nano-Fe0. Characterizing the intrinsic reactivity of individual Fe0 materials is a prerequisite to designing sustainable filters. Indeed, Fe0 ratio, Fe0 type, Fe0 shape, initial porosity, e.g. pore size and pore size distribution, and nature and size of admixing aggregates, e.g. pumice, pyrite and sand, are interrelated parameters which all influence the generation and accumulation of iron corrosion products. Fe0 should be characterized in long-term experiments, e.g. 12 months or longer, for Fe dissolution, H2 generation and removal of contaminants in three media, i.e., tap water, spring water and saline water, to allow reactivity comparison and designing field-scale filters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Metallic iron (Fe0) is a popular and low-cost material used in the water treatment industry for more than 160 years (Bischof 1973; Devonshire 1890; Baker 1934; van Craenenbroeck 1998; Cundy et al. 2008; Mwakabona et al. 2017). Fe0 has been exploited to remove various contaminants and groups of contaminants from polluted natural waters and wastewaters through various mechanisms such as adsorption, co-precipitation, degradation, ion exchange, reductive transformations and size exclusion (Henderson and Demond 2007; Hu et al. 2021; Lawrinenko et al. 2023a, b; Noubactep 2023). Relevant contaminants include chlorinated solvents (Guan et al. 2015), emerging contaminants (Stieber et al. 2011), halide salts (Antia 2022), metalloids (Lan et al. 2023), radionuclides (Zhang et al. 2021) and trace metals (Li and Liu 2022).
The literature unanimously reports a time-dependent decline in the performance of Fe0-based filters (Bigg and Judd 2000; Lee et al. 2004; Li et al. 2005, 2006; Henderson and Demond 2007; Bartzas and Komnitsas 2010; Li and Benson 2010; Guan et al. 2015). Henderson and Demond (2007) identified two related processes as the major causes of the system’s failure: (1) a decline of Fe0 reactivity (reactivity loss) and (2) a decline of the hydraulic conductivity (porosity loss and permeability loss) (Firdous and Devlin 2014, 2015; Cormican et al. 2020). Biofouling and hydrogen (H2) production were the other two possible causes of filter clogging (Noubactep 2013; Plessl et al. 2023; Singh et al. 2023).
Since 2007, the large majority of researchers has been attempting to solve both problems by applying various countermeasures (Bartzas and Komnitsas 2010; Li and Benson 2010; Guan et al. 2015). Addressing the two phenomena separately ignores the fact that the two are interrelated since both are linked to the inherent reactivity of Fe0 (Caré et al. 2013; Noubactep 2013; Domga et al. 2015). Reactivity loss and permeability loss are two inherent aspects of iron corrosion in porous media (Stefanoni et al. 2018; Angst 2019; Yang et al. 2021, 2022). In other words, reactivity loss and permeability loss occur in synergy in all Fe0-based filters. Consequently, increased preferential flow, reduced available porosity, residence time and reduced treatment performance over time are present in all Fe0 filters (Noubactep 2013; Cormican et al. 2020; Tao et al. 2022; Bilardi et al. 2023; Tao 2024).
Moreover, individual Fe0 materials respond differently to different environmental conditions including contamination, pH value, temperature, and water chemistry (Raychoudhury and Scheytt 2013; Sun et al. 2016). Therefore, to understand and predict the long-term performance of a Fe0 filter, it is essential to characterize the contaminant-free Fe0/H2O system in a holistic approach. This holistic approach is based on the law of conservation of mass and entails considering the Fe mass balance to discuss three aspects: (1) the time-dependent generation of iron corrosion products (FeCPs) (corrosion rate), (2) the extent to which the pore space is filled (porosity loss) and (3) how the two related processes are impacted by dynamic changes in the pore size distribution. Obviously, material selection (corrosion rate or intrinsic reactivity) has not received the due attention in earlier studies (Velimirovic et al. 2018; Li et al. 2019; Cui et al. 2023; Ren et al. 2023).
The science of the contaminant-free Fe0/H2O system has been presented a century ago in the iron corrosion literature (Whitney 1903) but has been largely overlooked by the pioneers of the Fe0 remediation technology (Gillham and O’Hannesin 1994; Matheson and Tratnyek 1994) and the majority of subsequent active researchers as summarized by Lawrinenko et al. (2023a) and Xiao et al. (2023). Whitney (1903) demonstrated that, at pH larger than 4.5, Fe0 immersed in water is corroded by water and water alone (H+ from the dissociation of water molecules) (Eq. 1). If molecular oxygen (O2) is available, Fe2+ is further oxidized to Fe3+ (Eq. 2). This makes the reaction after Eq. 3 impossible.
The demonstration by Whitney (1903) was later validated by corrosion scientists during the 1930s while establishing the electrochemical theory of aqueous metal corrosion (Evans et al. 1931; Evans and Hoar 1932; Evans and Mears 1934). Equation 1 clearly shows that the oxidative dissolution of one mole of Fe0 produces one mole of Fe2+ and one mole of H2 (gas). In the absence of any organic complexing agent (e.g. natural organic matter), Fe2+ and Fe3+ form salts, e.g. FeCl2, FeCl3, FeSO4, complexes (FeF63−), or precipitates, e.g. Fe2O3, Fe3O4 (Whitney 1903; Wang et al. 2018; Crespo et al. 2023; Tao 2024). Hydrogen (H2) either escapes or is used in biogeochemical processes within the system. In other words, before the advent of the Fe0 remediation technology, it was established that Fe0 corrodes in water, even when present as moisture (Stratmann and Müller 1994; Wilson 1995; Tratnyek 1996).
Iron corrosion by water (Eq. 1) is electrochemical in nature. Whitney (1903) also demonstrated that the presence of dissolved O2 and carbonic acid (H2CO3) accelerates iron corrosion by: (1) consuming Fe2+ for the chemical reduction of O2 (Eq. 2) and (2) supplying H+ for Fe0 oxidation (via H2CO3 dissociation). Clearly, O2 reduction in a Fe0/H2O system is a chemical reaction (electrons from Fe2+) (Eq. 2). The rationale for this is that the Fe0 surface is instantaneously shielded by an oxide scale, acting as a diffusion barrier for O2, and other potential dissolved oxidizing agents, including contaminants (Noubactep et al. 2012a; Noubactep 2016a; Konadu-Amoah et al. 2022). The oxide scale also acts as a conduction barrier for electrons from Fe0 (Fig. 1) (Hu et al. 2021; Xiao et al. 2023).
Here, we show that assumptions sustaining the design of current Fe0 filters are not valid. Equation 1 summarizes the alternative approach already adopted by some few scientists (Ghauch 2015; Vollprecht et al. 2020; Gheju and Balcu 2023; Bilardi et al. 2023; Lan et al. 2023), including our research group (Noubactep 2015, 2023; Noubactep 2023; Xiao et al. 2023; Tao 2024; Tao et al. 2024). Herein, Eq. 1 is used to demonstrate the inconsistency of the popular view summarized by Lawrinenko et al. (2023a, b). The following sections start with an overview description of Fe0 corrosion in porous media ("Metallic iron corrosion in porous media" section), followed by a summary of the fundamental flaws in designing Fe0 filters ("Fundamental flaws in designing metallic iron filters" section). The section "Next-generation metallic iron filters" discusses how to better design such filters by drawing lessons from earlier studies.
Metallic iron corrosion in porous media
A reliable approach for a knowledge-driven design of sustainable and stable Fe0 filters goes through a holistic analysis of the Fe0/H2O system (Noubactep 2009; Cao et al. 2021). A Fe0 filter is a porous bed containing granular Fe0 in a pure (Fe0 alone) or hybrid system (e.g. Fe0/pyrite, Fe0/sand) (Moraci and Calabrò 2010; Firdous and Devlin 2015; Tao et al. 2022). On the one hand, a porous bed is characterized by an initial porosity, e.g. 36% for compact spheres (Caré et al. 2013), which necessarily decreases over time as contaminants are eliminated from inflowing polluted waters. On the other hand, iron corrosion is a volumetric expansive process, meaning that the volume of each oxide (Voxide) is larger than that of the parent metallic iron (Viron): Voxide > Viron (Pilling and Bedworth 1923; Caré et al. 2008; Zhao et al. 2011). This inherent feature of Fe0 materials implies that Fe0 filters experience permeability loss even in the absence of any contaminants (Caré et al. 2013; Luo et al. 2013; Tao et al. 2022). Luo et al. (2013) have documented this evidence using X-ray computed tomography. The results of Luo et al. (2013) also recalled that Fe0 corrodes in deionized water (contaminant-free) as reported by Whitney (1903).
Iron corrosion in a Fe0 filter is an electrochemical process as discussed above (Eq. 1). The four components are: (1) iron dissolution at an anodic site of Fe0, i.e. releasing Fe2+ and leaving electrons behind, (2) transport of released Fe2+ through the electrolyte (polluted water) away from the Fe0 surface, (3) transfer of electrons to a cathodic site on the surface of uncorroded Fe0 and (4) H+ reduction at the cathode (Fig. 2). The corrosion rate is influenced by several factors including: (1) the rate with which species H+ and Fe2+ are consumed or released (intrinsic reactivity), (2) their rate of transport through the pore system within the filter (system porosity), (3) the occurrence of further chemical reactions such as complexation, oxidation, precipitation (i.e. pH value, solution chemistry), and (4) the occurrence of physical processes such as adsorption, ion exchange and size exclusion (pH value, solution chemistry) (Nesic 2007; Noubactep 2015; Sun et al. 2016; Tao 2024; Tao et al. 2024).
Components (1) to (4) locally affect the electrolyte chemistry at the Fe0 surface, which in turn affects the electrochemical reaction kinetics of the uncorroded Fe0. It is essential to mention that H2 and Fe2+ (Eq. 1) are stand-alone reducing agents for several organic (Jiao et al. 2009) and inorganic pollutants (Farrell et al. 2001). Moreover, several FeII and FeII/FeIII species generated in Fe0/H2O systems are stand-alone natural reducing agents (Charlet et al. 1998; Chaves 2005; Henderson and Demond 2007). For example, Fig. 2 shows that iron corrosion begins at a location where Fe2+ is generated (anode). Fe2+ goes into the aqueous solution and two electrons left behind migrate to another location (cathode), where they are taken up by H+ from water dissociation. The resulting hydroxide ions (OH−) react with the Fe2+ to initially form hydrous iron oxides that precipitate as Fe(OH)2. Depending on the environmental conditions, Fe(OH)2 is oxidized and transformed into various FeII/FeIII oxides that form the oxide scale (Charlet et al. 1998; Liger et al. 1999). The oxide scale acts as a barrier that restricts quantitative contaminant transfer to the Fe0 surface.
Another characteristic feature of Fe0 filters is the presence of a porous medium which significantly influences all processes involved in the progression of iron corrosion (Eq. 1). The most important being that: (1) there is limited space to store expansive corrosion products, and (2) transport processes are all limited, e.g. restricting convection. A potential beneficial role of a porous medium adjacent to the Fe0 surface is that different local properties at the Fe0/H2O interface create new electrochemical cells to sustain iron corrosion.
Summarized, to design a Fe0 filter, the following is needed: (1) a filter container, (2) a reactive Fe0 sample, and (3) one or several filling materials (e.g. gravel, pumice, sand) or reactive aggregates, e.g. manganese dioxide (MnO2), pyrite (FeS2) (Tao et al. 2022; Bilardi et al. 2023; Tao 2024). Because Fe0 corrosion is volumetric expansive in nature, a pure Fe0 filter (100% Fe0) can be efficient but not sustainable (Hussam 2009; Domga et al. 2015). In other words, sustainable Fe0 filters are hybrid in nature, e.g. Fe0/sand (Tepong-Tsindé et al. 2019; Tao et al. 2022).
In practice, Fe0 proportion, Fe0 type, Fe0 shape, initial porosity, nature, shape and size of admixing aggregates, and water quality are interrelated parameters which all influence the progressive generation and accumulation of iron corrosion products (FeCPs) within the pores of any Fe0 filter (Ruhl et al. 2014; Cormican et al. 2020; Tepong-Tsindé 2021). The filter function can also be influenced by sediment influx and deposition. Consequently, determining the rate of sedimentation and the period before sediment accumulation obstructs the filter's ability to function effectively is crucial in Fe0 filter design. While designing a Fe0 filter, enough storage capacity for FeCPs should be considered so that FeCPs accumulation does not affect the filter's function during its planned operational life. One proven efficient approach has been the use of porous Fe0 materials (Bischof 1973; Hussam and Munir 2007; Hussam 2009; Rahman et al. 2013; Bilardi et al. 2023; Plessl et al. 2023).
Fundamental flaws in designing metallic iron filters
The literature reports on "detailed analysis of the rate of surface precipitate buildup" (Wilkin et al. 2002) in Fe0 filters as a critical step in understanding how long-term systems can be sustainably designed (Li et al. 2005, 2006; Li and Benson 2010). However, the reasoning was rooted on the Fe0 amount available for the reductive transformation of selected contaminants (Sarr 2001; Wilkin et al. 2002). In this approach, depending on the environmental conditions (e.g. aquifer chemistry, composition of Fe0 filter), different types of minerals and surface coatings have been discussed (Vikesland et al. 2003, 2007; Li et al. 2006; Li and Benson 2010). The impacts of microbiological processes (e.g. presence of bacteria and their mediated processes) have also been largely discussed (Henderson and Demond 2007; Raychoudhury and Scheytt 2013; Fu et al. 2014; Obiri-Nyarko et al. 2014). In this context, Wilkin et al. (2002) present a subsurface Fe0 filter as a "large reservoir of iron" for contaminant transformation whose action can be enhanced by microorganisms including iron-reducing, sulphate-reducing or methanogenic bacteria. Wilkin et al. (2002) also suggested that such enhancements may occur at the "expense of faster corrosion," thereby inducing a faster clogging of the Fe0 filter.
The approach presented by Wilkin et al. (2002) is commonplace in the Fe0 literature and was mainly a recommendation of the Interstate Technology & Regulatory Council (ITRC 2005, 2011). Accordingly, to explore the biogeochemical processes occurring within a Fe0 filter, and thus understand the reasons for reactivity loss and permeability loss, sophisticated structural analytical techniques and detailed coring and water sampling programmes are needed (Gaspar et al. 2002; McGuire et al. 2003; Chekli et al. 2016; Li et al 2019; Wielinski et al. 2022). However, in the absence of any theory of the system ("Introduction" section), there is no guide to orient the discussion of the achieved results from such monitoring studies. In addition, most of the observations made from coring and water sampling programmes are just static snapshots and their measurements are inaccurate (Brenner 2010). Thus, they cannot enable the generation of non-trivial models of the dynamic processes within Fe0 filters, especially as these occur over an enormous range of timescales, for example, from a few days or weeks (laboratory experiments) to several months or years (pilot studies and field implementations). Moreover, each Fe0 material has its own intrinsic corrosion rate (Reardon 1995), meaning that two different materials will fill the same pore space within different timescales (Fig. 3).
Changes in the residual porosity in a bed filled with spherical Fe0 particles: left: at the start of the operation (t0 = 0) and right: at some time later (t > t0, but t < t∞) before Fe0 complete exhaustion at t∞. The time required for complete exhaustion (t∞) depends on various factors, including Fe0 intrinsic reactivity, initial porosity, water chemistry and Fe0 ratio
The approach recommended by the Interstate Technology & Regulatory Council (ITRC) for subsurface Fe0-based permeable reactive barriers (Fe0 PRBs) aimed at: (1) characterizing the surface precipitates and minerals formed within and downgradient of the Fe0 PRBs, (2) developing conceptual models to predict the type and rate of precipitate formation, (3) identifying relevant microbiological activities impacting the service life of Fe0 PRBs, and (4) developing applicable and cost-effective protocols for long-term performance monitoring of Fe0 PRBs (ITRC 2005; 2011).
Ideally, a Fe0 PRB should support complete groundwater remediation while maintaining an acceptable hydraulic conductivity over its planned service life (Bilardi et al. 2023). However, inherent temporal and spatial variability of chemical, microbial and physical processes within the Fe0 PRB is difficult to correlate with permeability loss and reactivity loss using the conventional approach (Wielinski et al. 2022; Tao et al. 2024). This correlation has not been successfully established by the ITRC approach (Santisukkasaem and Das 2019; Wielinski et al. 2022; Berardozzi et al. 2023), suggesting that an alternative approach is needed to design sustainable Fe0 filters, including Fe0 PRBs (Wielinski et al. 2022).
The service life of a Fe0 filter is essentially determined by how fast minerals precipitate within its pore space, which in turn determines the rate of porosity loss and permeability loss. This reveals the paramount importance of the corrosion rate (Fe0 intrinsic reactivity) of used materials in aqueous environments (Lufingo et al. 2019; Cui et al. 2023; Noubactep 2023; Noubactep et al. 2023a; b). Since 1995, the corrosion rate of Fe0 materials has been widely characterized (Reardon 1995, 2005; Velimirovic et al. 2018). However, this has been done in a too pragmatic approach (Lufingo et al. 2019) and for too short experimental durations (Li et al. 2019; Konadu-Amoah et al. 2023). This points to the need for: (1) an alternative approach based on the fundamentals of iron corrosion science and (2) considering reasonably long timescales representative of real field applications. The presentation herein insists on the fact that the oxidative dissolution of each individual Fe0 contributes to reducing the pore space (porosity loss) and inducing reactivity loss. This occurs to different extents depending on the prevailing environmental conditions, e.g. availability of molecular O2, Fe0 mass, Fe0 ratio, pH value, water chemistry (Sun et al. 2016; Tao et al. 2023, 2024).
A proof that the conventional approach has not worked is the development of artificial neural network for evaluating permeability decline in Fe0 PRBs (Santisukkasaem et al. 2015; Santisukkasaem and Das 2019; Berardozzi et al. 2023). These authors clearly stated that all other approaches were not successful as independently reported by Moraci et al. (2016) and Noubactep (2016b). However, the artificial neural network is not a physically based approach as it relies on the network’s ability to understand given information and outputs from physically based relationships derived using other methods (Santisukkasaem et al. 2015). Fortunately, a physical approach has been presented which is able to address both permeability loss and reactivity loss in a very simple and low-cost experimental setup: the methylene blue discoloration method (Btatkeu-K. et al. 2016; Konadu-Amoah et al. 2021; Tao 2024; Tao et al. 2024).
The methylene blue method uses methylene blue as an indicator of reactivity for the Fe0/H2O system (Btatkeu-K. et al. 2016; Konadu-Amoah et al. 2021; Konadu-Amoah 2023; Tao 2024; Tao et al. 2024). The methylene blue method is hinged on the historic findings by Mitchell et al. (1955) that the efficiency of pure sand to adsorb methylene blue declines as iron oxides progressively cover the surface of sand (Fig. 4). The suitability of the methylene blue method to characterize Fe0/H2O systems is based on the evidence that: (1) sand is a good adsorbent for methylene blue, and (2) iron oxide-coated sand has a poor affinity for methylene blue discoloration (Miyajima 2012). Zarime et al. (2024) have recently independently reproduced the results of Miyajima (2012) using granitic residual soil-supported nano-Fe0. The method characterizes methylene blue discoloration by sand as it is in-situ coated by iron corrosion products (FeCPs) in Fe0/H2O/sand system (Miyajima 2012). In other words, the methylene blue method enables the detection of the decline of Fe0 reactivity (reactivity loss), which is related to porosity loss. In fact, Miyajima (2012) has also demonstrated the ability of the methylene blue method to trace the clogging of Fe0 filters (permeability loss).
The time-dependent process of methylene blue (MB) discoloration in Fe0/sand systems. From the start of the experiment (t0 = 0), Fe0 corrosion produces solid iron corrosion products (FeCPs) which progressively coat the sand surface. At t1 higher than t0, available sand is completely coated with FeCPs and methylene blue is no longer attracted to sand particles. From t1 onwards, methylene blue is eliminated from the aqueous phase solely by occlusion within the matrix of precipitating FeCPs (co-precipitation)
Miyajima (2012) unequivocally demonstrated for the first time that admixing Fe0 with non-expansive aggregates is a prerequisite for sustainable Fe0 filters. Prior to 2012, admixing Fe0 with other aggregates was regarded as "material dilution" with possible adverse impacts on reaction rates and contaminant removal efficiency despite a beneficial cost reduction (Kenneke and McCutcheon 2003; Kaplan and Gilmore 2004; Song et al. 2005; Bi et al. 2009). This misconception originated from the false view that Fe0 is a reducing agent for many dissolved species (Matheson and Tratnyek 1994; O'Hannesin and Gillham 1998; Furukawa et al. 2002; Lawrinenko et al. 2023a, 2023b).
Next-generation metallic iron filters
A biased starting point
The presentation until now suggests that the Fe0 research community has been violating the first law of thermodynamics for three decades. It has been known since 1903 that, in aqueous solutions at pH 4.5 or above (quantitative Fe precipitation), Fe0 is corroded by H+ (from water dissociation or a weak acid) and only by H+ (Whitney 1903). According to Whitney (1903) and in tune with the laws of thermodynamics, O2 reduction in the presence of Fe0 (Eq. 2) is mediated by electrons from Fe2+ (Eq. 1). Fe0 corrosion is just accelerated as Fe2+ from Eq. 1 is consumed for O2 reduction (Le Chatelier's principle). In other words, it has been a mistake to introduce Fe0 as a stand-alone reducing agent for selected species (including O2) in polluted waters under environmental conditions (Matheson and Tratnyek 1994; Gillham and O’Hannesin 1994; O’Hannesin and Gillham 1998). Moreover, the pioneers of the Fe0 technology have not proved Whitney (1903) false. Instead, they were looking for a rationale "directly relevant" to "reductive dechlorination by Fe0" (Reynolds et al. 1990; Lee et al. 2004). However, as discussed above, reductive dechlorination occurs in the presence of Fe0 (Eq. 4) and not by Fe0 (Eq. 5) (Fig. 5). Clearly, the Fe0 remediation technology was introduced with a thinking mistake. This mistake has dragged active researchers very deep into confusion (Noubactep 2015; Makota et al. 2017; Cao et al. 2022; Xiao et al. 2023), to the extent that attempts to fix some confusionare now blamed and misconstrued as "isolated misconceptions" (Lawrinenko et al. 2023a).
The mechanism for contaminant (RCl) reduction in Fe0/H2O systems: left: Fe0 is oxidized by RCl and water (H+) (Path 1), and right: Fe0 is oxidized by water (H+) and RCl is reduced by FeII species and H2 (Path 2). Path 1 is not feasible because the oxide scale is a conduction barrier for electrons from Fe0. Path 2 is feasible and rooted on aqueous iron corrosion
In 1947, in recognition of the significance of the 1903 work of Dr. Whitney to the development of corrosion science, the National Association of Corrosion Engineers (NACE—Houston, USA) introduced the Willis Rodney Whitney Award with Dr. Whitney as the first recipient. The award is given "in recognition of significant contributions to corrosion science, such as the development or improvement of a theory that provides a fundamental understanding of corrosion phenomena and/or the prevention of corrosion." In other words, Dr. Whitney is regarded by corrosion experts as an individual who has made a major contribution to corrosion science through work that leads to a better understanding of the science of corrosion. Clearly, the consensus adopted to justify observed contaminant reduction in Fe0/H2O systems (O'Hannesin and Gillham 1998) has disproved and even distorted well-established knowledge of corrosion scientists (Hu et al. 2021; Konadu-Amoah et al. 2022; Xiao et al. 2023). The correction shall start at the origin, where possible, with little attention to the volume of scientific articles that have been propagating the named mistakes (Chen et al. 2019; Antia 2020; Li et al. 2021; Wu et al. 2024).
It is essential to recall that some individual researchers and research groups have constantly challenged the validity of the prevailing theory (Fe0 as a reducing agent) (Warren et al 1995; Fiedor et al. 1998; Farrell et al. 1999; Qiu et al. 2000; Lavine et al 2001; Lee et al. 2004; Noubactep 2007, 2008, 2015; Jiao et al. 2009; Gheju 2011, 2018; Ghauch et al. 2011). In particular, Lavine et al. (2001) investigated nitrobenzene reduction in Fe0/H2O systems using differential pulse polarography, a technique which can simultaneously monitor the disappearance of nitrobenzene and the appearance of Fe2+. Their results could not confirm any involvement of the Fe0 surface in the processes controlling nitrobenzene reduction within the system. This result is in tune with the seminal work of Whitney (1903) and recalls that despite a prevailing wrong theory, a science-based interpretation of one’s experimental observations is always possible (Vollprecht et al. 2020; Cao et al. 2021; Tao 2024; Tao et al. 2024).
Subsequent publications by several other research groups denying the named theory have been acknowledged in some overview papers like Giles et al (2011) and Sista et al. (2021). In other words, the wrong theory and the alternative have coexisted in the literature for the past 30 years. However, each individual researcher has opted for their preference (Lawrinenko et al. 2023a, 2023b; Noubactep 2023; Noubactep 2023); Xiao et al. 2023). As a result, a circular reasoning has been established, maintaining the Fe0 technology as a whole in the status of an innovative technology.
Roadmap for sustainable filters
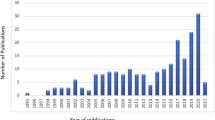
The misconception that Fe0 is an environmental reducing agent created unprecedented confusion within the environmental research community (Furukawa et al. 2002; Makota et al. 2017; Vollprecht et al. 2018; Hu et al. 2021; Lawrinenko et al. 2023a, b). The realization of this confusion around 2005 has prompted the exploration of new avenues of investigation (Noubactep 2007, 2012; Makota et al. 2017; Yang et al. 2022). A critical evaluation of the literature revealed three main pervasive issues in research methodology: (1) the lack of standardized procedures to characterize the Fe0/H2O system (Noubactep et al. 2006; Ghauch et al. 2011), (2) the lack of organized and reliable data (Noubactep 2023, 2005a), and (3) the lack of unified tools for Fe0 characterization, including the lack of a reference Fe0 material (Noubactep et al. 2005b; Reardon 2005). Moreover, past and current shortcomings in research methodology highlighted the need for a multifaceted approach to tackle the named confusion. Thus, from 2008 onwards, our research group set out to explore and develop affordable and applicable strategies to advance shared efforts in optimizing the application of Fe0 within the water treatment industry. Table 1 summarizes some milestones of this research effort.
One key phenomenon characterizing the Fe0/H2O system is the occurrence of some chemical reactions between dissolved Fe2+ from Eq. 1 and water constituents, including inorganic salts and molecular O2 (Wang et al. 2018; Tao et al. 2023, 2024). These interactions culminate in the coverage of the Fe0 surface by a film of iron corrosion products (e.g. oxide scale) (Nesic 2007). Thus, Fe0 oxidative dissolution (anodic dissolution) and contaminant removal (e.g. physical adsorption) and/or transformation (e.g. chemical reduction) occur at a different timescale (Schreier and Reinhard 1994; Hao et al. 2005; Naseri et al. 2017). This was one of the first experimental observations in investigating the Fe0/H2O system (Schreier and Reinhard 1994) and a clear proof that Fe0 dissolution and contaminant reduction are not simultaneous reactions (Lipczynska-Kochany et al. 1994; Lavine et al. 2001; Lan et al. 2023). This means that reducing electrons for any dissolved species are not from Fe0 or that contaminant reduction is not the cathodic reaction coupled to Fe0 oxidation (Noubactep 2016a; Hu et al. 2021; Xiao et al. 2023). This corresponds to the report of Whitney (1903) and disproves the theory introduced by the pioneers of the Fe0 remediation technology in the early 1990s. The question arises, how can this evidence be denied for so many years? The answer lies in the suitability of the used methodologies to investigate the Fe0/H2O system in both batch and column experiments (Naseri et al. 2017; Bilardi et al. 2023; Tao et al. 2023, 2024; Xiao et al. 2023).
There have been a large number of systematic studies designed to elucidate the mechanisms of contaminant removal in Fe0/H2O systems and the effects of many operational parameters, e.g. Cl−, Fe0 size, Fe0 treatment, Fe0 type, HCO3−, mixing intensity, O2, SO42−, on related processes (Sun et al. 2016; Abd El-Monaem et al. 2024; Tao et al. 2024). However, controversial results have been typically reported, even for the same probe contaminant and the same Fe0 material (Matheson and Tratnyek 1994; Jiao et al. 2009). This is usually justified by the evidence that varying experimental procedures are employed for the collection of data (Sun et al. 2016; Tao et al. 2023, 2024). For batch experiments, these procedures differ in Fe0 pre-treatment, Fe0 particle size (e.g. granular, powder, nanoscale), admixing agents used (e.g. FeS2, MnO2, pumice, sand), volume of the experimental vessels (mL), volume of solution added (mL), equilibration time allowed, and ambient temperature (Vidic et al. 1990; Polasek 2007; Naseri et al. 2017).
The reasoning in sections "Metallic iron corrosion in porous media" and "Fundamental flaws in designing metallic iron filters" suggests that the major pitfall in conventional batch experiments has been the disturbance of the "natural" layering sequence in Fe0/oxide/H2O in which the Fe0 surface is permanently shielded by an oxide scale. In fact, while performing batch experiments, mixing intensities of up to more than 400 rpm are used to explicitly keep all particles in suspension and avoid mass transfer limitations (Devlin et al. 1998; Miyajima and Noubactep 2012; Noubactep et al. 2012a). Such procedures certainly provide reproducible results, but without practical significance for filtration systems. In Fe0 filters, transport in the vicinity of Fe0 is diffusion-driven (Matheson and Tratnyek 1994; Nesic 2007). For this reason, quiescent batch experiments are recommended for treatability studies (Miyajima and Noubactep 2012; Noubactep 2012; Naseri et al. 2017). Our research group has been using exclusively quiescent batch experiments (mostly Fe0/sand mixtures) for the past 15 years (since 2009) for experimental durations of up to 90 days (3 months). A major result has been that expressing the performance of Fe0/H2O systems in terms of adsorption capacity (e.g. mg of contaminant per g of Fe0) is not acceptable (Naseri et al. 2017). This is because: (1) Fe0 is not depleted, (2) its corrosion rate is not known, and (3) no standard experimental protocol has been established.
The literature reports difficulties in understanding the long-term permeability of Fe0 filters (Domga et al 2015; Moraci et al 2016; Bilardi et al. 2023). Our research group has established around 2010 that sustainable Fe0 filters are hybrid in nature, wherein Fe0 is admixed with non-expansive aggregates (e.g. Fe0/MnO2, Fe0/pyrite, Fe0/sand) (Noubactep and Caré 2010a, b; Noubactep et al. 2011, 2012b). It is established that factors influencing the sustainability of Fe0 filters include: (1) reactive Fe0 (intrinsic reactivity), (2) Fe0 shape and size, (3) nature and extent of contamination, (4) biogeochemical and hydrogeological characteristics of the aquifer, and (5) groundwater flow velocity (Tao et al. 2022, 2023, 2024). Bilardi et al. (2023) analysed the relative importance of these parameters and showed that "admixing Fe0 with non-expansive granular materials" is the most suitable tool to design "long-term hydraulically efficient" Fe0 filters. This corresponds to our findings from 2010 (Noubactep and Caré 2010a, b), showing that despite hundreds of new peer-reviewed scientific publications, little progress has been made. The reason for this sad situation is the proliferation of pragmatic, but independent investigations without any opportunity to compare achieved results. In particular, concerning the impact of Fe0/sand ratio, the rationale for working with 50% mixing ratio (w/w or v/v) is rarely specified, while (1) the Fe0 intrinsic reactivity is not characterized (Cui et al 2023), (2) evidence exists that far lesser Fe0/sand ratios are efficient (Tepong-Tsindé 2021), and (3) experiments are performed for too short experimental durations (Alyoussef 2019; Yang 2022; Yang et al. 2022; Xiao et al. 2023; Tao et al. 2024).
This study recalled the following: (1) intensively homogenizing the experimental vessels has a significant influence on the decontamination efficiency of Fe0 while disturbing the real reaction mechanisms, (2) there have been no unified tools to characterize the intrinsic reactivity of Fe0 materials used, and (3) treatability studies have been performed for too short experimental durations. These three factors together explain differences in decontamination efficiencies reported in the literature for the same Fe0/contaminant pair, for example Fe0/CCl4 for which Matheson and Tratnyek (1994) favoured an electrochemical mechanism while Jiao et al. (2009) explicitly rule out any electron transfer from Fe0. Additionally, the Fe0 remediation technology involves immersing small Fe0 particles (less than 5 mm thick) in water-saturated zones for long times. This is a novel situation for environmental scientists since there are no comparable data in the scientific literature, for example, on the long-term corrosion kinetics (Yang et al. 2022).
Therefore, a new experimental procedure (long-term quiescent batch experiments) is recommended for obtaining better data reflecting the true Fe0 corrosion kinetics. Alternatively, the experimental vessels can be very gently homogenized, e.g. stirred at 75 rpm, such that the generation of an oxide scale on the Fe0 surface is not disturbed (Miyajima and Noubactep 2012). Universally employing this experimental procedure will enable the acquisition of reliable and comparable data to design and model better Fe0 filters for the water treatment industry. Since a parameter like an "adsorption capacity" for Fe0 materials cannot be defined, it is urgent to develop some standard experimental protocols to enable the comparison of results from independent investigations. The starting point is certainly the characterization of the intrinsic reactivity of available Fe0 materials, e.g. iPutec GmbH & Co. KG in Germany, Connelly-GPM in the USA or Högnäs in Sweden. Such Fe0 materials can be intensively characterized for their long-term ability to: (1) generate H2 under well-defined conditions, e.g. tap water, spring water and saline water, (2) release Fe2+ in selected solutions, e.g. ascorbic acid, ethylenediaminetetraacetic acid—EDTA, and (3) remove some selected probe contaminants under well-defined operational conditions.
The discussion of a large number of data, obtained under such controlled experimental conditions (e.g. items (1) to (3)), would reveal some common underlying trends for interactions within Fe0/H2O systems that provide confidence for a design that is non-material-specific. Accordingly, when a significant body of data exists on removal rates for selected contaminants by Fe0 materials depicting certain characteristics, site-specific treatability studies may only be required to fine-tune the design criteria for the optimal performance of Fe0 filters. Such data would provide a good starting point for the design of future laboratory-, pilot- and field-scale studies for investigating the remediation Fe0/H2O system (McGeough et al. 2007; Naseri et al. 2017; Xiao et al. 2023; Tao 2024).
The presentation herein has recalled that Fe0 is corroded by water (H+) and not by any contaminant. This means that polluted water contains one or several species that can enhance or impede Fe0 corrosion (Sun et al. 2016; Naseri et al. 2017; Xiao et al. 2023; Tao 2024; Tao et al. 2024). The question is: how are individual contaminants removed during Fe0 corrosion in the presence of polluted water? This approach is supported by reports on Fe0 corrosion in deionized water (Luo et al. 2013; Xin et al. 2016) and the removal of several species without redox reactivity in Fe0/H2O, such as methylene blue (Miyajima 2012; Btatkeu et al. 2016; Abd El-Monaem et al. 2024) pathogens (Bojić et al. 2001; You et al. 2005), phosphates (James et al. 1992; Erickson et al. 2012), triazoles (Jia et al. 2007a, b) and zinc (Bojic et al. 2009; Kishimoto et al. 2011).
Perspective
The Fe0 remediation research community has been working with the wrong concept for the past three decades. Thus, this presentation seeks to motivate colleagues to use the new information to design better remediation Fe0/H2O systems. In particular, the presentation has insisted on the relatively low significance of contaminants for the general design of such systems. In fact, the simplified dynamic Fe0/H2O system can be summarized in a surrogate reaction after Eq. 6:
The rationale for Eq. 6 is recalled in Sect. "Metallic iron corrosion in porous media" as resulting from the seminal work of Whitney (1903). Polluted water may be natural water or wastewaters from agricultural, domestic and industrial origins. Each water or wastewater is characterized by its chemical composition, including the concentration of protons (pH value) (Sun et al. 2016; Zhu et al. 2022; Li et al. 2023). The variability of water or wastewater sources makes the design of remediation Fe0/H2O systems highly site-specific. The presence of Fe complexes and Fe salts in Eq. 6 delays the availability of iron corrosion products (FeCPs), which are the contaminant scavengers (Fig. 6) (Tao et al. 2024). Once FeCPs are available, inorganic ligands, natural organic matter and polluting contaminants, e.g. arsenic, compete with each other to influence their further transformation, including the adsorptive removal of polluting contaminants (Tao 2024; Tao et al. 2024).
Iron corrosion in Fe0/H2O systems as influenced by the presence of diverse solutes, e.g. anions and O2. Fe0 is oxidized by water (H+), and O2 is eventually reduced by FeII species and H2. However, all solutes influence the process of iron oxidation and the subsequent precipitation of solid iron corrosion products (FeCPs) (co-precipitation). Once FeCPs are available, all solutes, including pollutants compete for adsorption at their surface
In other words, Tao (2024) has re-demonstrated that investigating the remediation Fe0/H2O system can be summarized by characterizing the process of Fe0 corrosion in the polluted water of concern (Eq. 6), while keeping in mind that each Fe0 reacts with its own intrinsic kinetics (corrosion rate) and that this kinetics declines with the time (reactivity loss) (Tao et al. 2024). The uniqueness of each reactive Fe0 material suggests that it is possible to manufacture and select appropriate Fe0 materials for specific applications. Two examples of such applications are safe drinking water supply and food fortification.
Safe drinking water supply
People throughout the world are afflicted by inadequate access to clean potable water, and this problem is expected to get worse in certain low-income regions. Since the early 2000s, a tremendous amount of research has been conducted to make Fe0-based filters affordable, efficient and robust methods of water treatment at domestic and community scales (Baumgartner et al. 2007; Shannon et al. 2008; Singh et al. 2014; Banerji and Chaudhari 2017; Kundu et al. 2018; Noubactep 2018, 2024; Ghosh et al. 2024). In other words, the affordability of such systems and their acceptability have already been largely discussed (Murcott 2007; Neumann et al. 2013; Kundu et al. 2018; Nguyen and Mulligan 2023; Ghosh et al. 2024). Here, we highlight how existing devices can be made more efficient based on the science of the system (Eq. 6, Fig. 2).
Two main types of Fe0 filters were developed for household water filters: (1) the Kanchan filter (Ngai et al. 2006, 2007) and (2) the Sono filter (Hussam and Munir 2007). Both systems were primarily developed for arsenic removal. However, Eq. 6 and field results demonstrated that far more species than arsenic were quantitatively removed from polluted waters (Naseri et al. 2017; Kundu et al. 2018; Huang et al. 2021a, b; Ndé-Tchoupé et al. 2022). A Sono filter uses a proprietary material and is regarded as one of the best available household water filters (Naseri et al. 2017), but has not been disseminated to the expected extent (Neumann et al. 2013; Kundu et al. 2016; Edward et al. 2023). As concerning the Kanchan filter, it has received continuous attention since 2007 (Singh et al. 2014; Wenk et al. 2014; Ogata et al. 2020; Mueller et al. 2021; Mueller et al. 2023; Nguyen and Mulligan 2023; Noubactep 2024), but is still not satisfactorily implemented. According to Huang et al. (2021a) and Ndé-Tchoupé et al. (2022), the original Kanchan filter was designed with a thinking mistake. Other researchers have rightly concluded that Sono filters are better than Kanchan ones (Singh et al. 2014; Wenk et al. 2014; Noubactep 2024). Equation 6 shows that where Kanchan filters were successful in supplying safe drinking water, the combination of "water quality, Fe0 reactivity, Fe0 mass, water flow velocity" was optimal to treat polluted water to drinking standards. At some other sites, the water chemistry was also responsible for the observed failures. However, the actual mistake is to design "one filter for all situations." Clearly, despite a design mistake (not addressed herein), a filter can be efficient but may be less or not sustainable. Community-scale filters, e.g. IIT Bombay filter, built on the principle of the Kanchan filter have been very efficient for the past 15 years (Banerji and Chaudhari 2017) and could serve communities of up to 8,000 people (Etmannski and Darton 2014; Ghosh et al. 2024).
Altogether, using the Kanchan filter and the IIT Bombay filter as reference systems, the whole world can design modified decentralized water treatment systems to achieve Goal 6.1 of the United Nations Sustainable Development Goals (SDGs) within the remaining six years to 2030. Since 2009, our research group has been actualizing related concepts almost on a yearly basis (Noubactep et al. 2009b; Noubactep 2010, 2013, 2018, 2020, 2024; Yang et al. 2020). However, more holistic research is needed, mainly at the pilot scale.
Metallic iron for the control of anaemia
Low-income countries are still suffering from iron deficiency anaemia (IDA). This is because iron fortification of food (e.g. table salt, wheat flour) has been the most important tool to control and defeat the IDA in the developed world where food is systematically processed (Hurrell 2021; Kumari and Chauhan 2022). Processed food is not often available in developing countries as a rule, and when it is available, it is not affordable for the poor (Charles 2012; Noubactep et al. 2023a; b). For these reasons, alternative iron sources have been sought for low-income countries. Relevant alternative solutions include: (1) cooking with iron pots (Arcanjo et al. 2020), (2) cooking in pots of other materials with iron ingots in the meat (Rodríguez-Vivaldi and Beerman 2018; Massey et al. 2023) and (3) fortifying potable water with FeII salts and ascorbic acid (Dutra-de-Oliveira et al. 2011).
Equation 6 suggests that all tools using iron metal (Fe0) to combat IDA are not necessarily reliable because of the unknown corrosion rates (Yang et al. 2020). On the other hand, the use of FeII salts is expensive. These two facts have motivated the development of a new approach to water fortification. The approach entails leaching iron from Fe0 with a solution of ascorbic acid (AA) and obtaining a concentrated FeII-AA solution (Solution 1). Solution 1 is then diluted to obtain drinking water with the desired Fe concentration, allowing the prevention or the healing of IDA. For details on this approach, interested readers are referred to Cui et al. (2023), Noubactep (2023), and Noubactep et al. (2023a; b). Clearly, the understanding of the operating mode of Fe0-based filters has inspired an innovation that can help the developing counties out of "the valley of tears" regarding the tragedy of IDA. In fact, the scientific community seems to be helpless in finding an affordable and universally applicable solution to control and defeat IDA (Rai et al. 2018; Pasupathy et al. 2023). More multidisciplinary research is needed to realize this concept for the benefit of the whole world particularly poor communities in low-income countries.
Coping with a reactive material
Understanding the corrosion process is crucial for designing novel Fe0-based technologies such as those for water treatment (“Safe drinking water supply” section) and combatting anaemia (“Metallic iron for the control of anaemia” section). These new technologies striving at "putting corrosion to use" (Tratnyek 1996) cannot just translate available results from research in corrosion science (Landolt 2007; Nesic 2007; El Ibrahimi et al. 2021; Li et al. 2024). This is because past research had mostly strived to avoid or mitigate corrosion (Holmes and Meadowcroft 1977; Nesic 2007; Balko et al. 2012; Chekli et al. 2016; El Ibrahimi et al. 2021). These efforts have already established the complexity of involved processes with at least two phases, for instance, solid (e.g. Fe0, FeCPs) and liquid (H2O); and solid and gas (e.g. H2). Therefore, corrosion research is a highly multidisciplinary task demanding the combination of several experimental methods to adequately monitor the corrosion process (Lavine et al. 2001; Balko et al. 2012; Wielinski et al. 2022). Traditionally, corrosion research is performed mainly by chemists, electrochemists, material scientists and physicists (Holmes and Meadowcroft 1977; Landolt 2007). These scientists express the corrosion rate in many different ways, including corroded depth per unit of time, current density, mass loss per unit time, number of moles transformed per unit of time, and volume of H2 generated per unit of time. However, for water treatment, the corrosion rate has to be correlated with the extent of contaminant removal (Lavine et al. 2001; Balko et al. 2012; Xiao et al. 2023; Tao et al. 2024).
The challenge of using Fe0 for water treatment entails considering two groups of phenomena and their uncertainties: (1) the Fe0 corrosion rate and the amount of FeCPs generated in-situ, and (2) the mineralogical constitution of FeCPs with their adsorptive affinities to the contaminants of concern. The question of how the whole process is influenced by the solution chemistry is still not properly addressed (Sun et al. 2016; Tao et al. 2024). The presentation above has demonstrated that the Fe0 reactivity is yet to be properly considered while the recent literature reports on fundamental flaws in designing and interpreting adsorption experiments (Tien 2007; Tran et al. 2017, 2023). In other words, while non-considering the amounts of contaminant scavengers generated in their systems, scientists have developed new models that are necessarily false. The viability of the Fe0 remediation as technology depends largely on the quality of the work produced by its investigators. Based on the fact that the problems exposed herein have been revealed since 2007 (Noubactep 2007, 2008), it is fair to state that, within the Fe0 research community, the majority of the engaged individuals are without adequate preparations or readiness to consider alternative views or theories. This must be a concern to the whole environmental research community. This issue cannot be resolved by a few individuals or research groups (Tien 2007; Ghauch et al 2011; Hu et al. 2021; Gheju and Balcu 2023; Lan et al. 2023). This paper was written to make the problem better known to colleagues and the general public.
Conclusion
An analysis of the Fe0/H2O system reveals that, for some 30 years, the Fe0 research community has misinterpreted the good experimental observation that some pollutants are chemically reduced in the presence of Fe0 as an electrochemical process (electrons from Fe0). In other words, inaccurate calculations, conclusions and discussions were drawn by scientists over a period equivalent to some 7 to 10 generations of PhD students. According to the viewpoints discussed above, the following key conclusions and perspectives are put forward:
-
Fe0 is not a stand-alone reducing agent under environmental conditions, at pH higher than 4.5;
-
The removal performance of Fe0 materials should not be expressed in mass per mass basis (e.g. mg pollutant per g Fe0), unless Fe0 is exhausted. This is because appropriate alternative parameters suitable for Fe0 are yet to be developed;
-
Permeability loss and reactivity loss are two inherent and related properties of Fe0 in a porous system. The occurrence of both processes is intimately related to the intrinsic reactivity of the materials used and the pore space available;
-
An in-depth characterization of the long-term reactivity of selected Fe0 materials under relevant operational conditions is regarded as a key step in designing the next-generation Fe0 filters.
-
In discussing experimental observations, more attention should be paid to understanding the unique properties of Fe0 corrosion products and target pollutants to avoid mistakes made when explaining removal mechanisms.
Data availability
Not applicable.
Code availability
Not applicable.
References
Abd El-Monaem EM, Omer AM, El-Subruiti GM, Mohy Eldin MS, Eltaweil AS (2024) Zero-valent iron supported-lemon derived biochar for ultra-fast adsorption of methylene blue. Biomass Conv Bioref 14:1697–1709. https://doi.org/10.1007/s13399-022-02362-y
Alyoussef G (2019) Characterizing the impact of contact time in investigating processes in Fe0/H2O systems. Master Dissertation, University of Göttingen, Germany.
Angst UM (2019) A critical review of the science and engineering of cathodic protection of steel in soil and concrete. Corrosion 75:1420–1433. https://doi.org/10.5006/3355
Antia DDJ (2022) Remediation of salinewastewater producing a fuel gas containing alkanes and hydrogen using zero valent iron (Fe0). Water 14:1926. https://doi.org/10.3390/w14121926
Antia DDJ (2020) Water treatment and desalination using the eco-materials n-Fe0 (ZVI), n-Fe3O4, n-FexOyHz[mH2O], and n-Fex[Cation]nOyHz[Anion]m [rH2O]. In: Kharissova OV et al (eds) Handbook of nanomaterials and nanocomposites for energy and environmental applications. Springer, Switzerland. https://doi.org/10.1007/978-3-030-11155-7_66-1
Arcanjo FPN, dos Anjos-Sena IV, Queiroz-de-Oliveira AF, Duarte-dos-Santos M, Chagas-de-Cavalcante JC, Cordeiro-Nascimento WM, Arcanjo CC, Santos PR (2020) Iron pots for the treatment of iron deficiency anemia: is there enough favorable evidence? Int J Health Sci 8:73–76. https://doi.org/10.19080/NFSIJ.2019.08.555738
Baker MN (1934) Sketch of the history of water treatment. J Am Water Works Assoc 26:902–938
Balko BA, Bossé SA, Cade AE, Jones-Landry EF, Amonette JE, Daschbach JL (2012) The effect of smectite on the corrosion of iron metal. Clays Clay Miner 60:136–152. https://doi.org/10.1346/CCMN.2012.0600204
Banerji T, Chaudhari S (2017) A cost-effective technology for arsenic removal: case study of zerovalent iron-based IIT Bombay arsenic filter in West Bengal. In: Nath K, Sharma V (eds) Water and sanitation in the New Millennium. Springer, New Delhi
Bartzas G, Komnitsas K (2010) Solid phase studies and geochemical modelling of low-cost permeable reactive barriers. J Hazard Mater 183:301–308. https://doi.org/10.1016/j.jhazmat.2010.07.024
Baumgartner J, Murcott S, Ezzati M (2007) Reconsidering ‘appropriate technology’: the effects of operating conditions on the bacterial removal performance of two household drinking-water filter systems. Environ Res Lett 2:024003. https://doi.org/10.1088/1748-9326/2/2/024003
Berardozzi E, Donadelli JA, Teixeira ACSC, Guardani R, Einschlag FSG (2023) Investigation of zero-valent iron (ZVI)/H2O continuous processes using multivariate analysis and artificial neural networks. Chem Eng J 453:139930. https://doi.org/10.1016/j.cej.2022.139930
Bi E, Devlin JF, Huang B (2009) Effects of mixing granular iron with sand on the kinetics of trichloroethylene reduction. Ground Water Monit Remed 29:56–62. https://doi.org/10.1111/j.1745-6592.2009.01234.x
Bigg T, Judd SJ (2000) Zero-valent iron for water treatment. Environ Technol 21:661–670. https://doi.org/10.1080/09593332108618077
Bilardi S, Calabrò PS, Moraci N (2023) A review of the hydraulic performance of permeable reactive barriers based on granular zero valent iron. Water 15:200. https://doi.org/10.3390/w15010200
Bischof G (1973) On the purification of water. Proc R Philos Soc Glasg 8:357–372
Bojić A, Purenović M, Kocić B, Perović J, Ursić-Janković J, Bojić D (2001) The inactivation of Escherichia coli by microalloyed aluminium based composite. Facta Univ 2:115–124
Bojic ALJ, Bojic D, Andjelkovic T (2009) Removal of Cu2+ and Zn2+ from model wastewaters by spontaneous reduction–coagulation process in flow conditions. J Hazard Mater 168:813–819. https://doi.org/10.1016/j.jhazmat.2009.02.096
Brenner S (2010) Sequences and consequences. Philos Trans R Soc B 365:207–212. https://doi.org/10.1098/rstb.2009.0221
Btatkeu-K BD, Miyajima K, Noubactep C, Caré S (2013) Testing the suitability of metallic iron for environmental remediation: Discoloration of methylene blue in column studies. Chem Eng J 215–216:959–968. https://doi.org/10.1016/j.cej.2012.11.072
Btatkeu-K BD, Tchatchueng JB, Noubactep C, Caré S (2016) Designing metallic iron based water filters: light from methylene blue discoloration. J Environ Manag 166:567–573. https://doi.org/10.1016/j.jenvman.2015.10.021
Btatkeu-K BD (2015) Optimizing the efficiency of household iron filters for safe drinking water. PhD Dissertation, University of Ngaoundéré, Ngaoundéré/Cameroon (in French)
Cao V, Ndé-Tchoupé AI, Hu R, Gwenzi W, Noubactep C (2021) The mechanism of contaminant removal in Fe(0)/H2O systems: The burden of a poor literature review. Chemosphere 280:130614. https://doi.org/10.1016/j.chemosphere.2021.130614
Cao V, Bakari O, Kenmogne-Tchidjo JF, Gatcha-Bandjun N, Ndé-Tchoupé AI, Gwenzi W, Njau KN, Noubactep C (2022) Conceptualizing the Fe0/H2O system: a call for collaboration to mark the 30th anniversary of the Fe0-based permeable reactive barrier technology. Water 14:3120. https://doi.org/10.3390/w14193120
Caré S, Nguyen QT, L’Hostis V, Berthaud Y (2008) Mechanical properties of the rust layer induced by impressed current method in reinforced mortar. Cement Concrete Res 38:1079–1091. https://doi.org/10.1016/j.cemconres.2008.03.016
Caré S, Crane R, Calabrò PS, Ghauch A, Temgoua E, Noubactep C (2013) Modeling the permeability loss of metallic iron water filtration systems. Clean Soil Air Water 41:275–282. https://doi.org/10.1002/clen.201200167
Charles CV (2012) Happy fish: a novel supplementation technique to prevent iron deficiency anemia in women in rural Cambodia. PhD Dissertation, The University of Guelph, Canada
Charlet L, Liger E, Gerasimo P (1998) Decontamination of TCE- and U-rich waters by granular iron: role of sorbed Fe (II). J Environ Eng 124:25–30. https://doi.org/10.1061/(ASCE)0733-9372(1998)124:1(25)
Chaves LHG (2005) The role of green rust in the environment: a review. Rev Bras Eng Agríc Ambient 9:284–288. https://doi.org/10.1590/S1415-43662005000200021
Chekli L, Bayatsarmadi B, Sekine R, Sarkar B, Maoz Shen A, Scheckel KG, Skinner W, Naidu R, Shon HK, Lombi E, Donner E (2016) Analytical characterisation of nanoscale zero-valent iron: a methodological review. Anal Chim Acta 903:13–35. https://doi.org/10.1016/j.aca.2015.10.040
Chen Q, Fan G, Na W, Liu J, Cui J, Li H (2019) Past, present, and future of groundwater remediation research: a scientometric analysis. Int J Environ Res Public Health 16:3975. https://doi.org/10.3390/ijerph16203975
Cormican A, Devlin JF, Divine C (2020) Grain size analysis and permeametry for estimating hydraulic conductivity in engineered porous media. Groundw Monit Remed 40:65–72. https://doi.org/10.1111/gwmr.12379
Crespo EB, Reichelt-Brushett A, Smith REW, Rose AL, Batley GE (2023) Improving the measurement of Iron(III) bioavailability in freshwater samples: methods and performance. Environ Toxicol Chem 42:303–316. https://doi.org/10.1002/etc.5530
Cui X, Xiao M, Tao R, Hu R, Ruppert H, Gwenzi W, Noubactep C (2023) Developing the ascorbic acid test: a candidate standard tool for characterizing the intrinsic reactivity of metallic iron for water remediation. Water 15:1930. https://doi.org/10.3390/w15101930
Cundy AB, Hopkinson L, Whitby RLD (2008) Use of iron-based technologies in contaminated land and groundwater remediation: a review. Sci Tot Environ 400:42–51. https://doi.org/10.1016/j.scitotenv.2008.07.002
Devlin JF, Klausen J, Schwarzenbach RP (1998) Kinetics of nitroaromatic reduction on granular iron in recirculating batch experiments. Environ Sci Technol 32:1941–1947. https://doi.org/10.1021/es970896g
Devonshire E (1890) The purification of water by means of metallic iron. J Frankl Inst 129:449–461. https://doi.org/10.1016/0016-0032(90)90189-P
Domga R, Togue-Kamga F, Noubactep C, Tchatchueng JB (2015) Discussing porosity loss of Fe0 packed water filters at ground level. Chem Eng J 263:127–134. https://doi.org/10.1016/j.cej.2014.10.105
Dutra-de-Oliveira JE, Marchini JS, Lamounier J, Almeida CA (2011) Iron-fortified drinking water studies for the prevention of children’s anemia in developing countries. Anemia. https://doi.org/10.1155/2011/815194
Edward D, Karungamye P, Nelson G, Selemani J, Njau KN (2023) Removal of arsenic in a sand filter coupled with zero valent iron. HydroResearch 6:228–234. https://doi.org/10.1016/j.hydres.2023.06.002
El Ibrahimi B, Nardeli JV, Guo L (2021) An overview of corrosion. In: Sustainable corrosion inhibitors I: fundamentals, methodologies, and industrial applications, vol 1403, pp. 1–19. https://doi.org/10.1021/bk-2021-1403.ch001
Erickson AJ, Gulliver JS, Weiss PT (2012) Capturing phosphates with iron enhanced sand filtration. Water Res 46:3032–3042. https://doi.org/10.1016/j.watres.2012.03.009
Etmannski TR, Darton RC (2014) A methodology for the sustainability assessment of arsenic mitigation technology for drinking water. Sci Tot Environ 488–489:505–511. https://doi.org/10.1016/j.scitotenv.2013.10.112
Evans UR, Hoar TP (1932) The velocity of corrosion from the electrochemical standpoint. Part II. Proc R Soc Lond A 137:343–365. https://doi.org/10.1098/rspa.1932.0140
Evans UR, Mears RB (1934) The velocity of corrosion from the electrochemical standpoint. Part III. Proc R Soc Lond A 146:153–165. https://doi.org/10.1098/rspa.1934.0144
Evans UR, Bannister LC, Britton SC (1931) The velocity of corrosion from the electrochemical standpoint. Proc R Soc Lond A 131:355–375. https://doi.org/10.1098/rspa.1931.0058
Farrell J, Bostick WD, Jarabeck RJ, Fiedor JN (1999) Uranium removal from ground water using zero valent iron media. Ground Water 34:618–624. https://doi.org/10.1111/j.1745-6584.1999.tb01150.x
Farrell J, Wang J, O’Day P, Conklin M (2001) Electrochemical and spectroscopic study of arsenate removal from water using zero-valent iron media. Environ Sci Technol 35:2026–2032. https://doi.org/10.1021/es0016710
Fiedor JN, Bostick WD, Jarabek RJ, Farrel J (1998) Understanding the mechanism, of uranium removal from groundwater by zero-valent iron using X-ray photoelectron spectroscopy. Environ Sci Technol 32:1466–1473. https://doi.org/10.1021/es970385u
Firdous R, Devlin JF (2014) Consideration of grain packing in granular iron treatability studies. J Contam Hydrol 164:230–239. https://doi.org/10.1016/j.jconhyd.2014.05.014
Firdous R, Devlin JF (2015) Visualizations and optimization of iron–sand mixtures for permeable reactive barriers. Groundw Monit Remed 35:78–84. https://doi.org/10.1111/gwmr.12123
Fu F, Dionysiou DD, Liu H (2014) The use of zero-valent iron for groundwater remediation and wastewater treatment: a review. J Hazard Mater 267:194–205. https://doi.org/10.1016/j.jhazmat.2013.12.062
Furukawa Y, Kim J-W, Watkins J, Wilkin RT (2002) Formation of ferrihydrite and associated iron corrosion products in permeable reactive barriers of zero-valent iron. Environ Sci Technol 36:5469–5475. https://doi.org/10.1021/es025533h
Gaspar DJ, Lea AS, Engelhard MH, Baer DR, Miehr R, Tratnyek PG (2002) Evidence for localization of reaction upon reduction of carbon tetrachloride by granular iron. Langmuir 18:7688–7693. https://doi.org/10.1021/la025798+
Ghauch A (2015) Iron-based metallic systems: An excellent choice for sustainable water treatment. Freiberg Online Geosci 32:1–80
Ghauch A, Abou Assi H, Baydoun H, Tuqan AM, Bejjani A (2011) Fe0-based trimetallic systems for the removal of aqueous diclofenac: mechanism and kinetics. Chem Eng J 172:1033–1044. https://doi.org/10.1016/j.cej.2011.07.020
Gheju M (2011) Hexavalent chromium reduction with zero-valent iron (ZVI) in aquatic systems. Water Air Soil Pollut 222:103–148. https://doi.org/10.1007/s11270-011-0812-y
Gheju M (2018) Progress in understanding the mechanism of CrVI removal in Fe0-based filtration systems. Water 10:651. https://doi.org/10.3390/w10050651
Gheju M, Balcu I (2023) Effect of sand co-presence on CrVI removal in Fe0-H2O system. Water 15:777. https://doi.org/10.3390/w15040777
Ghosh S, Singh S, Chaudhari S (2024) Arsenic and iron removal by zero-valent iron and electrocoagulation. In: Bandyopadhyay S (ed) Advances in drinking water purification: Small systems and emerging issues. Elsevier, Amsterdam, pp 69–90
Giles DE, Mohapatra M, Issa TB, Anand S, Singh P (2011) Iron and aluminium based adsorption strategies for removing arsenic from water. J Environ Manag 92:3011–3022. https://doi.org/10.1016/j.jenvman.2011.07.018
Gillham RW, O’Hannesin SF (1994) Enhanced degradation of halogenated aliphatics by zero-valent iron. Ground Water 32:958–967. https://doi.org/10.1111/j.1745-6584.1994.tb00935.x
Guan X, Sun Y, Qin H, Li J, Lo IMC, He D, Dong H (2015) The limitations of applying zero-valent iron technology in contaminants sequestration and the corresponding countermeasures: The development in zero-valent iron technology in the last two decades (1994–2014). Water Res 75:224–248. https://doi.org/10.1016/j.watres.2015.02.034
Hao Z, Xu X, Wang D (2005) Reductive denitrification of nitrate by scrap iron filings. J Zhejiang Univ Sci B 6:182–187. https://doi.org/10.1631/jzus.2005.B0182
Henderson AD, Demond AH (2007) Long-term performance of zero-valent iron permeable reactive barriers: a critical review. Environ Eng Sci 24:401–423. https://doi.org/10.1089/ees.2006.0071
Holmes DR, Meadowcroft DB (1977) Physical methods in corrosion technology. Phys Technol 8:2–9. https://doi.org/10.1088/0305-4624/8/1/I01
Hu R, Ndé-Tchoupé AI, Cao C, Gwenzi W, Noubactep C (2021) Metallic iron for environmental remediation: the fallacy of the electron efficiency concept. Front Environ Chem 2:677813. https://doi.org/10.3389/fenvc.2021.677813
Huang Z, Cao V, Nya EL, Gwenzi W, Noubactep C (2021a) Kanchan arsenic filters and the future of Fe0-based filtration systems for single household drinking water supply. Processes 9:58. https://doi.org/10.3390/pr9010058
Huang Z, Nya EL, Cao V, Gwenzi W, Rahman MA, Noubactep C (2021b) Universal access to safe drinking water: escaping the traps of non-frugal technologies. Sustainability 13:9645. https://doi.org/10.3390/su13179645
Hurrell RF (2021) Iron fortification practices and implications for iron addition to salt. J Nutr 151:3S-14S. https://doi.org/10.1093/jn/nxaa175
Hussam A (2009) Contending with a development disaster: sono filters remove arsenic from well water in Bangladesh. Innovations 4:89–102. https://doi.org/10.1162/itgg.2009.4.3.89
Hussam A, Munir AKM (2007) A simple and effective arsenic filter based on composite iron matrix: development and deployment studies for groundwater of Bangladesh. J Environ Sci Health A 42:1869–1878. https://doi.org/10.1080/10934520701567122
ITRC (Interstate Technology & Regulatory Council) (2005) Permeable reactive barriers: lessons learned/new directions. PRB-4. Washington: Interstate Technology & Regulatory Council, Permeable Reactive Barriers Team. www.itrcweb.org. Access 09 Mar 2012
ITRC (Interstate Technology & Regulatory Council) (2011) Permeable reactive barrier: technology update. PRB-5. Washington: Interstate Technology & Regulatory Council, PRB: Technology Update Team. www.itrcweb.org. Access 09 Mar 2012
James BR, Rabenhorst MC, Frigon GA (1992) Phosphorus sorption by peat and sand amended with iron oxides or steel wool. Water Environ Res 64:699–705. https://doi.org/10.2175/WER.64.5.6
Jia Y, Aagaard P, Breedveld GD (2007a) Sorption of triazoles to soil and iron minerals. Chemosphere 67:250–258. https://doi.org/10.1016/j.chemosphere.2006.10.021
Jia Y, Breedveld GD, Aagaard P (2007b) Column studies on transport of deicing additive benzotriazole in a sandy aquifer and a zerovalent iron barrier. Chemosphere 69:1409–1418. https://doi.org/10.1016/j.chemosphere.2007.04.074
Jiao Y, Qiu C, Huang L, Wu K, Ma H, Chen S, Ma L, Wu L (2009) Reductive dechlorination of carbon tetrachloride by zero-valent iron and related iron corrosion. Appl Catal B Environ 91:434–440. https://doi.org/10.1016/j.apcatb.2009.06.012
Kaplan DI, Gilmore TJ (2004) Zero-valent iron removal rates of aqueous Cr(VI) Measured under flow conditions. Water Air Soil Pollut 155:21–33. https://doi.org/10.1023/B:WATE.0000026518.23591.ac
Kenneke JF, McCutcheon SC (2003) Use of pretreatment zones and zero-valent iron for the remediation of chloroalkenes in an oxic aquifer. Environ Sci Technol 37:2829–2835. https://doi.org/10.1021/es0207302
Kishimoto N, Iwano S, Narazaki Y (2011) Mechanistic consideration of zinc ion removal by zero-valent iron. Water Air Soil Pollut 221:183–189. https://doi.org/10.1007/s11270-011-0781-1
Konadu-Amoah B, Ndé-Tchoupé AI, Hu R, Gwenzi W, Noubactep C (2021) Investigating the Fe0/H2O systems using the methylene blue method: validity, applications and future directions. Chemosphere 291:132913. https://doi.org/10.1016/j.chemosphere.2021.132913
Konadu-Amoah B, Hu R, Yang H, Cao V, Ndé-Tchoupé AI, Gwenzi W, Ruppert H, Noubactep C (2022) Realizing the potential of metallic iron for environmental remediation: Flee or adapt? Appl Water Sci 12:217. https://doi.org/10.1007/s13201-022-01738-9
Konadu-Amoah B, Hu R, Cui X, Tao R, Ndé-Tchoupé AI, Gwenzi W, Noubactep C (2023) Characterizing the process of phosphate removal in Fe0/H2O systems. Chem Eng J 465:143042. https://doi.org/10.1016/j.cej.2023.143042
Konadu-Amoah B (2023) Decontamination in Fe0-based systems: Understanding phosphate removal in Fe0/H2O system using the MB method. PhD Dissertation, Hohai University, Najing, China.
Kumari A, Chauhan AK (2022) Iron nanoparticles as a promising compound for food fortification in iron deficiency anemia: a review. J Food Sci Technol 59:3319–3335. https://doi.org/10.1007/s13197-021-05184-4
Kundu DK, Mol APJ, Gupta A (2016) Failing arsenic mitigation technology in rural Bangladesh: explaining stagnation in niche formation of the Sono filter. Water Pol 18:1490–1507. https://doi.org/10.2166/wp.2016.014
Kundu DK, Gupta A, Mol APJ, Rahman MM, van Halem D (2018) Experimenting with a novel technology for provision of safe drinking water in rural Bangladesh: the case of sub-surface arsenic removal (SAR). Technol Soc 53:161–172. https://doi.org/10.1016/j.techsoc.2018.01.010
Lan LE, Reina FD, De Seta GE, Meichtry JM, Litter MI (2023) Comparison between different technologies (zerovalent iron, coagulation-flocculation, adsorption) for arsenic treatment at high concentrations. Water 15:1481. https://doi.org/10.3390/w15081481
Landolt D (2007) Corrosion and surface chemistry of metals. EPEL Press, Lausanne
Lavine BK, Auslander G, Ritter J (2001) Polarographic studies of zero valent iron as a reductant for remediation of nitroaromatics in the environment. Microchem J 70:69–83. https://doi.org/10.1016/S0026-265X(01)00075-3
Lawrinenko M, Kurwadkar S, Wilkin RT (2023a) Long–term performance evaluation of zero-valent iron amended permeable reactive barriers for groundwater remediation—a mechanistic approach. Geosci Front 14:101494. https://doi.org/10.1016/j.gsf.2022.101494
Lawrinenko M, Kurwadkar S, Wilkin RT (2023b) Reply to comment by C. Noubactep on “Long-term performance evaluation of zero-valent iron amended permeable reactive barriers for groundwater remediation: a mechanistic approach.” Geosci Front 14:101583. https://doi.org/10.1016/j.gsf.2023.101583
Lee G, Rho S, Jahng D (2004) Design considerations for groundwater remediation using reduced metals. Korean J Chem Eng 21:621–628. https://doi.org/10.1007/BF02705496
Li L, Benson CH (2010) Evaluation of five strategies to limit the impact of fouling in permeable reactive barriers. J Hazard Mater 181:170–180. https://doi.org/10.1016/j.jhazmat.2010.04.113
Li H, Liu Q (2022) Reaction medium for permeable reactive barrier remediation of groundwater polluted by heavy metals. Front Environ Sci 10:968546. https://doi.org/10.3389/fenvs.2022.968546
Li L, Benson CH, Lawson EM (2005) Impact of mineral fouling on hydraulic behavior of permeable reactive barriers. Ground Water 43:582–596. https://doi.org/10.1111/j.1745-6584.2005.0042.x
Li L, Benson CH, Lawson EM (2006) Modeling porosity reductions caused by mineral fouling in continuous-wall permeable reactive barriers. J Contam Hydrol 83:89–121. https://doi.org/10.1016/j.jconhyd.2005.11.004
Li J, Dou X, Qin H, Sun Y, Yin D, Guan X (2019) Characterization methods of zerovalent iron for water treatment and remediation. Water Res 148:70–85. https://doi.org/10.1016/j.watres.2018.10.025
Li X, Li Z, Du C, Tian Z, Zhu Q, Li G, Shen Q, Li C, Li J, Li W, Zhao C, Zhang L (2021) Bibliometric analysis of zerovalent iron particles research for environmental remediation from 2000 to 2019. Environ Sci Pollut Res 28:4200–34210. https://doi.org/10.1007/s11356-021-13847-0
Li L, Li J, Ma T, Liao G, Gao F, Duan W, Luo K, Wang C (2023) Construction of core–shell Sb2S3@Cds nanorod with enhanced heterointerface interaction for chromium-containing wastewater treatment. Small 19:2302737. https://doi.org/10.1002/smll.202302737
Li S, Li C, Wang F (2024) Computational experiments of metal corrosion studies: a review. Mater Today Chem 37:101986. https://doi.org/10.1016/j.mtchem.2024.101986
Liger E, Charlet L, van Cappellen P (1999) Surface catalysis of uranium(VI) reduction by iron(II). Geochim Cosmochim Acta 63:2939–2955. https://doi.org/10.1016/S0016-7037(99)00265-3
Lipczynska-Kochany E, Harms S, Milburn R, Sprah G, Nadarajah N (1994) Degradation of carbon tetrachloride in the presence of iron and sulphur containing compounds. Chemosphere 29:1477–1489. https://doi.org/10.1016/0045-6535(94)90279-8
Lufingo M, Ndé-Tchoupé AI, Hu R, Njau KN, Noubactep C (2019) A novel and facile method to characterize the suitability of metallic iron for water treatment. Water 11:2465. https://doi.org/10.3390/w11122465
Luo P, Bailey EH, Mooney SJ (2013) Quantification of changes in zero valent iron morphology using X-ray computed tomography. J Environ Sci 25:2344–2351. https://doi.org/10.1016/S1001-0742(12)60237-X
Makota S, Ndé-Tchoupé AI, Mwakabona HT, Tepong-Tsindé R, Noubactep C, Nassi A, Njau KN (2017) Metallic iron for water treatment: Leaving the valley of confusion. Appl Water Sci 7:4177–4196. https://doi.org/10.1007/s13201-017-0601-x
Massey L, McGeehan S, Ratts M, Mitchell T, Cotten C, Perez A, Beerman K (2023) Can providing daily iron-fortified lunches to school-going children living in an impoverished Guatemalan community improve iron status? J Glob Health Rep 7:e2023018. https://doi.org/10.29392/001c.75318
Matheson LJ, Tratnyek P (1994) Reductive dehalogenation of chlorinated methanes by iron metal. Environ Sci Technol 28:2045–2053. https://doi.org/10.1021/es00061a012
McGeough KL, Kalin RM, Myles P (2007) Carbon disulfide removal by zero valent iron. Environ Sci Technol 41:4607–4612. https://doi.org/10.1021/es062936z
McGuire MM, Carlson DL, Vikesland PJ, Kohn T, Grenier AC, Langley LA, Roberts AL, Fairbrother DH (2003) Applications of surface analysis in the environmental sciences: dehalogenation of chlorocarbons with zero-valent iron and iron-containing mineral surfaces. Anal Chim Acta 496:301–313. https://doi.org/10.1016/S0003-2670(03)01009-2
Mitchell G, Poole P, Segrove HD (1955) Adsorption of methylene blue by high-silica sands. Nature 176:1025–1026. https://doi.org/10.1038/1761025a0
Miyajima K (2012) Optimizing the design of metallic iron filters for water treatment. Freiberg Online Geosci 32:1–60
Miyajima K, Noubactep C (2012) Effects of mixing granular iron with sand on the efficiency of methylene blue discoloration. Chem Eng J 200–202:433–438. https://doi.org/10.1016/j.cej.2012.06.069
Moraci N, Calabrò PS (2010) Heavy metals removal and hydraulic performance in zero-valent iron/pumice permeable reactive barriers. J Environ Manag 91:2336–2341. https://doi.org/10.1016/j.jenvman.2010.06.019
Moraci N, Lelo D, Bilardi S, Calabrò PS (2016) Modelling long-term hydraulic conductivity behaviour of zero valent iron column tests for permeable reactive barrier design. Can Geotech J 53:946–961. https://doi.org/10.1139/cgj-2015-0453
Mueller B, Dangol B, Ngai TKK, Hug SJ (2021) Kanchan arsenic filters in the lowlands of Nepal: mode of operation, arsenic removal, and future improvements. Environ Geochem Health 43:375–389. https://doi.org/10.1007/s10653-020-00718-9
Mueller B, Chan MCK, Hug SJ (2023) Unique geochemistry of arsenic-contaminated groundwater and corresponding mitigation efforts in Southern Nepal. ACS EST Water 3:1527–1535. https://doi.org/10.1021/acsestwater.2c00404
Murcott S (2007) Co-evolutionary design for development: influences shaping engineering design and implementation in Nepal and the global village. J Int Dev 19:123–144. https://doi.org/10.1002/jid.1353
Mwakabona HT, Ndé-Tchoupé AI, Njau KN, Noubactep C, Wydra KD (2017) Metallic iron for safe drinking water provision: Considering a lost knowledge. Water Res 117:127–142. https://doi.org/10.1016/j.watres.2017.03.001
Naseri E, Ndé-Tchoupé AI, Mwakabona HT, Nanseu-Njiki CP, Noubactep C, Njau KN, Wydra KD (2017) Making Fe0-based filters a universal solution for safe drinking water provision. Sustainability 9:1224. https://doi.org/10.3390/su9071224
Ndé-Tchoupé AI, Konadu-Amoah B, Gatcha-Bandjun N, Hu R, Gwenzi W, Noubactep C (2022) Kanchan arsenic filters for household water treatment: Unsuitable or unsustainable? Water 14:2318. https://doi.org/10.3390/w14152318
Nesic S (2007) Key issues related to modelling of internal corrosion of oil and gas pipelines—a review. Corros Sci 49:4308–4338. https://doi.org/10.1016/j.corsci.2007.06.006
Neumann A, Kaegi R, Voegelin A, Hussam A, Munir AKM, Hug SJ (2013) Arsenic removal with composite iron matrix filters in Bangladesh: a field and laboratory study. Environ Sci Technol 47:4544–4554. https://doi.org/10.1021/es305176x
Ngai TKK, Murcott S, Shrestha RR, Dangol B, Maharjan M (2006) Development and dissemination of Kanchan™ Arsenic Filter in rural Nepal. Water Sci Technol Water Supply 6:137–146. https://doi.org/10.2166/ws.2006.807
Ngai TKK, Shrestha RR, Dangol B, Maharjan M, Murcott SE (2007) Design for sustainable development—household drinking water filter for arsenic and pathogen treatment in Nepal. J Environ Sci Health A 42:1879–1888. https://doi.org/10.1080/10934520701567148
Nguyen PM, Mulligan CN (2023) Study on the influence of water composition on iron nail corrosion and arsenic removal performance of the Kanchan arsenic filter (KAF). J Hazard Mater Adv 10:100285. https://doi.org/10.1016/j.hazadv.2023.100285
Noubactep C (2007) Processes of contaminant removal in “Fe0–H2O” systems revisited. The importance of co-precipitation. Open Environ Sci 1:9–13. https://doi.org/10.23689/FIDGEO-823
Noubactep C (2008) A critical review on the mechanism of contaminant removal in Fe0–H2O systems. Environ Technol 29:909–920. https://doi.org/10.1080/09593330802131602
Noubactep C (2009) An analysis of the evolution of reactive species in Fe0/H2O systems. J Hazard Mater 168:1626–1631. https://doi.org/10.1016/j.jhazmat.2009.02.143
Noubactep C (2010) Metallic iron for safe drinking water worldwide. Chem Eng J 165:740–749. https://doi.org/10.1016/j.cej.2010.09.065
Noubactep C (2011) Aqueous contaminant removal by metallic iron: is the paradigm shifting? Water SA 37:419–426. https://doi.org/10.4314/wsa.v37i3.68493
Noubactep C (2012) Investigating the processes of contaminant removal in Fe0/H2O systems. Korean J Chem Eng 29:1050–1056. https://doi.org/10.1007/s11814-011-0298-8
Noubactep C (2013) Metallic iron for water treatment: a critical review. Clean Soil Air Water 41:702–710. https://doi.org/10.1002/clen.201200502
Noubactep C (2015) Metallic iron for environmental remediation: a review of reviews. Water Res 85:114–123. https://doi.org/10.1016/j.watres.2015.08.023
Noubactep C (2016a) Research on metallic iron for environmental remediation: stopping growing sloppy science. Chemosphere 153:528–530. https://doi.org/10.1016/j.chemosphere.2016.03.088
Noubactep C (2016b) Predicting the hydraulic conductivity of metallic iron filters: Modeling gone astray. Water 8:162. https://doi.org/10.3390/w8040162
Noubactep C (2018) Metallic iron (Fe0) provide possible solution to universal safe drinking water provision. J Water Technol Treat Methods 1:102. https://doi.org/10.31021/jwt.20181102
Noubactep C (2020) Metallic iron for environmental remediation: prospects and limitations. Chap 36. In: D’Mello JPF (ed) A handbook of environmental toxicology: human disorders and ecotoxicology. CAB International, Wallingford, pp 531–544
Noubactep C (2023) Comments on "Long-term performance evaluation of zero-valent iron amended permeable reactive barriers for groundwater remediation: a mechanistic approach" by Lawrinenko et al. Geosci Frontiers 14:101494. Geosci Front 14:101582. https://doi.org/10.1016/j.gsf.2023.101583
Noubactep C (2024) Metallic iron for decentralized safe drinking water supply: self-reliance is possible. In: Bandyopadhyay S (ed) Advances in drinking water purification: small systems and emerging issues. Elsevier, Amsterdam, pp 231–251
Noubactep C, Caré S (2010a) Dimensioning metallic iron beds for efficient contaminant removal. Chem Eng J 163:454–460. https://doi.org/10.1016/j.cej.2010.07.051
Noubactep C, Caré S (2010b) Enhancing sustainability of household water filters by mixing metallic iron with porous materials. Chem Eng J 162:635–642. https://doi.org/10.1016/j.cej.2010.06.012
Noubactep C, Meinrath G, Dietrich P, Sauter M, Merkel B (2005a) Testing the suitability of zerovalent iron materials for reactive walls. Environ Chem 2:71–76. https://doi.org/10.1071/EN04014
Noubactep C, Meinrath G, Merkel JB (2005b) Investigating the mechanism of uranium removal by zerovalent iron materials. Environ Chem 2:235–242. https://doi.org/10.1071/EN05003
Noubactep C, Schöner A, Meinrath G (2006) Mechanism of uranium (VI) fixation by elemental iron. J Hazard Mater 132:202–212. https://doi.org/10.1016/j.jhazmat.2005.08.047
Noubactep C, Licha T, Scott TB, Fall M, Sauter M (2009a) Exploring the influence of operational parameters on the reactivity of elemental iron materials. J Hazard Mater 172:943–951. https://doi.org/10.1016/j.jhazmat.2009.07.097
Noubactep C, Schöner A, Woafo P (2009b) Metallic iron filters for universal access to safe drinking water. Clean Soil Air Water 37:930–937. https://doi.org/10.1002/clen.200900114
Noubactep C, Btatkeu-K BD, Tchatchueng JB (2011) Impact of MnO2 on the efficiency of metallic iron for the removal of dissolved metal. Chem Eng J 178:78–84. https://doi.org/10.1016/j.cej.2011.10.014
Noubactep C, Schöner A, Sauter M (2012a) Significance of oxide-film in discussing the mechanism of contaminant removal by elemental iron materials. In: Photo-electrochemistry & photo-biology for the sustainability. Union Press, Osaka. ISBN-10: 4946428615; ISBN-13: 978-4946428616, pp. 97–122.
Noubactep C, Caré S, Btatkeu KBD, Nanseu-Njiki CP (2012b) Enhancing the sustainability of household Fe0/sand filters by using bimetallics and MnO2. Clean Soil Air Water 40:100–109. https://doi.org/10.1002/clen.201100014
Noubactep C, Kenmogne-Tchidjo JF, Vollmer S (2023a) Iron-fortified water: a new approach for reducing iron deficiency anemia in resource-constrained settings. Sci Rep 13:13565. https://doi.org/10.1038/s41598-023-40600-z
Noubactep C, Meinrath G, Dietrich P, Merkel B (2003b) Mitigating uranium in groundwater: prospects and limitations. Environ Sci Technol 37:4304–4308. https://doi.org/10.1021/es034296v
Obiri-Nyarko F, Grajales-Mesa SJ, Malina G (2014) An overview of permeable reactive barriers for in situ sustainable groundwater remediation. Chemosphere 111:243–259. https://doi.org/10.1016/j.chemosphere.2014.03.112
Ogata R, Dangol B, Sakamoto M (2020) Sustainability assessment of long-term, widely used household Kanchan Arsenic Filters in Nepal. J Environ Sci Health A 55:517–527. https://doi.org/10.1080/10934529.2019.1710414
O’Hannesin SF, Gillham RW (1998) Long-term performance of an in situ “iron wall” for remediation of VOCs. Ground Water 36:164–170. https://doi.org/10.1111/j.1745-6584.1998.tb01077.x
Pasupathy E, Kandasamy R, Thomas K, Basheer A (2023) Alternate day versus daily oral iron for treatment of iron deficiency anemia: a randomized controlled trial. Sci Rep 13:1818. https://doi.org/10.1038/s41598-023-29034-9
Phukan M (2015) Characterizing the Fe0/sand system by the extent of dye discoloration. Freiberg Online Geosci 40:1–70
Pilling NB, Bedworth RE (1923) The oxidation of metals at high temperatures. J Inst Metals 29:529–591
Plessl K, Russ A, Vollprecht D (2023) Application and development of zero-valent iron (ZVI) for groundwater and wastewater treatment. Int J Environ Sci Technol 20:6913–6928. https://doi.org/10.1007/s13762-022-04536-7
Polasek P (2007) Differentiation between different kinds of mixing in water purification—back to basics. Water SA 33:249–251. https://doi.org/10.4314/wsa.v33i2.49082
Qiu SR, Lai H-F, Roberson MJ, Hunt ML, Amrhein C, Giancarlo LC, Flynn GW, Yarmoff (2000) Removal of contaminants from aqueous solution by reaction with iron surfaces. Langmuir 16:2230–2236. https://doi.org/10.1021/la990902h
Rahman MA, Karmakar S, Salama H, Gactha-Bandjun N, Btatkeu-K BD, Noubactep C (2013) Optimising the design of Fe0-based filtration systems for water treatment: the suitability of porous iron composites. J Appl Solut Chem Model 2:165–177. https://doi.org/10.6000/1929-5030.2013.02.03.2
Rai RK, Fawzi WW, Barik A, Chowdhury A (2018) The burden of iron-deficiency anaemia among women in India: how have iron and folic acid interventions fared? WHO South East Asia J Public Health 7:18–23. https://doi.org/10.4103/2224-3151.228423
Raychoudhury T, Scheytt T (2013) Potential of zerovalent iron nanoparticles for remediation of environmental organic contaminants in water: a review. Water Sci Technol 68:1425–1439. https://doi.org/10.2166/wst.2013.358
Reardon EJ (1995) Anaerobic corrosion of granular iron: measurement and interpretation of hydrogen evolution rates. Environ Sci Technol 29:2936–2945. https://doi.org/10.1021/es00012a008
Reardon EJ (2005) Zerovalent irons: Styles of corrosion and inorganic control on hydrogen pressure buildup. Environ Sci Tchnol 39:7311–7317. https://doi.org/10.1021/es050507f
Ren Y, Cui M, Zhou Y, Lee Y, Ma J, Han Z, Khim J (2023) Zero-valent iron based materials selection for permeable reactive barrier using machine learning. J Hazard Mater 453:131349. https://doi.org/10.1016/j.jhazmat.2023.131349
Reynolds GW, Hoff JT, Gillham RW (1990) Sampling bias caused by materials used to monitor halocarbons in groundwater. Environ Sci Technol 24:135–142. https://doi.org/10.1021/es00071a017
Rodríguez-Vivaldi AM, Beerman K (2018) Testing the efficacy of the Lucky Iron Fish® in reversing iron deficiency anemia in rural, impoverished regions of Guatemala. J Global Health Rep 2:e2018014. https://doi.org/10.29392/joghr.2.e2018014
Ruhl AS, Franz G, Gernert U, Jekel M (2014) Corrosion product and precipitate distribution in two-component Fe(0) permeable reactive barriers. Chem Eng J 239:26–32. https://doi.org/10.1016/j.cej.2013.11.017
Santisukkasaem U, Das DB (2019) A non-dimensional analysis of permeability loss in zero-valent iron permeable reactive barrier (PRB). Transp Porous Med 126:139–159. https://doi.org/10.1007/s11242-018-1096-0
Santisukkasaem U, Olawuyi F, Oye P, Das DB (2015) Artificial neural network (ANN) for evaluating permeability decline in permeable reactive barrier (PRB). Environ Process 2:291–307. https://doi.org/10.1007/s40710-015-0076-4
Sarr D (2001) Zero-valent-iron permeable reactive barriers - how long will they last? Remediation 11:1–18
Schreier CG, Reinhard M (1994) Transformation of chlorinated organic compounds by iron and manganese powders in buffered water and in landfill leachate. Chemosphere 29:1743–1753. https://doi.org/10.1016/0045-6535(94)90320-4
Shannon MA, Bohn PW, Elimelech M, Georgiadis JG, Marinas BJ, Mayes AM (2008) Science and technology for water purification in the coming decades. Nature 452:301–310. https://doi.org/10.1038/nature06599
Singh A, Smith LS, Shrestha S, Maden N (2014) Efficacy of arsenic filtration by Kanchan Arsenic Filter in Nepal. J Water Health 12:596–599. https://doi.org/10.2166/wh.2014.148
Singh R, Chakma S, Birke V (2023) Performance of field-scale permeable reactive barriers: an overview on potentials and possible implications for in-situ groundwater remediation applications. Sci Tot Environ 858:158838. https://doi.org/10.1016/j.scitotenv.2022.158838
Sista KS, Kumar D, Sinha GR, Moon AP, Dwarapudi S (2021) Iron powders as a potential material for arsenic removal in aqueous systems. ISIJ Int 61:2687–2702. https://doi.org/10.2355/isijinternational.ISIJINT-2021-258
Song D-I, Kim YH, Shin WS (2005) A simple mathematical analysis on the effect of sand in Cr(VI) reduction using zero valent iron. Korean J Chem Eng 22:67–69. https://doi.org/10.1007/BF02701464
Stefanoni M, Angst U, Elsener B (2018) Electrochemistry and capillary condensation theory reveal the mechanism of corrosion in dense porous media. Sci Rep 8:7407. https://doi.org/10.1038/s41598-018-25794-x
Stieber M, Putschew A, Jekel M (2011) Treatment of pharmaceuticals and diagnostic agents using zero-valent iron—kinetic studies and assessment of transformation products assay. Environ Sci Technol 45:4944–4950. https://doi.org/10.1021/es200034j
Stratmann M, Müller J (1994) The mechanism of the oxygen reduction on rust-covered metal substrates. Corros Sci 36:327–359. https://doi.org/10.1016/0010-938X(94)90161-9
Sun Y, Li J, Huang T, Guan X (2016) The influences of iron characteristics, operating conditions and solution chemistry on contaminants removal by zero-valent iron: a review. Water Res 100:277–295. https://doi.org/10.1016/j.watres.2016.05.031
Tao R, Yang H, Cui X, Xiao M, Gatcha-Bandjun N, Kenmogne-Tchidjo JF, Lufingo M, Konadu-Amoah B, Tepong-Tsindé R, Ndé-Tchoupé AI, Touomo-Wouafo M, Btatkeu-K BD, Gwenzi W, Hu R, Tchatchueng JB, Ruppert H, Noubactep C (2022) The suitability of hybrid Fe0/aggregate filtration systems for water treatment. Water 14:260. https://doi.org/10.3390/w14020260
Tao R, Cui X, Xiao M, Hu R, Gwenzi W, Ruppert H, Noubactep C (2023) Influence of water salinity on the efficiency on Fe0-based systems for water treatment. Water 15:2466. https://doi.org/10.3390/w15132466
Tao R, Hu R, Gwenzi W, Ruppert H, Noubactep C (2024) Effects of common dissolved anions on the efficiency of Fe0-based remediation systems. J Environ Manage (Accepted JEMA-D-24–01739)
Tao R (2024) Effects of solid additives and inorganic ligands on the efficiency of metallic iron-based water treatment systems. PhD Dissertation, University of Göttingen, Germany.
Tepong-Tsindé R (2021) Designing and piloting a household filter for the peri-urban population of Douala (Cameroon). Freiberg Online Geosci 61:1–80
Tepong-Tsindé R, Ndé-Tchoupé AI, Noubactep C, Nassi A, Ruppert H (2019) Characterizing a newly designed steel-wool-based household filter for safe drinking water provision: Hydraulic conductivity and efficiency for pathogen removal. Processes 7:966. https://doi.org/10.3390/pr7120966
Tien C (2007) Remarks on adsorption manuscripts revised and declined: an editorial. Sep Purif Technol 54:277–278. https://doi.org/10.1016/j.jhazmat.2007.04.015
Tran HN, You S-J, Hosseini-Bandegharaei A, Chao H-P (2017) Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: a critical review. Water Res 120:88–116. https://doi.org/10.1016/j.watres.2017.04.014
Tran HN, Bollinger J-C, Lima EC, Juang R-S (2023) How to avoid mistakes in treating adsorption isotherm data (liquid and solid phases): Some comments about correctly using Radke-Prausnitz nonlinear model and Langmuir equilibrium constant. J Environ Manag 325:116475. https://doi.org/10.1016/j.jenvman.2022.116475
Tratnyek PG (1996) Putting corrosion to use: remediating contaminated groundwater with zero-valent metals. Chem Ind 13:499–503
van Craenenbroeck W (1998) Easton & Anderson and the water supply of Antwerp (Belgium). Ind Archaeol Rev 20:105–116. https://doi.org/10.1179/iar.1998.20.1.105
Velimirovic M, Auffan M, Carniato L, Batka VM, Schmid D, Wagner S, Borschneck D, Proux O, von der Kammer F, Hofmann T (2018) Effect of field site hydrogeochemical conditions on the corrosion of milled zerovalent iron particles and their dechlorination efficiency. Sci Tot Environ 618:1619–1627. https://doi.org/10.1016/j.scitotenv.2017.10.002
Vidic RD, Suidan MT, Traegner UK, Nakhla GF (1990) Adsorption isotherms: illusive capacity and role of oxygen. Water Res 24:1187–1195. https://doi.org/10.1016/0043-1354(90)90041-4
Vikesland PJ, Klausen J, Zimmermann H, Roberts AL, Ball PW (2003) Longevity of granular iron in groundwater treatment processes: changes in solute transport properties over time. J Contam Hydrol 64:3–33. https://doi.org/10.1016/S0169-7722(02)00150-X
Vikesland PJ, Heathcock AM, Rebodos RL, Makus KE (2007) Particle size and aggregation effects on magnetite reactivity toward carbon tetrachloride. Environ Sci Technol 41:5277–5283. https://doi.org/10.1021/es062082i
Vollprecht D, Krois L-M, Sedlazeck KP, Müller P, Mischitz R, Olbrich T, Pomberger R (2018) Removal of critical metals from waste water by zero-valent iron. J Clean Prod 208:1409–1420. https://doi.org/10.1016/j.jclepro.2018.10.180
Vollprecht D, Plessl K, Neuhold S, Kittinger F, Öfner W, Müller P, Mischitz R, Sedlazeck KP (2020) Recovery of molybdenum, chromium, tungsten, copper, silver, and zinc from industrial waste waters using zero-valent iron and tailored beneficiation processes. Processes 8:279. https://doi.org/10.3390/pr8030279
Wang D, Gilliland SE III, Yi X, Logan K, Heitger DR, Lucas HR, Wang W-N (2018) Iron mesh-based metal organic framework filter for efficient arsenic removal. Environ Sci Technol 52:4275–4284. https://doi.org/10.1021/acs.est.7b06212
Warren KD, Arnold RG, Bishop TL, Lindholm LC, Betterton EA (1995) Kinetics and mechanism of reductive dehalogenation of carbon tetrachloride using zero-valence metals. J Hazard Mater 41:217–227. https://doi.org/10.1016/0304-3894(94)00117-Y
Wenk CB, Kaegi R, Hug SJ (2014) Factors affecting arsenic and uranium removal with zero-valent iron: laboratory tests with Kanchan-type iron nail filter columns with different groundwaters. Environ Chem 11:547–557. https://doi.org/10.1071/EN14020
Whitney WR (1903) The corrosion of iron. J Am Chem Soc 25:394–406. https://doi.org/10.1021/ja02006a008
Wielinski J, Jimenez-Martinez J, Göttlicher J, Steininger R, Mangold S, Hug SJ, Berg M, Voegelin A (2022) Spatiotemporal mineral phase evolution and arsenic retention in microfluidic models of zerovalent iron-based water treatment. Environ Sci Technol 56:13696–13708. https://doi.org/10.1021/acs.est.2c02189
Wilkin RT, Puls RW, Sewell GW (2002) Long-term performance of permeable reactive barriers using zero-valent iron: an evaluation at two sites. EPA 600-S-02-001, Environmental Research Brief. United States Environmental Protection Agency, Cincinnati, OH
Wilson EK (1995) Zero-valent metals provide possible solution to groundwater problems. Chem Eng News 73:19–22
Wu Q, Sun Y, Luo Z, Li X, Wen Y, Shi Y, Wu X, Huang X, Zhu Y, Huang C (2024) Application and development of zero-valent iron (ZVI)-based materials for environmental remediation: a scientometric and visualization analysis. Environ Res 241:117659. https://doi.org/10.1016/j.envres.2023.117659
Xiao M, Hu R, Tao R, Cui X, Konadu-Amoah B, Yang H, Ndé-Tchoupé AI, Gwenzi W, Noubactep C, Ruppert H (2023) Metallic iron for environmental remediation: the still overlooked iron chemistry. Appl Water Sci 13:222. https://doi.org/10.1007/s13201-023-02028-8
Xin J, Tang F, Zheng X, Shao H, Koldito O, Lu X (2016) Distinct kinetics and mechanisms of mZVI particles aging in saline and fresh groundwater: H2 evolution and surface passivation. Water Res 100:80–87. https://doi.org/10.1016/j.watres.2016.04.074
Yang H, Hu R, Ndé-Tchoupé AI, Gwenzi W, Ruppert H, Noubactep C (2020) Designing the next generation of Fe0-based filters for decentralized safe drinking water treatment. Processes 8:745. https://doi.org/10.3390/pr8060745
Yang H, Hu R, Ruppert H, Noubactep C (2021) Modeling porosity loss in Fe0-based permeable reactive barriers with Faraday’s law. Sci Rep 11:16998. https://doi.org/10.1038/s41598-021-96599-8
Yang H, Liu Q, Hu R, Ptak T, Taherdangkoo R, Liu Y, Noubactep C (2022) Numerical case studies on long-term effectiveness of metallic iron based permeable reactive barriers: importance of porosity heterogeneity of the barrier. J Hydrol 612:128148. https://doi.org/10.1016/j.jhydrol.2022.128148
Yang H (2022) Modelling the porosity loss of metallic iron beds for water treatment. PhD Dissertation, University of Göttingen, Germany.
You Y, Han J, Chiu PC, Jin Y (2005) Removal and inactivation of waterborne viruses using zerovalent iron. Environ Sci Technol 39:9263–9269. https://doi.org/10.1021/es050829j
Zarime NA, Solemon B, Yaacob WZW, Jamil H, Omara RC, Wahab WA, Isac AAM (2024) Removal of methylene blue by granitic residual soil supported nano zero valent iron (Gr-nZVI): kinetic and equilibrium studies. Desalin Water Treat 317:100099. https://doi.org/10.1016/j.dwt.2024.100099
Zhang W, Dong Y, Wang H, Guo Y, Zeng H, Zan J (2021) Removal of uranium from groundwater using zero-valent-iron coated quartz sands. J Radioanal Nucl Chem 327:1–12. https://doi.org/10.1007/s10967-020-07523-9
Zhao Y, Ren H, Dai H, Jin W (2011) Composition and expansion coefficient of rust based on X-ray diffraction and thermal analysis. Corros Sci 53:1646–1658. https://doi.org/10.1016/j.corsci.2011.01.007
Zhu Z-y, Li J-y, Li W, Liu X-y, Dang Y-y, Ma T-h, Wang C-y (2022) Simulated-sunlight-driven Cr(VI) reduction on a type-II heterostructured Sb2S3/CdS photocatalyst. Environ Sci Nano 9:1738–1747. https://doi.org/10.1039/D2EN00050D
Acknowledgements
The authors gratefully acknowledge the technical support for improving the illustrations by Junjie Qi (School of Earth Science and Engineering, Hohai University, Nanjing, China). We would like to thank the peer reviewers for their valuable suggestions and comments on improving this paper.
Funding
Open Access funding enabled and organized by Projekt DEAL. Funding for this study was received from the National Natural Science Foundation of China (NSFC Grant No. 42372280), “Investigating the permeability loss and the spatial heterogenous permeability change of zero-valent iron permeable reactive barrier for groundwater remediation.”
Author information
Authors and Affiliations
Contributions
MX, RT, XC and HY contributed to conceptualization, critical thinking and writing original draft. CN, WG and RH contributed to discussion and review editing. MX, RT, XC and CN contributed to data collection. CN, WG and RH contributed reviewing, proofreading and supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have given consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xiao, M., Hu, R., Gwenzi, W. et al. Materials for sustainable metallic iron-based water filters: a review. Environ Chem Lett 22, 2113–2131 (2024). https://doi.org/10.1007/s10311-024-01736-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-024-01736-0